Abstract
Enhanced external counterpulsation (EECP) is a FDA-approved treatment for patients with coronary artery disease and unstable angina. Although beneficial effects of EECP have been linked to central/cardiac adaptations, recent findings have shown peripheral/vascular effects. Here, we sought to determine EECP-induced blood flow patterns and their association with vascular function. The present study was designed to investigate endothelium-mediated arterial vasodilation changes after one 45-min session of either EECP or Sham EECP in 18 randomly assigned apparently healthy, young men (25±4 years). Brachial (b) and femoral (f) flow-mediated dilation (FMD) were assessed before and within 10 min after completing EECP or Sham. After 20 min of EECP, peak blood flow velocity (V) and brachial and femoral artery diameters (D) were recorded live for 2 min. In addition, a blood sample was drawn from the earlobe to determine hematocrit and then to calculate blood viscosity (μ) and density (ρ), Reynolds number (Re=V*D*ρ/μ), and endothelial shear stress (ESS=2μ*V/D). EECP increased retrograde shear stress and retrograde-turbulent blood flow in the femoral artery and antegrade-laminar shear stress in the brachial artery. fFMD was increased after EECP compared with Sham and baseline (fFMD=13.1±3.7 vs. 7.9±4.6% and 7.8±4.5%, respectively, P<0.05) and bFMD was increased after EECP compared with baseline (bFMD=10.6±4.8 vs. 7.0±3.5%, P<0.05), despite different blood flow patterns. These results provide novel evidence that a single session of EECP-induced blood flow patterns improve endothelial function in peripheral muscular conduit arteries.
Similar content being viewed by others
Introduction
Enhanced external counterpulsation (EECP) is an FDA-approved treatment for patients with coronary artery disease and unstable angina.1, 2, 3, 4, 5, 6 EECP involves sequential inflation and deflation of compressible cuffs wrapped around the subject’s calves, lower thighs and upper thighs. Compressed air pressure is applied via the cuffs to the lower extremities in a sequence synchronized with the cardiac cycle via microprocessor-interpreted ECG signals. Although several mechanisms have been proposed for the long-term effects of EECP in coronary artery disease patients,2, 4, 5, 6 the acute blood flow patterns created by the compressive cuffs and the immediate effects on peripheral vascular function have not been carefully studied in humans.
The ECG-synchronized inflation of the lower-body cuffs augments coronary diastolic pressure.7 This cardiac, or central, adaptation has been the main research focus of EECP; however, current studies have shown vascular adaptations as well.4, 8, 9 In fact, our laboratory has shown reduced stiffness in central elastic conduit arteries after 35 1-h sessions of EECP.4, 8, 10
Although changes in aortic and coronary hemodynamics have been proposed as primary hypotheses for the central changes after EECP, just a few studies have focused on the peripheral hemodynamics.11, 12, 13, 14 More specifically, acute blood flow patterns created by cuff compressions and their impact on peripheral endothelial function have not been studied in humans. The present study aims to determine if there is any acute peripheral vascular adaptation after a single 45-min session of EECP.
Finally, animal and human studies,11, 12, 13 and mathematical models14 have shown that EECP produces an increased retrograde blood flow and blood flow velocity in the lower extremities. Interestingly, there are some contradictory results when measuring endothelial function after retrograde flow. For example, a group of investigators15, 16 reported that brachial artery flow-mediated dilation (FMD) was impaired after the brachial artery was exposed to increased retrograde flow. In contrast, recent findings from our lab showed that 35 1-h sessions of EECP improved femoral FMD.4
Thus, the purpose of the present study was to characterize blood flow patterns in the femoral and brachial arteries during a single session of EECP and their relationship with endothelium-dependent vasodilation, measured via FMD. We hypothesized that blood flow patterns would be retrograde-turbulent and antegrade-laminar in the femoral and brachial arteries, respectively, and that both blood flow patterns would acutely improve vascular function.
