Abstract
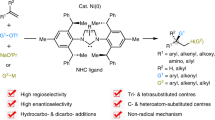
Alkenes are the most ubiquitous prochiral functional groups—those that can be converted from achiral to chiral in a single step—that are accessible to synthetic chemists. For this reason, difunctionalization reactions of alkenes (whereby two functional groups are added to the same double bond) are particularly important, as they can be used to produce highly complex molecular architectures1,2. Stereoselective oxidation reactions, including dihydroxylation, aminohydroxylation and halogenation3,4,5,6, are well established methods for functionalizing alkenes. However, the intermolecular incorporation of both carbon- and nitrogen-based functionalities stereoselectively across an alkene has not been reported. Here we describe the rhodium-catalysed carboamination of alkenes at the same (syn) face of a double bond, initiated by a carbon–hydrogen activation event that uses enoxyphthalimides as the source of both the carbon and the nitrogen functionalities. The reaction methodology allows for the intermolecular, stereospecific formation of one carbon–carbon and one carbon–nitrogen bond across an alkene, which is, to our knowledge, unprecedented. The reaction design involves the in situ generation of a bidentate directing group and the use of a new cyclopentadienyl ligand to control the reactivity of rhodium. The results provide a new way of synthesizing functionalized alkenes, and should lead to the convergent and stereoselective assembly of amine-containing acyclic molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
McDonald, R. I., Liu, G. & Stahl, S. S. Palladium(II)-catalyzed alkene functionalization via nucleopalladation: stereochemical pathways and enantioselective catalytic applications. Chem. Rev. 111, 2981–3019 (2011)
Chemler, S. R. & Bovino, M. T. Catalytic aminohalogenation of alkenes and alkynes. Am. Chem. Soc. Catal. 3, 1076–1091 (2013)
Berkesell, A. & Gröger, H. Asymmetric Organocatalysis (Wiley-VCH, 2005)
Jacobsen, E. N. & Wu, M. H. in Comprehensive Asymmetric Catalysis (eds Jacobsen, E. N., Pfaltz, A. & Yamamoto, H. ) 1309–1326 (Springer, 1999)
Hennecke, U. New catalytic approaches towards the enantioselective halogenation of alkenes. Chem. Asian J. 7, 456–465 (2012)
Tan, C. K., Yu, W. Z. & Yeung, Y. Y. Stereoselective bromofunctionalization of alkenes. Chirality 26, 328–343 (2014)
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014)
Zhou, J. & Hartwig, J. F. Intermolecular, catalytic asymmetric hydroamination of bicyclic alkenes and dienes in high yield and enantioselectivity. J. Am. Chem. Soc. 130, 12220–12221 (2008)
Shen, X. & Buchwald, S. L. Rhodium-catalyzed asymmetric intramolecular hydroamination of unactivated alkenes. Angew. Chem. Int. Edn 49, 564–567 (2010)
Beller, M., Seayad, J., Tillack, A. & Jiao, H. Catalytic Markovnikov and anti-Markovnikov functionalization of alkenes and alkynes: recent developments and trends. Angew. Chem. Int. Edn 43, 3368–3398 (2004)
Beletskaya, I. P. & Cheprakov, A. V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 100, 3009–3066 (2000)
Werner, E. W., Mei, T.-S., Burckle, A. J. & Sigman, M. S. Enantioselective Heck arylations of acyclic alkenyl alcohols using a redox-relay strategy. Science 338, 1455–1458 (2012)
Coldham, I. & Hufton, R. Intramolecular dipolar cycloaddition reactions of azomethine ylides. Chem. Rev. 105, 2765–2810 (2005)
Nakamura, I. & Yamamoto, Y. Transition-metal-catalyzed reactions in heterocyclic synthesis. Chem. Rev. 104, 2127–2198 (2004)
