Abstract
Aneurysmal bone cyst (ABC) is a benign osteolytic bone lesion in which there are blood-filled spaces separated by fibrous septa containing giant cells. The nature of the giant cells in this lesion and the mechanism of bone destruction in ABC is not certain. In this study, we have analysed several characteristics of mononuclear and multinucleated cells in the ABC and examined the cellular and molecular mechanisms of ABC osteolysis. The antigenic and functional phenotype of giant cells in ABC was determined by histochemistry/immunohistochemistry using antibodies to macrophage and osteoclast markers. Giant cells and CD14+ and CD14− mononuclear cells were isolated from ABC specimens and cultured on dentine slices and coverslips with receptor activator of nuclear factor κB ligand (RANKL)+/− macrophage-colony stimulating factor (M-CSF) and functional and cytochemical evidence of osteoclast differentiation sought. Giant cells in ABC expressed an osteoclast-like phenotype (CD51+, CD14−, cathepsin K+, TRAP+) and were capable of lacunar resorption, which was inhibited by zoledronate, calcitonin and osteoprotegerin (OPG). When cultured with RANKL±M-CSF, CD14+, but not CD14−, mononuclear cells differentiated into TRAP+ multinucleated cells that were capable of lacunar resorption. M-CSF was not necessary for osteoclast formation from CD14+ cell cultures. CD14− cells variably expressed RANKL, OPG and M-CSF but supported osteoclast differentiation. Our findings show that the giant cells in ABC express an osteoclast-like phenotype and are formed from CD14+ macrophage precursors. CD14− mononuclear stromal cells express osteoclastogenic factors and most likely interact with CD14+ cells to form osteoclast-like giant cells by a RANKL-dependent mechanism.
Similar content being viewed by others
Main
Benign and malignant tumours that contain numerous osteoclast-like giant cells frequently arise in bone and are categorised on the basis of tissue location and histomorphology.1, 2 The mechanism whereby giant cells accumulate and contribute to the osteolysis that accompanies growth of these tumours in bone is not certain. Primary aneurysmal bone cyst (ABC) is a benign cystic giant cell-rich tumour that arises most commonly in the metaphyseal region of a long bone;1, 2 the cysts are filled with blood and the fibrous cyst wall contains fibroblast-like stromal cells, scattered macrophages and osteoclast-like giant cells. The lesion most commonly arises in the first two decades of life and is a clonal proliferation associated with a characteristic rearrangement of the short arm of chromosome 17.3, 4, 5
Osteoclasts are formed from marrow-derived circulating precursors, which express a monocyte/macrophage phenotype.6, 7 These mononuclear phagocyte precursors express the receptor activator of nuclear factor κB (RANK) and, in the presence of macrophage-colony stimulating factor (M-CSF) and the ligand for RANK (RANKL), differentiate into multinucleated cells that express the specific cytochemical and functional phenotype of mature bone-resorbing osteoclasts.8, 9, 10 It has been shown that in giant cell tumour of bone (GCTB), a distinct clinicopathological entity that most commonly arises in the epiphysis of a long bone, the giant cells express the phenotypic and functional features of mature osteoclasts.11 Whether ABC and other giant cell-rich lesions of bone that arise in other anatomical locations also contain osteoclast-like giant cells has not been determined.
In this study, our aim has been to determine the nature of mononuclear and multinucleated giant cells in ABC. We have determined the phenotypic characteristics of giant cells in ABC and, in cell culture studies, sought to identify the mononuclear precursor population that gives rise to them. We have also determined the cellular and molecular mechanisms required for giant cell formation in ABC and evaluated whether pharmacological inhibitors of osteoclast formation and bone resorption could have a role in the ABC treatment.
MATERIALS AND METHODS
Alpha minimum essential medium (αMEM) and foetal bovine serum (FBS) were purchased from Gibco Laboratories (Paisley, Scotland); αMEM containing 10% FBS, 100 U/ml penicillin and 100 μg/ml streptomycin (MEM/FBS) was used for cell culture experiments. M-CSF and osteoprotegerin (OPG) were purchased from R&D Cytokines (Abingdon, UK). Collagenase and the diagnostic tartrate-resistant acid phosphatase (TRAP) kit were from Sigma Diagnostics (St Louis, MO, USA). All reagents used in reverse transcription and DNA amplification were from Invitrogen, Life Technologies (Paisley, UK). Zoledronate was obtained from Novartis (Basel, Switzerland). This study was approved by the Oxford Clinical Research Ethics Committee.