Methods
Subjects
Eighteen young men, 19–34 years of age, were enrolled in the study. All subjects were apparently healthy with no known cardiovascular disease or major cardiovascular risk factors. Exclusion criteria included currently exercising three times per week or more, known cardiovascular disease including cardiac arrhythmias, prescription medication, ‘over-the-counter’ painkillers, such as NSAIDs or aspirin, or nutritional supplements containing antioxidants. The study was approved by the Institutional Review Board at the University of Florida, and written informed consent was obtained from all subjects. Subjects were placed in a supine resting position on the EECP gurney in a quiet temperature-controlled environment. Following a 10-min rest period, brachial blood pressure was measured in triplicate via an automated non-invasive device (BpTRU BPM-100, VSM MedTech, Coquitlam, BC, Canada). All subjects were tested at the same time of day to avoid any diurnal variations following at least 8 h of fasting and with no caffeine intake for at least 12 h. Female subjects were not recruited due to significant variations in vascular function during the four phases of the menstrual cycle.17
Experimental protocol
Subjects were randomly assigned to one of the two groups: group 1: active EECP (EECP, n=9) with cuffs inflated to 250 mm Hg (Angio New-IV, Vasomedical, Westbury, NY, USA); group 2: sham-EECP (Sham, n=9) with cuffs inflated to 50 mm Hg, which give the sensation of treatment without altering arterial hemodynamics.7 All subjects received a single, 45-min session of EECP or Sham. Hematocrit was measured in duplicate before the EECP session. Before and during the EECP or Sham session, central aortic blood pressure was assessed non-invasively via applanation tonomtery (SphygmoCor, AtCor Medical, West Ryde, NSW, Australia) as previously described.18 Finally, brachial and femoral FMD was performed before and within 10 min after the EECP or Sham session. During EECP or Sham, brachial and femoral artery diameters and blood flow velocities were assessed using high-resolution ultrasound and Doppler, respectively.
Brachial and femoral artery FMD
Brachial (b) and femoral (f) endothelial-dependent arterial vasodilation was performed using high-resolution ultrasound (HDI 3000, ATL, Bothell, WA, USA), and following international guidelines.19 After lying quietly for 15 min, a 10.5-MHz linear phase array ultrasound transducer was used to image the left brachial and femoral arteries longitudinally and recorded directly to a digital storage device via a super video interface (Pinnacle System, Avid Technology, Tewksbury, MA, USA). Imaging was performed with the ultrasound probe fixed approximately 5 cm above the antecubital fossa and approximately 2 cm below the inguinal ligament for brachial and femoral measurement sites, respectively. Skin of the brachial and femoral sites were marked with a permanent marker to keep the site consistent between imaging acquisitions. After obtaining resting baseline end diastolic diameters and blood flow velocity, a blood pressure cuff placed on the upper forearm, 1–2 cm below the elbow or on the lower thigh, or 2–3 cm above kneecap, was inflated to 200 mm Hg for 5 min. The transducer was held in the same position for the duration of cuff inflation to ensure the same section of the brachial or femoral artery was measured before and after cuff inflation. Additionally, distal cuff placement has been suggested to serve as a more accurate bioassay of endothelial nitric oxide availability.20
Brachial and femoral artery diameters were determined every 5 s for 150 s from 30 s before cuff deflation using automated edge-detection software (Vascular Research Tools, Medical Imaging Applications LLC, Coralville, IA, USA). Peak systolic blood flow velocity was also recorded every 5 s for 150 s from 30 s before cuff deflation. Brachial and femoral peak diameters were identified as the single peak diameter observed during the plateau phase after cuff deflation.21 Brachial and femoral FMD were calculated as relative (%) and absolute change in brachial and femoral artery diameters in response to the forearm hyperemic stimulus. The hyperemic stimulus was calculated as the area under the shear rate curve (AUC) using the trapezoidal rule from the time-point of cuff deflation to the time-point of maximal post-deflation diameter, and it was used to normalize brachial and femoral FMD.22, 23 In our laboratory, coefficient of variation for peak %FMD using this method is ∼8%.24
Brachial and femoral artery diameters and blood flow velocities during EECP
After 20 min of the 45-min EECP or Sham session, femoral and brachial artery diameters and peak systolic and diastolic blood flow velocities were measured. These measurements were performed with high-resolution ultrasound and Doppler using the same procedure described for arterial FMD testing. Data was continuously recorded for at least 2 min. All frames included for analysis had to meet the following inclusion criteria: (1) more than 5 s apart from any contiguous selected frame, (2) contain a clean vessel diameter and (3) contain averaged peak systolic and diastolic velocities directly calculated by Doppler. Data analysis involving blood flow velocity was averaged among at least 10 different time points within the 2-min acquisition period.
