Abstract
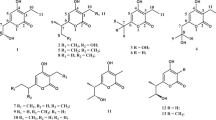
Density functional theory calculations of 1H and 13C chemical shifts are used to assign the relative configurations of isohirsut-1-ene, isohirsut-4-ene and tsukubadiene, three terpenes previously isolated from an engineered Streptomyces.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Tantillo, D. J. Biosynthesis via carbocations: theoretical studies on terpene formation (review). Nat. Prod. Rep. 28, 1035–1053 (2011).
Cane, D. E. Enzymatic formation of sesquiterpenes. Chem. Rev. 90, 1089–1103 (1990).
Yamada, Y., Cane, D. E. & Ikeda, H. Diversity and analysis of bacterial terpene synthases (review). Methods Enzymol. 515, 123–162 (2012).
Hong, Y. J. & Tantillo, D. J. How cyclobutanes are assembled in nature – insights from quantum chemistry (review). Chem. Soc. Rev. 43, 5042–5050 (2014).
Zerbe, P. & Bohlmann, J. Enzymes for synthetic biology of ambroxide-related diterpenoid fragrance compounds (review). Adv. Biochem. Eng. Biotechnol. 148, 427–447 (2015).
Chen, F., Tholl, D., Bohlmann, J. & Pichersky, E. The family of terpene synthases in plants: a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom (review). Plant J. 66, 212–229 (2011).
Cane, D. E. Isoprenoids including carotenoids and steroids. Compr. Nat. Prod. Chem. 2, 155–200 (1999).
Blunt, J. W., Copp, B. R., Keyzers, R. A., Munro, M. H. G. & Prinsep, M. R. Marine natural products (review). Nat. Prod. Rep. 33, 382–431 (2016).
Christianson, D. W. Unearthing the roots of terpenome. Curr. Opin. Chem. Biol. 12, 141–150 (2008).
Davis, E. M. & Croteau, R. Cyclization enzymes in the biosynthesis of monoterpenes, sesquiterpenes, and diterpenes. Top. Cur. Chem. 209, 53–95 (2000).
Yamada, Y. et al. Novel terepenes generated by heterologous expression of bacterial terpene synthase genes in an engineered Streptomyces host. J. Antibiot. 68, 385–394 (2015).
Nguyen, Q. N. N. & Tantillo, D. J. The many roles of quantum chemical predictions in synthetic organic chemistry (a focus review). Chem. Asian J. 9, 674–680 (2014).
Lodewyk, M. W., Siebert, M. R. & Tantillo, D. J. Computational prediction of 1H and 13C chemical shifts: a useful tool for natural product, mechanistic and synthetic organic chemistry. Chem. Rev. 112, 1839–1862 (2012).
Willoughby, P. H., Jansma, M. J. & Hoye, T. R. A Guide to Small-Molecule Structure Assignment through Computation of (1H and 13C) NMR Chemical Shifts. Nat. Protoc. 9, 643–660 (2014).
Vaughan, M. M. et al. Formation of the unusual semivolatile diterpene rhizathalene by the Arabidopsis Class I terpene synthase TPS08 in the root stele is involved in defense against belowground herbivory. Plant Cell 25, 1108–1125 (2013).
Hong, Y. J. & Tantillo, D. J. How many secondary carbocations are involved in the biosynthesis of avermitilol? Org. Lett. 13, 1294–1297 (2011).
Nguyen, Q. N. N. & Tantillo, D. J. Caryolene-forming carbocation rearrangements. Beilstein J. Org. Chem. 9, 323–331 (2013).
Lodewyk, M. W. et al. The correct structure of aquatolide – experimental validation of a theoretically-predicted structural revision. J. Am. Chem. Soc. 134, 18550–18553 (2012).
Quasdorf, K. W., Huters, A. D., Lodewyk, M. W., Tantillo, D. J. & Garg, N. K. Total synthesis of oxidized Welwitindolinones and (-)-N-methylwelwitindolinone C isonitrile. J. Am, Chem. Soc. 134, 1396–1399 (2012).
Stephens, P. J., McCann, D. M., Devlin, F. J. & Smith, A. B. Determination of the absolute configurations of natural products via density functional theory calculations of optical rotation, electronic circular dichroism, and vibrational circular dichroism: the cytotoxic suberosanone, and suberosenol A acetate. J. Nat. Prod. 69, 1055–1064 (2006).
Gaussian 09, Revision D. 01 (Gaussian, Inc.: Wallingford, CT, USA, 2004).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Adamo, C. & Barone, V. Exchange functionals with improved long-range behavior and adiabatic connection methods without adjustable parameters: the mPW and mPW1PW models. J. Chem. Phys. 108, 664–675 (1998).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Jain, R., Bally, T. & Rablen, P. R. Calculating accurate proton chemical shifts of organic molecules with density functional methods and modest basis sets. J. Org. Chem. 74, 4017–4023 (2009).
CHESHIRE. CHEmical SHift REpository with Coupling Constants Added Too. Available at http://cheshirenmr.info. Accessed on 23 March 2016.
Smith, S. G. & Goodman, J. M. Assigning stereochemistry to single diastereoisomers by GIAO NMR calculation: the DP4 probability. J. Am. Chem. Soc. 132, 12946–12959 (2010).
Spartan’10 (Wavefunction, Ind.: Irvine, CA, USA (2010).
Acknowledgements
We gratefully acknowledge support from the US National Science Foundation (CHE-1361807 and CHE030089 (XSEDE program)). This work is dedicated to David Cane, an inspiration to mechanistically inclined natural products chemists and natural product-loving mechanistic chemists alike!
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Nguyen, Q., Tantillo, D. Using quantum chemical computations of NMR chemical shifts to assign relative configurations of terpenes from an engineered Streptomyces host. J Antibiot 69, 534–540 (2016). https://doi.org/10.1038/ja.2016.51
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ja.2016.51