Abstract
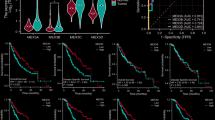
The pathogenesis of coxsackieviral infection is a multifactorial process involving host genetics, viral genetics and the environment in which they interact. We have used a mouse model of Coxsackievirus B3 infection to characterize the contribution of host genetics to infection survival and to viral hepatitis. Twenty-five AcB/BcA recombinant congenic mouse strains were screened. One, BcA86, was found to be particularly susceptible to early mortality; 100% of BcA86 mice died by day 6 compared with 0% of B6 mice (P=0.0012). This increased mortality was accompanied by an increased hepatic necrosis as measured by serum alanine aminotransferase (ALT) levels (19547±10556 vs 769±109, P=0.0055). This occurred despite a predominantly resistant (C57BL/6) genetic background. Linkage analysis in a cohort (n=210) of (BcA86x C56Bl/10)F2 animals revealed a new locus on chromosome 13 (peak linkage 101.2 Mbp, lod 4.50 and P=0.003). This locus controlled serum ALT levels as early as 48 h following the infection, and led to an elevated expression of type I interferon. Another locus on chromosome 17 (peak linkage 57.2 Mbp) was significantly linked to heart viral titer (lod 3.4 and P=0.046). These results provide new evidence for the presence of genetic loci contributing to the susceptibility of mice to viral hepatitis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Melnick JL, Godman GC . Pathogenesis of coxsackie virus infection; multiplication of virus and evolution of the muscle lesion in mice. J Exp Med 1951; 93: 247–266.
Gkrania-Klotsas E, Langenberg C, Tauriainen S, Sharp SJ, Luben R, Forouhi NG et al. The association between prior infection with five serotypes of Coxsackievirus B and incident type 2 diabetes mellitus in the EPIC-Norfolk study. Diabetologia 2012; 55: 967–970.
Cheng LL, Ng PC, Chan PK, Wong HL, Cheng FW, Tang JW . Probable intrafamilial transmission of coxsackievirus b3 with vertical transmission, severe early-onset neonatal hepatitis, and prolonged viral RNA shedding. Pediatrics 2006; 118: e929–e933.
Tseng F-C, Huang H-C, Chi C-Y, Lin T-L, Liu C-C, Jian J-W et al. Epidemiological survey of enterovirus infections occurring in Taiwan between 2000 and 2005: analysis of sentinel physician surveillance data. J Med Virol 2007; 79: 1850–1860.
Wang SM, Liu CC, Yang YJ, Yang HB, Lin CH, Wang JR . Fatal coxsackievirus B infection in early infancy characterized by fulminant hepatitis. J Infect 1998; 37: 270–273.
Yajima T, Knowlton KU . Viral myocarditis: from the perspective of the virus. Circulation 2009; 119: 2615–2624.
Gauntt C, Huber S . Coxsackievirus experimental heart diseases. Front Biosci 2003; 8: e23–e35.
Wolfgram L, Beisel K, Herskowitz A, Rose NR . Variations in the susceptibility to Coxsackievirus B3-induced myocarditis among different strains of mice. J Immunol 1986; 136: 1846–1852.
Gauntt C, Gomez P, Duffey P, Grant J, Trent D, Witherspoon S et al. Characterization and myocarditic capabilities of coxsackievirus B3 variants in selected mouse strains. J Virol 1984; 52: 598–605.
Wessely R, Klingel K, Knowlton KU, Kandolf R . Cardioselective infection with coxsackievirus B3 requires intact type I interferon signaling: implications for mortality and early viral replication. Circulation 2001; 103: 756–761.
Deonarain R, Cerullo D, Fuse K, Liu P, Fish E . Protective role for interferon-beta in coxsackievirus B3 infection. Circulation 2004; 110: 3540–3543.
Yasukawa H, Yajima T, Duplain H, Iwatate M, Kido M, Hoshijima M et al. The suppressor of cytokine signaling-1 (SOCS1) is a novel therapeutic target for enterovirus-induced cardiac injury. J Clin Invest 2003; 111: 469–478.
Richer MJ, Lavallee DJ, Shanina I, Horwitz MS . Toll-like receptor 3 signaling on macrophages is required for survival following coxsackievirus B4 infection. PLoS One 2009; 4: e4127.
Wang JP, Cerny A, Asher DR, Kurt-Jones EA, Bronson RT, MDA5 Finberg RW . and MAVS mediate type I interferon responses to coxsackie B virus. J Virol 2010; 84: 254–260.
