Abstract
Objective:
To investigate the association between maternal pregnancy and estimated postnatal serum concentrations of the organochlorines 2,2′,4,4′,5,5′-hexachlorobiphenyl (CB-153) and 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (p,p'-DDE) and body mass index (BMI) z-scores in 5- to 9-year-old children.
Methods:
Maternal sera from the INUENDO birth cohort (2002–2004) comprising mother–child pairs (N=1109) from Greenland, Warsaw (Poland), and Kharkiv (Ukraine) were analysed for CB-153 and p,p'-DDE, using gas chromatography-mass-spectrometry, and were grouped into tertiles for statistical analyses. A toxicokinetic model was used to estimate the first 12 months cumulative exposure to the compounds. Associations between these compounds and child age- and sex-specific BMI z-scores were calculated at follow-up (2010–2012), using multiple linear regression analysis.
Results:
No clear associations between pregnancy CB-153 and p,p'-DDE and child BMI were observed (the pooled differences in BMI z-score (95% confidence interval) comparing 3rd tertile to 1st tertile were −0.07 (−0.32 to 0.18) and −0.10 (−0.30 to 0.10) kg m−2, respectively). For postnatal CB-153 and p,p'-DDE and BMI, the overall differences in BMI z-score comparing 3rd tertile to 1st tertile were 0.12 (−0.15 to 0.39) and −0.03 (−0.20 to 0.27) kg m−2, respectively.
Conclusions:
This follow-up study of Greenlandic, Polish and Ukrainian populations showed no clear association between pregnancy and postnatal exposure to p,p'-DDE and CB-153 and BMI at the age of 5–9 years.
Similar content being viewed by others
Introduction
The man-made chemicals dichlorodiphenyltrichloroethane (DDT) and polychlorinated biphenyls (PCBs) are ubiquitous in the environment, although the use of them has been banned (PCBs) or restricted (DDT). PCBs and DDT (and thereby the degradation product 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDE)) bio-accumulate within the food chain and are measurable in human serum and milk around the world.1, 2, 3, 4 The compounds pass the placenta, causing exposure of the fetus to the chemicals.1,5 This is of major concern, as exposure during prenatal development and early life may be related to a wide range of health outcomes in children, such as perturbation of the endocrine system, attention deficit hyperactivity disorder-like behaviour, decreased motor skills and cognitive impairments.6, 7, 8, 9
Prenatal PCBs have been found to downregulate thyroid function in mice and rats10 and to be associated with lower levels of free and total thyroxine (T4) in human cord and neonatal blood,11 which could possibly lead to an increase in adipocyte lipid accumulation.12 An upregulation of adipocytes in newborn mice has been reported in relation to prenatal exposure to the oestrogen diethylstilbestrol,13 and as o,p'-DDT is known to have weak oestrogenic properties, mechanisms could be similar in p,p'-DDE. Also, the relation between other environmental compounds with supposed oestrogenic or antiandrogenic properties, such as bisphenol A and phthalates, and overweight has been discussed.14,15 Few studies have examined the relation between pregnancy PCB and DDE exposures and body mass index (BMI) standardized by age and sex, and findings are inconsistent.16, 17, 18, 19, 20
To our knowledge, only one study examined postnatal exposure to these compounds in relation to BMI z-scores or related metrics and found no association.16 Measurement of the child’s postnatal exposure is rarely performed, and therefore a toxicokinetic model, estimating the postnatal levels of persistent organic pollutants was developed by Verner et al.21 Using this model, we were able to estimate the child’s serum level of 2,2′,4,4′,5,5′-hexachlorobiphenyl (CB-153) and 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (p,p'-DDE) and thereby take the postnatal exposure of the compounds into account. Thus, the aim of this study was to investigate the associations between maternal pregnancy CB-153 and p,p'-DDE serum concentrations and the estimated accumulated concentrations of the compounds during the first 12 months of life in the offspring, and age- and sex-standardized BMI z-scores when the children reached young school-age. We hypothesized that higher exposure to CB-153 and p,p'-DDE in utero and early life would be associated with higher BMI.
