Abstract
Infant Acute Lymphoblastic Leukemia (ALL) driven by the KMT2A::AFF1 onco-fusion is an aggressive, poor prognosis disease with few co-operative mutations. The fusion originates in utero, yet the embryonic initiating steps of disease development remain poorly understood. Here, we present a novel murine KMT2A::AFF1 model, that provides key insights into KMT2A::AFF1 pre-leukemia, relevant to human disease. The model enables precise oncogene induction, and upon targeting hematopoietic stem and progenitor cells (HSPCs) a selective negative impact on proliferation of hematopoietic stem cells (HSCs) was observed, regardless of developmental state during induction. However, a unique CD24+PreProB subset expanded exclusively within the KMT2A::AFF1 embryonic context. This population was absent when targeting lymphoid progenitors, highlighting the importance of the cell of origin for leukemic development. The CD24+PreProB subset displayed key features of pre-leukemic stem cells, including lineage plasticity and aberrant engraftment ability. In line with their pre-malignant phenotype, single-cell transcriptomics revealed a signature consistent with stemness, and notable, up-regulation of Hmga2, a regulator of self-renewal. The signature was critically transferable to human KMT2A::AFF1 patients. Furthermore, given that CD24 is a potential therapeutic target, our findings uncover a distinct embryonic pre-leukemic state with direct relevance to human disease.

Similar content being viewed by others
Introduction
Despite a remarkable progress in treating children with leukemia, infants with acute lymphoblastic leukemia (ALL) still have a poor prognosis, with survival rates of about fifty percent [1, 2]. These infant leukemias are often driven by chromosomal translocations of the KMT2A gene with AFF1 (t(4;11)), and the resulting KMT2A::AFF1 oncogene (MLL::AF4) gives rise to an aggressive, poor prognosis B cell leukemia [1,2,3,4]. KMT2A is a histone methyltransferase with key roles in epigenetic regulation, and the fusion drives transcriptional activation and aberrant expression of Homeobox (HOX) genes, contributing to leukemogenesis [5, 6]. KMT2A::AFF1 leukemia is seen in all age groups, but the disease in infants can be regarded as its own entity, with a particularly low mutational burden, and a rapid and therapy resistant disease [3, 7].
There is abundant evidence that infant ALL originates prenatally [8, 9], and after a pre-leukemic phase, disease becomes evident before one year of age. Embryonic hematopoiesis occurs in overlapping waves, and in contrast to adult hematopoiesis, embryonic progenitors emerge independently of definitive hematopoietic stem cells (HSCs) [10]. Definitive HSCs are produced in the aorta–gonad–mesonephros (AGM) region at embryonic day (E) 10.5 in mice, and then migrate to the fetal liver (FL), which serves as the main hematopoietic organ for most of development, whereas the bone marrow (BM) becomes active primarily after birth [10].
Infant ALL originates within this embryonic context, and a relevant model of the disease onset requires that the initiating onco-fusion is expressed at the appropriate developmental stage and in the relevant cell type(s). Earlier studies have mainly focused on the adult setting [11,12,13,14,15] and when investigations on early embryonic hematopoietic cells were performed, these findings were limited by the fact that no phenotype was observed in the embryos [16]. The impact of the oncogene was detectable only ex vivo, and in adult mice, it manifested as a lymphoma-like condition rather than leukemia [16]. Recent advancements in transforming human cells have shown promise [17, 18], though these models also face limitations in exploring the pre-leukemic state of the disease. Consequently, as of yet, the prenatal initiating phase of infant ALL remains a challenge to model in situ, and the events preceding leukemia development remains largely unexplored.
To investigate the prenatal events driving disease development, we here developed and utilized a novel inducible murine KMT2A::AFF1 model, enabling precise targeting of the fusion gene to hematopoietic stem and progenitor cells. This approach uncovered a distinct fetal pre-leukemic state and identified a transcriptional signature transferable to human KMT2A::AFF1 leukemia. Our findings provide critical insights into the ontogeny and cell-type-specific effects of KMT2A::AFF1, and the transcriptional alterations underlying the pre-leukemic state.
Methods
Please see supplementary methods for details.
Animals
Col1a1-tetO-KMT2A::AFF1 (KMT2A::AFF1Col1a1-tetO), Rosa26-ZtTA [19] and different Cre strains were used to enable cell type specific induction of KMT2A::AFF1 (Vav-Cre [20]; Rag1-Cre [21]; Mb1-Cre [22]). Embryos were obtained by timed mating overnight. Adult mice were analyzed 8–30 weeks of age if not otherwise stated. All animal experiments were performed in accordance with the relevant guidelines and regulations and were approved by the Malmö/Lund Ethics Committee on Animal Testing at the Lund District Court.
Colony-forming assays
In vitro semi solid cultures were performed with ~100-250 purified cells that were cultured in Methocult (Stemcell Technologies) supplemented with cytokines. Colonies were scored and replated weekly.
Transplantation assay
Recipient NOD-SCID-GAMMA (Il2rg–/–) (NSG) mice (CD45.1) were sub-lethally irradiated (250 cGy) and donor CD24+ PreProBs cells ( ~ 500–1300cells/recipient) or LSK (Lin-; SCA1+; cKIT+) cells ( ~ 1500–2000 cells/ recipient) were purified using Fluorescence-Activated Cell Sorting (FACS) and subsequently transplanted.
Single-cell cellular indexing of transcriptomes and epitopes by sequencing
Cells were purified and loaded onto the Chromium platform Next GEM Single Cell 3’ Kit v3.1 (10x Genomics) and subjected to library preparation, according to the manufacturer’s instruction [23]. The resulting libraries were sequenced on a NovaSeq system (Illumina).
Data analysis
Flow cytometry data were analyzed using FlowJo_v10 (BD). Graphs and statistical analysis were performed using GraphPadPrism_v10. Single-cell RNA-sequencing data were visualized in ShinyCell [24].
Results
KMT2A::AFF1 induction in embryonic hematopoietic stem and progenitor cells
To study the KMT2A::AFF1-associated pre-leukemic state, we created a novel inducible mouse model. For this, we placed a human KMT2A::AFF1 fusion gene under the control of a Tet-op promoter (KMT2A::AFF1Col1a1-tetO), as described [25]. The fusion was made Cre inducible by crossing with the Rosa26-ZtTA strain [19], in which the tetracycline-trans-activator (tTA) contains a STOP cassette flanked by LoxP sites. Subsequently, we induced the expression of KMT2A::AFF1 in embryonic hematopoietic stem and progenitor cells (HSPCs) using Vav-Cre recombinase [20], resulting in KMT2A::AFF1Col1a1-tetO /ZtTA/Vav-Cre+ strain (named KMT2A::AFF1Vav-Cre+ throughout) (Fig. 1A). Cre-positive FLs from embryonic day (E)18.5 expressed KMT2A::AFF1 at ~2.5 to 4.5-fold higher levels than endogenous Kmt2a, and Homeobox A9 (Hoxa9), a known KMT2A::AFF1 target gene [26], was significantly elevated (Fig. 1B; Supplementary Fig. 1A, B).