Blood flow-induced endothelial shear stress (ESS) and Reynolds number (Re)
ESS is the tangential force derived by the friction of the flowing blood on the endothelial surface.25, 26 It was determined in brachial and femoral arteries during antegrade and retrograde blood flows using ESS=2μ*V/D, where μ is blood viscosity, V is peak blood flow velocity and D is artery diameter. ESS was expressed in dynes cm−2.27
Re is a dimensionless ratio of blood inertial forces to viscous forces. During laminar blood flow, Re values are low, typically lower than 1000.25, 26 During turbulent blood flow, Re values are higher, typically above 2000.25, 26 Re was calculated using Re=(V*D*ρ)/μ, where V is peak blood flow velocity; D is artery diameter; ρ is blood density; and μ is blood viscosity.28 Blood density was calculated using the following equation:29, 30, 31

where Hct is hematocrit expressed as a fraction. Blood viscosity was determined using the following equations:29, 30, 31

where μplasma is plasma viscosity expressed in 10−1 N m−2 per s; T is temperature expressed in °C; SR is shear rate (if SR<100 s−1, then SR=calculated SR (V/D); if SR⩾100 s−1, then SR=100).
Statistical analysis
Descriptive statistics, including mean, standard deviations (s.d.), standard error of the means (s.e.m.), and minimum and maximum values were obtained. Normal distribution for all dependent variables was confirmed using Shapiro–Wilkins and Smirnoff tests (at least one test P>0.05). T-tests were performed for comparisons of single-time variables at baseline. Two-way repeated measurements–ANOVAs comparing dependent variables before and during or after EECP session, and between EECP and Sham groups were performed. Fisher’s least significant difference was used as post-hoc analysis. Data is expressed as mean±s.d. unless otherwise stated. All statistical analyses were performed using SPSS (version 19.0, Chicago, IL, USA), and statistical significance was considered when P<0.05.
Results
Table 1 shows general characteristics from the Sham and EECP groups at resting conditions before intervention. There were no significant differences in age, height, weight, body mass index, peripheral blood pressure, hematocrit and blood density between groups at baseline.
Central blood pressure
Central aortic diastolic and mean arterial pressures were higher during EECP compared with Sham (111±9 vs. 71±8 mm Hg and 98±8 vs. 81±7 mm Hg, P<0.05, respectively), but central aortic systolic blood pressure did not change during the 45-min EECP session.
Blood flow patterns
Ultrasound pictures and Doppler spectrum of brachial and femoral arteries are presented in Figures 1 and 2, respectively. During EECP, brachial artery blood flow velocity shows two peaks of antegrade flow per cardiac cycle compared with Sham (Figure 1b, bottom), whereas femoral artery blood flow velocity is increased and mainly retrograde during late diastole and early systole (Figure 2b, bottom). ESS was increased during EECP compared with Sham only in the brachial antegrade and femoral retrograde flows (29.0±6.6 vs. 16.6±4.1 dynes cm−2 and −19.1±5.1 vs. −3.8±0.3 dynes cm−2, P<0.05, respectively) (Figure 3).
Shear stress before and during EECP. (a) Brachial artery, Sham group; (b) Brachial artery, EECP group; (c) Femoral artery, Sham group; (d) Femoral artery, EECP group. Closed boxes (▪), antegrade flow; open boxes (□), retrograde flow. Values are mean±s.e.m. (*P<0.05 EECP vs. Sham. †P<0.05 during vs. before).
Flow turbulence
Re was significantly increased in both brachial and femoral retrograde flows during EECP compared with resting conditions (Figure 4). However, blood flow was turbulent only in femoral retrograde flow (Re>2000) during EECP compared with Sham (Figure 4d).
Re before and during EECP. (a) Brachial artery, Sham group; (b) Brachial artery, EECP group; (c) Femoral artery, Sham group; (d) Femoral artery, EECP group. Closed boxes (▪), antegrade flow; open boxes (□), retrograde flow. Re⩽−2000, retrograde turbulent flow; Re⩾2000, antegrade turbulent flow. Values are mean±s.e.m. (*P<0.05 EECP vs. Sham. †P<0.05 during vs. before).