Materials and methods
Study population
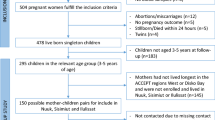
A total of 1441 pregnant women from Greenland, Warsaw (Poland) and Kharkiv (Ukraine) were enroled from antenatal health-care clinics and provided a blood sample in the INUENDO birth cohort between May 2002 and February 2004. At baseline, 567 (85%) of 665 eligible women participated in Greenland; 258 (37%) of 690 participated in Poland and 612 (25%) of 2478 eligible women participated in the study in Ukraine. Further details on the baseline study population are described elsewhere.22 The follow-up of the children in the cohort was conducted between January 2010 and May 2012, when the children were between 5 and 9 years old. Parents were interviewed using an interview-based questionnaire with regard to child physical development and lifestyle. All participating parents signed informed consent, and the local ethical committees approved the study.
Data collection
The follow-up interviews were primarily conducted face-to-face at the participants’ residence or at the local hospitals. A medical doctor, assisted by local health workers, was the main interviewer in Greenland. In Poland, interviews were conducted at local meeting points or at the participant’s residence by a team of four interviewers. In Ukraine, all interviews were conducted by a team of 59 paediatricians at eight paediatric polyclinics. Telephone interviews were performed when families lived in remote areas of Greenland (n=130) or had moved to Denmark (n=34). Also, in Greenland, a proportion of the questionnaires was filled in by the parents without an interview.
Assessment of prenatal CB-153 and p,p'-DDE exposure
At baseline, the pregnant women had a blood sample drawn in order to identify the fetal exposures through placental diffusion. In Greenland, the blood samples were drawn when the women were (median (10–90 percentile)) in gestational week 24 (13–37); in Poland, in gestational week 33 (27–37); and in Ukraine, in gestational week 23 (9–40). Ten millilitre cubital vein blood was drawn into vacuum tubes for serum collection without additives (Becton Dickinson, Meylan, France). All centres used the same ethanol-rinsed sampling device obtained from Lund University, Sweden. Sera were stored at −80 °C until analyses, and all chemical analyses were performed at the Department of Occupational and Environmental Medicine at Lund University Hospital. The sera were analysed for CB-153 and p,p'-DDE and used as bio-markers of the prenatal exposure to PCBs and DDT/DDE, respectively. The sera were analysed by gas chromatography-mass-spectrometry following solid phase extraction.23 Samples were analysed twice on different days and the mean concentration of these two determinations was used. The detection limits were 0.05 ng ml−1 for CB-153 and 0.1 ng ml−1 for p,p'-DDE. For CB-153, 85 samples were below the detection limit (LOD) and for p,p'-DDE, 10 samples were below the LOD. When concentrations were below the LOD, they were set to half the LOD based on fresh weight concentrations for all subsequent analyses. In-house prepared quality control samples were included in all analysed series. The relative standard deviations (s.d.) were calculated from samples analysed in duplicate at different days. These were used to estimate the imprecision. For this, the results of the analysed sera were divided into three groups: low, medium and high level, and the s.d. and the mean concentration for each group were calculated.24 These were 18% in the range 0.05–0.8 ng ml−1 (n=990), 10% in the range 0.2–0.8 ng ml−1 (n=990) and 10% in the range 0.7–19 ng ml−1 (n=990) for CB-153 and 11% in the range 0.1–2 ng ml−1 (n=1 058), 8% in the range 2–5 ng ml−1 (n=1 058) and 7% in the range 3–37 ng ml−1 (n=1 058) for p,p'-DDE. CB-153 and p,p'-DDE levels were adjusted for serum concentrations of triglycerides and cholesterol, which were determined by enzymatic methods and were expressed as ng g−1 lipids. The inter-assay coefficients of variation for cholesterol and triglycerides were 1.5–2.0%. Further details are described elsewhere.23
Estimation of postnatal CB-153 and p,p'-DDE exposure
To estimate the postnatal cumulative contribution of the compounds for the first 12 months after birth, a toxicokinetic model developed by Verner et al. was used.21 The model was validated in a Canadian Inuit birth cohort and a Slovakian birth cohort, leading us to assume validity in our populations. The model inputs were: age of the mother at delivery, maternal pre-pregnancy weight, duration of exclusive breast feeding, duration of partial breastfeeding, gestational age, child sex, birth weight, child’s weight at follow-up and age at measurement, and up to two previous recorded weight measurements, maternal levels of CB-153 and p,p'-DDE during pregnancy (placental diffusion), gestational age at blood sampling and half-life of the compounds.21 The estimate of the postnatal exposure used in the regression models was the area under the curve, which is equivalent to cumulative accumulated CB-153 and p,p'-DDE levels during the first 12 months after birth. We used this exposure metric, as the contribution of persistent organic pollutants via breastfeeding is the most prevalent and important source of a child’s postnatal exposure and we had breastfeeding data up until the age of 12 months.21 As the toxicokinetic model used total and exclusive duration of breastfeeding as predictors, only children with this information were included in these analyses (n=1047). The toxicokinetic model was performed using acslX software (Aegis Technologies Group, Inc., Huntsville, AL, USA).