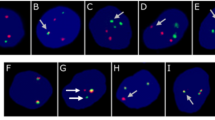
A Schematic illustration of the model. Human KMT2A::AFF1 was placed under the control of a Tet-OP promotor; the tissue recombinase (tTA) had a STOP cassette flanked by LoxP sites. Strain specific Cre-recombinase was used to induce expression of KMT2A::AFF1. B Relative expression (normalized to B-actin) of KMT2A::AFF1 in purified HSCs, LMPPs and ProBs from E18.5 KMT2A::AFF1Vav-Cre+ fetal livers (FLs). Box plots define lower and upper quartiles, and whiskers min to max values (4 FACS experiments). C, D Frequencies of LSK, HSCs and LMPPs as percent of CD45+ cells in control and KMT2A::AFF1Vav-Cre+ FLs at (C) E14.5 (3 experiments) and (D) E18.5 (6 experiments). E Flow cytometry plots of LSK compartment in control and KMT2A::AFF1Vav-Cre+ FLs at E18.5. Cells were gated CD45+Lin–CD19–. Further gating is indicated in the figure showing LSK (top) and LMPPs (bottom). Numbers are mean percentages of total CD45+ cells. F Representative photos of E18.5 HSCs (left) and LMPPs (right) bulk liquid cultures at day 7. Genotypes (control and KMT2A::AFF1Vav-Cre+) are indicated. G Schematic illustration of single-cell OP9 co-culture workflow and subsequent evaluation of B and myeloid linage potentials using flow cytometry. H, I Single HSCs (H) and LMPPs (I) were co-cultured on OP9 stroma. Frequency of colonies (left) and linage output (right) for control and KMT2A::AFF1Vav-Cre+ FLs at E18.5 are shown. Colonies were defined as B, myeloid; B and myeloid (B & My) or not designated to any of these (none) (3 experiments). J Cell cycle analysis of HSCs (left) and MPPs (LSKCD150-CD48+)(right) at E18.5. Mean percentages of cells in G0/G1 and S/G2/M for each population are shown (1–2 embryos per genotype,1 experiment). Bars show means ± SD, and each dot represents an individual embryo. *p ≤ 0.05; ** p ≤ 0.01; ****p ≤ 0.0001; n.s. not significant.
We observed that KMT2A::AFF1Vav-Cre+ pups were born at slightly lower Mendelian ratios than littermate controls. They were also smaller in size and did not survive beyond the neonatal period. The spleens of these pups were significantly reduced and contained less mature lymphoid and erythroid cells, whereas the number of myeloid CD11B+GR1+ cells were comparable to controls (Supplementary Fig. 1C–E). In addition, HSPCs were clearly affected, with the LSK compartment reduced almost twofold (LSK; negative for mature lineage markers Lin-; SCA1+; cKIT+) (Supplementary Fig. 1F).
Thus, KMT2A::AFF1 induction with Vav-Cre recombinase triggered a postnatal hematopoietic phenotype affecting HSPCs as well as more mature blood cell lineages.
KMT2A::AFF1 impacts proliferation and lineage output of HSPCs
Human KMT2A::AFF1 disease initiation is recognized to occur in utero; therefore, we next explored the consequences of KMT2A::AFF1 during embryogenesis. As a window of susceptibility to KMT2A::AFF1 has been suggested between E12-14 in mice [16], FLs from E14.5 were investigated. Here we observed a mild phenotype within the HSPC compartment, mainly seen as a reduction in lympho-myeloid primed progenitors (LMPPs; LSKFLT3+) [27] (Fig. 1C; Supplementary Fig. 2A). As the phenotype was mild compared to what was seen postnatally, later fetal stages were investigated. At E18.5, a more pronounced effect of the oncogene was seen, with a reduction in the LSK compartment, both in frequency and absolute number, along with a continued decrease in LMPPs, as seen at E14.5 (Fig. 1D, E; Supplementary Fig. 2B, C). Functionally, these LMPPs proliferated similar to control cells in vitro, whereas KMT2A::AFF1+ HSCs (LSKFLT3-CD150+CD48-) [28, 29] were found to expand less than controls (Fig. 1F). Additional experiments evaluating proliferation and lineage output at the single-cell level were therefore performed. In agreement with the results from the bulk cultures, a significant reduction in colony formation of single KMT2A::AFF1+ HSCs compared to control was observed (2.9% vs 27.5%) (Fig. 1G, H). In the colonies produced from KMT2A::AFF1+ HSCs, B lineage output was sparse, and clones were biased towards the myeloid lineage (Fig. 1H). No difference in colony formation was observed between KMT2A::AFF1+ LMPPs and control cells, however, as observed for HSCs, lineage output was skewed towards the myeloid lineage (Fig. 1I). In agreement with our in vitro data, cell cycle analysis showed that KMT2A::AFF1+ HSCs had a higher proportion of cells in G0/G1 compared to control (91% vs 85%) (Fig. 1J).
Together, our findings show that KMT2A::AFF1 exerts differential effects across early hematopoietic populations. While HSC proliferation was impaired, LMPPs retained their colony-forming capacity. Both populations, however, exhibited a bias towards myeloid output compared to controls.
A unique CD24+ PreProB population emerges upon KMT2A::AFF1 induction in embryonic HSPCs
We next explored the impact of the KMT2A::AFF1 oncogene on the B progenitor compartment at E18.5 [30, 31] (gating strategy in Supplementary Fig. 3A). Using flow cytometry, both PreProB (B220+CD43+CD19-) and ProB (B220+CD43+CD19+IGM-cKIT+) progenitors were significantly increased in KMT2A::AFF1+ embryos compared to littermate controls. Furthermore, within the B220+CD43+ population, cells with a CD24+CD19- phenotype were observed; a population hardly detected in the control animals (Fig. 2A, B; Supplementary Fig. 3B). CD24 is a surface marker that is typically dim on PreProB progenitors and is first upregulated at the CD19+ ProB cell stage [30, 31]. When the B220+CD93+ compartment was visualized on a flow cytometry UMAP, the KMT2A::AFF1 PreProBs clustered with control, despite disparities in CD24 expression (Fig. 2C). The majority of the KMT2A::AFF1+ PreProB cells (independent of CD24 status) also had surface expression of the progenitor marker cKIT, which was lower on control cells (Supplementary Fig. 3C). Of note, an increase in CD24+ PreProB cells could be observed already at E14.5 (Supplementary Fig. 3D). Functionally, the KMT2A::AFF1 CD24+ PreProBs at E18.5 displayed an increased cloning capacity (6% vs 18%, respectively) and notable lineage plasticity, giving rise to both B and myeloid colonies, whereas control cells predominantly generated B cell colonies (Fig. 2D).