Brachial and femoral FMD and hyperemic stimulus
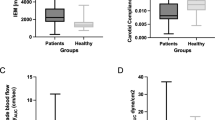
Both brachial and femoral artery FMD increased after EECP compared with baseline (Figures 5a and c). Femoral FMD increased after EECP compared with Sham, whereas the time to peak femoral dilation was reduced (Figures 5c and d). There was no difference in the hyperemic stimulus before and after EECP what produced similar increases in brachial and femoral FMD when normalized by AUC (Tables 2 and 3).
Absolute diameter changes
Brachial artery baseline and peak diameters did not change with EECP; however, the absolute change was greatest after EECP (Table 2). Interestingly, femoral baseline diameter decreased after EECP, whereas femoral peak diameter remained unchanged. These differences are consistent with a greatest absolute diameter change after EECP (Table 3).
Discussion
The present study was designed to characterize peripheral blood flow patterns during a single 45-min session of EECP and determine the acute effects on endothelium-dependent vasodilation. The major findings in this study are threefold: first, peripheral blood flow patterns during EECP are antegrade-laminar and retrograde-turbulent in brachial and femoral arteries, respectively; second, both blood flow patterns create acute dramatic increases in ESS; and third, a single 45-min session of EECP produces immediate improvement on endothelial-dependent vasodilation in both brachial and femoral arteries.
Few studies have investigated acute EECP-induced blood flow patterns.11, 12, 13, 14 Using a porcine EECP model, Zhang et al.11 found that brachial artery blood flow velocity and wall shear stress increased by 1.3- and 2.1-fold, respectively, during EECP. However, these authors did not study hind limb blood flow patterns. Using a numerical simulation of EECP, Ozawa et al.14 showed significant increases in retrograde shear stress in the lower abdomen and femoral cuff sites (0 vs. −35 dynes cm−2 and 0 vs. −15 dynes cm−2, respectively, P<0.05). Although this study was able to predict a biphasic pulse pressure in the radial artery during EECP, the authors did not report shear stress or acute blood flow patterns in the upper limb vasculature.14 A decade ago, Cai et al.12 studied the changes in blood flow velocity during EECP using an ultrasonic Doppler flowmeter. They observed a 1.2-fold increase in femoral artery retrograde blood flow velocity. However, the authors did not report either shear stress or blood flow patterns in the upper limb vasculature.12 In a more recent study, Werner et al.13 assessed blood flow velocity in the posterior tibial artery during EECP and brachial FMD after EECP. Although the authors observed ∼fourfold increase in the pulsatility index, showing a significant increase in retrograde blood flow, there was no change in brachial FMD after EECP.13
The present study, to the best of our knowledge, is the most comprehensive non-invasive investigation of acute EECP-induced blood flow patterns to date. We designed a placebo-controlled study where we measured acute blood flow velocities and vessel diameters in both the lower (femoral artery) and upper (brachial artery) extremities. In addition, we determined blood flow patterns in both vascular sites with the additional calculations of shear stress and flow turbulence. Furthermore, we performed endothelium-dependent vasodilation assessment in both vascular sites to determine the relationship between acute blood flow patterns during EECP and endothelial function immediately after EECP. Blood flow-induced ESS is the primary physiological stimulus that regulates vascular endothelial function.25, 26, 32 The present study showed that ESS was increased by 1.75- and 5.02-fold in both brachial and femoral arteries during antegrade and retrograde flows, respectively (Figure 3). In addition, we showed for the first time that this increase in ESS was associated with improvement in both brachial (51%) and femoral FMD (68%) (Figure 5). Interestingly, blood flow patterns in femoral and brachial vessels were opposite. Whereas brachial blood flow was mainly antegrade-laminar, femoral blood flow was mainly retrograde-turbulent (Figures 1, 2, 3, 4).