Assessment of anthropometric measures
The child’s height was measured with the child standing barefoot against a wall, marking the top of the head and measuring the height to the nearest centimetre by use of ordinary measure tape. The child’s weight was measured to the nearest 0.1 kg by a weighing scale available at the family’s home or at the clinics. All measurements were performed by the interviewer except for those who were telephone-interviewed. Child BMI was calculated from weight (in kilograms)/ height × height (in metres). BMI was expressed as z-score representing the deviation in s.d. units from the mean of a standard normal distribution of BMI specific to age (1 month intervals) and sex. Positive z-values are above the 50 percentile and negative z-values are below the 50 percentile. The standards were based on the World Health Organization (WHO) Growth Standards 2007, which are applicable regardless of ethnicity or country of origin,25 as population-specific growth data were not available for all populations.
Covariates
Variables that according to the literature might influence child growth were obtained from questionnaires. These variables were: maternal pre-pregnancy BMI (continuous), paternal BMI (continuous), maternal smoking (serum cotinine in pregnancy ⩽10/>10 ng ml−1), maternal pre-pregnancy alcohol intake (<7, ⩾7 drinks per week), maternal educational level (left school at or before the age of 15 years, at the age of 16–17 years, at or above the age of 18 years), parity (1, 2–3, 4+ child births), maternal age at baseline (continuous), total breastfeeding duration (0, <6, 6–12, >12 months), child physical activity level (<2.75 h a day, ⩾2.75 h per day) and child diet (predominantly healthy versus predominately unhealthy; derived as sugary drinks or deserts <6–7 versus ⩾6–7 times a week).26, 27, 28, 29, 30, 31, 32 Gestational age at blood sampling in weeks (continuous) was included as it was correlated with the exposure levels.
Statistical analyses
Missing information
The number of missing values on height, weight and covariates ranged from 0 to 27%. As complete case analysis may lead to selection bias, we addressed the missing information problem, using chained multiple imputation allowing us to include participants with incomplete data in the statistical analyses.33 Assuming the missing information to be missing at random (systematic differences between observed and missing values can be explained by differences in observed data), this approach will result in more precise and unbiased estimates.33,34
Briefly, multiple different imputed data sets (m>1) are created, and each missing value is replaced with a set of random plausible values based on known subject characteristics and other predictors in the complete data set, incorporating the appropriate variability across the m data sets. The new m complete data sets are analysed, producing a single set of results accounting for the variability of the missing data.33
In a combined imputation across country, we generated 100 imputed data sets based on the following predictors: CB-153, p,p'-DDE, maternal height, maternal pre-pregnancy weight, paternal height, paternal weight, maternal educational level, maternal age at baseline, maternal smoking status during pregnancy, maternal pre-pregnancy alcohol consumption, parity, duration of breastfeeding, preterm birth, child sex, gestational age at blood drawing, child physical activity level, child diet, child age at interview, birth weight, birth length, z-score of weight at follow-up and z-score of height at follow-up.
