Abstract
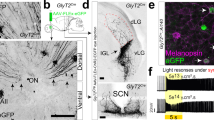
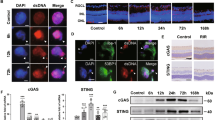
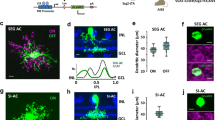
Synaptic circuits in the retina transform visual input gathered by photoreceptors into messages that retinal ganglion cells (RGCs) send to the brain. Processes of retinal interneurons (amacrine and bipolar cells) form synapses on dendrites of RGCs in the inner plexiform layer (IPL). The IPL is divided into at least 10 parallel sublaminae; subsets of interneurons and RGCs arborize and form synapses in just one or a few of them1,2,3. These lamina-specific circuits determine the visual features to which RGC subtypes respond3,4,5. Here we show that four closely related immunoglobulin superfamily (IgSF) adhesion molecules—Dscam (Down’s syndrome cell adhesion molecule), DscamL (refs 6–9), Sidekick-1 and Sidekick-2 (ref. 10)—are expressed in chick by non-overlapping subsets of interneurons and RGCs that form synapses in distinct IPL sublaminae. Moreover, each protein is concentrated within the appropriate sublaminae and each mediates homophilic adhesion. Loss- and gain-of-function studies in vivo indicate that these IgSF members participate in determining the IPL sublaminae in which synaptic partners arborize and connect. Thus, vertebrate Dscams, like Drosophila Dscams11,12,13,14,15,16,17,18,19, play roles in neural connectivity. Together, our results on Dscams and Sidekicks suggest the existence of an IgSF code for laminar specificity in retina and, by implication, in other parts of the central nervous system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ramón y Cajal, S. La rétine des vertébrés. La Cellule 9, 119–257 (1892); reprinted in The Vertebrate Retina (ed. Rodieck, R. W.) 775–904 (Freeman, San Francisco, 1974)
Masland, R. H. The fundamental plan of the retina. Nature Neurosci. 4, 877–886 (2001)
Wässle, H. Parallel processing in the mammalian retina. Nature Rev. Neurosci. 5, 747–757 (2004)
Roska, B. & Werblin, F. Vertical interactions across ten parallel, stacked representations in the mammalian retina. Nature 410, 583–587 (2001)
Pang, J. J., Gao, F. & Wu, S. M. Segregation and integration of visual channels: layer-by layer computation of ON-OFF signals by amacrine cell dendrites. J. Neurosci. 22, 4693–4701 (2002)
Yamakawa, K. et al. DSCAM: a novel member of the immunoglobulin superfamily maps in a Down syndrome region and is involved in the development of the nervous system. Hum. Mol. Genet. 7, 227–237 (1998)
Agarwala, K. L. et al. Down syndrome cell adhesion molecule DSCAM mediates homophilic intercellular adhesion. Brain Res. Mol. Brain Res. 79, 118–126 (2000)
Agarwala, K. L. et al. Cloning and functional characterization of DSCAML1, a novel DSCAM-like cell adhesion molecule that mediates homophilic intercellular adhesion. Biochem. Biophys. Res. Commun. 285, 760–772 (2001)
Yimlamai, D. et al. The zebrafish down syndrome cell adhesion molecule is involved in cell movement during embryogenesis. Dev. Biol. 279, 44–57 (2005)
Yamagata, M., Weiner, J. A. & Sanes, J. R. Sidekicks: synaptic adhesion molecules that promote lamina-specific connectivity in the retina. Cell 110, 649–660 (2002)
Schmucker, D. et al. Drosophila Dscam is an axon guidance receptor exhibiting extraordinary molecular diversity. Cell 101, 671–684 (2000)
Wang, J., Zugates, C. T., Liang, I. H., Lee, C. H. & Lee, T. Drosophila Dscam is required for divergent segregation of sister branches and suppresses ectopic bifurcation of axons. Neuron 33, 559–571 (2002)
Hummel, T. et al. Axonal targeting of olfactory receptor neurons in Drosophila is controlled by Dscam. Neuron 37, 221–231 (2003)
Zhan, X. L. et al. Analysis of Dscam diversity in regulating axon guidance in Drosophila mushroom bodies. Neuron 43, 673–686 (2004)
Zhu, H. et al. Dendritic patterning by Dscam and synaptic partner matching in the Drosophila antennal lobe. Nature Neurosci. 9, 349–355 (2006)
Chen, B. E. et al. The molecular diversity of Dscam is functionally required for neuronal wiring specificity in Drosophila . Cell 125, 607–620 (2006)