A total of 20 cases of ABC (formalin-fixed paraffin-embedded tissue) were retrieved from the files of the Nuffield Orthopaedic Centre (NOC) Histopathology Department, Oxford. All cases presented as a benign osteolytic lesion with characteristic radiological and morphological features of ABC. Clinical features of the cases analysed is shown in Supplementary Table S1 (Supplementary material).
Immunohistochemistry
Antigens were detected by incubation with labelled polymer and diaminobenzidine. The sections were then counterstained with haematoxylin, dehydrated, cleared and mounted. Immunohistochemical staining of 5 μm sections was carried out by an indirect immunoperoxidase technique. (ChemMate Envision, Dako, UK). Tissue sections were dewaxed and rehydrated by successive immersion in xylene, graded ethanols and water. Antigen retrieval was performed by microwave treatment (700 W, 2–4 min) in Target Retrieval Solution (Dako, UK). Endogenous peroxidase was blocked with 0.2% v/v hydrogen peroxide in 80% ethanol and protein block serum prior to 30 min incubation with the mouse monoclonal antibodies PD7/26, KP1 (Dakopatts, UK), NCL-CD51 and NCL-L-CD14-223 (Novocastra, UK) and 182–12G5 (Daiichi Chemical Company Limited, Toyama, Japan). These antibodies are directed against CD68, CD51 (vitronectin receptor), CD14 and cathepsin K, respectively.
Isolation and Culture of Giant Cells from ABC
Fresh tissue from five cases of ABC, all of which showed radiological and morphological evidence of osteolysis, was obtained at the time of surgery at the NOC, Oxford. ABC tumour tissue was washed in sterile phosphate-buffered saline (PBS). Fragments of the tumour were then placed in collagenase (diluted 1 mg/ml in αMEM) and incubated at 37°C for up to 45 min. The digested tissue suspension was then passed through a 70-μm pore size cell strainer (Falcon, Becton Dickinson, Oxford, UK). The number of giant cells was counted in a haematocytometer and the volume adjusted to obtain approximately 30 giant cells. The cell suspension was then added to dentine slices, prepared as previously described,11 or glass coverslips in a 96-well plate. After 3 h incubation at 37°C in a humidified atmosphere of 5% CO2 and 95% air, the dentine slices and glass coverslips were washed in MEM/FBS to remove any non-adherent cells. The slices were then transferred into 24 well plates containing fresh MEM/FBS and incubated in the presence and absence of the resorption inhibitors calcitonin (10 ng/ml), OPG (500 ng/ml) and zoledronate (10−8 M/ml). Cultures were maintained for 24 h and 72 h at 37°C. Cultures on glass coverslips were fixed in acetone and stained histochemically for the expression of the osteoclast marker TRAP using a commercially available kit (Sigma Diagnostics) and counterstained with haematoxylin.12 Cultures on dentine slices were treated with 1 M ammonium hydroxide, washed in distilled water and ultrasonicated to remove adherent cells; these slices were then stained with 0.5% (w/v) toluidine blue to reveal areas of lacunar resorption and examined by light microscopy. The number of resorption pits was counted and the percentage surface area of lacunar resorption on each dentine slice measured using image analysis software (Adobe Photoshop, USA) as previously described.11
Isolation of Giant Cells and CD14+ and CD14− Mononuclear Cells from ABC Tissue
Isolation of specific mononuclear cell populations was carried out following collagenase digestion of ABC tissue. The filtrate was centrifuged at 1300 rpm for 10 min and the cells then resuspended in PBS, 2 mM EDTA, 0.5% BSA and incubated with MACS, CD14 microbeads (Miltenyi Biotec, UK) for 15 min at 4°C before being passed through a Minimax magnetic cell separator. ABC-derived mononuclear cells were sorted into CD14+ and CD14− cell populations and the respective populations added (1 × 105 cells/well) to 6 mm glass coverslips and dentine slices in a 96-well tissue culture plate. After 3 h incubation, the dentine slices and glass coverslips were washed in MEM/FBS to remove any non-adherent cells and then transferred to 24-well tissue culture plates.