Data analysis
Study subjects were divided into population-specific and total population tertiles of exposure. A non-response analysis was performed to check for inconsistencies between responders and non-responders. Spearman’s rank correlation was used to assess the correlation between maternal pregnancy levels of CB-153 and p,p'-DDE as well as the correlation between maternal pregnancy levels and postnatal levels of the two compounds. Crude associations between the exposures and age-standardized z-scores of BMI at follow-up were examined with lowest exposure tertile as the reference category, using univariate linear regression. Adjusted associations of pre- and postnatal exposure to CB-153 and p,p'-DDE on BMI z-score were examined, using multiple linear regression with lowest exposure tertile as reference category. We chose a priori to adjust for covariates, which were previously demonstrated to be associated with child growth and the exposures, as described above. In addition, odds ratios for the associations between organochlorines and overweight were calculated, using logistic regression. Overweight (defined as >+1 s.d. from the mean BMI of the WHO Growth Standards) was compared with normal weight (>−2 s.d. to ⩽1 s.d. from the mean of the WHO Growth Reference). Additional analyses were performed to check the robustness of the results: (a) stratified by sex, (b) birth weight and preterm birth. These were included in the model but were not included in the main analysis, as they may well be mediators of the relation between prenatal and postnatal exposure to CB-153 and p,p'-DDE and growth, (c) excluding maternal BMI from the model, as this also may be a mediator of the associations under study, (d) prenatal and postnatal exposures kept in the same model, (e) only adjusting for maternal age and maternal smoking and, finally, (f) sensitivity analyses using different chained imputation models and by creating a varying number of data sets (20 and 150) were performed to check the robustness of the imputation model.
All analyses were performed stratified by population as well as pooled (adjusted for population). A P-value less than 0.05 was considered statistically significant. The Stata statistical package (version 12.1, StataCorp, College Station, TX, USA) was used for all analyses.
Results
At baseline, 1441 pregnant women participated in an interview and donated a blood sample. A total of 1117 (78%) women and their children were followed up when the median age of the children was 8 years in Greenland and Poland and 7 years in Ukraine. Twins (n=8) were excluded, leaving a total study population of 1109 at follow-up. The study population was distributed across Greenland (525 (47.3% of the study population)), Poland (92 (8.3%)) and Ukraine (492 children (44.4%)). The participation rates at follow-up were 89% in Greenland, 36% in Poland and 80% in Ukraine. The Spearman’s correlation coefficient between maternal pregnancy levels of CB-153 and p,p'-DDE was 0.92 in Greenland, 0.47 in Poland and 0.46 in Ukraine. In Greenland, the Spearman’s correlation coefficient between maternal pregnancy levels and estimated postnatal levels was 0.81 and 0.82 for CB-153 and p,p'-DDE, respectively. In Poland, the equivalents were 0.78 and 0.68, and in Ukraine, the correlations were 0.56 and 0.49, respectively. The non-response analysis showed only modest differences between responders and non-responders at follow-up concerning exposure levels and no difference in maternal pre-pregnancy BMI and paternal BMI (Supplementary Table 1). Characteristics of the study population are presented in Table 1. The median BMI (10–90 percentile) of the Greenlandic children was 17 kg m−2 (15–21) compared with 15 (13–19) and 16 (14–18) in Poland and Ukraine, respectively. In Greenland, 27% of the children were overweight or obese according to WHO standards, whereas the equivalents in Poland and Ukraine were 19% and 18%, respectively. In comparison with women from Greenland, women from Ukraine were younger, more often primiparous, less often smokers, more often breastfed for >12 months, had a lower pre-pregnancy BMI and their children were younger at follow-up. Women from Poland were older at enrolment, had lower pre-pregnancy BMI, were more often primiparous, had higher educational level and were less often smokers in comparison with the Greenlandic population. In Greenland, maternal median (10–90 percentile) serum CB-153 was 107 (30–369) ng g−1 lipids, which was considerably higher than in Poland and Ukraine. Maternal p,p'-DDE concentrations were highest in Ukraine, with maternal median (10–90 percentile) serum p,p'-DDE at 639 (329–1303) ng g−1 lipids. The estimated postnatal CB-153 exposure was highest in Greenland, whereas estimated postnatal p,p'-DDE exposure was highest in Ukraine, closely followed by Poland. The difference in prenatal and estimated postnatal exposure profiles are further presented in Table 2, describing the exposure tertiles stratified by country. The pattern of missing data in relation to country is presented in Table 3.
