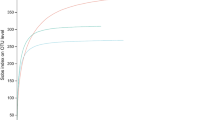Abstract
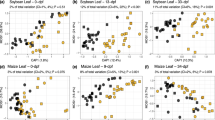
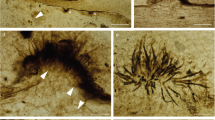
Fungi are considered the primary decomposers of dead plant biomass in terrestrial ecosystems. However, current knowledge regarding the successive changes in fungal communities during litter decomposition is limited. Here we explored the development of the fungal community over 24 months of litter decomposition in a temperate forest with dominant Quercus petraea using 454-pyrosequencing of the fungal internal transcribed spacer (ITS) region and cellobiohydrolase I (cbhI) genes, which encode exocellulases, to specifically address cellulose decomposers. To quantify the involvement of phyllosphere fungi in litter decomposition, the fungal communities in live leaves and leaves immediately before abscission were also analysed. The results showed rapid succession of fungi with dramatic changes in the composition of the fungal community. Furthermore, most of the abundant taxa only temporarily dominated in the substrate. Fungal diversity was lowest at leaf senescence, increased until month 4 and did not significantly change during subsequent decomposition. Highly diverse community of phyllosphere fungi inhabits live oak leaves 2 months before abscission, and these phyllosphere taxa comprise a significant share of the fungal community during early decomposition up to the fourth month. Sequences assigned to the Ascomycota showed highest relative abundances in live leaves and during the early stages of decomposition. In contrast, the relative abundance of sequences assigned to the Basidiomycota phylum, particularly basidiomycetous yeasts, increased with time. Although cellulose was available in the litter during all stages of decomposition, the community of cellulolytic fungi changed substantially over time. The results indicate that litter decomposition is a highly complex process mediated by various fungal taxa.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Amann R, Ludwig W, Schleifer K . (1995). Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol Rev 59: 143–169.
Amend AS, Seifert KA, Bruns TD . (2010). Quantifying microbial communities with 454 pyrosequencing: does read abundance count? Mol Ecol 19: 5555–5565.
Aneja MK, Sharma S, Fleischmann F, Stich S, Heller W, Bahnweg G et al. (2006). Microbial colonization of beech and spruce litter - influence of decomposition site and plant litter species on the diversity of microbial community. Microb Ecol 52: 127–135.
Baldrian P . (2008). Enzymes of saprotrophic basidiomycetes. In: Boddy L, Frankland J, van West P (eds). Ecology of Saprotrophic Basidiomycetes. Academic Press: New York, pp 19–41.
Baldrian P, Valášková V . (2008). Degradation of cellulose by basidiomycetous fungi. FEMS Microbiol Rev 32: 501–521.
Baldrian P, Voříšková J, Dobiášová P, Merhautová V, Lisá L, Valášková V . (2011). Production of extracellular enzymes and degradation of biopolymers by saprotrophic microfungi from the upper layers of forest soil. Plant Soil 338: 111–125.
Baldrian P, Kolařík M, Štursová M, Kopecký J, Valášková V, Větrovský T et al. (2012). Active and total microbial communities in forest soil are largely different and highly stratified during decomposition. ISME J 6: 248–258.
Baldrian P, Šnajdr J, Merhautová V, Dobiášová P, Cajthaml T, Valášková V (in press). Responses of the extracellular enzyme activities in hardwood forest to soil temperature and seasonality and the potential effects of climate change. Soil Biol Biochem: doi:10.1016/j.soilbio.2012.01.020.
Berg B, McClaugherty C, Santo AVD, Johnson D . (2001). Humus buildup in boreal forests: effects of litter fall and its N concentration. Can J Forest Res 31: 988–998.
Boberg JB, Ihrmark K, Lindahl BD . (2011). Decomposing capacity of fungi commonly detected in Pinus sylvestris needle litter. Fungal Ecol 4: 110–114.
Bodeker ITM, Nygren CMR, Taylor AFS, Olson A, Lindahl BD . (2009). ClassII peroxidase-encoding genes are present in a phylogenetically wide range of ectomycorrhizal fungi. ISME J 3: 1387–1395.
Carrino-Kyker SR, Swanson AK . (2008). Temporal and spatial patterns of eukaryotic and bacterial communities found in vernal pools. Appl Environ Microbiol 74: 2554–2557.
de Boer W, Folman LB, Summerbell RC, Boddy L . (2005). Living in a fungal world: impact of fungi on soil bacterial niche development. FEMS Microbiol Rev 29: 795–811.
Di Marino E, Scattolin L, Bodensteiner P, Agerer R . (2008). Sistotrema is a genus with ectomycorrhizal species - confirmation of what sequence studies already suggested. Mycol Prog 7: 169–176.
Dickie IA, Fukami T, Wilkie JP, Allen RB, Buchanan PK . (2012). Do assembly history effects attenuate from species to ecosystem properties? A field test with wood-inhabiting fungi. Ecol Lett 15: 133–141.
