Abstract
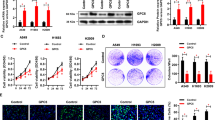
Glypican-5 (GPC5) is a member of the heparin sulfate proteoglycans. Previous studies of GPC5 in lung tumorigenesis showed conflicting results. In this study, we confirmed that GPC5 was downregulated in lung adenocarcinoma tissues compared with adjacent normal lung tissues. The low expression of GPC5 was significantly associated with poor outcome in lung adenocarcinoma. To understand the biological mechanism of the downregulation, we examined the promoter methylation status of GPC5 gene. We found that GPC5 was significantly hypermethylated in lung cancer tissues and lung cancer cell lines compared with normal lung tissues. The methylation level of GPC5 was negatively correlated with its transcriptional expression. De-methylation experiments further confirmed that the loss of GPC5 expression was regulated by its hypermethylation. Overexpression of GPC5 inhibited proliferation, migration and invasion of lung cancer cells in vitro, and repressed tumor growth in vivo, whereas knockdown of GPC5 was able to reverse the effect. Furthermore, we demonstrated that GPC5 could suppress the Wnt/β-catenin signaling by binding to Wnt3a at the cell surface, which mediated its function as a tumor suppressor. Overall, these findings demonstrate that GPC5 is a novel epigenetically silenced tumor suppressor, which inhibits tumor growth by suppressing Wnt/β-catenin signaling in lung adenocarcinoma. Our findings substantially expand our understanding about the role and the molecular mechanism of GPC5 in tumorigenesis of lung cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A . Cancer statistics, 2015. CA Cancer J Clin 2015; 65: 5–29.
Dela Cruz CS, Tanoue LT, Matthay RA . Lung cancer: epidemiology, etiology, and prevention. Clin Chest Med 2011; 32: 605–644.
Zhang J, Gold KA, Lin HY, Swisher SG, Xing Y, Lee JJ et al. Relationship between tumor size and survival in non-small-cell lung cancer (NSCLC): an analysis of the surveillance, epidemiology, and end results (SEER) registry. J Thorac Oncol 2015; 10: 682–690.
Wanders R, Steevens J, Botterweck A, Dingemans AM, Reymen B, Baardwijk A et al. Treatment with curative intent of stage III non-small cell lung cancer patients of 75 years: a prospective population-based study. Eur J Cancer 2011; 47: 2691–2697.
Neri S, Yoshida J, Ishii G, Matsumura Y, Aokage K, Hishida T et al. Prognostic impact of microscopic vessel invasion and visceral pleural invasion in non-small cell lung cancer: a retrospective analysis of 2657 patients. Ann Surg 2014; 260: 383–388.
Veugelers M, Vermeesch J, Reekmans G, Steinfeld R, Marynen P, David G . Characterization of glypican-5 and chromosomal localization of human GPC5, a new member of the glypican gene family. Genomics 1997; 40: 24–30.
Filmus J, Capurro M, Rast J . Glypicans. Genome Biol 2008; 9: 224.
Williamson D, Selfe J, Gordon T, Lu YJ, Pritchard-Jones K, Murai K et al. Role for amplification and expression of glypican-5 in rhabdomyosarcoma. Cancer Res 2007; 67: 57–65.
Nishimura R, Takita J, Sato-Otsubo A, Kato M, Koh K, Hanada R et al. Characterization of genetic lesions in rhabdomyosarcoma using a high-density single nucleotide polymorphism array. Cancer Sci 2013; 104: 856–864.
Zhang C, Zhang S, Zhang D, Zhang Z, Xu Y, Liu S . A lung cancer gene GPC5 could also be crucial in breast cancer. Mol Genet Metab 2011; 103: 104–105.
Li F, Shi W, Capurro M, Filmus J . Glypican-5 stimulates rhabdomyosarcoma cell proliferation by activating Hedgehog signaling. J Cell Biol 2011; 192: 691–704.
Li Y, Sheu CC, Ye Y, de Andrade M, Wang L, Chang SC et al. Genetic variants and risk of lung cancer in never smokers: a genome-wide association study. Lancet Oncol 2010; 11: 321–330.