Huber SA, Roberts B, Moussawi M, Boyson JE . Slam haplotype 2 promotes NKT but suppresses Vgamma4+ T-cell activation in coxsackievirus B3 infection leading to increased liver damage but reduced myocarditis. Am J Pathol 2013; 182: 401–409.
Huhn MH, McCartney SA, Lind K, Svedin E, Colonna M, Flodstrom-Tullberg M . Melanoma differentiation-associated protein-5 (MDA-5) limits early viral replication but is not essential for the induction of type 1 interferons after Coxsackievirus infection. Virology 2010; 401: 42–48.
Chow LH, Gauntt CJ, McManus BM . Differential effects of myocarditic variants of Coxsackievirus B3 in inbred mice. A pathologic characterization of heart tissue damage. Lab Invest 1991; 64: 55–64.
Wiltshire SA, Leiva-Torres GA, Vidal SM . Quantitative trait locus analysis, pathway analysis, and consomic mapping show genetic variants of Tnni3k, Fpgt, or H28 control susceptibility to viral myocarditis. J Immunol 2011; 186: 6398–6405.
Aly M, Wiltshire SA, Chahrour G, Osti J-CL, Vidal SM . Complex genetic control of host susceptibility to coxsackievirus B3-induced myocarditis. Genes Immun 2007; 8: 193–204.
Herskowitz A, Wolfgram L, Rose NR, Beisel K . Coxsackievirus B3 murine myocarditis: a pathologic spectrum of myocarditis in genetically defined inbred strains. J Am Coll Cardiol 1987; 9: 1311–1319.
Wiltshire SA, Diez E, Miao Q, Dubé M-P, Gagné M, Paquette O et al. Genetic control of high density lipoprotein-cholesterol in AcB/BcA recombinant congenic strains of mice. Physiol Genomics 2012; 44: 843–852.
Min-Oo G, Fortin A, Tam M, Nantel A, Stevenson M, Gros P . Pyruvate kinase deficiency in mice protects against malaria. Nat Genet 2003; 35: 357–362.
Chow L, Gauntt C, McManus B . Differential effects of myocarditic variants of Coxsackievirus B3 in inbred mice. A pathologic characterization of heart tissue damage. Lab Invest 1991; 64: 55–64.
Wessely R, Klingel K, Knowlton K, Kandolf R . Cardioselective infection with coxsackievirus B3 requires intact type I interferon signaling: implications for mortality and early viral replication. Circulation 2001; 103: 756–761.
Yang RQ, Yi NJ, Xu SZ . Box-Cox transformation for QTL mapping. Genetica 2006; 128: 133–143.
Xia Y, Won S, Du X, Lin P, Ross C, La Vine D et al. Bulk segregation mapping of mutations in closely related strains of mice. Genetics 2010; 186: 1139–1146.
Bongfen SE, Rodrigue-Gervais I-G, Berghout J, Torre S, Cingolani P, Wiltshire SA et al. An N-Ethyl-N-Nitrosourea (ENU)-Induced Dominant Negative Mutation in the JAK3 Kinase Protects against Cerebral Malaria. PloS One 2012; 7: e31012.
Khetsuriani N, Lamonte-Fowlkes A, Oberst S, Pallansch M . Enterovirus surveillance—United States, 1970-2005. MMWR Surveill Summ 2006; 55: 1–20.
CfDCaP. CDC . Nonpolio enterovirus and human parechovirus surveillance— United States, 2006–2008. MMWR Morb Mortal Wkly Rep 2010; 59: 1577–1580.
Traystman MD, Chow LH, McManus BM, Herskowitz A, Nesbitt MN, Beisel KW . Susceptibility to Coxsackievirus B3-induced chronic myocarditis maps near the murine Tcr alpha and Myhc alpha loci on chromosome 14. Am J Pathol 1991; 138: 721–726.
Knowlton KU, Jeon ES, Berkley N, Wessely R, Huber S . A mutation in the puff region of VP2 attenuates the myocarditic phenotype of an infectious cDNA of the Woodruff variant of coxsackievirus B3. J Virol 1996; 70: 7811–7818.
Stadnick E, Dan M, Sadeghi A, Chantler JK . Attenuating mutations in coxsackievirus B3 map to a conformational epitope that comprises the puff region of VP2 and the knob of VP3. J Virol 2004; 78: 13987–14002.
Schmidtke M, Merkle I, Klingel K, Hammerschmidt E, Zautner AE, Wutzler P . The viral genetic background determines the outcome of coxsackievirus B3 infection in outbred NMRI mice. J Med Virol 2007; 79: 1334–1342.
Gay RT, Belisle S, Beck MA, Meydani SN . An aged host promotes the evolution of avirulent coxsackievirus into a virulent strain. Proc Natl Acad Sci USA 2006; 103: 13825–13830.