A Flow cytometry plots of B progenitor compartment in control and KMT2A::AFF1Vav-Cre+ embryos at E18.5. Numbers show mean percentages of total CD45+ cells. B Frequencies of B progenitors as percentage of CD45+ cells in control and KMT2A::AFF1Vav-Cre+ FLs at E18.5 (6 experiments). C UMAP visualizations of flow cytometry data from control and KMT2A::AFF1Vav-Cre+ embryos, displaying different B progenitors in E18.5 FLs. Combined UMAP is shown to the left, and per genotype to the right (3–5 embryos/genotype, down sampled to 10.000cells/embryo, one experiment). D Single CD24+ PreProBs were co-cultured on OP9 stroma. Frequency of colonies (left) and linage output (right) for control and KMT2A::AFF1Vav-Cre+ FLs at E18.5 are shown. Colonies were defined as B (CD19+B220+), myeloid (CD11B+GR1+); B and myeloid (B & My) or not described to any of these (none) (3 experiments). E, F B progenitor compartment in control and (E) KMT2A::AFF1Rag1-Cre+ or (F) KMT2A::AFF1Mb1-Cre+ embryos at E18.5. Flow cytometry plots (left) and frequencies of CD24+ PreProBs as percentage of CD45+ cells (right) (3–4 experiments). G Relative expression (normalized to B-actin) of Hoxa9 in purified E18.5 CD24+ PreProBs from controls and KMT2A::AFF1 Vav-Cre; Rag1-Cre and Mb1-Cre respectively. Box plots define lower and upper quartiles, and whiskers min to max values. (KMT2A::AFF1Rag1-Cre+ embryos were heterozygous or homozygous for KMT2A::AFF1Cola1-tetO.) Bars show means ± SD and each dot represents an individual embryo. *p ≤ 0.05; ****p ≤ 0.0001. n.s. not significant.
In an earlier study, using a different murine KMT2A::AFF1 model, long latency lymphomas developed when the fusion was induced in lymphoid progenitors [14]. Thus, we sought to assess whether the CD24+ PreProB population would emerge in our model if KMT2A::AFF1 was induced in lymphoid committed progenitors in the embryo. Rag-1 (Recombination activating gene 1) is involved in VDJ recombination and expressed in lymphoid progenitors in adults, and lympho-myeloid committed progenitors in the embryo [32, 33], whereas Mb1 (also named Cd79a) is part of the B cell antigen receptor. Hence, KMT2A::AFF1 expression was induced using the Rag1-Cre [21] and Mb1-Cre [22] strains (Supplementary Fig. 4A, B). Analysis of the PreProB compartment at E18.5 showed no significant difference between KMT2A::AFF1+ and control cells at this stage and Hoxa9 expression was only marginally increased compared to control (Fig. 2E–G; Supplementary Fig. 4C–F). Thus, a unique CD24+ PreProB population emerges specifically upon KMT2A::AFF1 induction in HSPCs.
B progenitor compartment is unaffected by postnatal induction of KMT2A::AFF1
To explore the consequences of a postnatal induction of the oncogene, pregnant females were given doxycycline (dox) to delay the induction of KMT2A::AFF1 (Fig. 3A). Expression of KMT2A::AFF1 was analyzed after birth and confirmed at ∼ day 40. In adult mice ( > 8 weeks) a mild phenotype was observed, including a reduction in LMPPs, whereas HSCs were unaffected (Fig. 3B–D; Supplementary Fig. 5A). Functionally, KMT2A::AFF1 induced myeloid bias in both HSCs and LMPPs, and KMT2A::AFF1+ HSCs formed fewer colonies, and a smaller proportion of cells were actively cycling (Fig. 3E–G). Thus, the phenotype observed in the LSK compartment in postnatally induced mice resembled that of their embryonically induced counterparts.
A Schematic illustration of postnatal induction of KMT2A::AFF1 by administration of Doxycycline (Dox) food to the female from approx. day 5 to 18 of pregnancy. KMT2A::AFF1 expression was analyzed after ∼6-weeks and phenotype at 8–30 weeks. B Relative expression (normalized to B-actin) of KMT2A::AFF1 in purified HSCs, LMPPs, PreProBs (CD24-/+) and ProBs in bone marrows (BMs) from ∼6-week-old mice, treated with Doxycycline as shown in panel (A) (1 FACS experiment). C Frequencies of LSK, HSCs and LMPPs as percent of CD45+ cells in control and KMT2A::AFF1Vav-Cre+ postnatally induced mice (6 experiments, nmales = 7; nfemales = 18). D Flow cytometry plots of LSK compartment in control and KMT2A::AFF1Vav-Cre+ postnatally induced mice. Numbers are mean percentages of total CD45+ cells. E Single HSCs were co-cultured on OP9 stroma. Frequency of colonies (left) and linage output (right) for control and KMT2A::AFF1Vav-Cre+ postnatally induced mice are shown. Colony distribution defined as B (CD19+B220+), myeloid (CD11B+GR1+); B and myeloid (B & My) or not described to any of these (none) (3 experiments). F Cell cycle analysis of HSCs from control and KMT2A::AFF1Vav-Cre+ postnatally induced mice. Mean percentages of cells in G0, G1 and S/G2/M are shown (6–8 mice per genotype, 3 experiments). G Single LMPPs were co-cultured on OP9 stroma. Frequency of colonies (left) and linage output (right) for control and KMT2A::AFF1Vav-Cre+ postnatally induced mice are shown. Colony distribution defined as in panel (E) (5 experiments). H Flow cytometry plots of B progenitor compartment in control and KMT2A::AFF1Vav-Cre+ postnatally induced mice. Numbers show mean percentages of total CD45+ cells. I Frequencies of B progenitors as percentage of CD45+ cells in control and KMT2A::AFF1Vav-Cre+ postnatally induced mice (7 experiments, nmales = 8; nfemales = 13–20) (ProB not defined by cKIT expression). J Single CD24+ PreProBs were co-cultured on OP9 stroma. Frequency of colonies (left) and linage output (right) for control and KMT2A::AFF1Vav-Cre+ postnatally induced mice are shown. Colony distribution as defined in panel E (3 experiments). Bars show means ± SD and each dot represents an individual mouse. **p ≤ 0.01; ***p ≤ 0.001; n.s. not significant.
However, when we assessed the B progenitor compartment in these mice, it was largely unaffected, and specifically no expansion of the CD24+ PreProB subset was observed (Fig. 3H–I; Supplementary Fig. 5B, C). The few KMT2A::AFF1 CD24+ PreProB cells seen were purified and analyzed in single-cell stromal co-cultures. However, their cloning capacity and lineage output were comparable to those of their control counterparts (Fig. 3J).
These findings highlight a developmental susceptibility to the oncogene, with the observed expansion of CD24+ PreProBs strictly dependent on KMT2A::AFF1 expression in embryonic HSPCs.
Embryonic CD24+ PreProBs emerging in response to KMT2A::AFF1 display self-renewal capacity
The unique PreProB population observed in the embryo could potentially represent leukemia initiating cells. As such, the cells should possess self-renewal capacity, which was subsequently investigated by serial replating in a semi-solid culture assay (Fig. 4A). After one week of culture the KMT2A::AFF1+ expressing CD24+ PreProBs produced significantly more colonies than control cells and KMT2A::AFF1+ cells were successfully replated for more than four rounds (Fig. 4B). The colonies were primarily of B (B220+CD19+) phenotype, but some myeloid (CD11B+GR1+) cells were also detected during the first weeks of culture (Fig. 4C). Cells with an immunophenotype compatible with CD24+ PreProBs (B220+CD43+CD24+CD19-) were observed during the early replating rounds but gradually decreased over time, consistent with a decline in colony numbers (Supplementary Fig. 6A).