Li Y, Miao L, Cai H, Ding J, Xiao Y, Yang J et al. The overexpression of glypican-5 promotes cancer cell migration and is associated with shorter overall survival in non-small cell lung cancer. Oncol Lett 2013; 6: 1565–1572.
Yang X, Zhang Z, Qiu M, Hu J, Fan X, Wang J et al. Glypican-5 is a novel metastasis suppressor gene in non-small cell lung cancer. Cancer Lett 2013; 341: 265–273.
Li Y, Yang P . GPC5 gene and its related pathways in lung cancer. J Thorac Oncol 2011; 6: 2–5.
Gyorffy B, Surowiak P, Budczies J, Lanczky A . Online survival analysis software to assess the prognostic value of biomarkers using transcriptomic data in non-small-cell lung cancer. PLoS One 2013; 8: e82241.
De Cat B, David G . Developmental roles of the glypicans. Semin Cell Dev Biol 2001; 12: 117–125.
Okolicsanyi RK, van Wijnen AJ, Cool SM, Stein GS, Griffiths LR, Haupt LM . Heparan sulfate proteoglycans and human breast cancer epithelial cell tumorigenicity. J Cell Biochem 2014; 115: 967–976.
Clevers H, Nusse R . Wnt/beta-catenin signaling and disease. Cell 2012; 149: 1192–1205.
Blitzer JT, Nusse R . A critical role for endocytosis in Wnt signaling. BMC Cell Biol 2006; 7: 28.
Grumolato L, Liu G, Mong P, Mudbhary R, Biswas R, Arroyave R et al. Canonical and noncanonical Wnts use a common mechanism to activate completely unrelated coreceptors. Genes Dev 2010; 24: 2517–2530.
Stewart DJ . Wnt signaling pathway in non-small cell lung cancer. J Natl Cancer Inst 2014; 106: djt356.
Wang HQ, Xu ML, Ma J, Zhang Y, Xie CH . Frizzled-8 as a putative therapeutic target in human lung cancer. Biochem Biophys Res Commun 2012; 417: 62–66.
Kumar S, Zigman M, Patel TR, Trageser B, Gross JC, Rahm K et al. Molecular dissection of Wnt3a-Frizzled8 interaction reveals essential and modulatory determinants of Wnt signaling activity. BMC Biol 2014; 12: 44.
Bourhis E, Tam C, Franke Y, Bazan JF, Ernst J, Hwang J et al. Reconstitution of a frizzled8.Wnt3a.LRP6 signaling complex reveals multiple Wnt and Dkk1 binding sites on LRP6. J Biol Chem 2010; 285: 9172–9179.
Hong SW, Jiang Y, Kim S, Li CJ, Lee DK . Target gene abundance contributes to the efficiency of siRNA-mediated gene silencing. Nucleic Acid Ther 2014; 24: 192–198.
Hu X, Hipolito S, Lynn R, Abraham V, Ramos S, Wong-Staal F . Relative gene-silencing efficiencies of small interfering RNAs targeting sense and antisense transcripts from the same genetic locus. Nucleic Acids Res 2004; 32: 4609–4617.
Song HH, Filmus J . The role of glypicans in mammalian development. Biochim Biophys Acta 2002; 1573: 241–246.
Christianson HC, Belting M . Heparan sulfate proteoglycan as a cell-surface endocytosis receptor. Matrix Biol 2014; 35: 51–55.
Knelson EH, Nee JC, Blobe GC . Heparan sulfate signaling in cancer. Trends Biochem Sci 2014; 39: 277–288.
Filmus J, Capurro M . The role of glypicans in Hedgehog signaling. Matrix Biol 2014; 35: 248–252.
Capurro MI, Xiang YY, Lobe C, Filmus J . Glypican-3 promotes the growth of hepatocellular carcinoma by stimulating canonical Wnt signaling. Cancer Res 2005; 65: 6245–6254.
Capurro M, Martin T, Shi W, Filmus J . Glypican-3 binds to Frizzled and plays a direct role in the stimulation of canonical Wnt signaling. J Cell Sci 2014; 127: 1565–1575.
Capurro MI, Xu P, Shi W, Li F, Jia A, Filmus J . Glypican-3 inhibits Hedgehog signaling during development by competing with patched for Hedgehog binding. Dev Cell 2008; 14: 700–711.