Mai, D. N. & Wolfe, J. P. Asymmetric palladium-catalyzed carboamination reactions for the synthesis of enantiomerically enriched 2-(arylmethyl)- and 2-(alkenylmethyl)pyrrolidines. J. Am. Chem. Soc. 132, 12157–12159 (2010)
Wolfe, J. P. Synthesis of saturated heterocycles via metal-catalyzed alkene carboamination or carboalkoxylation reactions. Top. Heterocycl. Chem. 32, 1–37 (2013)
Zeng, W. & Chemler, S. R. Copper(II)-catalyzed enantioselective intramolecular carboamination of alkenes. J. Am. Chem. Soc. 129, 12948–12949 (2007)
Weidner, K., Giroult, A., Panchaud, P. & Renaud, P. Efficient carboazidation of alkenes using a radical desulfonylative azide transfer process. J. Am. Chem. Soc. 132, 17511–17515 (2010)
Zhang, H. et al. Copper-catalyzed intermolecular aminocyanation and diamination of alkenes. Angew. Chem. Int. Edn 52, 2529–2533 (2013)
Piou, T. & Rovis, T. Rh(III)-catalyzed cyclopropanation initiated by C–H activation: ligand development enables a diastereoselective [2 + 1] annulation of N-enoxyphthalimides and alkenes. J. Am. Chem. Soc. 136, 11292–11295 (2014)
Mo, J., Wang, L., Liu, Y. & Cui, X. Transition-metal-catalyzed direct C–H functionalization under external-oxidant-free conditions. Synthesis 439–459 (2015)
Neely, J. M. & Rovis, T. Rh(III)-catalyzed regioselective synthesis of pyridines from alkenes and α,β-unsaturated oxime esters. J. Am. Chem. Soc. 135, 66–69 (2013)
Hyster, T. K. & Rovis, T. An improved catalyst architecture for rhodium(III) catalyzed C–H activation and its application to pyridone synthesis. Chem. Sci. (Camb.) 2, 1606–1610 (2011)
Webb, N. J., Marsden, S. P. & Raw, S. A. Rhodium(III)-catalyzed C–H activation/annulation with vinyl esters as an acetylene equivalent. Org. Lett. 16, 4718–4721 (2014)
Guimond, N., Gorelsky, S. I. & Fagnou, K. Rhodium(III)-catalyzed heterocycle synthesis using an internal oxidant: improved reactivity and mechanistic studies. J. Am. Chem. Soc. 133, 6449–6457 (2011)
Rakshit, S., Grohmann, C., Besset, T. & Glorius, F. Rh(III)-catalyzed directed C−H olefination using an oxidizing directing group: mild, efficient, and versatile. J. Am. Chem. Soc. 133, 2350–2353 (2011)
Acknowledgements
We thank the National Institute of General Medical Sciences (grant no. GM80442) for support. We thank Johnson Matthey for rhodium salts, and J. Chu and B. Newell (at Colorado State University) for solving X-ray structures.
Author information
Authors and Affiliations
Contributions
T.P. and T.R. conceived the concept and prepared the manuscript. T.R. directed the investigations. T.P. developed and studied the reaction.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data and Supplementary References. (PDF 13428 kb)
Rights and permissions
About this article
Cite this article
Piou, T., Rovis, T. Rhodium-catalysed syn-carboamination of alkenes via a transient directing group. Nature 527, 86–90 (2015). https://doi.org/10.1038/nature15691
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/nature15691
This article is cited by
-
Tuning light-driven oxidation of styrene inside water-soluble nanocages
Nature Communications (2024)
-
Cobalt(III)-catalyzed asymmetric ring-opening of 7-oxabenzonorbornadienes via indole C–H functionalization
Nature Communications (2023)
-
Rhodium-catalyzed enantio- and diastereoselective carboamidation of bicyclic olefins toward construction of remote chiral centers and axis
Science China Chemistry (2023)
-
Bifunctional reagents in organic synthesis
Nature Reviews Chemistry (2021)
-
Iron-catalyzed carboazidation of alkenes and alkynes
Nature Communications (2019)