Abstract
Background:
Non-alcoholic fatty liver disease (NAFLD) is the most common chronic liver disease worldwide and is strongly associated with obesity, dyslipidemia and insulin resistance. NAFLD often presents as simple steatosis (NAFL) but can progress to non-alcoholic steatohepatitis (NASH) and fibrosis. Current non-invasive biomarkers are not tailored to identify significant (⩾F2) fibrosis, although recent guidelines recommend a stringent follow-up of this patient population. We and others have reported on the role of pathological angiogenesis in the pathogenesis of NAFLD, highlighting pro-angiogenic factors as potential diagnostic markers.
Objective:
To investigate the applicability of angiogenic and endothelial dysfunction markers as non-invasive diagnostic tools for NASH or NASH-associated fibrosis in obese patients.
Methods:
In a prospective cross-sectional study, male patients undergoing bariatric surgery (n=61) and control patients (n=35) were recruited. Serum protein levels and visceral adipose tissue gene expression of endothelial dysfunction and angiogenic markers were analyzed by multiplex bead-based assay and quantitative RT-PCR, respectively. For validation, we recruited a second cohort of patients undergoing bariatric surgery (n=40) and a cohort of NAFLD patients from our outpatient clinic (n=30).
Results:
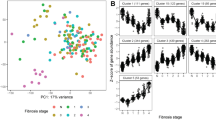
We identified serum vascular cell adhesion molecule-1 (VCAM-1) as an independent predictor for ⩾F2 fibrosis (median 14.0 vs 8.7 ng ml−1 in patients with and without significant fibrosis; P<0.0001) with an area under the receiver-operating characteristics (AUROC) curve of 0.80. The cutoff point of 13.2 ng ml−1 showed a sensitivity of 80% and specificity of 83%. In line with these results, VCAM-1 visceral adipose tissue gene expression was also elevated in patients with fibrosis (P=0.030). In the bariatric surgery and clinical validation cohorts, VCAM-1 displayed similar AUROCs of 0.89 and 0.85, respectively.
Conclusions:
VCAM-1 levels are able to accurately predict significant (⩾F2) fibrosis in NAFLD patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Yki-Jarvinen H . Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol 2014; 2: 901–910.
Lonardo A, Ballestri S, Marchesini G, Angulo P, Loria P . Nonalcoholic fatty liver disease: a precursor of the metabolic syndrome. Dig Liver Dis 2015; 47: 181–190.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M . Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016; 64: 73–84.
Ekstedt M, Hagstrom H, Nasr P, Fredrikson M, Stal P, Kechagias S et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 2015; 61: 1547–1554.
Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2015; 149: 389–397.e10.
Vernon G, Baranova A, Younossi ZM . Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 2011; 34: 274–285.
European Association for the Study of the Liver, European Association for the Study of Diabetes, European Association for the Study of Obesity. EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 2016; 64: 1388–1402.
Machado MV, Cortez-Pinto H . Non-invasive diagnosis of non-alcoholic fatty liver disease. A critical appraisal. J Hepatol 2013; 58: 1007–1019.
Guha IN, Parkes J, Roderick P, Chattopadhyay D, Cross R, Harris S et al. Noninvasive markers of fibrosis in nonalcoholic fatty liver disease: validating the European Liver Fibrosis Panel and exploring simple markers. Hepatology 2008; 47: 455–460.
McPherson S, Anstee QM, Henderson E, Day CP, Burt AD . Are simple noninvasive scoring systems for fibrosis reliable in patients with NAFLD and normal ALT levels? Eur J Gastroenterol Hepatol 2013; 25: 652–658.
Villanova N, Moscatiello S, Ramilli S, Bugianesi E, Magalotti D, Vanni E et al. Endothelial dysfunction and cardiovascular risk profile in nonalcoholic fatty liver disease. Hepatology 2005; 42: 473–480.
Long MT, Wang N, Larson MG, Mitchell GF, Palmisano J, Vasan RS et al. Nonalcoholic fatty liver disease and vascular function: cross-sectional analysis in the Framingham heart study. Arterioscler Thromb Vasc Biol 2015; 35: 1284–1291.
Kasumov T, Edmison JM, Dasarathy S, Bennett C, Lopez R, Kalhan SC . Plasma levels of asymmetric dimethylarginine in patients with biopsy-proven nonalcoholic fatty liver disease. Metabolism 2011; 60: 776–781.
Verrijken A, Francque S, Mertens I, Prawitt J, Caron S, Hubens G et al. Prothrombotic factors in histologically proven nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology 2014; 59: 121–129.