Table 4 presents crude and adjusted results of child BMI z-score in relation to prenatal exposure to the compounds based on the imputation-based analyses. Overall, there was no crude or adjusted dose–response relation between prenatal CB-153 and p,p'-DDE exposure and BMI z-scores. In Ukraine, crude and adjusted associations of maternal p,p'-DDE concentrations in the third tertile (>791 ng g−1 lipids) compared with the first tertile (<488 ng g−1 lipids) were inversely associated with child BMI z-scores (adjusted β=−0.30 (95% confidence interval (CI): −0.55 to −0.04)). Results from the complete case analyses were similar (Supplementary Table 2).
The results on the imputation-based associations between estimated first 12 months postnatal exposures and child BMI z-scores are presented in Table 5. Neither crude nor adjusted analyses indicated associations apart from a statistically significant association in Ukraine, where adjusted medium p,p'-DDE concentrations compared with low p,p'-DDE concentrations were associated with lower BMI z-scores (adjusted β=−0.35 (95% CI: −0.67 to −0.03)). Results from the complete case analyses were similar (Supplementary Table 3). ORs of the association between maternal and estimated postnatal organochlorine concentrations and overweight were consistent with the results of the linear regression (data not shown).
Several sensitivity analyses showed generally similar results as seen in Tables 4 and 5 (data not shown): (a) no difference was observed when the results were stratified by sex, (b) including preterm birth and birth weight in the model lead to similar results, (c) removing maternal BMI from the model had no impact on results, (d) having both prenatal and postnatal exposure in the model gave similar results, except for a nonsignificant result for the medium exposed group of postnatal p,p'-DDE in Ukraine and a statistically significant P for trend in Poland on postnatal CB-153 and DDE and BMI z-score (Supplementary Table 4), (e) analysing smaller models with only maternal age and smoking as covariates made the Ukrainian result on high prenatal p,p'-DDE and BMI z-score significant (β=−0.35 (95% CI: −0.60 to −0.10)) but did not change the results otherwise (Supplementary Table 5) and finally, (f) the four sensitivity analyses on the imputation model proved the imputation model robust, as the results did not materially change.
Discussion
Overall, this study suggests no strong association between prenatal and early-life p,p'-DDE and CB-153 exposures and BMI z-scores at 5–9 years of age.
Our hypothesis was that higher early-life exposure to DDE and PCBs would be associated with higher BMI. However, the results did not support this. Our results are in line with a recent study within the US Collaborative Perinatal Study, which reported no associations between high levels of prenatal p,p’-DDE or total-PCB exposures and overweight or obesity at 7 years.35 We observed no association between prenatal CB-153 exposure and BMI z-score, whereas a study reported higher BMI s.d. scores in relation to cord blood total-PCBs at 1–3 years.17 The difference in results could be caused by duration of follow-up, as the children in this cohort are somewhat older and potential associations earlier in life have not been investigated. Also, the observed association could be due to congeners other than CB-153. Another well-performed but rather small study found an association between high prenatal total-PCB exposures and risk of overweight in girls at 6.5 years compared with low exposures.16 This was also observed in a cohort study of 5-year olds from the 1960s with high PCB exposure levels,36 but these results are inconsistent with our study results. Also, in contrast to our study, a small Spanish study reported an association between medium prenatal DDE exposure and increased risk of overweight compared with low exposure,16 whereas another study with rather high prenatal DDE exposure levels observed no independent association between prenatal DDE exposure and BMI or risk of overweight.37 Our null finding for postnatal exposure to CB-153 supports a previous null finding in a small but well-performed Dutch cohort study examining weight and height as outcomes.38 However, one small informative study has reported increased risk of overweight in relation to postnatal PCB and DDE levels.16 Results of all the above discussed follow-up studies have reached inconsistent conclusions, which could be due to different exposure levels, measured congeners, choice of covariates in the model and age at the follow-up.
The results of this study should be interpreted in light of some study limitations. In the main analyses, we chose to use the WHO Growth Standards for BMI z-scores as internally derived standardized BMI z-scores might have caused unreliable coefficients because of the small sample in Poland. As the WHO Growth Standards are generally representative across ethnicities,39 we deemed this approach preferable, and sensitivity analyses proved the results to be robust.