Findings of the present study support the hypothesis that both antegrade and retrograde flow patterns can improve endothelial function when ESS is increased; however, they contradict previous studies.15, 16 Thijssen et al.15 and Tinken et al.16 used brachial FMD to determine the effects of retrograde flow, induced by blood flow restriction15 and forearm heating/handgrip exercise,16 on endothelial function. In general, they found a dose-dependent decrease in brachial artery FMD with increased brachial artery retrograde blood flow. However, ESS data were not reported either at rest or during exercise. Even though retrograde shear rate significantly increased during blood restriction and handgrip exercise, ESS could have remained constant. The mechanism responsible for this phenomenon is the exponential increase of blood viscosity when shear rate decreases below 100 s−1, which produces a buffer effect.30, 33 This non-Newtonian fluid behavior, characteristic of human blood, would exponentially increase resting retrograde ESS. Therefore, it is conceivable that retrograde ESS during blood flow restriction15 and/or handgrip exercise16 was not significantly greater than during resting conditions. In addition, when using reported data from these studies,15, 16 for example, average brachial artery diameter of 4 mm,16 peak retrograde velocity15, 16 and average hematocrit of 50% in young men, peak retrograde Re calculated for both studies would be lower than ∼500, indicating the presence of laminar flow. Therefore, based upon at least two dimensions of blood flow, direction and turbulence, the decrease in brachial FMD observed in those studies was likely produced by a retrograde-laminar blood flow, with unknown shear stress.15, 16 The present study has shown an increased femoral FMD when blood flow is retrograde-turbulent and ESS is increased. Determining if retrograde-turbulent flow without increased ESS would improve FMD needs further study.
Our results are consistent, at least in part, with in vitro and animal studies.34, 35, 36, 37 Davies et al.34 observed that beneficial changes in bovine aortic endothelial cells were produced by turbulent flow in vitro. In the present study, we observed improvement in femoral artery FMD after EECP, despite turbulent blood flow in the femoral artery during EECP. Duchene et al.37 observed that vascular anti-inflammatory changes in human umbilical vein endothelial cells produced by laminar flow in vitro were dependent upon increased ESS. In the present study, EECP caused significant increases in brachial ESS and we observed improvement in brachial artery FMD after the EECP stimulus. Moreover, Cheng et al.35, 36 have shown that turbulent flow could promote atherosclerotic plaque stability, rather than the often-reported endothelial dysfunction, confirming than turbulent blood flow is not always detrimental to vascular health. In the present study, EECP caused retrograde turbulent flow in the femoral artery, but we observed improvement in femoral FMD after the EECP stimulus.
Our findings may be associated with the endothelial mechanotransduction system. According to current theories, endothelial cells are capable of sensing blood flow via a complex mechanical system.26, 38 Although antegrade laminar flow upregulates endothelial function, antegrade turbulent downregulates it.26, 38 However, and based on Thijssen et al.15 and Tijken et al.16 studies, laminar flow could also downregulate endothelial function when flow is retrograde. The present study showed that retrograde turbulent flow could produce the same beneficial effects as antegrade laminar flow; although further studies are needed to confirm this in vivo finding.
Although our results showed clear improvement in both brachial and femoral artery FMD after EECP, we cannot overlook that reactive hyperemic responses are artery dependent.22, 23, 39, 40 Although vascular reactivity to the hyperemic stimulus is higher in smaller than bigger vessels (for example, brachial vs. femoral), the present study showed comparable brachial and femoral artery FMDs (Figure 5). The enhanced femoral FMD after EECP could be attributed to a significant acute decrease in the femoral baseline diameter observed immediately after EECP (Table 3). It is possible that the 250 mm Hg-external compressions elicit an increased femoral artery vascular tone, which increases shear rate. Thus, endothelium-dependent vasodilation is enhanced and accelerated. We did not measured the femoral baseline diameter recovery time. However, if this smaller femoral artery diameter is maintained for a period of time (for example, 2–3 h), it would enhance resting shear stress after EECP and could explain, at least in part, the femoral artery functional adaptations observed after chronic exposure to EECP.4
Although there are several methodological differences, our results are in agreement with some of the aforementioned studies.11, 12, 14 We observed increases in brachial artery shear stress during EECP similar to Zhang et al.11 (1.75-fold vs. 2.1-fold increase, respectively) and similar significant increases in retrograde flow in the femoral artery during EECP compared to both Cai et al.12 and the mathematical estimations by Ozawa et al.14 Interestingly, Werner et al.13 did not find any significant improvement in brachial FMD after one 1-h session of EECP. The main reason might be that the authors measured brachial artery diameter only 1 min after deflation, potentially missing the peak diameter.22
The direct mechanical stimuli produced by acute EECP-induced blood flow patterns appear to have additive chronic effects after 35 1-h sessions of EECP. We recently reported peripheral vascular adaptations associated with increased nitric oxide bioavailability4 and improved arterial stiffness8, 10 after 35 1-h sessions of EECP. Similar findings were observed in a recent study where chronic intermittent external compressions were applied to the forearm.41 The EECP-induced creation of shear stimulus and subsequent acute improvements in brachial and femoral FMD observed in the present study support the hypothesis that peripheral arterial function may be the therapeutic target of chronic EECP treatment.2, 6 Indeed, there is inadequate data to support the hypothesis that EECP increases myocardial perfusion and oxygen supply. An alternative hypothesis, supported by novel data from the present study, is that EECP improves peripheral arterial function, which serves to reduce left ventricular afterload and myocardial oxygen demand.