Zipursky, S. L., Wojtowicz, W. M. & Hattori, D. Got diversity? Wiring the fly brain with Dscam. Trends Biochem. Sci. 31, 581–588 (2006)
Kidd, T. & Condron, B. Avoiding the SCAMs. Neuron 54, 350–352 (2007)
Millard, S. S. et al. Dscam2 mediates axonal tiling in the Drosophila visual system. Nature 447, 720–724 (2007)
Zeng, Y., Cai, X. & Cullen, B. R. Use of RNA polymerase II to transcribe artificial microRNAs. Methods Enzymol. 392, 371–380 (2005)
Chang, K., Elledge, S. J. & Hannon, G. J. Lessons from Nature: microRNA-based shRNA libraries. Nature Methods 3, 707–714 (2006)
Hughes, S. H. The RCAS vector system. Folia Biol. (Praha) 50, 107–119 (2004)
Wohrn, J. C. et al. Cadherin expression in the retina and retinofugal pathways of the chicken embryo. J. Comp. Neurol. 396, 20–38 (1998)
Yamagata, M. et al. Labeled lines in the retinotectal system: markers for retinorecipient sublaminae and the retinal ganglion cell subsets that innervate them. Mol. Cell. Neurosci. 33, 296–310 (2006)
Li, X. et al. The minimum internal and external sequence requirements for transposition of the eukaryotic transformation vector piggyBac. Mol. Genet. Genomics 266, 190–198 (2001)
Ding, S. et al. Efficient transposition of the piggyBac (PB) transposon in mammalian cells and mice. Cell 122, 473–483 (2005)
Poliakov, A., Cotrina, M. & Wilkinson, D. G. Diverse roles of eph receptors and ephrins in the regulation of cell migration and tissue assembly. Dev. Cell 7, 465–480 (2004)
Hong, K. et al. A ligand-gated association between cytoplasmic domains of UNC5 and DCC family receptors converts netrin-induced growth cone attraction to repulsion. Cell 97, 927–941 (1999)
Fuerst, P. G., Koizumi, A., Masland, R. H. & Burgess, R. W. Neurite arborization and mosaic spacing in the mouse retina require DSCAM. Nature doi: 10.1038/nature06514 (this issue)
Sanes, J. R. & Yamagata, M. Formation of lamina-specific synaptic connections. Curr. Opin. Neurobiol. 9, 79–87 (1999)
Spira, A. W. et al. Localization of choline acetyltransferase-like immunoreactivity in the embryonic chick retina. J. Comp. Neurol. 260, 526–538 (1987)
Barres, B. A. et al. Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron 1, 791–803 (1988)
Inuzuka, H., Miyatani, S. & Takeichi, M. R-cadherin: a novel Ca(2+)-dependent cell-cell adhesion molecule expressed in the retina. Neuron 7, 69–79 (1991)
Johnson, C. D. & Epstein, M. L. Monoclonal antibodies and polyvalent antiserum to chicken choline acetyltransferase. J. Neurochem. 46, 968–976 (1986)
Acknowledgements
We thank R. Burgess and colleagues for sharing data on mouse Dscam before submission, K. Hong for anti-sidekick-1 antiserum and M. J. Fraser (University of Notre Dame) for piggyBac plasmids. This work was supported by a grant from the NIH to J.R.S.
Author Contributions M.Y. and J.R.S. conceived the experiments. M.Y. performed the experiments. J.R.S. and M.Y. wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-10 with Legends. (PDF 40766 kb)
Rights and permissions
About this article
Cite this article
Yamagata, M., Sanes, J. Dscam and Sidekick proteins direct lamina-specific synaptic connections in vertebrate retina. Nature 451, 465–469 (2008). https://doi.org/10.1038/nature06469
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1038/nature06469
This article is cited by
-
Neuronal DSCAM regulates the peri-synaptic localization of GLAST in Bergmann glia for functional synapse formation
Nature Communications (2024)
-
DSCAM is differentially patterned along the optic axon pathway in the developing Xenopus visual system and guides axon termination at the target
Neural Development (2022)
-
Identification of female-specific genetic variants for metabolic syndrome and its component traits to improve the prediction of metabolic syndrome in females
BMC Medical Genetics (2019)
-
The Use of Whole Exome Sequencing in a Cohort of Transgender Individuals to Identify Rare Genetic Variants
Scientific Reports (2019)
-
The cell adhesion protein CAR is a negative regulator of synaptic transmission
Scientific Reports (2019)