Kitade M, Yoshiji H, Noguchi R, Ikenaka Y, Kaji K, Shirai Y et al. Crosstalk between angiogenesis, cytokeratin-18, and insulin resistance in the progression of non-alcoholic steatohepatitis. World J Gastroenterol 2009; 15: 5193–5199.
Kitade M, Yoshiji H, Kojima H, Ikenaka Y, Noguchi R, Kaji K et al. Leptin-mediated neovascularization is a prerequisite for progression of nonalcoholic steatohepatitis in rats. Hepatology 2006; 44: 983–991.
Coulon S, Legry V, Heindryckx F, Van Steenkiste C, Casteleyn C, Olievier K et al. Role of vascular endothelial growth factor in the pathophysiology of nonalcoholic steatohepatitis in two rodent models. Hepatology 2013; 57: 1793–1805.
Francque S, Laleman W, Verbeke L, Van Steenkiste C, Casteleyn C, Kwanten W et al. Increased intrahepatic resistance in severe steatosis: endothelial dysfunction, vasoconstrictor overproduction and altered microvascular architecture. Lab Invest 2012; 92: 1428–1439.
Lefere S, Van Steenkiste C, Verhelst X, Van Vlierberghe H, Devisscher L, Geerts A . Hypoxia-regulated mechanisms in the pathogenesis of obesity and non-alcoholic fatty liver disease. Cell Mol Life Sci 2016; 73: 3419–3431.
Pasarin M, Abraldes JG, Rodriguez-Vilarrupla A, La Mura V, Garcia-Pagan JC, Bosch J . Insulin resistance and liver microcirculation in a rat model of early NAFLD. J Hepatol 2011; 55: 1095–1102.
Francque SM, van der Graaff D, Kwanten WJ . Non-alcoholic fatty liver disease and cardiovascular risk: pathophysiological mechanisms and implications. J Hepatol 2016; 65: 425–443.
Bekaert M, Ouwens DM, Horbelt T, Van de Velde F, Fahlbusch P, Herzfeld de Wiza D et al. Reduced expression of chemerin in visceral adipose tissue associates with hepatic steatosis in patients with obesity. Obesity 2016; 24: 2544–2552.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC . Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412–419.
American Diabetes Association. Standards of Medical Care in Diabetes-2016. Diabetes Care 2016; 39 (Suppl 1): S1–112.
Ratziu V, Giral P, Charlotte F, Bruckert E, Thibault V, Theodorou I et al. Liver fibrosis in overweight patients. Gastroenterology 2000; 118: 1117–1123.
Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006; 43: 1317–1325.
Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005; 41: 1313–1321.
Kleiner DE, Brunt EM . Nonalcoholic fatty liver disease: pathologic patterns and biopsy evaluation in clinical research. Semin Liver Dis 2012; 32: 3–13.
Bedossa P, Consortium FP . Utility and appropriateness of the fatty liver inhibition of progression (FLIP) algorithm and steatosis, activity, and fibrosis (SAF) score in the evaluation of biopsies of nonalcoholic fatty liver disease. Hepatology 2014; 60: 565–575.
Loomba R, Abraham M, Unalp A, Wilson L, Lavine J, Doo E et al. Association between diabetes, family history of diabetes, and risk of nonalcoholic steatohepatitis and fibrosis. Hepatology 2012; 56: 943–951.
du Plessis J, van Pelt J, Korf H, Mathieu C, van der Schueren B, Lannoo M et al. Association of adipose tissue inflammation with histologic severity of nonalcoholic fatty liver disease. Gastroenterology 2015; 149: 635–648.e14.
Sung HK, Doh KO, Son JE, Park JG, Bae Y, Choi S et al. Adipose vascular endothelial growth factor regulates metabolic homeostasis through angiogenesis. Cell Metab 2013; 17: 61–72.
Ratziu V . Novel pharmacotherapy options for NASH. Dig Dis Sci 2016; 61: 1398–1405.
Kwok R, Tse YK, Wong GL, Ha Y, Lee AU, Ngu MC et al. Systematic review with meta-analysis: non-invasive assessment of non-alcoholic fatty liver disease—the role of transient elastography and plasma cytokeratin-18 fragments. Aliment Pharmacol Ther 2014; 39: 254–269.
Sun K, Wernstedt Asterholm I, Kusminski CM, Bueno AC, Wang ZV, Pollard JW et al. Dichotomous effects of VEGF-A on adipose tissue dysfunction. Proc Natl Acad Sci USA 2012; 109: 5874–5879.