We used different scales for weighing children at the follow-up examination. However, any misclassification most likely did not differ between exposure groups and therefore was non-differential. Further, we had no measure of the children’s postnatal organochlorines exposure and used a toxicokinetic model to estimate the accumulated concentrations during the first 12 months of life. The toxicokinetic model has proven robust in a validation study in similar settings, and a study suggests that the method is superior to the often used method of duration of breastfeeding multiplied by the prenatal exposure.21 As exclusive breastfeeding has been reported as having the largest relative influence on estimates of postnatal exposure,21 we chose the first 12 months of life, where exclusive breastfeeding is present, as the postnatal period. An exact measure of exposure of each child would, however, have been desirable. Blood samples were collected throughout pregnancy and as organochlorine concentrations tend to decrease during pregnancy, there is a risk of exposure misclassification. However, we addressed this issue by adjusting for gestational age at blood sampling, although we recognize that this has not completely eliminated misclassification. Also, analysis of only complete cases in a follow-up study with missing data could cause selection bias and to overcome this challenge, we performed multiple imputation analyses. The considerable loss to follow-up in Poland (follow-up participation rate 36%) could introduce selection bias. However, non-response analysis showed only modest difference according to exposure levels, and no difference on maternal pre-pregnacy BMI and paternal BMI between responders and non-responders indicating no high risk of selection bias by the loss to follow-up.
Exposure levels vary according to population. Still, the results are somewhat consistent across population apart from the inverse association observed in Ukraine, which could be spurious. The lack of associations, however, may be a result of generally low exposure levels as both exposures are receding.
This study has some important strengths. First, the follow-up was continued for up till 9 years, enabling analyses of associations for a considerable period of time. Second, the sample size in Greenland and Ukraine made it possible to model the associations, including adjustment for a number of covariates, with substantial power in these two countries and in the pooled analyses. Third, the varying exposure levels in the three countries enhanced our chance to observe an association, if any. Finally, we used a standardized measure of BMI, which facilitates comparison across studies with populations of different ages. Our results were generally robust, as several sensitivity analyses showed similar results with few exceptions.
In conclusion, in this prospective birth cohort study of Greenlandic, Polish and Ukrainian populations, we found no overall association between pre- or postnatal exposure to p,p'-DDE and CB-153 and BMI at the age of 5–9 years.
References
Jacobson JL, Fein GG, Jacobson SW, Schwartz PM, Dowler JK . The transfer of polychlorinated biphenyls (PCBs) and polybrominated biphenyls (PBBs) across the human placenta and into maternal milk. Am J Public Health 1984; 74: 378–379.
Gladen BC, Schecter AJ, Papke O, Shkyryak-Nyzhnyk ZA, Hryhorczuk DO, Little RE . Polychlorinated dibenzo-p-dioxins, polychlorinated dibenzofurans, and coplanar polychlorinated biphenyls in breast milk from two cities in Ukraine. J Toxicol Environ Health A 1999; 58: 119–127.
Gladen BC, Monaghan SC, Lukyanova EM, Hulchiy OP, Shkyryak-Nyzhnyk ZA, Sericano JL et al. Organochlorines in breast milk from two cities in Ukraine. Environ Health Perspect 1999; 107: 459–462.
Czaja K, Ludwicki JK, Goralczyk K, Strucinski P . Organochlorine pesticides, HCB, and PCBs in human milk in Poland. Bull Environ Contam Toxicol 1997; 58: 769–775.
Dorea JG, Cruz-Granja AC, Lacayo-Romero ML, Cuadra-Leal J . Perinatal metabolism of dichlorodiphenyldichloroethylene in Nicaraguan mothers. Environ Res 2001; 86: 229–237.
Rogan WJ, Ragan NB . Some evidence of effects of environmental chemicals on the endocrine system in children. Int J Hyg Environ Health 2007; 210: 659–667.
Eubig PA, Aguiar A, Schantz SL . Lead and PCBs as risk factors for attention deficit/hyperactivity disorder. Environ Health Perspect 2010; 118: 1654–1667.
Boucher O, Muckle G, Bastien CH . Prenatal exposure to polychlorinated biphenyls: a neuropsychologic analysis. Environ Health Perspect 2009; 117: 7–16.
Ribas-Fito N, Sala M, Kogevinas M, Sunyer J . Polychlorinated biphenyls (PCBs) and neurological development in children: a systematic review. J Epidemiol Community Health 2001; 55: 537–546.
