Abstract
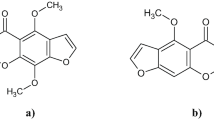
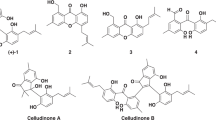
A new cytotoxic viriditoxin derivative, cladosporinone (1), along with the known viriditoxin (2) and two viriditoxin derivatives (3 and 4) were obtained from the fungus C ladosporium cladosporioides isolated from the sediment of a hypersaline lake in Egypt. The structure of the new compound (1) was determined by 1D and 2D NMR measurements as well as by high-resolution ESIMS and electronic circular dichroism spectroscopy. All isolated compounds were studied for their cytotoxicity against the murine lymphoma cell line L5187Y and for their antibiotic activity against several pathogenic bacteria. Viriditoxin (2) was the most active compound in both bioassays. Compound 1 also exhibited strong cytotoxicity against the murine lymphoma cell line L5187Y with an IC50 value of 0.88 μm, whereas its antibiotic activity was weak.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Aoki, H. & Okuhara, M. Natural β-lactam antibiotics. Ann. Rev. Microbiol. 34, 159–181 (1980).
Abraham, E. Selective reminiscences of β-lactam antibiotics: early research on penicillin and cephalosporins. BioEssays 12, 601–606 (1990).
Muñiz, C. C., Zelaya, T. E. C., Esquivel, G. R. & Fernández, F. J. Penicillin and cephalosporin production: a historical perspective. Rev. Latinoam. Microbiol. 49, 88–98 (2007).
Traber, R., Hofmann, H. & Kobel, H. Cyclosporins—new analogues by precursor directed biosynthesis. J. Antibiot. 42, 591–597 (1989).
Murthy, M. V. R., Mohan, E. V. S. & Sadhukhan, A. K. Cyclosporin-A production by Tolypocladium inflatum using solid state fermentation. Process Biochem 34, 269–280 (1999).
Alberts, A. W. Discovery, biochemistry and biology of lovastatin. Am. J. Cardiol. 62, 10J–15J (1988).
Wilson, Z. E. & Brimble, M. A. Molecules derived from the extremes of life. Nat. Prod. Rep. 26, 44–71 (2009).
Kingston, D. G. I. Modern natural products drug discovery and its relevance to biodiversity conservation. J. Nat. Prod. 74, 496–511 (2011).
Maezato, Y. & Blum, P. Survival of the fittest: overcoming oxidative stress at the extremes of acid, heat and metal. Life 2, 229–242 (2012).
Ma, Y., Galinski, E. A., Grant, W. D., Oren, A. & Ventosa, A. Halophiles 2010: life in saline environments. Appl. Environ. Microbiol. 76, 6971–6981 (2010).
Satyanarayana, T., Raghukumar, C. & Shivaji, S. Extremophilic microbes: diversity and perspectives. Curr. Sci. 89, 78–90p (2005).
Jalgaonwala, R. E., Mohite, B. V. & Mahajan, R. T. A review: natural products from plant associated endophytic fungi. J. Microbiol. Biotech. Res 1, 21–32 (2011).
Demain, A. L. Importance of microbial natural products and the need to revitalize their discovery. J. Ind. Microbiol. Biotechnol. 41, 185–201 (2014).
Liu, Y. et al. Tetrahydroanthraquinone derivatives from the endophytic fungus Stemphylium globuliferum. Eur. J. Org. Chem 12, 2646–2653 (2015).
Liu, Y. et al. Trimeric anthracenes from the endophytic fungus Stemphylium globuliferum. J. Nat. Prod. 77, 1734–1738 (2014).
Okoye, F. B. C. et al. A phenyldilactone, bisnorsesquiterpene, and cytotoxic phenolics from Maytenus senegalensis leaves. Tetrahedron Lett. 55, 3756–3760 (2014).
Chen, H. et al. A new cytotoxic cytochalasin from the endophytic fungus Trichoderma harzianum. Nat. Prod. Commun. 10, 585–587 (2015).