Culture of CD14– Mononuclear Stromal Cells from ABC
CD14− cells isolated from ABC tissue as detailed above were washed in PBS. Then cultured in tissue culture flasks, covered with a small amount of αMEM/FBS and incubated at 37°C in a humidified atmosphere containing 5% CO2. The medium was changed after 24 h and then at 5–7 day intervals. Outgrowth cultures of cells from these fragments were continuously observed and the morphology of the cells noted. Cultures were passaged after 2 weeks, re-grown and then passaged after a further 2–4 weeks in culture. Human monocytes isolated from peripheral blood were co-cultured on coverslips with the above CD14− cells, as previously described,11 and evidence of osteoclast differentiation by TRAP staining was determined.
PCR Analysis of CD14− Mononuclear Cells Derived from ABC
To determine whether CD14− mononuclear cells expressed the mRNA for osteoclastogenesis-associated factors, RT-PCR was carried out on three cases of ABC to identify mRNA transcripts for RANKL, M-CSF and OPG. GCTB stromal cells, which supported osteoclastogenesis in our previous study,11 were employed as positive control. RNA was extracted from cultured cells in TRI Reagent (Sigma) and treated with DNAase I (Invitrogen). RNA was tested for quality and concentration using a NanoDrop ND1000 spectrophotometer. Single-strand cDNA was synthesised using the SuperScript VILO cDNA Synthesis Kit (Invitrogen) and oligo dT primers. RT-PCR was performed with the Rotor-Gene 3000 (Corbett Research) and the Express SYBR GreenER Kit (qPCR Supermix Universal) (Invitrogen). The PCR cycling conditions were as follows: 50°C 2 min, 95°C 2 min, 95°C 15 s, and 60°C 1 min for 40 cycles. QuantiTect real-time primer sets were designed by Qiagen. The catalogue numbers were as follows; RANKL (QT00215614), M-CSF (QT00035224), OPG (QT00014294) and β-actin (QT00095431). β-actin mRNA expression served as an internal control. The experiments were repeated at least two times. All the samples were amplified in triplicate. One-way analysis of variance (ANOVA) test followed by Dunnet post hoc test was performed in order to compare the data between control GCTB cells and CD14− ABC cells using Graphpad Prism. (Graphpad Software, La Jolla, CA, USA).
RESULTS
Morphological and Cytochemical Features of Mononuclear and Multinucleated Cells of ABC
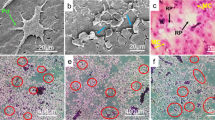
Haematoxylin and eosin-stained sections of 20 ABC cases showed the typical features of a cystic bone lesion in which there were blood-filled spaces separated by fibrous septa containing numerous macrophages and scattered osteoclast-like multinucleated giant cells. The giant cells showed no marked cytological atypia or mitotic activity. Reactive bone formation was seen in the fibrous septa of some tumours. Immunohistochemical staining of paraffin sections of ABC showed that the giant cells expressed an osteoclast phenotype being CD14−, CD51+, CD68+ and cathepsin K+ (Figure 1).13, 14 A few scattered round or ovoid macrophage-like cells in the mononuclear component had a similar antigenic phenotype. There were also spindle-shaped fibroblast-like stromal cells that were negative for the above macrophage/ osteoclast markers in the fibrous septa.
Phenotypic Characteristics of Cultured Multinucleated Giant Cells Isolated from ABC
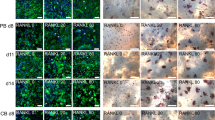
The multinucleated giant cells isolated from ABC were large (TRAP+) cells, which had abundant cytoplasm with broad pseudopodia. Lacunar resorption is the functional criterion whereby a multinucleated cell is defined as an osteoclast,6, 15 and in 24 h cultures of unsorted cells derived from ABC, it was noted that isolated giant cells formed numerous lacunar resorption pits, both in the presence and absence of RANKL and M-CSF. (Figure 2) This lacunar resorption was inhibited by the addition of calcitonin, OPG and zoledronate. (Figure 3) The addition of zoledronate at 10−8 mol/ml, was sufficient to abolish osteoclast resorption in vitro on some dentine slices.
