Abstract
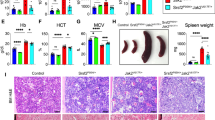
The JAK2V617F mutation commonly found in myeloproliferative neoplasms (MPNs) induces constitutive phosphorylation/activation of the signal transducer and activator of transcription 3 (Stat3). However, the contribution of Stat3 in MPN evoked by JAK2V617F remains unknown. To determine the role of Stat3 in JAK2V617F-induced MPN, we generated Stat3-deficient Jak2V617F-expressing mice. Whereas expression of Jak2V617F resulted in a polycythemia vera-like disease characterized by increased red blood cells (RBCs), hematocrit, neutrophils and platelets in the peripheral blood of Jak2V617F knock-in mice, deletion of Stat3 slightly reduced RBC and hematocrit parameters and modestly increased platelet numbers in Jak2V617F knock-in mice. Moreover, deletion of Stat3 significantly increased the neutrophil counts/percentages and markedly reduced the survival of mice expressing Jak2V617F. These phenotypic manifestations were reproduced upon bone marrow (BM) transplantation into wild-type animals. Flow cytometric analysis showed increased hematopoietic stem cell and granulocyte-macrophage progenitor populations in the BM and spleens of Stat3-deficient Jak2V617F mice. Stat3 deficiency also caused a marked expansion of Gr-1+/Mac-1+ myeloid cells in Jak2V617F knock-in mice. Histopathologic analysis revealed marked increase in granulocytes in the BM, spleens and livers of Stat3-deficient Jak2V617F-expressing mice. Together, these results suggest that deletion of Stat3 increases the severity of MPN induced by Jak2V617F.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
James C, Ugo V, Le Couédic JP, Staerk J, Delhommeau F, Lacout C et al. A unique clonal JAK2 mutation leading to constitutive signaling causes polycythemia vera. Nature 2005; 434: 1144–1148.
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJP et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005; 7: 387–397.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005; 365: 1054–1061.
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Eng J Med 2005; 352: 1779–1790.
Zhao R, Xing S, Li Z, Fu X, Li Q, Krantz SB et al. Identification of an acquired JAK2 mutation in polycythemia vera. J Biol Chem 2005; 280: 22788–22792.
Akada H, Yan D, Zou H, Fiering S, Hutchison RE, Mohi MG . Conditional expression of heterozygous or homozygous Jak2V617F from its endogenous promoter induces a polycythemia vera-like disease. Blood 2010; 115: 3589–3597.
Wernig G, Mercher T, Okabe R, Levine RL, Lee BH, Gilliland DG . Expression of V617F causes a polycythemia vera-like disease with associated myelofibrosis in a murine bone marrow transplant model. Blood 2006; 107: 4274–4281.
Tiedt R, Hao-Shen H, Sobas MA, Looser R, Dirnhofer S, Schwaller J et al. Ratio of mutant JAK2-V617F to wild-type Jak2 determines the MPD phenotypes in transgenic mice. Blood 2008; 111: 3931–3940.
Shide K, Shimoda HK, Kumano T, Karube K, Kameda T, Takenaka K et al. Development of ET, primary myelofibrosis and PV in mice expressing JAK2 V617F. Leukemia 2008; 22: 87–95.
Xing S, Wanting TH, Zhao W, Ma J, Wang S, Xu X et al. Transgenic expression of JAK2V617F causes myeloproliferative disorders in mice. Blood 2008; 111: 5109–5117.
Marty C, Lacout C, Martin A, Hasan S, Jacquot S, Birling MC et al. Myeloproliferative neoplasm induced by constitutive expression of JAK2V617F in knock-in mice. Blood 2010; 116: 783–787.
Mullally A, Lane SW, Ball B, Megerdichian C, Okabe R, Al-Shahrour F et al. Physiological Jak2V617F expression causes a lethal myeloproliferative neoplasm with differential effects on hematopoietic stem and progenitor cells. Cancer Cell 2010; 17: 584–596.
Li J, Spensberger D, Ahn JS, Anand S, Beer PA, Ghevaert C et al. JAK2 V617F impairs hematopoietic stem cell function in a conditional knock-in mouse model of JAK2 V617F-positive essential thrombocythemia. Blood 2010; 116: 1528–1538.