Flemming S, Burkard N, Renschler M, Vielmuth F, Meir M, Schick MA et al. Soluble VE-cadherin is involved in endothelial barrier breakdown in systemic inflammation and sepsis. Cardiovasc Res 2015; 107: 32–44.
Galkina E, Ley K . Vascular adhesion molecules in atherosclerosis. Arterioscler Thromb Vasc Biol 2007; 27: 2292–2301.
Coulon S, Francque S, Colle I, Verrijken A, Blomme B, Heindryckx F et al. Evaluation of inflammatory and angiogenic factors in patients with non-alcoholic fatty liver disease. Cytokine 2012; 59: 442–449.
Dewerchin M, Carmeliet P . PlGF: a multitasking cytokine with disease-restricted activity. Cold Spring Harb Perspect Med 2012; 2: a011056.
Tarantino G, Conca P, Pasanisi F, Ariello M, Mastrolia M, Arena A et al. Could inflammatory markers help diagnose nonalcoholic steatohepatitis? Eur J Gastroenterol Hepatol 2009; 21: 504–511.
Yilmaz Y, Yonal O, Kurt R, Alahdab YO, Ozdogan O, Celikel CA et al. Circulating levels of vascular endothelial growth factor A and its soluble receptor in patients with biopsy-proven nonalcoholic fatty liver disease. Arch Med Res 2011; 42: 38–43.
Yilmaz Y, Dolar E, Ulukaya E, Akgoz S, Keskin M, Kiyici M et al. Soluble forms of extracellular cytokeratin 18 may differentiate simple steatosis from nonalcoholic steatohepatitis. World J Gastroenterol 2007; 13: 837–844.
Cusi K, Chang Z, Harrison S, Lomonaco R, Bril F, Orsak B et al. Limited value of plasma cytokeratin-18 as a biomarker for NASH and fibrosis in patients with non-alcoholic fatty liver disease. J Hepatol 2014; 60: 167–174.
Yoshimura K, Okanoue T, Ebise H, Iwasaki T, Mizuno M, Shima T et al. Identification of novel noninvasive markers for diagnosing nonalcoholic steatohepatitis and related fibrosis by data mining. Hepatology 2016; 63: 462–473.
Raevens S, Coulon S, Van Steenkiste C, Colman R, Verhelst X, Van Vlierberghe H et al. Role of angiogenic factors/cell adhesion markers in serum of cirrhotic patients with hepatopulmonary syndrome. Liver Int 2015; 35: 1499–1507.
Lo Iacono O, Rincon D, Hernando A, Ripoll C, Catalina MV, Salcedo M et al. Serum levels of soluble vascular cell adhesion molecule are related to hyperdynamic circulation in patients with liver cirrhosis. Liver Int 2008; 28: 1129–1135.
Richards TJ, Kaminski N, Baribaud F, Flavin S, Brodmerkel C, Horowitz D et al. Peripheral blood proteins predict mortality in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 2012; 185: 67–76.
Acknowledgements
We thank Petra van Wassenhove and Hilde Devlies for their excellent technical assistance, Elien Glorieus for her accurate management of the patient database, and Roos Coolman from the Statistics unit for her helpful advice. Guarantor of the article: Anja Geerts. SL, SR and XV received a research grant from the Fund for Scientific Research Flanders (FWO15/ASP/146, FWO14/ASP/200 and 1700214N, respectively). HVV is a senior clinical researcher of the FWO Flanders.
Author contributions
SL, LD, BL and AG conceived and designed the study; SL, FVdV, MB, YVN, MP and AH were involved in data acquisition; SL, LD, SR, CVS, BL and AG were involved in the data analysis and interpretation; SL drafted the paper; LD, XV, HVV, BL and AG critically revised the manuscript for intellectual content.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Lefere, S., Van de Velde, F., Devisscher, L. et al. Serum vascular cell adhesion molecule-1 predicts significant liver fibrosis in non-alcoholic fatty liver disease. Int J Obes 41, 1207–1213 (2017). https://doi.org/10.1038/ijo.2017.102
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ijo.2017.102
This article is cited by
-
Intra and inter-organ communication through extracellular vesicles in obesity: functional role of obesesomes and steatosomes
Journal of Translational Medicine (2025)
-
Endothelial dysfunction markers predict short-term mortality in patients with severe alcoholic hepatitis
Hepatology International (2021)
-
Wet Biomarker-Based Assessment of Steatosis, Inflammation, and Fibrosis in NAFLD
Current Hepatology Reports (2017)