Hallgren S, Sinjari T, Hakansson H, Darnerud PO . Effects of polybrominated diphenyl ethers (PBDEs) and polychlorinated biphenyls (PCBs) on thyroid hormone and vitamin A levels in rats and mice. Arch Toxicol 2001; 75: 200–208.
Herbstman JB, Sjodin A, Apelberg BJ, Witter FR, Halden RU, Patterson DG et al. Birth delivery mode modifies the associations between prenatal polychlorinated biphenyl (PCB) and polybrominated diphenyl ether (PBDE) and neonatal thyroid hormone levels. Environ Health Perspect 2008; 116: 1376–1382.
Janesick A, Blumberg B . Endocrine disrupting chemicals and the developmental programming of adipogenesis and obesity. Birth Defects Res C Embryo Today 2011; 93: 34–50.
Newbold RR, Padilla-Banks E, Jefferson WN . Environmental estrogens and obesity. Mol Cell Endocrinol 2009; 304: 84–89.
Vom Saal FS, Nagel SC, Coe BL, Angle BM, Taylor JA . The estrogenic endocrine disrupting chemical bisphenol A (BPA) and obesity. Mol Cell Endocrinol 2012; 354: 74–84.
Goodman M, Lakind JS, Mattison DR . Do phthalates act as obesogens in humans? A systematic review of the epidemiological literature. Crit Rev Toxicol 2014; 44: 151–175.
Valvi D, Mendez MA, Martinez D, Grimalt JO, Torrent M, Sunyer J et al. Prenatal concentrations of polychlorinated biphenyls, DDE, and DDT and overweight in children: a prospective birth cohort study. Environ Health Perspect 2012; 120: 451–457.
Verhulst SL, Nelen V, Hond ED, Koppen G, Beunckens C, Vael C et al. Intrauterine exposure to environmental pollutants and body mass index during the first 3 years of life. Environ Health Perspect 2009; 117: 122–126.
Warner M, Aguilar Schall R, Harley KG, Bradman A, Barr D, Eskenazi B . In utero DDT and DDE exposure and obesity status of 7-year-old Mexican-American children in the CHAMACOS cohort. Environ Health Perspect 2013; 121: 631–636.
Mendez MA, Garcia-Esteban R, Guxens M, Vrijheid M, Kogevinas M, Goni F et al. Prenatal organochlorine compound exposure, rapid weight gain, and overweight in infancy. Environ Health Perspect 2011; 119: 272–278.
Cupul-Uicab LA, Hernandez-Avila M, Terrazas-Medina EA, Pennell ML, Longnecker MP . Prenatal exposure to the major DDT metabolite 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDE) and growth in boys from Mexico. Environ Res 2010; 110: 595–603.
Verner MA, Sonneborn D, Lancz K, Muckle G, Ayotte P, Dewailly E et al. Toxicokinetic modeling of persistent organic pollutant levels in blood from birth to 45 months of age in longitudinal birth cohort studies. Environ Health Perspect 2013; 121: 131–137.
Toft G, Axmon A, Giwercman A, Thulstrup AM, Rignell-Hydbom A, Pedersen HS et al. Fertility in four regions spanning large contrasts in serum levels of widespread persistent organochlorines: a cross-sectional study. Environ Health 2005; 4: 26.
Jonsson BA, Rylander L, Lindh C, Rignell-Hydbom A, Giwercman A, Toft G et al. Inter-population variations in concentrations, determinants of and correlations between 2,2',4,4',5,5'-hexachlorobiphenyl (CB-153) and 1,1-dichloro-2,2-bis (p-chlorophenyl)-ethylene (p,p'-DDE): a cross-sectional study of 3161 men and women from Inuit and European populations. Environ Health 2005; 4: 27.
Sennbro CJ, Lindh CH, Tinnerberg H, Gustavsson C, Littorin M, Welinder H et al. Development, validation and characterization of an analytical method for the quantification of hydrolysable urinary metabolites and plasma protein adducts of 2,4- and 2,6-toluene diisocyanate, 1,5-naphthalene diisocyanate and 4,4'-methylenediphenyl diisocyanate. Biomarkers 2003; 8: 204–217.
de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J . Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ 2007; 85: 660–667.