The present study was not without limitations. Our studied population was healthy, young individuals, whereas EECP is normally prescribed for patients with coronary artery disease. Further studies investigating EECP-induced blood flow patterns and acute vascular effects of EECP in clinical populations are appropriate. In addition, our sham group received 50 mm Hg compressions, which could increase venous return. Venous return could increase stroke volume via Frank-Starling law, increasing mean arterial pressure. Although this cascade of events is possible, invasive studies measuring central pressure during EECP have shown that pressure cuff of 100 mm Hg did not altered mean arterial pressure.7 Finally, and to prevent further complexity, calculations of shear stress and turbulence were based on Poiseuille’s laws approximation avoiding Womersley’s pulsatile flow frequency analysis, which should have considered a two-pulse analysis (that is, heart rate and EECP). Nevertheless, the main findings on ESS of the present study should not change as we have recently reported that using Poiseuille approximation only underestimate ESS.42
In summary, the present study showed that EECP acutely improves endothelium-dependent vasodilation in both femoral and brachial arteries. EECP creates opposite blood flow patterns in the femoral (retrograde-turbulent) and brachial (antegrade-laminar) arteries. However, both flow patterns dramatically increase ESS, suggesting that an increased ESS is needed to improve endothelial function. These findings support the hypothesis that peripheral arterial function could be considered another therapeutic target of chronic EECP treatment.
References
Arora RR, Shah AG . The role of enhanced external counterpulsation in the treatment of angina and heart failure. Can J Cardiol 2007; 23: 779–781.
Feldman AM . Enhanced external counterpulsation: mechanism of action. Clin Cardiol 2002; 25 (Suppl 2): Ii11–Ii15.
Taguchi I, Ogawa K, Kanaya T, Matsuda R, Kuga H, Nakatsugawa M . Effects of enhanced external counterpulsation on hemodynamics and its mechanism. Circ J 2004; 68: 1030–1034.
Braith RW, Conti CR, Nichols WW, Choi CY, Khuddus MA, Beck DT, Casey DP . Enhanced external counterpulsation improves peripheral artery flow-mediated dilation in patients with chronic angina: a randomized sham-controlled study. Circulation 2010; 122: 1612–1620.
Kitsou V, Xanthos T, Roberts R, Karlis GM, Padadimitriou L . Enhanced external counterpulsation: mechanisms of action and clinical applications. Acta Cardiol 2010; 65: 239–247.
Bonetti PO, Holmes DR, Lerman A, Barsness GW . Enhanced external counterpulsation for ischemic heart disease: what’s behind the curtain? J Am Coll Cardiol 2003; 41: 1918–1925.
Michaels AD, Accad M, Ports TA, Grossman W . Left ventricular systolic unloading and augmentation of intracoronary pressure and doppler flow during enhanced external counterpulsation. Circulation 2002; 106: 1237–1242.
Casey DP, Beck DT, Nichols WW, Conti CR, Choi CY, Khuddus MA, Braith RW . Effects of enhanced external counterpulsation on arterial stiffness and myocardial oxygen demand in patients with chronic angina pectoris. Am J Cardiol 2011; 107: 1466–1472.
Zhang Y, He X, Liu D, Wu G, Chen X, Ma H, Du Z, Dong Y, Jin Y, He W, Wang K, Lawson WE, Hui JC, Zheng Z . Enhanced external counterpulsation attenuates atherosclerosis progression through modulation of proinflammatory signal pathway. Arterioscler Thromb Vasc Biol 2010; 30: 773–780.
Nichols WW, Estrada JC, Braith RW, Owens K, Conti CR . Enhanced external counterpulsation treatment improves arterial wall properties and wave reflection characteristics in patients with refractory angina. J Am Coll Cardiol 2006; 48: 1208–1214.