A Schematic illustration of the replating assay in semi-solid media. B Semi-solid in vitro cultures of CD24+ PreProBs control and KMT2A::AFF1Vav-Cre+ fetal livers (FLs) from E18.5. Scoring and replating was done weekly. Total number of colonies/1.000 cells for individual embryos are shown. Means ± SD (4 experiments). C Frequencies of B (CD19+B220+) (blue) and myeloid (CD11B+GR1+) (red) cells detected by flow cytometry at different time points in the replating culture of KMT2A::AFF1Vav-Cre+ CD24+ PreProBs. Mean percentages of total CD45+ cells (left) and representative cytospin (right) of cells at 2nd (top) and 3rd (bottom) replating. D Schematic illustration of in vivo assay. E, F LSK and CD24+ PreProBs from control and KMT2A::AFF1Vav-Cre+ were transplanted into sub-lethally irradiated NSG mice and donor cells assessed in peripheral blood at the indicated time-points. E Mean frequencies of CD45.2 donor cells displayed as percentages of total CD45+ cells. F Lineage distribution (B/ Myeloid/ T) of reconstituted KMT2A::AFF1+ PreProBs and LSK mice shown as mean percentages of total CD45.2+ cells (nLSK= 1–3; nPreProB = 1–9). G Photos (left) and violin plot of white blood cell (WBC) counts (right) of recipient´s spleens at final readout, 2-8 months after transplantation. Immunophenotype (CD24+ PreProBs or LSK) and genotype of donor cells are indicated. H Immunophenotype of recipient´s bone marrow (BM) transplanted with CD24+ PreProBs from KMT2A::AFF1Vav-Cre+ embryos. Cells were gated for donor CD45.2+ cells and negative for lineage markers. CD19 and IL7R are displayed for individual mice. Numbers show percentage of total CD45.2+ cells. I UMAP visualizations of flow cytometry data from transplanted KMT2A::AFF1Vav-Cre+ embryos, displaying different B progenitors. Combined UMAP of CD45.2+7-AAD-Lin- cells (4 PreProBs, 2 LSKs) (top, left), one KMT2A::AFF1+ LSK (top, right) and three different KMT2A::AFF1+ CD24+ PreProB recipients (bottom). Down sampled to 30.000 cells/mouse. **p ≤ 0.01; ****p ≤ 0.0001.
The observed self-renewal capacity in vitro led to further investigations in vivo. Since the KMT2A::AFF1Vav-Cre+ mice did not survive the postnatal period, these investigations were conducted in an adult transplantation setting. NOD-SCID-GAMMA (Il2rg-/-) (NSG) mice were used as recipients, as these lack lymphoid cells, allowing for donor B cells to expand without competition. LSKs and CD24+ PreProBs from KMT2A::AFF1+ and control embryos were subsequently transplanted into sub-lethally irradiated NSG mice (Fig. 4D). None of the control PreProB recipients showed any long-term reconstitution in peripheral blood, whereas a majority of the KMT2A::AFF1+ CD24+ PreProB transplanted mice displayed long-term engraftment, with levels close to those obtained with LSKs (Fig. 4E). Reconstituted KMT2A::AFF1+ CD24+ PreProB mice also displayed multilineage engraftment, with donor-derived B (CD19+), T (CD3+) and myeloid (CD11B+GR1+) cells detected in peripheral blood (Fig. 4F). High engraftment levels were also observed in the BM (5 out of 7 analyzed mice; Supplementary Fig. 6B) and several of these mice had enlarged spleens (Fig. 4G).
Additionally, despite transplanting an immunophenotypically strict population, the CD24+ PreProB reconstituted mice exhibited diverse phenotypes. Some displayed an increase in an interleukin 7 receptor (IL7R) expressing population, that was negative for mature lymphoid markers (CD19, CD3, NK1.1) as highlighted in an unsupervised UMAP (Fig. 4H–I; Supplementary Fig. 6C). IL7R is known to define common lymphoid progenitors in mice, and essential for the development of B and T cells [34,35,36]. Hence, these mice exhibit a phenotype indicative of a more immature lymphoid population. Of note, mice #6 and #8 displayed signs of illness and were sacrificed prematurely at 2 and 5 months respectively. Mouse (#6) had low engraftment levels in the bone marrow (4.6%). Despite this, the Lin-CD19-IL7R+ population was observed. Some mice also displayed a partial block in B cell differentiation, and mouse #7 had an aberrant CD24+ PreProB population (Supplementary Fig. 6D). While the immature lymphoid population observed is reminiscent of findings in a previous murine ALL model [37], we did not observe any differentiation block in the mice transplanted with KMT2A::AFF1+ LSK cells.
Taken together, our results demonstrate distinct properties of the oncogene in different cell populations. Notably, the PreProB subset, marked by surface CD24 expression, exhibits self-renewal capacity and reduced differentiation potential, suggesting a role as leukemia-initiating cells.
KMT2A::AFF1 induces transcriptional changes in fetal and adult HSPCs
To assess the molecular impact of KMT2A::AFF1 induction, multimodal single-cell RNA-sequencing (RNA-seq) using CITE-seq (cellular-indexing-of-transcriptomes-and-epitopes) was performed [23]. The analysis included PreProB cells (irrespective of CD24 expression), as well as LSKs and ProB cells from both E18.5 embryos and postnatally induced adult mice (Fig. 5A–C; Supplementary Fig. 7A–C; Supplementary Table 1). The UMAPs, divided by developmental stage, had distinct cell states based on expression of established marker genes such as Mllt3, Il7r and Mpo (Fig. 5C; Supplementary Fig. 7C–F).
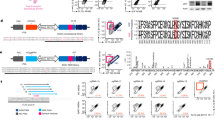
A Schematic illustration of the single-cell RNA-seq experimental workflow. B, C UMAPs of LSK, PreProB and ProB cells from control and KMT2A::AFF1Vav-Cre+ E18.5 embryos. B Color coded based on purified population and genotype and (C) based on cluster. D Lineage affiliation of shared up- (blue) and down-regulated (brown) DEGs in KMT2A::AFF1Vav-Cre+ LSKs compared to control, analyzed with CellRadar (p adj<0.01; |log2 foldchange | >0.5). E UMAPs of LSK, PreProBs and ProB cells from control and KMT2A::AFF1Vav-Cre+ E18.5 embryos (top) and adult (bottom), highlighting Gata2 expressing LSK cells. Down sampled to equal cell number. F Dot plot displaying the top up- and down-regulated DEGs in KMT2A::AFF1Vav-Cre+ PreProBs compared to control at E18.5 for all purified populations. Red indicates up- and blue down-regulated genes. G Lineage affiliation of significantly up- (purple) and down-regulated (blue) DEGs in KMT2A::AFF1Vav-Cre+ PreProBs compared to control at E18.5, analyzed with CellRadar (p adj < 0.01; |log2 foldchange | >1.0).