Strauss RS . Effects of the intrauterine environment on childhood growth. Br Med Bull 1997; 53: 81–95.
Pala V, Lissner L, Hebestreit A, Lanfer A, Sieri S, Siani A et al. Dietary patterns and longitudinal change in body mass in European children: a follow-up study on the IDEFICS multicenter cohort. Eur J Clin Nutr 2013; 67: 1042–1049.
Timmermans SH, Mommers M, Gubbels JS, Kremers SP, Stafleu A, Stehouwer CD et al. Maternal smoking during pregnancy and childhood overweight and fat distribution: the KOALA Birth Cohort Study. Pediatr Obes 2013; 9: e14–e25.
Shi Y, De Groh M, Morrison H . Perinatal and early childhood factors for overweight and obesity in young Canadian children. Can J Public Health 2013; 104: e69–e74.
Haugaard LK, Ajslev TA, Zimmermann E, Angquist L, Sorensen TI . Being an only or last-born child increases later risk of obesity. PLoS One 2013; 8: e56357.
Dubois L, Girard M . Early determinants of overweight at 4.5 years in a population-based longitudinal study. Int J Obes (Lond) 2006; 30: 610–617.
Schuch I, Castro TG, Vasconcelos Fde A, Dutra CL, Goldani MZ . Excess weight in preschoolers: prevalence and associated factors. J Pediatr (Rio J) 2013; 89: 179–188.
Sterne JA, White IR, Carlin JB, Spratt M, Royston P, Kenward MG et al. Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ 2009; 338: b2393.
Donders AR, van der Heijden GJ, Stijnen T, Moons KG . Review: a gentle introduction to imputation of missing values. J Clin Epidemiol 2006; 59: 1087–1091.
Cupul-Uicab LA, Klebanoff MA, Brock JW, Longnecker MP . Prenatal exposure to persistent organochlorines and childhood obesity in the U.S. Collaborative perinatal project. Environ Health Perspect 2013; 121: 1103–1109.
Hertz-Picciotto I, Charles MJ, James RA, Keller JA, Willman E, Teplin S . In utero polychlorinated biphenyl exposures in relation to fetal and early childhood growth. Epidemiology 2005; 16: 648–656.
Jusko TA, Koepsell TD, Baker RJ, Greenfield TA, Willman EJ, Charles MJ et al. Maternal DDT exposures in relation to fetal and 5-year growth. Epidemiology 2006; 17: 692–700.
Patandin S, Koopman-Esseboom C, de Ridder MA, Weisglas-Kuperus N, Sauer PJ . Effects of environmental exposure to polychlorinated biphenyls and dioxins on birth size and growth in Dutch children. Pediatr Res 1998; 44: 538–545.
WHO Multicentre Growth Reference Study Group. WHO Child Growth Standards based on length/height, weight and age. Acta Paediatr Suppl 2006; 450: 76–85.
Acknowledgements
The CLEAR (Climate change, environmental contaminants and reproductive health, http://www.inuendo.dk/clear) and INUENDO (Biopersistent organochlorines in diet and human fertility) studies were funded by the European Commission’s 7th and 5th Framework Programmes, respectively (grants FP7-ENV-2008-1-226217 and QLK4-CT-2001-00202).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Rights and permissions
About this article
Cite this article
Høyer, B., Ramlau-Hansen, C., Henriksen, T. et al. Body mass index in young school-age children in relation to organochlorine compounds in early life: a prospective study. Int J Obes 38, 919–925 (2014). https://doi.org/10.1038/ijo.2014.58
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ijo.2014.58
Keywords
This article is cited by
-
Dichlorodiphenyltrichloroethane for Malaria and Agricultural Uses and Its Impacts on Human Health
Bulletin of Environmental Contamination and Toxicology (2023)
-
Prenatal exposure to persistent organic pollutants and metals and problematic child behavior at 3–5 years of age: a Greenlandic cohort study
Scientific Reports (2021)
-
Chronic oral exposure to pesticides and their consequences on metabolic regulation: role of the microbiota
European Journal of Nutrition (2021)
-
Maternal Exposure to Synthetic Chemicals and Obesity in the Offspring: Recent Findings
Current Environmental Health Reports (2015)