Dilly O, Bartsch S, Rosenbrock P, Buscot F, Munch JC . (2001). Shifts in physiological capabilities of the microbiota during the decomposition of leaf litter in a black alder (Alnus glutinosa (Gaertn.) L.) forest. Soil Biol Biochem 33: 921–930.
Duong LM, McKenzie EHC, Lumyong S, Hyde KD . (2008). Fungal succession on senescent leaves of Castanopsis diversifolia in Doi Suthep-Pui National Park, Thailand. Fungal Divers 30: 23–36.
Edgar RC . (2010). Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26: 2460–2461.
Edwards IP, Upchurch RA, Zak DR . (2008). Isolation of fungal cellobiohydrolase I genes from sporocarps and forest soils by PCR. Appl Environ Microbiol 74: 3481–3489.
Frankland JC . (1998). Fungal succession - unravelling the unpredictable. Mycol Res 102: 1–15.
Hering TF . (1967). Fungal decomposition of oak leaf litter. Trans Brit Mycol Soc 50: 267–273.
Jumpponen A, Jones KL . (2009a). Seasonally dynamic fungal communities in the Quercus macrocarpa phyllosphere differ between urban and nonurban environments. New Phytol 186: 496–513.
Jumpponen A, Jones KL . (2009b). Massively parallel 454 sequencing indicates hyperdiverse fungal communities in temperate Quercus macrocarpa phyllosphere. New Phytol 184: 438–448.
Kjoller A, Struwe S . (1982). Microfungi in ecosystems: fungal occurrence and activity in litter and soil. Oikos 39: 289–422.
Koide K, Osono T, Takeda H . (2005). Colonization and lignin decomposition of Camellia japonica leaf litter by endophytic fungi. Mycoscience 46: 280–286.
Korkama-Rajala T, Mueller MM, Pennanen T . (2008). Decomposition and fungi of needle litter from slow- and fast-growing norway spruce (Picea abies) clones. Microb Ecol 56: 76–89.
Kubartova A, Ranger J, Berthelin J, Beguiristain T . (2009). Diversity and decomposing ability of Saprophytic fungi from temperate forest litter. Microb Ecol 58: 98–107.
Li W, Godzik A . (2006). Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22: 1658–1659.
Lindahl BD, Finlay RD . (2006). Activities of chitinolytic enzymes during primary and secondary colonization of wood by basidiomycetous fungi. New Phytol 169: 389–397.
Melillo J, Aber J, Linkins A, Ricca A, Fry B, Nadelhoffer K . (1989). Carbon and nitrogen dynamics along the decay continuum: plant litter to soil organic matter. Plant Soil 115: 189–198.
O‘Brien HE, Parrent JL, Jackson JA, Moncalvo JM, Vilgalys R . (2005). Fungal community analysis by large-scale sequencing of environmental samples. Appl Environ Microbiol 71: 5544–5550.
Osono T, Takeda H . (2001). Organic chemical and nutrient dynamics in decomposing beech leaf litter in relation to fungal ingrowth and succession during 3-year decomposition processes in a cool temperate deciduous forest in Japan. Ecol Res 16: 649–670.
Osono T . (2002). Phyllosphere fungi on leaf litter of Fagus crenata: occurrence, colonization, and succession. Can J Bot 80: 460–469.
Osono T . (2005). Colonization and succession of fungi during decomposition of Swida controversa leaf litter. Mycologia 97: 589–597.
Osono T . (2006). Role of phyllosphere fungi of forest trees in the development of decomposer fungal communities and decomposition processes of leaf litter. Can J Microbiol 52: 701–716.
Osono T . (2007). Ecology of ligninolytic fungi associated with leaf litter decomposition. Ecol Res 22: 955–974.
Osono T, Hirose D, Fujimaki R . (2006). Fungal colonization as affected by litter depth and decomposition stage of needle litter. Soil Biol Biochem 38: 2743–2752.
Osono T, Ishii Y, Takeda H, Seramethakun T, Khamyong S, To-Anun C et al. (2009). Fungal succession and lignin decomposition on Shorea obutsa leaves in a tropical seasonal forest in northern Thailand. Fungal Divers 36: 101–119.
Promputtha I, Lumyong S, Dhanasekaran V, McKenzie E, Hyde K, Jeewon R . (2007). A phylogenetic evaluation of whether endophytes become saprotrophs at host senescence. Microb Ecol 53: 579–590.
Rajala T, Peltoniemi M, Hantula J, Mäkipää R, Pennanen T . (2011). RNA reveals a succession of active fungi during the decay of Norway spruce logs. Fungal Ecol 4: 437–448.
Reeder J, Knight R . (2010). Rapidly denoising pyrosequencing amplicon reads by exploiting rank-abundance distributions. Nat Methods 7: 668–669.
Sadaka N, Ponge JF . (2003). Fungal colonization of phyllosphere and litter of Quercus rotundifolia Lam. in a holm oak forest (High Atlas, Morocco). Biol Fertil Soils 39: 30–36.