The most striking functional consequences of the oncogene in the HSPC compartment were a general myeloid bias and a reduced cloning capacity of HSCs, and these phenotypes were independent of developmental states of induction (Fig. 1H–J; Fig. 3E–G). We therefore extracted developmentally shared differentially expressed genes (DEGs) in KMT2A::AFF1+ LSKs compared to control (Supplementary Fig. 7G; Supplementary Table 2). The up-regulated shared DEGs were associated with ‘LT-HSC’, whereas the down-regulated genes were linked to a more lymphoid signature, in agreement with the myeloid bias observed functionally (Fig. 5D). Among the joint up-regulated genes we also observed the transcription factor Gata2 (Fig. 5E; Supplementary Table 2), which might be relevant as increased activity of Gata2 induces quiescence of HSCs in both mice and human [38]. Thus, the molecular landscape of the HSPC compartment underscores the impact of KMT2A::AFF1 on HSCs proliferation, as well as its general effect on lineage affiliation, regardless of developmental state.
Embryonic KMT2A::AFF1+ PreProBs display a pre-leukemic stem cell signature
Subsequently, we compared KMT2A::AFF1+ PreProBs and ProBs from fetal and adult stages to their age-matched normal counterparts. The fetal PreProB stood out as most different to normal counterparts with >250 DEGs (p adj<0.01; |log2 fold change | >1.0; Supplementary Fig. 7H). KMT2A::AFF1+ target genes, such as Hoxa9 and Meis1 were highly expressed [26], and genes associated with stemness such as Hlf and Mecom (Evi1) were up-regulated [39, 40] (Fig. 5F; Supplementary Fig. 7H). Furthermore, the up-regulated genes were predominantly affiliated with ‘LT-HSC’ while the down-regulated genes were more closely associated with ‘ProB’ (Fig. 5G), together indicative of a differentiation block.
We next visualized the PreProBs alongside LSK and ProB cells in the combined UMAP. Adult PreProBs, regardless of KMT2A::AFF1+ status, and fetal control cells all clustered together. In contrast, a fraction of the fetal KMT2A::AFF1+ PreProBs clustered with molecularly more primitive cells within the LSK compartment (Fig. 6A; Supplementary Fig. 8A).
A UMAPs of LSK, PreProBs and ProBs from control and KMT2A::AFF1Vav-Cre+ embryos, highlighting PreProBs in control (top) and KMT2A::AFF1Vav-cre+ (bottom), respectively. B UMAP of PreProBs from control and KMT2A::AFF1Vav-Cre+ embryos, with the different clusters (left) and displaying the cells from cluster #5 on the UMAP from Fig. 5B (right). C Venn diagram of up- and downregulated genes in cluster #5 compared to control PreProBs and control cells in cluster #0 from the combined UMAP (“molecular HSC cluster”). The universal up- and downregulated genes formed the pre-leukemic signature. D UMAP displaying co-expression of Hmga2 and Cd24a in KMT2A::AFF1 expressing cells at E18.5 (left) and in postnatally induced mice (right). Pie charts show percentage of cells expressing each gene in Cre+ and Cre- cells, respectively. E Boxplot showing z-scored mean expression of the up-regulated genes from the pre-leukemic signature in different subtypes of ALL (n = 67–254) from bulk RNA-seq samples [46]. The box includes first to third quartile and the line indicates the median. F UMAP of healthy BM [48]. G Expression of the pre-leukemic signature in individual KMT2A::AFF1 infant ALL samples [47], mapped onto the UMAP in panel (F). Color scale set to the same range for all samples (0.35). Ages of the patients are listed on top. H Lack of expression of the pre-leukemic signature in the healthy BM reference. I Bar plots displaying predicted cell type (from healthy BM in F) of the signature positive cells in KMT2A::AFF1 leukemic patient samples [47]. J Violin plot displaying the UCell score of the pre-leukemic signature among different KMT2A rearranged infant ALLs [48]. K Boxplot of HMGA2 expression in different subtypes of ALL (n = 67–254) [46] displayed per age group. The box includes first to third quartile and the line indicates the median. L Heatmap displaying expression of selected genes in CD24+CD19- cells from KMT2A::AFF1 infants [47]. ****p adj ≤ 0.0001.
Due to the molecular heterogeneity seen, a separate UMAP displaying only PreProBs was generated. Specifically, one cluster in this UMAP (#5) contained primarily KMT2A::AFF1+ cells. When assessed in the combined UMAP, these cells mapped to the primitive cells and importantly expressed Cd24a (Fig. 6B; Supplementary Fig. 8B, C), linking it to the expanded unique population seen by flow cytometry.
To obtain the molecular signature of the pre-malignant cells, the PreProB cluster #5 cells were compared to their normal PreProB counterparts as well as to the control cells in cluster 0 (molecular ‘HSCs’) from the combined UMAP (Fig. 5C). The comparison rendered 48 commonly up-regulated genes, including KMT2A::AFF1 target genes such as Meis1 and Hoxa genes (Fig. 6C; Supplementary Table 3). As the murine pre-leukemic clone was unique to ontogeny, we assessed the signature for genes characteristic to fetal hematopoiesis. One such gene is high-mobility-group-AT-hook-2 (Hmga2), which is higher expressed during ontogeny in both mouse and human [41,42,43,44]. It has also been reported as a direct target of KMT2A::AFF1 [26, 45]. As the expanded PreProB population was defined by surface expression of CD24, the relationship between Hmga2 and Cd24a was assessed. We found that the genes were largely co-expressed in the embryo (48-59%), in contrast to adults, where only a small fraction of the cells (9-12%) co-expressed the genes (Fig. 6D).
Collectively, these data define the transcriptional signature of the pre-leukemic cells and links it to the expanded CD24+ population seen in the embryo.
The pre-leukemic KMT2A::AFF1 signature is transferable to leukemic patient samples
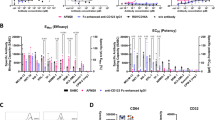
Finally, to explore the relevance of the identified signature in human disease, the genes were converted to their human counterparts and expression investigated in patient ALL samples. When analyzing bulk RNA-seq data from different subtypes of ALL [46], the pre-leukemic signature was found significantly enriched in KMT2A::AFF1 patients (Fig. 6E). To evaluate expression patterns of the signature in infant KMT2A::AFF1 ALL, two single-cell RNA-seq data sets [47, 48] were investigated. To resolve the hierarchy among the leukemic blasts, cells were first mapped onto a healthy BM reference [48] (Fig. 6F). Signature positive cells were found to variable degree in the patients, but not in the healthy control. Overall, the positive cells primarily projected to Common Lymphoid Progenitor (CLP) and PreProB cell types on the healthy reference, linking it to the murine pre-leukemic population (Fig. 6G–I, Supplementary Fig. 8D–F, Supplementary Table 4). The frequency of positive cells however differed between the datasets investigated, which could be due to differences in purification or methodology. We also detected the signature in some patients with KMT2A::MLLT1 or KMT2A::MLLT3 rearrangements (Fig. 6J; Supplementary Table 4), suggesting that the signature can be broadly applicable to KMT2A rearranged leukemia.