Zhang Y, He X, Chen X, Ma H, Liu D, Luo J, Du Z, Jin Y, Xiong Y, He J, Fang D, Wang K, Lawson WE, Hui JC, Zheng Z, Wu G . Enhanced external counterpulsation inhibits intimal hyperplasia by modifying shear stress responsive gene expression in hypercholesterolemic pigs. Circulation 2007; 116: 526–534.
Cai D, Wu R, Shao Y . Experimental study of the effect of external counterpulsation on blood circulation in the lower extremities. Clin Invest Med 2000; 23: 239–247.
Werner D, Michalk F, Hinz B, Werner U, Voigt JU, Daniel WG . Impact of enhanced external counterpulsation on peripheral circulation. Angiology 2007; 58: 185–190.
Ozawa ET, Bottom KE, Xiao X, Kamm RD . Numerical simulation of enhanced external counterpulsation. Ann Biomed Eng 2001; 29: 284–297.
Thijssen DH, Dawson EA, Tinken TM, Cable NT, Green DJ . Retrograde flow and shear rate acutely impair endothelial function in humans. Hypertension 2009; 53: 986–992.
Tinken TM, Thijssen DH, Hopkins N, Black MA, Dawson EA, Minson CT, Newcomer SC, Laughlin MH, Cable NT, Green DJ . Impact of shear rate modulation on vascular function in humans. Hypertension 2009; 54: 278–285.
Adkisson EJ, Casey DP, Beck DT, Gurovich AN, Martin JS, Braith RW . Central, peripheral, and resistance arterial reactivity fluctuates during the phases of the menstrual cycle. Exp Biol Med 2010; 235: 111–118.
Gurovich AN, Beck DT, Braith RW . Aortic pulse wave analysis is not a surrogate for central arterial pulse wave velocity. Exp Biol Med 2009; 234: 1339–1344.
Corretti MC, Anderson TJ, Benjamin EJ, Celermajer D, Charbonneau F, Creager MA, Deanfield J, Drexler H, Gerhard-Herman M, Herrington D, Vallance P, Vita J, Vogel R . Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the international brachial artery reactivity task force. J Am Coll Cardiol 2002; 39: 257–265.
Pyke KE, Dwyer EM, Tschakovsky ME . Impact of controlling shear rate on flow-mediated dilation responses in the brachial artery of humans. J Appl Physiol 2004; 97: 499–508.
Sonka M, Liang W, Lauer RM . Automated analysis of brachial ultrasound image sequences: early detection of cardiovascular disease via surrogates of endothelial function. IEEE Trans Med Imaging 2002; 21: 1271–1279.
Harris RA, Nishiyama SK, Wray DW, Richardson RS . Ultrasound assessment of flow-mediated dilation. Hypertension 2010; 55: 1075–1085.
Thijssen DH, Dawson EA, Black MA, Hopman MT, Cable NT, Green DJ . Heterogeneity in conduit artery function in humans: impact of arterial size. Am J Physiol Heart Circ Physiol 2008; 295: H1927–H1934.
Avery J, Gurovich A, Braith R . Reproducibility of brachial and femoral arterial diameter and flow-mediated dilation via automatic edge-detection software. Med Sci Sports Exercise 2010; 42: 315.
Chatzizisis YS, Coskun AU, Jonas M, Edelman ER, Feldman CL, Stone PH . Role of endothelial shear stress in the natural history of coronary atherosclerosis and vascular remodeling: molecular, cellular, and vascular behavior. J Am Coll Cardiol 2007; 49: 2379–2393.
Davies PF . Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat Clin Pract 2009; 6: 16–26.
Parker BA, Trehearn TL, Meendering JR . Pick your poiseuille: normalizing the shear stimulus in studies of flow-mediated dilation. J Appl Physiol 2009; 107: 1357–1359.
Nichols WW, O’rourke MF . Mcdonald’s Blood Flow In Arteries. Hodder Arnold: London. 2005.
Charlesworth D . Relationship of blood rheology to blood flow. In: Lowe G, Barbenel J, Forbes C, (eds). Clinical Aspects Of Blood Viscosity And Cell Deformability. Springer-Verlag: New York. 1981 pp 91–96.
Dormandy J . Measurement of whole-blood viscosity. In: Lowe G, Barbenel J, Forbes C, (eds.). Clinical Aspects Of Blood Viscosity And Cell Deformability. Springer-Verlag: New York. 1981 pp 67–78.