Chen E, Beer PA, Godfrey AL, Ortmann CA, Li J, Costa-Pereira AP et al. Distinct clinical phenotypes associated with JAK2V617F reflect differential STAT1 signaling. Cancer Cell 2010; 18: 524–535.
Anand S, Stedham F, Gudgin E, Campbell P, Beer P, Green AR et al. Increased basal intracellular signaling patterns do not correlate with JAK2 genotype in human myeloproliferative neoplasms. Blood 2011; 118: 1610–1621.
Yan D, Hutchison RE, Mohi G . Critical requirement for Stat5 in a mouse model of polycythemia vera. Blood 2012; 119: 3539–3549.
Walz C, Ahmed W, Lazarides K, Betancur M, Patel N, Hennighausen L et al. Essential role for Stat5a/b in myeloproliferative neoplasms induced by BCR-ABL1 and JAK2(V617F) in mice. Blood 2012; 119: 3550–3560.
Duek A, Lundberg P, Shimizu T, Grisouard J, Karow A, Kubovcakova L et al. Loss of Stat1 decreases megakaryopoiesis and favors erythropoiesis in a JAK2-V617F-driven mouse model of MPNs. Blood 2014; 123: 3943–3950.
Baker SJ, Rane SG, Reddy EP . Hematopoietic cytokine receptor signaling. Oncogene 2007; 26: 6724–6737.
Lee CK, Raz R, Gimeno R, Gertner R, Wistinghausen B, Takeshita K et al. STAT3 is a negative regulator of granulopoiesis but is not required for G-CSF-dependent differentiation. Immunity 2002; 17: 63–72.
Chou WC, Levy D, Lee CK . STAT3 positively regulates an early step in B-cell development. Blood 2006; 108: 3005–3011.
Mantel C, Messina-Graham S, Moh A, Cooper S, Hangoc G, Fu XY et al. Mouse hematopoietic cell-targeted STAT3 deletion: stem/progenitor cell defects, mitochondrial dysfunction, ROS overproduction, and a rapid aging-like phenotype. Blood 2012; 120: 2589–2599.
Yu H, Pardoll D, Jove R . STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 2009; 9: 798–809.
Corcoran RB, Contino G, Deshpande V, Tzatsos A, Conrad C, Benes CH et al. STAT3 plays a critical role in KRAS-induced pancreatic tumorigenesis. Cancer Res 2011; 71: 5020–5029.
Gough DJ, Marié IJ, Lobry C, Aifantis I, Levy DE . STAT3 supports experimental K-RasG12D-induced murine myeloproliferative neoplasms dependent on serine phosphorylation. Blood 2014; 124: 2252–2261.
Bronte-Tinkew DM, Terebiznik M, Franco A, Ang M, Ahn D, Mimuro H et al. Helicobacter pylori cytotoxin-associated gene A activates the signal transducer and activator of transcription 3 pathway in vitro and in vivo. Cancer Res 2009; 69: 632–639.
Couto JP, Daly L, Almeida A, Knauf JA, Fagin JA, Sobrinho-Simões M et al. STAT3 negatively regulates thyroid tumorigenesis. Proc Natl Acad Sci USA 2012; 109: E2361–E2370.
de la Iglesia N, Konopka G, Puram SV, Chan JA, Bachoo RM, You MJ et al. Identification of a PTEN-regulated STAT3 brain tumor suppressor pathway. Genes Dev 2008; 22: 449–462.
Alonzi T, Maritano D, Gorgoni B, Rizzuto G, Libert C, Poli V . Essential role of STAT3 in the control of the acute-phase response as revealed by inducible gene activation in the liver. Mol Cell Biol 2001; 21: 1621–1632.
Kühn R, Schwenk F, Aguet M, Rajewsky K . Inducible gene targeting in mice. Science 1995; 269: 1427–1429.
Prchal JF, Axelrad AA . Bone-marrow responses in polycythemia vera. N Engl J Med 1974; 290: 1382.
Fourouclas N, Li J, Gilby DC, Campbell PJ, Beer PA, Boyd EM et al. Methylation of the suppressor of cytokine signaling 3 gene (SOCS3) in myeloproliferative disorders. Haematologica 2008; 93: 1635–1644.
Qing Y, Stark GR . Alternative activation of STAT1 and STAT3 in response to interferon-gamma. J Biol Chem 2004; 279: 41679–41685.