Osteoclast-Like Giant Cells in ABC are Formed from CD14+ Mononuclear Cells
In all 24 h cultures of selected CD14+ and CD14− ABC-derived mononuclear cells on dentine slices, it was noted that there was no evidence of lacunar resorption. After incubation for 21 days in the presence of M-CSF and RANKL or RANKL alone, CD14+ cell cultures on glass coverslips contained TRAP+ and CD51+ multinucleated cells. Parallel cultures on dentine slices showed functional evidence of osteoclast differentiation with the formation of numerous single and compound lacunar resorption pits (Figure 4). The mean number of pits formed on the dentine slices in the presence of RANKL and M-CSF was 46. M-CSF was not required for the formation of osteoclast-like giant cells capable of lacunar resorption. Resorption pits were not formed when RANKL was omitted. Formation of TRAP+/CD51+ multinucleated cells and resorption pits were not seen in 21 day CD14− cell cultures (± added RANKL and M-CSF).
ABC-Derived CD14− Cells Express RANKL, OPG and M-CSF
RT-PCR studies showed that cultured CD14− cells derived from three cases of ABC expressed mRNA for the osteoclastogenesis-related factors RANKL, M-CSF and OPG. In ABC case no. 2, RANKL expression was more than 2.5-folds higher than in control GCTB stromal cells with M-CSF expression approximately the same as the control (Figure 3). In ABC cases no. 1 and no. 3, M-CSF expression was less than half that of control GCTB stromal cells and RANKL expression was approximately the same as the control. Expression of OPG was upregulated (2.5–22-fold) in CD14− cells derived from all three ABC cases (Figure 5).
DISCUSSION
In this study, we have shown that the giant cells in ABC express the phenotypic features of osteoclasts and that these cells form from the CD14+ macrophages present within the lesion. CD14− mononuclear cells in ABC expressed the osteoclastogenic factors RANKL and M-CSF, and were capable of supporting the formation of TRAP+ multinucleated cells from mononuclear phagocyte osteoclast precursors found in the monocyte fraction of peripheral blood mononuclear cells. These findings suggest that CD14− mononuclear cells in ABC have a crucial role in inducing the RANKL-dependent formation of osteoclast-like giant cells and that these giant cells are responsible for the bone destruction seen in this condition.
Giant cells in ABC exhibited both the antigenic and functional phenotype of osteoclasts, expressing CD51, CD68 and cathepsin K but not CD14.13, 14 In short-term (24 h) cultures of unsorted cells from ABC on dentine slices, these giant cells were capable of lacunar resorption, confirming the osteoclastic nature of these cells.15 To determine the mechanism whereby these osteoclastic giant cells form in ABC, we isolated CD14+ and CD14− cell populations from ABC tissue and cultured them in the presence of the osteoclastogenic factors RANKL and M-CSF. Under these conditions, only CD14+/TRAP−/CD51− mononuclear cells differentiated into CD14−/TRAP+/CD51+ multinucleated cells that were capable of resorption. CD14+ osteoclast precursors have been shown to lose and acquire macrophage and osteoclast markers respectively in the process of osteoclast differentiation.14, 16 Our findings indicate that the formation of giant cells in ABC is likely to occur by a similar step-wise process from CD14+ mononuclear phagocyte precursors.
ABC is one of a number of giant cell-rich lesions of bone associated with excessive bone resorption. Our RT-PCR findings indicate that CD14− stromal cells in ABC express the osteoclastogenic factors RANKL and M-CSF.11 CD14− stromal cells in GCTB are known to express RANKL and our findings suggest that, as in GCTB, formation of osteoclast-like giant cells in ABC occurs by a RANKL-dependent mechanism whereby CD14+ mononuclear phagocyte osteoclast precursors interact with RANKL-expressing CD14− mononuclear stromal cells.11 Osteoid and bone formation by mononuclear stromal cells is not uncommonly seen in the fibrous cyst wall of an ABC and it is likely that, like normal osteoblasts, these bone-forming cells express RANKL. It should be noted that mononuclear stromal cells have been shown to express RANKL in a number of other giant cell-rich lesions of bone and soft tissue including giant cell tumour of soft parts, giant cell granuloma of the jaw and chondroblastoma.11, 17, 18, 19 Variable expression of RANKL and other growth factors influencing osteoclastogenesis may account for differences in the number of osteoclast-like giant cells as well as other morphological features that can be seen in ABC.
M-CSF is also an essential osteoclastogenic factor and mRNA for M-CSF was also expressed by CD14− mononuclear stromal cells in ABC. M-CSF is required for macrophage formation and survival; it has a role in osteoclast formation and is produced by osteoblasts.20, 21 M-CSF is known to be produced by tumour cells and stromal cells in several tumours, as well as by activated macrophages, and it is notable that ABC-derived mononuclear stromal cells did not require exogenous M-CSF for osteoclast differentiation. CD14− mononuclear cells also induced the formation of TRAP+ multinucleated cells from CD14+ monocytes in the absence of M-CSF, suggesting that these cells produce this osteoclastogenic factor.