Hmga2 is, as mentioned, associated with fetal hematopoiesis [41,42,43,44] and a potential target of KMT2A::AFF1 [26, 45], making it an essential part of the pre-leukemic signature. We therefore explored expression of HMGA2 in different sub-types of ALL [46], and found it upregulated in KMT2A::AFF1-driven leukemia and enriched in infants. HMGA2 also played an important role in expansion of leukemic cells as its knock down in KMT2A::AFF1 cell lines reduced proliferation, consistent with a previous study [45] (Fig. 6K; Supplementary Fig. 8G). As the murine pre-leukemic PreProB cells could be defined by CD24+CD19- and expression of Hmga2, we explored the existence of an equivalent subset in the human setting. In the healthy BM control, HMGA2 expression was confined to the primitive CD34 positive HSPC cluster, and CD24 was mainly seen in the B cell compartment, in line with the expression pattern in human B cell development [49] (Supplementary Fig. 8H). Next, in infant ALLs, transcriptionally CD24+CD19- cells were selected from the single-cell data sets and HMGA2 expression investigated. Four KMT2A::AFF1 patients (n = 11) had a sub-fraction of variable size that also expressed HMGA2, a feature that was rare in the KMT2A::MLLT1 and KMT2A::MLLT3 infants (Fig. 6L; Supplementary Fig. 8I, Supplementary Table 4).
Thus, the ontogeny-specific transcriptional program identified in our murine model is enriched in KMT2A::AFF1 patients, providing critical insights into the transcriptional alterations that drive human disease progression.
Discussion
KMT2A::AFF1 infant ALL stands out as a disease of its own, with a specific clinical manifestation and unfavorable prognosis [3]. Adding to that, murine models have not been able to fully recapitulate the disease [13]. Here, using a novel KMT2A::AFF1 murine model we obtained novel insights into the disease initiating steps in the embryo of relevance to human disease. By combining the Tet-op promoter with Cre-LoxP technology, we generated an inducible system that allowed for cell-specific expression within the hematopoietic system. Induction of the onco-fusion in the HSPC compartment revealed a bias towards myeloid output in vitro and a selective negative impact on the cloning capacity of HSCs. The former is interesting from a clinical perspective as KMT2A::AFF1 ALL is known to be able to switch lineage upon relapse [50]. The latter feature may be linked to increased levels of Gata2, known to induce quiescence in HSCs [38]. Gata2 activity was most prominent in the more primitive clusters, highlighting that this effect could be cell type specific, which would be in line with our functional data. Additionally, the finding is not specific to the KMT2A::AFF1 onco-fusion but has also been reported for KMT2A::MLLT1 [25]. The cell cycle status of cancer cells is a crucial factor in therapy response, as current treatments primarily target actively dividing cells.
The impact of KMT2A::AFF1 on the HSPC compartment was independent of developmental stage as we observed similar outcomes in both embryonic and postnatally induced mice. The expansion of the B compartment was however specific to the embryo. The immunophenotype was consistent with PreProBs (B220+CD43+CD19-), except for surface expression of CD24 (also known as heat stable antigen; HSA), a cell adhesion marker normally upregulated later in the B cell hierarchy, on CD19+ ProBs [30, 31]. The pre-malignant potential of the KMT2A::AFF1+ CD24+ PreProBs was further explored in vivo. As only ∼500-1300 CD24+ PreProBs were transplanted to each recipient, the immune-deficient NSG model was used, thereby allowing the cells to expand without competition. In this setting the cells displayed lineage plasticity and self-renewal potential. Other pathological features, such as long-term engraftment, splenomegaly and a block in differentiation were observed. Thus, our model allowed for identification of a novel pre-malignant state, that we extensively characterized. Notably, we did not observe development of acute leukemia in our transplantation setting, and it is likely that additional factors are needed for transformation, at least in the murine setting. Two micro RNAs, miR-130b and miR-128a, have been identified as important co-drivers for the conversion [37], and inflammatory stimulus [12, 51] may also be important for the progression to fulminant disease.
The pre-leukemic cells were exclusive to induction with Vav-Cre, and were not observed upon targeting more lymphoid progenitors. This suggests that the cell of origin of pre-leukemia is found within the HSPC compartment. One could speculate as to why these committed cells were not susceptible to transformation. One possible explanation could be that the accessibility to chromatin of key target genes of KMT2A::AFF1 was restricted at the progenitor stages. In line with this, Hoxa9 was only marginally increased in CD24+ PreProBs from the KMT2A::AFF1 Rag1-Cre and Mb1-Cre models. Notably, the pre-leukemic cells were absent in postnatally induced adult mice. In contrast, the myeloid skewing observed within the HSPC compartment occurred regardless of developmental stage, suggesting it is a consequence of oncogenic effects on definitive HSPCs. Meanwhile, the expansion of the B cell compartment was specific to the embryonic context and likely driven by fetal, transient progenitors. In line with that, Vav-Cre is expressed early in hematopoiesis, targeting definitive HSCs but also cells from the intermediate wave, including pre-HSCs and MPPs [20]. A recent publication highlights that most cells populating the FL during gestation are from the non-definitive wave, with little contribution from definitive HSCs [52]. These findings suggest that the pre-leukemic cells may originate early, potentially from pre-HSCs or even an earlier wave of hematopoiesis.
Our transcriptional analysis revealed an unexpected heterogeneity in the PreProB subset expressing the onco-fusion. A subset of the KMT2A::AFF1+ PreProBs clustered together, exhibiting upregulation of KMT2A::AFF1 target genes and stemness genes. This cluster expressed Cd24a, linking the molecular profile of a self-renewing early B progenitor to the distinct pre-leukemic population we identified functionally and prospectively through our study. Thus, the unique CD24+ PreProB population identified here displayed stem cell properties in vivo, as well as a transcriptional stemness signature, likely representing the initiating step of disease development. Furthermore, the pre-leukemic signature was detected to varying degrees in KMT2A-rearranged patient samples, including blasts aligning with the CLP and PreProB compartments in a normal reference. This observation aligns with the pre-leukemic cells from which the signature was derived. The characteristic of Cd24a-Hmga2 co-expression in the murine model could to some extent be observed in the KMT2A::AFF1 infants, and is interesting from a therapeutic perspective, where knockdown of HMGA2 reduced proliferation of KMT2A::AFF1 ALL cell lines [45]. Additionally, CD24 is a surface marker explored as a promising cancer immunotherapy target [53, 54], and has also been linked to leukemia initiating cell (LICs) in KMT2A::AFF1 ALL [55]. Thus, the distinct CD24 expression on the pre-leukemic cells presents an attractive therapeutic opportunity to explore further.
Altogether, our model captures an embryonic, in situ pre-leukemic state driven by KMT2A::AFF1, providing unique insight into the earliest stages of leukemogenesis. These findings offer a foundation for future strategies aimed at intercepting infant ALL at its developmental origin.
Data availability
The data generated within this study have been deposited on GEO under GSE251658. The human samples used in the study can be found here: single-cell RNA-seq of healthy BM and KMT2A rearranged samples [48] at the Human Tumor Atlas Network (HTAN) (https://humantumoratlas.org/); single-cell RNA-seq of the 6 KMT2A::AFF1 patient samples [47] are all deposited together in the European Genome-phenome Archive (EGA) under EGAS00001003986 (and available upon approval by the Princess Máxima Data Access Committee); bulk RNA-seq of ALL patient samples [46] https://pecan.stjude.cloud/static/hg19/pan-all/BALL-1988S-HTSeq.zip. The code can be found in a Github repository: https://github.com/sara-palo/calderon_et_al.