Harkness J . Measurement of plasma viscosity. In: Lowe G, Barbenel J, Forbes C, (eds). Clinical Aspects Of Blood Viscosity And Cell Deformability. Springer-Verlag: New York. 1981 pp 79–87.
Cunningham KS, Gotlieb AI . The role of shear stress in the pathogenesis of atherosclerosis. Lab Invest 2005; 85: 9–23.
Soulis JV, Giannoglou GD, Chatzizisis YS, Seralidou KV, Parcharidis GE, Louridas GE . Non-newtonian models for molecular viscosity and wall shear stress in a 3d reconstructed human left coronary artery. Med Eng Phys 2008; 30: 9–19.
Davies PF, Remuzzi A, Gordon EJ, Dewey CF, Gimbrone MA . Turbulent fluid shear stress induces vascular endothelial cell turnover in vitro. Proc Natl Acad Sci USA 1986; 83: 2114–2117.
Cheng C, Tempel D, Van Haperen R, Van Der Baan A, Grosveld F, Mj Daemen, Krams R, De Crom R . Atherosclerotic lesion size and vulnerability are determined by patterns of fluid shear stress. Circulation 2006; 113: 2744–2753.
Cheng C, Van Haperen R, De Waard M, Van Damme LC, Tempel D, Hanemaaijer L, Van Cappellen GW, Bos J, Slager CJ, Duncker DJ, Van Der Steen AF, De Crom R, Krams R . Shear stress affects the intracellular distribution of enos: direct demonstration by a novel in vivo technique. Blood 2005; 106: 3691–3698.
Duchene J, Cayla C, Vessillier S, Scotland R, Yamashiro K, Lecomte F, Syed I, Vo P, Marrelli A, Pitzalis C, Cipollone F, Schanstra J, Bascands J-L, Hobbs AJ, Perretti M, Ahluwalia A . Laminar shear stress regulates endothelial kinin b1 receptor expression and function. potential implication in atherogenesis. Arterioscler Thromb Vasc Biol 2009; 29: 1757–1763 ATVBAHA.109.191775.
Thi MM, Tarbell JM, Weinbaum S, Spray DC . The role of the glycocalyx in reorganization of the actin cytoskeleton under fluid shear stress: a ‘bumper-car’ model. Proc Natl Acad Sci USA 2004; 101: 16483–16488.
Thijssen DH, Van Bemmel MM, Bullens LM, Dawson EA, Hopkins ND, Tinken TM, Black MA, Hopman MT, Cable NT, Green DJ . The impact of baseline diameter on flow-mediated dilation differs in young and older humans. Am J Physiol Heart Circ Physiol 2008; 295: H1594–H1598.
Wray DW, Nishiyama SK, Donato AJ, Richardson RS . Human vascular aging: limb-specific lessons. Exerc Sport Sci Rev 2010; 38: 177–185.
Roseguini BT, Sheldon R, Stroup A, Bell JW, Maurer D, Crist BD, Laughlin MH, Newcomer SC . Impact of chronic intermittent external compressions on forearm blood flow capacity in humans. Eur J Appl Physiol 2011; 111: 509–519.
Gurovich AN, Braith RW . Analysis of both pulsatile and streamline blood flow patterns during aerobic and resistance exercise. Eur J Appl Physiol, (e-pub ahead of print 1 March 2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gurovich, A., Braith, R. Enhanced external counterpulsation creates acute blood flow patterns responsible for improved flow-mediated dilation in humans. Hypertens Res 36, 297–305 (2013). https://doi.org/10.1038/hr.2012.169
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/hr.2012.169
Keywords
This article is cited by
-
The Effect of EECP on Ischemic Heart Failure: a Systematic Review
Current Cardiology Reports (2023)
-
Effects of an external pneumatic compression device vs static compression garment on peripheral circulation and markers of sports performance and recovery
European Journal of Applied Physiology (2022)
-
Individual shear rate therapy (ISRT)—further development of external counterpulsation for decreasing blood pressure in patients with symptomatic coronary artery disease (CAD)
Hypertension Research (2020)
-
Internal validation of an automated system for brachial and femoral flow mediated dilation
Clinical Hypertension (2017)
-
Endothelial function does not improve with high-intensity continuous exercise training in SHR: implications of eNOS uncoupling
Hypertension Research (2016)