Santamaría J, Bayman P . (2005). Fungal epiphytes and endophytes of coffee leaves (Coffea arabica). Microb Ecol 50: 1–8.
Schneider T, Keiblinger KM, Schmid E, Sterflinger-Gleixner K, Ellersdorfer G, Roschitzki B et al. (2012). Who is who in litter decomposition? Metaproteomics reveals major microbial players and their biogeochemical functions. ISME J 6: 1749–1762.
Slavikova E, Vadkertiova R, Vranova D . (2007). Yeasts colonizing the leaf surfaces. J Basic Microbiol 47: 344–350.
Stone JK . (1987). Initiation and development of latent infections by Rhabdocline parkeri on Douglas-fir. Can J Bot 65: 2614–2621.
Suto Y . (1999). Mycosphaerella chaenomelis sp. nov.: the teleomorph of Cercosporella sp., the causal fungus of frosty mildew in Chaenomeles sinensis, and its role as the primary infection source. Mycoscience 40: 509–516.
Šnajdr J, Valášková V, Merhautová V, Herinková J, Cajthaml T, Baldrian P . (2008). Spatial variability of enzyme activities and microbial biomass in the upper layers of Quercus petraea forest soil. Soil Biol Biochem 40: 2068–2075.
Šnajdr J, Cajthaml T, Valášková V, Merhautová V, Petránková M, Spetz P et al. (2011). Transformation of Quercus petraea litter: successive changes in litter chemistry are reflected in differential enzyme activity and changes in the microbial community composition. FEMS Microbiol Ecol 75: 291–303.
Štursová M, Žifčáková L, Leigh MB, Burgess R, Baldrian P . (2012). Cellulose utilisation in forest litter and soil: identification of bacterial and fungal decomposers. FEMS Microbiol Ecol 80: 735–746.
Tang AMC, Jeewon R, Hyde KD . (2005). Succession of microfungal communities on decaying leaves of Castanopsis fissa. Can J Microbiol 51: 967–974.
Tedersoo L, Nilsson RH, Abarenkov K, Jairus T, Sadam A, Saar I et al. (2010). 454 Pyrosequencing and Sanger sequencing of tropical mycorrhizal fungi provide similar results but reveal substantial methodological biases. New Phytol 188: 291–301.
Unterseher M, Schnittler M . (2009). Dilution-to-extinction cultivation of leaf-inhabiting endophytic fungi in beech (Fagus sylvatica L.) - different cultivation techniques influence fungal biodiversity assessment. Mycol Res 113: 645–654.
Valášková V, Šnajdr J, Bittner B, Cajthaml T, Merhautová V, Hoffichter M et al. (2007). Production of lignocellulose-degrading enzymes and degradation of leaf litter by saprotrophic basidiomycetes isolated from a Quercus petraea forest. Soil Biol Biochem 39: 2651–2660.
Weber CF, Zak DR, Hungate BA, Jackson RB, Vilgalys R, Evans RD et al. (2011). Responses of soil cellulolytic fungal communities to elevated atmospheric CO2 are complex and variable across five ecosystems. Environ Microbiol 13: 2778–2793.
White TJ, Bruns TD, Lee SB, Taylor JW . (1990). Analysis of phylogenetic relationships by amplification and direct sequencing of ribosomal RNA genes. In: Innis MA, Gelfand DH, Sninsky JN, White TJ (eds). PCR Protocols: A Guide to Methods and Applications. Academic Press: New York, pp 315–322.
Zhang P, Tian X, He X, Song F, Ren L, Jiang P . (2008). Effect of litter quality on its decomposition in broadleaf and coniferous forest. Eur J Soil Biol 44: 392–399.
Žifčáková L, Dobiášová P, Kolářová Z, Koukol O, Baldrian P . (2011). Enzyme activities of fungi associated with Picea abies needles. Fungal Ecol 4: 427–436.
Acknowledgements
This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic (ME10152, LD12050) and by the Czech Science Foundation (P504/12/0709).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The ISME Journal website
Rights and permissions
About this article
Cite this article
Voříšková, J., Baldrian, P. Fungal community on decomposing leaf litter undergoes rapid successional changes. ISME J 7, 477–486 (2013). https://doi.org/10.1038/ismej.2012.116
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ismej.2012.116
Keywords
This article is cited by
-
Different regulation processes of litter phosphorus loss for leaf and root under subtropical afforestation
Plant and Soil (2023)
-
Fungal communities mediate but do not control leaf litter chemical transformation in a temperate oak forest
Plant and Soil (2023)
-
The succession of microbial communities after residue returning in a Solonchak
Plant and Soil (2023)
-
Rhizosphere Microorganisms and Soil Physicochemical Properties of Restored Wetland Plant Communities at Cutting Slash of Populus deltoides in Dongting Lake
Wetlands (2023)
-
Metabarcoding analysis of the soil fungal community to aid the conservation of underexplored church forests in Ethiopia
Scientific Reports (2022)