Suzuki, K., Nozawa, K., Nakajima, S. & Kawai, K. Structure revision of mycotoxin, viriditoxin, and its derivatives. Chem. Pharm. Bull. 38, 3180–3181 (1990).
Mizuba, S., Hsu, C. & Jiu, J. A third metabolite from Spicaria divaricata NRRL 5771. J. Antibiot. 30, 670–672 (1977).
Jiu, J. & Mizuba, S. Metabolic products from Spicaria divaricata NRRL 5771. J. Antibiot. 27, 760–765 (1974).
Ayer, W. A., Craw, P. A. & Nozawa, K. Two 1H-naphtho[2,3-c]pyran-1-one metabolites from the fungus Paecilomyces variotii. Can. J. Chem. 69, 189–191 (1991).
Tan, N. P. H. & Donner, C. D. Total synthesis and confirmation of the absolute stereochemistry of semiviriditoxin, a naphthopyranone metabolite from the fungus Paecilomyces variotii. Tetrahedron 65, 4007–4012 (2009).
Bode, H. B., Bethe, B., Höfs, R. & Zeeck, A. Big effects from small changes: possible ways to explore nature's chemical diversity. ChemBioChem 3, 619–627 (2002).
Chen, H. et al. Inducing secondary metabolite production by the soil-dwelling fungus Aspergillus terreus through bacterial co-culture. Phytochem. Lett. 12, 35–41 (2015).
Ola, A. R. B., Thomy, D., Lai, D., Brötz-Oesterhelt, H. & Proksch, P. Inducing secondary metabolite production by the endophytic fungus Fusarium tricinctum through coculture with Bacillus subtilis. J. Nat. Prod. 76, 2094–2099 (2013).
Donner, C. D. Naphthopyranones—isolation, bioactivity, biosynthesis and synthesis. Nat. Prod. Rep. 32, 578–604 (2015).
Anderson, D. E. et al. Comparison of small molecule inhibitors of the bacterial cell division protein FtsZ and identification of a reliable cross-species inhibitor. ACS Chem. Biol. 7, 1918–1928 (2012).
Foss, M. H. et al. Inhibitors of bacterial tubulin target bacterial membranes in vivo. Med. Chem. Comm 4, 112–119 (2013).
Wang, J. et al. Discovery of a small molecule that inhibits cell division by blocking FtsZ, a novel therapeutic target of antibiotics. J. Biol. Chem. 278, 44424–44428 (2003).
Okoyea, F. B. C., Nworuc, C. S., Debbaba, A., Esimone, C. O. & Proksch, P. Two new cytochalasins from an endophytic fungus, KL-1.1 isolated from Psidium guajava leaves. Phytochem. Lett. 14, 51–55 (2015).
Wang, S. et al. Chaetopyranin, a benzaldehyde derivative, and other related metabolites from Chaetomium globosum, an endophytic fungus derived from the marine red alga Polysiphonia urceolata. J. Nat. Prod. 69, 1622–1625 (2006).
Acknowledgements
The financial support by the German Federal Ministry of Education and Support (BMBF; Bundesministerium für Bildung und Forschung) granted to PP and by the Ministry of Science and Technology (MOST) to WHL are gratefully acknowledged. YL wishes to thank the China Scholarship Council and the Ministry of Education of China for awarding her with a scholarship. The authors are indebted to Prof. MS Abdel-Aziz (National Research Center, Egypt) for his help during the collection of sediment from the hypersaline lake El Hamra in Egypt and to Prof. H Brötz-Oesterhelt (University of Tuebingen, Germany) for performing the antibacterial assays.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, Y., Kurtán, T., Yun Wang, C. et al. Cladosporinone, a new viriditoxin derivative from the hypersaline lake derived fungus Cladosporium cladosporioides. J Antibiot 69, 702–706 (2016). https://doi.org/10.1038/ja.2016.11
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ja.2016.11
This article is cited by
-
Subcellular localization of fungal specialized metabolites
Fungal Biology and Biotechnology (2022)
-
The mycotoxin viriditoxin induces leukemia- and lymphoma-specific apoptosis by targeting mitochondrial metabolism
Cell Death & Disease (2022)