The origin and nature of ABC is not certain but rearrangement of the short arm of chromosome 17 has been reported, most commonly as a balanced translocation involving the long arm of chromosome 16.3, 4 Overexpression of TRE17/USP6 in the stromal cells of ABC is thought to have a key role in the pathogenesis of ABC; promoters of TRE17/USP6 fusion partners, such as cadherin-11, are active during osteoblast differentiation and bone formation. The chimaeric TRE17/USP6 gene product in ABC is also known to induce the expression of matrix metalloproteinases 9 and 10 via the RhoA-ROCK signalling pathway, leading to activation of NF-kappa B.22 Rho-ROCK signalling pathway is a critical regulator of bone remodelling,21, 23 and it is tempting to speculate that overexpression of TRE17/USP6 related to the expression of osteoclastogenic factors noted in ABC.
Our findings show that the giant cells in ABC are osteoclast-like and that these cells form by a RANKL-dependent mechanism. It was therefore not surprising to find that resorption inhibitors reduced lacunar resorption by ABC giant cells. Zoledronate, an aminobisphosphonate that inhibits osteoclast resorption activity and survival,24 markedly reduced lacunar resorption by giant cells isolated from ABC. OPG binds to RANKL; it competitively inhibits the RANKL–RANK interaction that leads to osteoclast formation and reduces osteoclastic resorption activity.25 Our findings suggest that pharmacological inhibitors of osteoclast formation and activity may be useful in the treatment of ABC. It is possible that bisphosphonates and the anti-RANKL antibody denosumab, both of which have been used to treat GCTB,26, 27 may also be useful in inhibiting osteoclast formation and osteolysis in ABC.
References
Rosenberg AE, GP N, Fletcher J . Aneurysmal bone cyst. In: Fletcher C, Unni K, Mertens F (eds) Pathology and Genetics of Tumours of Soft Tissue and Bone. IARC: Lyon, 2002, pp 338–339.
Unni KK, Inwards YI, Bridge AN, et al. Tumors of the bones and joints. In: Silverberg SG (ed.) AFIP Atlas of Tumor Pathology, 4th edn. AFIP: Washington DC, 2005, pp 281–298.
Oliveira AM, Hsi BL, Weremowicz S, et al. USP6 (Tre2) fusion oncogenes in aneurysmal bone cyst. Cancer Res 2004;64:1920–1923.
Oliveira AM, Perez-Atayde AR, Inwards CY, et al. USP6 and CDH11 oncogenes identify the neoplastic cell in primary aneurysmal bone cysts and are absent in so-called secondary aneurysmal bone cysts. Am J Pathol 2004;165:1773–1780.
Panoutsakopoulos G, Pandis N, Kyriazoglou I, et al. Recurrent t(16;17)(q22;p13) in aneurysmal bone cysts. Genes Chromosomes Cancer 1999;26:265–266.
Athanasou NA . Cellular biology of bone-resorbing cells. J Bone Joint Surg Am 1996;78:1096–1112.
Fujikawa Y, Quinn JM, Sabokbar A, et al. The human osteoclast precursor circulates in the monocyte fraction. Endocrinology 1996;137:4058–4060.
Quinn JM, Elliott J, Gillespie MT, et al. A combination of osteoclast differentiation factor and macrophage-colony stimulating factor is sufficient for both human and mouse osteoclast formation in vitro. Endocrinology 1998;139:4424–4427.
Tanaka S, Takahashi N, Udagawa N, et al. Macrophage colony-stimulating factor is indispensable for both proliferation and differentiation of osteoclast progenitors. J Clin Invest 1993;91:257–263.
Yasuda H, Shima N, Nakagawa N, et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 1998;95:3597–3602.
Lau YS, Sabokbar A, Gibbons CL, et al. Phenotypic and molecular studies of giant-cell tumors of bone and soft tissue. Hum Pathol 2005;36:945–954.
Minkin C . Bone acid phosphatase: tartrate-resistant acid phosphatase as a marker of osteoclast function. Calcif Tissue Int 1982;34:285–290.