References
Pieters R, De Lorenzo P, Ancliffe P, Aversa LA, Brethon B, Biondi A, et al. Outcome of infants younger than 1 year with acute lymphoblastic leukemia treated with the interfant-06 protocol: results from an international phase III randomized study. J Clin Oncol. 2019;37:2246–56.
Pieters R, Schrappe M, De Lorenzo P, Hann I, De Rossi G, Felice M, et al. A treatment protocol for infants younger than 1 year with acute lymphoblastic leukaemia (Interfant-99): an observational study and a multicentre randomised trial. Lancet. 2007;370:240–50.
Rice S, Roy A. MLL-rearranged infant leukaemia: A ‘thorn in the side’ of a remarkable success story. Biochim Biophys Acta Gene Regul Mech. 2020;1863:194564.
Meyer C, Larghero P, Almeida Lopes B, Burmeister T, Groger D, Sutton R, et al. The KMT2A recombinome of acute leukemias in 2023. Leukemia. 2023;37:988–1005.
Winters AC, Bernt KM. MLL-rearranged leukemias-an update on science and clinical approaches. Front Pediatr. 2017;5:4.
Armstrong SA, Staunton JE, Silverman LB, Pieters R, den Boer ML, Minden MD, et al. MLL translocations specify a distinct gene expression profile that distinguishes a unique leukemia. Nat Genet. 2002;30:41–47.
Andersson AK, Ma J, Wang J, Chen X, Gedman AL, Dang J, et al. The landscape of somatic mutations in infant MLL-rearranged acute lymphoblastic leukemias. Nat Genet. 2015;47:330–7.
Hein D, Borkhardt A, Fischer U. Insights into the prenatal origin of childhood acute lymphoblastic leukemia. Cancer Metastasis Rev. 2020;39:161–71.
Gale KB, Ford AM, Repp R, Borkhardt A, Keller C, Eden OB, et al. Backtracking leukemia to birth: identification of clonotypic gene fusion sequences in neonatal blood spots. Proc Natl Acad Sci USA. 1997;94:13950–4.
Dzierzak E, Bigas A. Blood development: hematopoietic stem cell dependence and independence. Cell Stem Cell. 2018;22:639–51.
Chen W, Li Q, Hudson WA, Kumar A, Kirchhof N, Kersey JH. A murine Mll-AF4 knock-in model results in lymphoid and myeloid deregulation and hematologic malignancy. Blood. 2006;108:669–77.
Krivtsov AV, Feng Z, Lemieux ME, Faber J, Vempati S, Sinha AU, et al. H3K79 methylation profiles define murine and human MLL-AF4 leukemias. Cancer cell. 2008;14:355–68.
Milne TA. Mouse models of MLL leukemia: recapitulating the human disease. Blood. 2017;129:2217–23.
Metzler M, Forster A, Pannell R, Arends MJ, Daser A, Lobato MN, et al. A conditional model of MLL-AF4 B-cell tumourigenesis using invertor technology. Oncogene. 2006;25:3093–103.
Duguid A, Mattiucci D, Ottersbach K. Infant leukaemia - faithful models, cell of origin and the niche. Dis Model Mech. 2021;14:dmm049189.
Barrett NA, Malouf C, Kapeni C, Bacon WA, Giotopoulos G, Jacobsen SEW, et al. Mll-AF4 confers enhanced self-renewal and lymphoid potential during a restricted window in development. Cell Rep. 2016;16:1039–54.
Lin S, Luo RT, Ptasinska A, Kerry J, Assi SA, Wunderlich M, et al. Instructive role of MLL-fusion proteins revealed by a model of t(4;11) Pro-B acute lymphoblastic leukemia. Cancer cell. 2016;30:737–49.
Rice S, Jackson T, Crump NT, Fordham N, Elliott N, O’Byrne S, et al. A human fetal liver-derived infant MLL-AF4 acute lymphoblastic leukemia model reveals a distinct fetal gene expression program. Nat Commun. 2021;12:6905.
Li L, Tasic B, Micheva KD, Ivanov VM, Spletter ML, Smith SJ, et al. Visualizing the distribution of synapses from individual neurons in the mouse brain. PLoS One. 2010;5:e11503.
Stadtfeld M, Graf T. Assessing the role of hematopoietic plasticity for endothelial and hepatocyte development by non-invasive lineage tracing. Development. 2005;132:203–13.
McCormack MP, Forster A, Drynan L, Pannell R, Rabbitts TH. The LMO2 T-cell oncogene is activated via chromosomal translocations or retroviral insertion during gene therapy but has no mandatory role in normal T-cell development. Mol Cell Biol. 2003;23:9003–13.
Hobeika E, Thiemann S, Storch B, Jumaa H, Nielsen PJ, Pelanda R, et al. Testing gene function early in the B cell lineage in mb1-cre mice. Proc Natl Acad Sci USA. 2006;103:13789–94.
Stoeckius M, Hafemeister C, Stephenson W, Houck-Loomis B, Chattopadhyay PK, Swerdlow H, et al. Simultaneous epitope and transcriptome measurement in single cells. Nat methods. 2017;14:865–8.
Ouyang JF, Kamaraj US, Cao EY, Rackham OJL. ShinyCell: simple and sharable visualization of single-cell gene expression data. Bioinformatics. 2021;37:3374–6.
Ugale A, Norddahl GL, Wahlestedt M, Sawen P, Jaako P, Pronk CJ, et al. Hematopoietic stem cells are intrinsically protected against MLL-ENL-mediated transformation. Cell Rep. 2014;9:1246–55.
Guenther MG, Lawton LN, Rozovskaia T, Frampton GM, Levine SS, Volkert TL, et al. Aberrant chromatin at genes encoding stem cell regulators in human mixed-lineage leukemia. Genes Dev. 2008;22:3403–8.
Adolfsson J, Mansson R, Buza-Vidas N, Hultquist A, Liuba K, Jensen CT, et al. Identification of Flt3+ lympho-myeloid stem cells lacking erythro-megakaryocytic potential a revised road map for adult blood lineage commitment. Cell. 2005;121:295–306.
Kiel MJ, Yilmaz OH, Iwashita T, Yilmaz OH, Terhorst C, Morrison SJ. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell. 2005;121:1109–21.
Kim I, He S, Yilmaz OH, Kiel MJ, Morrison SJ. Enhanced purification of fetal liver hematopoietic stem cells using SLAM family receptors. Blood. 2006;108:737–44.
Rumfelt LL, Zhou Y, Rowley BM, Shinton SA, Hardy RR. Lineage specification and plasticity in CD19- early B cell precursors. J Exp Med. 2006;203:675–87.
Hardy RR, Carmack CE, Shinton SA, Kemp JD, Hayakawa K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J Exp Med. 1991;173:1213–25.
Boiers C, Carrelha J, Lutteropp M, Luc S, Green JC, Azzoni E, et al. Lymphomyeloid contribution of an immune-restricted progenitor emerging prior to definitive hematopoietic stem cells. Cell Stem Cell. 2013;13:535–48.
Igarashi H, Gregory SC, Yokota T, Sakaguchi N, Kincade PW. Transcription from the RAG1 locus marks the earliest lymphocyte progenitors in bone marrow. Immunity. 2002;17:117–30.