Frisancho-Kiss S, Davis SE, Nyland JF, Frisancho JA, Cihakova D, Barrett MA et al. Cutting edge: cross-regulation by TLR4 and T cell Ig mucin-3 determines sex differences in inflammatory heart disease. J Immunol 2007; 178: 6710–6714.
Frisancho-Kiss S, Nyland JF, Davis SE, Frisancho JA, Barrett MA, Rose NR et al. Sex differences in coxsackievirus B3-induced myocarditis: IL-12Rbeta1 signaling and IFN-gamma increase inflammation in males independent from STAT4. Brain Res 2006; 1126: 139–147.
Woodruff JF . The influence of quantitated post-weaning undernutrition on coxsackievirus B3 infection of adult mice. II. Alteration of host defense mechanisms. J Infect Dis 1970; 121: 164–181.
Woodruff JF, Woodruff JJ . Modification of severe coxsackievirus B 3 infection in marasmic mice by transfer of immune lymphoid cells. Proc Natl Acad Sci USA 1971; 68: 2108–2111.
Fortin A, Diez E, Rochefort D, Laroche L, Malo D, Rouleau G et al. Recombinant congenic strains derived from A/J and C57BL/6 J: a tool for genetic dissection of complex traits. Genomics 2001; 74: 21–35.
Boivin GA, Pothlichet J, Skamene E, Brown EG, Loredo-Osti JC, Sladek R et al. Mapping of clinical and expression quantitative trait loci in a sex-dependent effect of host susceptibility to mouse-adapted influenza H3N2/HK/1/68. J Immunol 2012; 188: 3949–3960.
Coyne CB, Bozym R, Morosky SA, Hanna SL, Mukherjee A, Tudor M et al. Comparative RNAi screening reveals host factors involved in enterovirus infection of polarized endothelial monolayers. Cell Host Microbe 2011; 9: 70–82.
Antoniak S, Owens AP 3rd, Baunacke M, Williams JC, Lee RD, Weithauser A et al. PAR-1 contributes to the innate immune response during viral infection. J Clin Investig 2013; 123: 1310–1322.
Ilnytska O, Santiana M, Hsu NY, Du WL, Chen YH, Viktorova EG et al. Enteroviruses harness the cellular endocytic machinery to remodel the host cell cholesterol landscape for effective viral replication. Cell Host Microbe 2013; 14: 281–293.
Esfandiarei M, Boroomand S, Suarez A, Si X, Rahmani M, McManus B . Coxsackievirus B3 activates nuclear factor kappa B transcription factor via a phosphatidylinositol-3 kinase/protein kinase B-dependent pathway to improve host cell viability. Cell Microbiol 2007; 9: 2358–2371.
Esfandiarei M, Luo H, Yanagawa B, Suarez A, Dabiri D, Zhang J et al. Protein kinase B/Akt regulates coxsackievirus B3 replication through a mechanism which is not caspase dependent. J Virol 2004; 78: 4289–4298.
Diez E, Lee SH, Gauthier S, Yaraghi Z, Tremblay M, Vidal S et al. Birc1e is the gene within the Lgn1 locus associated with resistance to Legionella pneumophila. Nat Genet 2003; 33: 55–60.
Bortoluci KR, Medzhitov R . Control of infection by pyroptosis and autophagy: role of TLR and NLR. Cell Mol Life Sci 2010; 67: 1643–1651.
Suppiah V, Moldovan M, Ahlenstiel G, Berg T, Weltman M, Abate ML et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet 2009; 41: 1100–1104.
Aly M, Wiltshire S, Chahrour G, Osti JC, Vidal SM . Complex genetic control of host susceptibility to coxsackievirus B3-induced myocarditis. Genes Immun 2007; 8: 193–204.
Rozen S, Skaletsky H . Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 2000; 132: 365–386.
Yang H, Ding Y, Hutchins L, Szatkiewicz J, Bell T, Paigen B et al. A customized and versatile high-density genotyping array for the mouse. Nat Methods 2009; 6: 663–666.
Box GCD . An Analysis of Transformations. J R Stat Soc Ser B 1964; 16: 211–252.
Acknowledgements
This work was supported by grants from the Canadian Institutes of Health Research (CIHR) SAW, GAL and JM were supported by FRSQ Scholarships and SMV by the Canada Research Chair program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Wiltshire, S., Marton, J., Leiva-Torres, G. et al. Mapping of a quantitative trait locus controlling susceptibility to Coxsackievirus B3-induced viral hepatitis. Genes Immun 16, 261–267 (2015). https://doi.org/10.1038/gene.2015.5
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/gene.2015.5