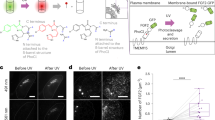
Abstract
We constructed a novel peptide-array format system for cellular toxicity analysis. In this system, a peptide was immobilized on a conventional 96-well plate bottom via a photo-cleavable linker. Once UV light irradiated the desired wells, the peptide was released from the bottom. As a result, the cytotoxic behavior of the peptide could be monitored. Immobilization and light-irradiation conditions were optimized. The immobilized peptide showed no cytotoxicity; therefore, the cells could be cultured on the peptide-immobilizing plate from the beginning of the experiment. Cell-toxicity assays with this system for three cell types were performed. All cell types showed ∼25% lowering of viability with the photo-released 5-(and-6)-carboxytetramethylrhodamine (TMR)-GKLAKLAKKLAKLAKKLAKLAKGC (TMR-KLA-C) peptide compared with the non-coated plate. This relative toxicity nearly corresponded to that of ∼10 μM TMR-KLA-C in solution, and we found that the released peptide concentration per well was ∼10 μM at 60 min irradiation. Throughout this study, we successfully immobilized peptides via the photo-cleavable linker, released them by UV irradiation spatiotemporally and conducted the cell-toxicity assay. This study implies that the peptide photo-releasing array system will allow the realization of high-throughput cell arrays for cellomics analyses and cell-based phenotypic drug screenings.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Stockwell, B. R. Exploring biology with small organic molecules. Nature 432, 846–854 (2004).
Kandpal, R., Saviola, B. & Felton, J. The era of 'omics unlimited. Biotechniques 46, 351–355 (2009).
Fernandes, T. G., Diogo, M. M., Clark, D. S., Dordick, J. S. & Cabral, J. M. High-throughput cellular microarray platforms: applications in drug discovery, toxicology and stem cell research. Trends Biotechnol. 27, 342–349 (2009).
Usui, K., Takahashi, M., Nokihara, K. & Mihara, H. Peptide arrays with designed alpha-helical structures for characterization of proteins from FRET fingerprint patterns. Mol. Divers. 8, 209–218 (2004).
Usui, K., Ojima, T., Takahashi, M., Nokihara, K. & Mihara, H. Peptide arrays with designed secondary structures for protein characterization using fluorescent fingerprint patterns. Biopolymers 76, 129–139 (2004).
Usui, K., Tomizaki, K. -Y., Ohyama, T., Nokihara, K. & Mihara, H. A novel peptide microarray for protein detection and analysis utilizing a dry peptide array system. Mol. Biosyst. 2, 113–121 (2006).
Usui, K., Tomizaki, K. -Y. & Mihara, H. Protein-fingerprint data mining of a designed alpha-helical peptide array. Mol. Biosyst. 2, 417–420 (2006).
Usui, K., Tomizaki, K.-Y. & Mihara, H. Screening of alpha-helical peptide ligands controlling a calcineurin-phosphatase activity. Bioorg. Med. Chem. Lett. 17, 167–171 (2007).
Usui, K., Tomizaki, K. -Y. & Mihara, H. A designed peptide chip: protein fingerprinting technology with a dry peptide array and statistical data mining. Methods Mol. Biol. 570, 273–284 (2009).
Kodadek, T. Development of protein-detecting microarrays and related devices. Trends Biochem. Sci. 27, 295–300 (2002).
Tomizaki, K. -Y., Usui, K. & Mihara, H. Protein-detecting microarrays: current accomplishments and requirements. Chembiochem 6, 782–799 (2005).
Uttamchandani, M. & Yao, S. Q. Peptide microarrays: next generation biochips for detection, diagnostics and high-throughput screening. Curr. Pharm. Des. 14, 2428–2438 (2008).
Tomizaki, K.-Y., Usui, K. & Mihara, H. Proteins: Array-Based Techniques. (ed. Begley, T. P.) 144–158 (Wiley Encyclopedia of Chemical Biology (2009).
Tomizaki, K. Y., Usui, K. & Mihara, H. Protein-protein interactions and selection: array-based techniques for screening disease-associated biomarkers in predictive/early diagnosis. FEBS J 277, 1996–2005 (2010).
Usui, K., Kakiyama, T., Tomizaki, K.-Y., Mie, M., Kobatake, E. & Mihara, H. Cell fingerprint patterns using designed α-helical peptides to screen for cell-specific toxicity. Bioorg. Med. Chem. Lett. 21, 6281–3284 (2011).