Kondo M, Weissman IL, Akashi K. Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell. 1997;91:661–72.
von Freeden-Jeffry U, Vieira P, Lucian LA, McNeil T, Burdach SE, Murray R. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J Exp Med. 1995;181:1519–26.
Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med. 1994;180:1955–60.
Malouf C, Antunes ETB, O’Dwyer M, Jakobczyk H, Sahm F, Landua SL, et al. miR-130b and miR-128a are essential lineage-specific codrivers of t(4;11) MLL-AF4 acute leukemia. Blood. 2021;138:2066–92.
Tipping AJ, Pina C, Castor A, Hong D, Rodrigues NP, Lazzari L, et al. High GATA-2 expression inhibits human hematopoietic stem and progenitor cell function by effects on cell cycle. Blood. 2009;113:2661–72.
Kataoka K, Sato T, Yoshimi A, Goyama S, Tsuruta T, Kobayashi H, et al. Evi1 is essential for hematopoietic stem cell self-renewal, and its expression marks hematopoietic cells with long-term multilineage repopulating activity. J Exp Med. 2011;208:2403–16.
Komorowska K, Doyle A, Wahlestedt M, Subramaniam A, Debnath S, Chen J, et al. Hepatic leukemia factor maintains quiescence of hematopoietic stem cells and protects the stem cell pool during regeneration. Cell Rep. 2017;21:3514–23.
Yuan J, Nguyen CK, Liu X, Kanellopoulou C, Muljo SA. Lin28b reprograms adult bone marrow hematopoietic progenitors to mediate fetal-like lymphopoiesis. Science. 2012;335:1195–1200.
Copley MR, Babovic S, Benz C, Knapp DJ, Beer PA, Kent DG, et al. The Lin28b-let-7-Hmga2 axis determines the higher self-renewal potential of fetal haematopoietic stem cells. Nat Cell Biol. 2013;15:916–25.
Kumar P, Beck D, Galeev R, Thoms JAI, Talkhoncheh MS, de Jong I, et al. HMGA2 promotes long-term engraftment and myeloerythroid differentiation of human hematopoietic stem and progenitor cells. Blood Adv. 2019;3:681–91.
Sommarin MNE, Olofzon R, Palo S, Dhapola P, Soneji S, Karlsson G, et al. Single-cell multiomics of human fetal hematopoiesis defines a developmental specific population and a fetal signature. Blood Adv. 2023;7:5325–40.
Wu Z, Eguchi-Ishimae M, Yagi C, Iwabuki H, Gao W, Tauchi H, et al. HMGA2 as a potential molecular target in KMT2A-AFF1-positive infant acute lymphoblastic leukaemia. Br J Haematol. 2015;171:818–29.
Gu Z, Churchman ML, Roberts KG, Moore I, Zhou X, Nakitandwe J, et al. PAX5-driven subtypes of B-progenitor acute lymphoblastic leukemia. Nat Genet. 2019;51:296–307.
Candelli T, Schneider P, Garrido Castro P, Jones LA, Bodewes E, Rockx-Brouwer D, et al. Identification and characterization of relapse-initiating cells in MLL-rearranged infant ALL by single-cell transcriptomics. Leukemia. 2022;36:58–67.
Chen C, Yu W, Alikarami F, Qiu Q, Chen CH, Flournoy J, et al. Single-cell multiomics reveals increased plasticity, resistant populations, and stem-cell-like blasts in KMT2A-rearranged leukemia. Blood. 2022;139:2198–211.
Bendall SC, Davis KL, Amir el AD, Tadmor MD, Simonds EF, Chen TJ, et al. Single-cell trajectory detection uncovers progression and regulatory coordination in human B cell development. Cell. 2014;157:714–25.
Rossi JG, Bernasconi AR, Alonso CN, Rubio PL, Gallego MS, Carrara CA, et al. Lineage switch in childhood acute leukemia: an unusual event with poor outcome. Am J Hematol. 2012;87:890–7.
Malouf C, Ottersbach K. Fetal liver Mll-AF4+ hematopoietic stem and progenitor cells respond directly to poly(I:C), but not to a single maternal immune activation. Exp Hematol. 2019;76:49–59.
Yokomizo T, Ideue T, Morino-Koga S, Tham CY, Sato T, Takeda N, et al. Independent origins of fetal liver haematopoietic stem and progenitor cells. Nature. 2022;609:779–84.
Christian SL. CD24 as a potential therapeutic target in patients with B-cell leukemia and lymphoma: current insights. Onco Targets Ther. 2022;15:1391–402.
Barkal AA, Brewer RE, Markovic M, Kowarsky M, Barkal SA, Zaro BW, et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature. 2019;572:392–6.
Aoki Y, Watanabe T, Saito Y, Kuroki Y, Hijikata A, Takagi M, et al. Identification of CD34+ and CD34- leukemia-initiating cells in MLL-rearranged human acute lymphoblastic leukemia. Blood. 2015;125:967–80.
Acknowledgements
We acknowledge Dr Mikael Sommarin for valuable input and discussion for experimental design, Monika Dudenhoffer-Pfeiffer for cloning of the KMT2A::AFF1 fusion gene and Gerd Sten for targeting of ES cells. The Lund Stem Cell Center (SSC) FACS Facility, SSC Cell and Gene Therapy Core, the Lund University Transgenic Animal Facility, Clinical Genomics Lund, SciLifeLab and the Center for Translational Genomics at Lund University are acknowledged for technical support with experiments. This work was supported by the Swedish Childhood Cancer Foundation (TJ2018-0015; HFT2023-0006; PR2018-0111; PR2021-0032; PR2023-0065; CB; PR2022-0091; DB), the Ragnar Söderberg Foundation (M34/18; CB), the Swedish Research Council (2019-01913, 2023-02095 CB; 2022-00932; DB), Swedish Cancer Society (23 3084 Pj CB; 24 3386 Pj DB), the Swedish Society of Medicine (CB), the Märta Winkler Foundation for Medical Research (CB) and the national strategic research area grant StemTherapy.
Funding
Open access funding provided by Lund University.
Author information
Authors and Affiliations
Contributions
ASC performed all the experiments with contributions from SK and RG; DB generated the KMT2A::AFF1 mouse model and revised the manuscript; ME provided input on experiments; KŽ and AS contributed to the shRNA knock down experiments; RWS provided patient data; ZM and SP did the comparison to human data; SL and SS performed the bioinformatic analysis of the sequencing data; ASC analyzed, visualized the data, and wrote the manuscript. CB conceived, design and supervised the study, analyzed data, and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests. Current affiliation for Shabnam Kharazi is NextCell Pharrma AB, Huddinge, Sweden, for Mohamed Eldeeb Acrivon Therapeutics, Lund, Sweden and for Zahra Masoumi Department of Biology, University of York, York, United Kingdom.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Calderón, A.S., Ghazanfari, R., Masoumi, Z. et al. Ontogeny-specific induction of the KMT2A::AFF1-fusion drives development of a distinct CD24 positive pre-leukemic state. Leukemia 39, 2099–2111 (2025). https://doi.org/10.1038/s41375-025-02665-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41375-025-02665-9