Athanasou NA, Quinn J . Immunophenotypic differences between osteoclasts and macrophage polykaryons: immunohistological distinction and implications for osteoclast ontogeny and function. J Clin Pathol 1990;43:997–1003.
Horton MA, Lewis D, McNulty K, et al. Monoclonal antibodies to osteoclastomas (giant cell bone tumors): definition of osteoclast-specific cellular antigens. Cancer Res 1985;45 (Pt 2):5663–5669.
Chambers TJ, Horton MA . Failure of cells of the mononuclear phagocyte series to resorb bone. Calcif Tissue Int 1984;36:556–558.
Faust J, Lacey DL, Hunt P, et al. Osteoclast markers accumulate on cells developing from human peripheral blood mononuclear precursors. J Cell Biochem 1999;72:67–80.
Huang L, Cheng YY, Chow LT, et al. Receptor activator of NF-kappaB ligand (RANKL) is expressed in chondroblastoma: possible involvement in osteoclastic giant cell recruitment. Mol Pathol 2003;56:116–120.
Itonaga I, Hussein I, Kudo O, et al. Cellular mechanisms of osteoclast formation and lacunar resorption in giant cell granuloma of the jaw. J Oral Pathol Med 2003;32:224–231.
Won KY, Kalil RK, Kim YW, et al. RANK signalling in bone lesions with osteoclast-like giant cells. Pathology 2011;43:318–321.
Motoyoshi K . Biological activities and clinical application of M-CSF. Int J Hematol 1998;67:109–122.
Tsurukai T, Udagawa N, Matsuzaki K, et al. Roles of macrophage-colony stimulating factor and osteoclast differentiation factor in osteoclastogenesis. J Bone Miner Metab 2000;18:177–184.
Ye Y, Pringle LM, Lau AW, et al. TRE17/USP6 oncogene translocated in aneurysmal bone cyst induces matrix metalloproteinase production via activation of NF-kappaB. Oncogene 2010;29:3619–3629.
Harmey D, Stenbeck G, Nobes CD, et al. Regulation of osteoblast differentiation by Pasteurella multocida toxin (PMT): a role for Rho GTPase in bone formation. J Bone Miner Res 2004;19:661–670.
Reid IR . Bisphosphonates. Skeletal Radiol 2007;36:711–714.
Hofbauer LC, Khosla S, Dunstan CR, et al. The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption. J Bone Miner Res 2000;15:2–12.
Balke M, Campanacci L, Gebert C, et al. Bisphosphonate treatment of aggressive primary, recurrent and metastatic giant cell tumour of bone. BMC Cancer 2010;10:462.
Thomas D, Henshaw R, Skubitz K, et al. Denosumab in patients with giant-cell tumour of bone: an open-label, phase 2 study. Lancet Oncol 2010;11:275–280.
Acknowledgements
We would like to thank Chris Lowe for typing the manuscript. This study was supported by the Oxford NIHR, BRU, and was undertaken as part of EuroBoNet, a EU-funded Network of Excellence.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Author contributions: NAA conceived the study; RMIT, TGK, FH, AD and HJK carried out experiments and analysed samples. All authors were involved in writing the paper and had final approval of the submitted version.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
Bone destruction in aneurysmal bone cysts (ABC) is due to giant cells, which express an osteoclast-like phenotype and are formed from RANKL-expressing CD14+ macrophage precursors by interaction with CD14− mononuclear stromal cells. The molecular mechanisms of ABC osteolysis suggest that pharmacological inhibitors of osteoclasts are a potential treatment for ABC.
Supplementary information
Rights and permissions
About this article
Cite this article
Taylor, R., Kashima, T., Hemingway, F. et al. CD14− mononuclear stromal cells support (CD14+) monocyte–osteoclast differentiation in aneurysmal bone cyst. Lab Invest 92, 600–605 (2012). https://doi.org/10.1038/labinvest.2012.5
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/labinvest.2012.5
Keywords
This article is cited by
-
Unusual Multinucleated Giant Cell Reaction in a Tongue Squamous Cell Carcinoma: Histopathological and Immunohistochemical Features
Head and Neck Pathology (2018)
-
Giant cell tumour of the distal radius/ulna: response to pre-operative treatment with short-term denosumab
Clinical Sarcoma Research (2017)
-
Distinct H3F3A and H3F3B driver mutations define chondroblastoma and giant cell tumor of bone
Nature Genetics (2013)