Jiang, X., Ferrigno, R., Mrksich, M. & Whitesides, G. M. Electrochemical desorption of self-assembled monolayers noninvasively releases patterned cells from geometrical confinements. J. Am. Chem. Soc. 125, 2366–2367 (2003).
Ryan, D., Parviz, B. A., Linder, V., Semetey, V., Sia, S. K., Su, J., Mrksich, M. & Whitesides, G. M. Patterning multiple aligned self-assembled monolayers using light. Langmuir 20, 9080–9088 (2004).
Carrico, I. S., Maskarinec, S. A., Heilshorn, S. C., Mock, M. L., Liu, J. C., Nowatzki, P. J., Franck, C., Ravichandran, G. & Tirrell, D. A. Lithographic patterning of photoreactive cell-adhesive proteins. J. Am. Chem. Soc. 129, 4874–4875 (2007).
Veiseh, M., Veiseh, O., Martin, M. C., Asphahani, F. & Zhang, M. Short peptides enhance single cell adhesion and viability on microarrays. Langmuir 23, 4472–4479 (2007).
Holmes, C. P. & Jones, D. G. Reagents for combinatorial organic synthesis: development of a new o-nitrobenzyl photolabile linker for solid phase synthesis. J. Org. Chem. 60, 2318–2319 (1995).
Whitehouse, D. L., Savinov, S. N. & Austin, D. J. An improved synthesis and selective coupling of a hydroxy based photolabile linker for solid phase organic synthesis. Tetrahedron Lett. 38, 7851–7852 (1997).
Rinnová, M., Nováková, M., Kasicka, V. & Jirácek, J. Side reactions during photochemical cleavage of an alpha-methyl-6-nitroveratryl-based photolabile linker. J. Pept. Sci. 6, 355–365 (2000).
Nakayama, K., Tachikawa, T. & Majima, T. Protein recording material: photorecord/erasable protein array using a UV-eliminative linker. Langmuir 24, 1625–1628 (2008).
Zhang, Y., Dong, Z., Nomura, M., Zhong, S., Chen, N., Bode, A. M. & Dong, Z. Signal transduction pathways involved in phosphorylation and activation of p70S6K following exposure to UVA irradiation. J. Biol. Chem. 276, 20913–20923 (2001).
Soughayer, J. S., Wang, Y., Li, H., Cheung, S. -H., Rossi, F. M., Stanbridge, E. J., Sims, C. E. & Allbritton, N. L. Characterization of TAT-mediated transport of detachable kinase substrates. Biochemistry 43, 8528–8540 (2004).
Hoff, A., André, T., Fischer, R., Voss, S., Hulko, M., Marquardt, U., Wiesmüller, K. -H. & Brock, R. Chemolabile cellular microarrays for screening small molecules and peptides. Mol. Divers. 8, 311–320 (2004).
Chan, W. C. & White, P. D. Fmoc Solid Phase Peptide Synthesis: A Practical Approach, (Oxford University Press, New York, 2000).
Ellerby, H. M., Arap, W., Ellerby, L. M., Kain, R., Andrusiak, R., Rio, G. D., Krajewski, S., Lombardo, C. R., Rao, R., Ruoslahti, E., Bredesen, D. E. & Pasqualini, R. Anti-cancer activity of targeted pro-apoptotic peptides. Nat. Med. 5, 1032–1038 (1999).
Acknowledgements
We thank Dr M Nakamura (Tokyo University of Pharmacy and Life Sciences, Tokyo, Japan) and Dr T Takahashi (Gunma University, Japan) for valuable discussions and generous support. This study was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Japan Science and Technology Agency. K.U. is also grateful to the Grants-in-Aid for Scientific Research, the ‘Core Research’ project (2009-2014) from MEXT. K.-y.T. acknowledges the Ryukoku University Science and Technology Fund.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on Polymer Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Kakiyama, T., Usui, K., Tomizaki, Ky. et al. A peptide release system using a photo-cleavable linker in a cell array format for cell-toxicity analysis. Polym J 45, 535–539 (2013). https://doi.org/10.1038/pj.2013.20
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pj.2013.20
Keywords
This article is cited by
-
Exploring the flexible chemistry of 4-fluoro-3-nitrophenyl azide for biomolecule immobilization and bioconjugation
Analytical and Bioanalytical Chemistry (2016)
-
Effects of the properties of short peptides conjugated with cell-penetrating peptides on their internalization into cells
Scientific Reports (2015)