Xi Y, Chen Y . Wnt signaling pathway: implications for therapy in lung cancer and bone metastasis. Cancer Lett 2014; 353: 8–16.
Teo JL, Kahn M . The Wnt signaling pathway in cellular proliferation and differentiation: a tale of two coactivators. Adv Drug Deliv Rev 2010; 62: 1149–1155.
Liu D, Shriver Z, Venkataraman G, El Shabrawi Y, Sasisekharan R . Tumor cell surface heparan sulfate as cryptic promoters or inhibitors of tumor growth and metastasis. Proc Natl Acad Sci USA 2002; 99: 568–573.
Janda CY, Waghray D, Levin AM, Thomas C, Garcia KC . Structural basis of Wnt recognition by Frizzled. Science 2012; 337: 59–64.
Avanesov A, Honeyager SM, Malicki J, Blair SS . The role of glypicans in Wnt inhibitory factor-1 activity and the structural basis of Wif1's effects on Wnt and Hedgehog signaling. PLoS Genet 2012; 8: e1002503.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001; 25: 402–408.
Lu L, Katsaros D, de la Longrais IA, Sochirca O, Yu H . Hypermethylation of let-7a-3 in epithelial ovarian cancer is associated with low insulin-like growth factor-II expression and favorable prognosis. Cancer Res 2007; 67: 10117–10122.
Han F, Liu W, Jiang X, Shi X, Yin L, Ao L et al. SOX30, a novel epigenetic silenced tumor suppressor, promotes tumor cell apoptosis by transcriptional activating p53 in lung cancer. Oncogene 2014; 34: 4391–4402.
Liu WB, Han F, Jiang X, Yang LJ, Li YH, Liu Y et al. ANKRD18A as a novel epigenetic regulation gene in lung cancer. Biochem Biophys Res Commun 2012; 429: 180–185.
Lv J, Zhu P, Yang Z, Li M, Zhang X, Cheng J et al. PCDH20 functions as a tumour-suppressor gene through antagonizing the Wnt/beta-catenin signalling pathway in hepatocellular carcinoma. J Viral Hepat 2015; 22: 201–211.
Cline MS, Craft B, Swatloski T, Goldman M, Ma S, Haussler D et al. Exploring TCGA Pan-Cancer data at the UCSC Cancer Genomics Browser. Sci Rep 2013; 3: 2652.
Labidi-Galy SI, Clauss A, Ng V, Duraisamy S, Elias KM, Piao HY et al. Elafin drives poor outcome in high-grade serous ovarian cancers and basal-like breast tumors. Oncogene 2015; 34: 373–383.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (no. 81171903 and no. 81472190 to Y Li; no. 81370139 to L Bai), the Chongqing Natural Science Foundation of China (no. cstc2015jcyjBX0110 to Y Li), the Discipline Construction and Development Foundation from Xinqiao Hospital, Third Military Medical University (to L Bai), and Foundation for Clinical Research from Xinqiao Hospital, Third Military Medical University (no. 2014YLC13 to L Bai).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yuan, S., Yu, Z., Liu, Q. et al. GPC5, a novel epigenetically silenced tumor suppressor, inhibits tumor growth by suppressing Wnt/β-catenin signaling in lung adenocarcinoma. Oncogene 35, 6120–6131 (2016). https://doi.org/10.1038/onc.2016.149
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/onc.2016.149
This article is cited by
-
RBM14 as a novel epigenetic-activated tumor oncogene is implicated in the reprogramming of glycolysis in lung cancer
World Journal of Surgical Oncology (2023)
-
A novel NET-related gene signature for predicting DLBCL prognosis
Journal of Translational Medicine (2023)
-
A potassium-chloride co-transporter promotes tumor progression and castration resistance of prostate cancer through m6A reader YTHDC1
Cell Death & Disease (2023)
-
WNT ligands in non-small cell lung cancer: from pathogenesis to clinical practice
Discover Oncology (2023)
-
The Glypican proteoglycans show intrinsic interactions with Wnt-3a in human prostate cancer cells that are not always associated with cascade activation
BMC Molecular and Cell Biology (2021)