Croker BA, Krebs DL, Zhang JG, Wormald S, Willson TA, Stanley EG et al. SOCS3 negatively regulates IL-6 signaling in vivo. Nat Immunol 2003; 4: 540–545.
Vainchenker W, Constantinescu SN . JAK/STAT signaling in hematological malignancies. Oncogene 2012; 32: 2601–2613.
Vainchenker W, Delhommeau F, Constantinescu SN, Bernard OA . New mutations and pathogenesis of myeloproliferative neoplasms. Blood 2011; 118: 1723–1735.
Schuringa JJ, Wierenga AT, Kruijer W, Vellenga E . Constitutive Stat3, Tyr705, and Ser727 phosphorylation in acute myeloid leukemia cells caused by the autocrine secretion of interleukin-6. Blood 2000; 95: 3765–3770.
Koskela HL, Eldfors S, Ellonen P, van Adrichem AJ, Kuusanmäki H, Andersson EI et al. Somatic STAT3 mutations in large granular lymphocytic leukemia. N Engl J Med 2012; 366: 1905–1913.
White CA, Nicola N . SOCS3: an essential physiological inhibitor of signaling by interleukin-6 and G-CSF family cytokines. JAKSTAT 2013; 2: e25045.
Hookham MB, Elliott J, Suessmuth Y, Staerk J, Ward AC, Vainchenker W et al. The myeloproliferative disorder-associated JAK2 V617F mutant escapes negative regulation by suppressor of cytokine signaling 3. Blood 2007; 109: 4924–4929.
Croker BA, Mstcalf D, Robb L, Wei W, Mifsud S, DiRago L et al. SOCS3 is a critical physiological negative regulator of G-CSF signaling and emergency granulopoiesis. Immunity 2004; 20: 153–165.
Kimura A, Kinjyo I, Matsumura Y, Mori H, Mashima R, Harada M et al. SOCS3 is a physiological negative regulator for granulopoiesis and granulocyte colony-stimulating factor receptor signaling. J Biol Chem 2004; 279: 6905–6910.
Lobry C, Oh P, Mansour MR, Look AT, Aifantis I . Notch signaling: switching an oncogene to a tumor suppressor. Blood 2014; 123: 2451–2459.
Klinakis A, Lobry C, Abdel-Wahab O, Oh P, Haeno H, Buonamici S et al. A novel tumour-suppressor function for the Notch pathway in myeloid leukaemia. Nature 2011; 473: 230–233.
Friedman AD . Transcriptional control of granulocyte and monocyte development. Oncogene 2007; 26: 6816–6828.
Zhang DE, Zang P, Wang ND, Hetherington CJ, Darlington GJ, Tenen DG . Absence of granulocyte colony-stimulating factor signaling and neutrophil development in CCAAT enhancer binding protein alpha-deficient mice. Proc Natl Acad Sci USA 1997; 94: 569–574.
Wang QF, Friedman AD . CCAAT/enhancer-binding proteins are required for granulopoiesis independent of their induction of the granulocyte colony-stimulating factor receptor. Blood 2002; 99: 2776–2785.
Acknowledgements
We thank Dr Valeria Poli (University of Turin, Italy) for providing the Stat3-floxed mouse. This work was supported by the grants from the Leukemia and Lymphoma Society and US National Institute of Health (NIH; R01 HL095685) awarded to GM. GM is a scholar of the Leukemia and Lymphoma Society.
Author Contributions
DY performed research, analyzed data and wrote the manuscript; FJ performed research; REH performed histopathologic analysis and revised the manuscript; GM designed the research, analyzed data and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Yan, D., Jobe, F., Hutchison, R. et al. Deletion of Stat3 enhances myeloid cell expansion and increases the severity of myeloproliferative neoplasms in Jak2V617F knock-in mice. Leukemia 29, 2050–2061 (2015). https://doi.org/10.1038/leu.2015.116
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/leu.2015.116
This article is cited by
-
Progress in elucidation of molecular pathophysiology of myeloproliferative neoplasms and its application to therapeutic decisions
International Journal of Hematology (2020)
-
CD300f:IL-5 cross-talk inhibits adipose tissue eosinophil homing and subsequent IL-4 production
Scientific Reports (2017)