Abstract
Maximizing sensitivity while minimizing manufacturing complexity and costs remains a challenge in the development of flexible gas sensors. Two-dimensional transition metal dichalcogenides (TMDCs) have emerged as potential candidates; however, further enhancement of their gas sensitivity is required without increasing fabrication complexity. Here, we report ultrasensitive and selective gas sensors fabricated by mechanically drawing compressed TMDCs on flexible cellulose paper. This method rapidly and cost-effectively integrates nanolayered TMDCs, such as WSe₂ and MoS₂, onto the rough paper surface while abundantly exposing their edge sites. Notably, WSe2 exhibits a high response of ~1978.63% toward 10 ppm NO₂ with exceptional sensitivity (~139.9% ppm⁻¹ at 0.1 to 1 ppm NO2) and an ultra-low detection limit (1.25 ppb), far exceeding the responses to NH3, CO, and six species of volatile organic compounds. These high responses remain stable under severe mechanical deformations. Furthermore, we present a portable wireless sensing device that enables real-time gas monitoring on a smartphone, using the TMDC-drawn paper as a replaceable sensor in a 3D-printed frame. Compared to conventional flexible gas sensors, our approach not only simplifies fabrication but also offers outstanding sensitivity.
Similar content being viewed by others
Introduction
Recent progress in two-dimensional (2D) materials has highlighted their ability to sensitively recognize various analytes1. Among many 2D materials, transition metal dichalcogenides (TMDCs) have attracted significant interest as emerging sensing materials, because of their large surface-to-volume ratio, tunable band structure, rich redox chemistry, and high chemical adsorbability2. These unique properties lead to integrating TMDCs on various classes of substrates for exploring the interaction between TMDCs and target vapors, thereby resulting in the development of gas sensors that function on the basis of charge transfer upon the adsorption of molecules3,4,5,6,7,8,9,10,11. The gas sensitivity of TMDCs has been further improved by controlling the number of layers3,12, engineering defects and edges13,14,15,16, functionalizing the surface17,18,19,20,21, forming Schottky junction22, and irradiating light during sensing cycles23,24. In parallel, theoretical studies predict that NO2 molecules, which can cause serious chronic respiratory disease, can be strongly bound to TMDCs25,26,27. As supported by theory, several TMDC-based sensors enable the sensitive detection of NO2 down to the sub-ppm level6,8,9,11,13,14,15,24.
Based on their superior sensing properties and atomically thin 2D structures, TMDCs are also considered as promising candidates for flexible gas sensors28,29,30,31,32,33. As these sensors can be seamlessly mounted on non-planar surfaces, they would be suitable for constructing wearable sensors that provide environmental information to users. However, despite their potential utility, the reported NO2 sensitivity of TMDC-based flexible sensors34 is typically not as high as that of their rigid counterparts1. In addition, flexible sensors are manufactured by time-consuming, expensive, and sophisticated processes. Specifically, the preparation and integration of TMDCs onto deformable substrates usually require a series of high-temperature synthesis and transfer processes31,32 and routinely involve interactions with various solvents28,30, which can undesirably change the properties of TMDCs35. Thus, it is necessary to develop a sensor with high gas sensitivity that can be fabricated in a simpler and more cost-effective manner.
To address the low sensitivity and complex fabrication of flexible TMDC sensors, we present a highly sensitive device prepared by directly drawing TMDCs onto cellulose paper. Mechanical abrasion of a TMDC pellet leaves nanolayered TMDCs on the rough surface of the paper with abundantly exposed edges, which can provide many adsorption sites, resulting in exceptional chemical sensitivity. Recently, the abraded van der Waals materials have proven to be applicable as high-performance electronic devices, sensors, and energy storage devices36,37,38,39,40,41,42,43. Thus, the mechanical drawing of TMDCs provides a way to integrate them on the paper using a safe, simple, cost-effective, and solvent-free procedure, as previously demonstrated for carbon nanomaterials44,45,46,47,48,49,50,51,52. In addition, the paper substrate would be suitable for realizing foldable and disposable devices, because of its lightweightness, porosity, biodegradability, and flexibility53,54,55. In particular, the large surface area of an entangled fibrous network of cellulose would provide a favorable platform for the integration of nanomaterials and the detection of gases44,45,49,56,57,58,59,60.
In this work, we demonstrate highly sensitive, selective, and flexible gas sensors based on 2D TMDCs mechanically drawn onto cellulose paper. The rapid (~1 min once the pellet is ready) and cost-effective (~$0.05 per sensor for materials) fabrication process involves simple handwriting using silver ink for electrodes and a TMDC pellet for the sensing materials. As exemplary TMDCs, we applied WSe2 or MoS2 nanolayers to paper for the selective detection of NO2 over NH3, CO, and six species of volatile organic compounds (VOCs), and demonstrated significantly higher NO2 responses compared to those of identically drawn carbon nanotubes (CNTs). Particularly, 2D WSe2 shows an ultrahigh sensing response of ~1978.63% toward 10 ppm NO2 exposure in air at room temperature, which is 10.04 and 73.8 times higher than those of MoS2 and CNTs, respectively. In addition, the sensitivity of WSe2 reaches ~139.9% ppm−1 even at exposure to low NO2 concentrations ranging from 0.1 to 1 ppm. All these characteristics enable stable NO2 detection under severe mechanical deformations without compromising the sensitivity, as confirmed by reversible 180° folding and unfolding (bending radius of ~80 μm) with high cyclic durability. Our approach not only outperforms conventional flexible gas sensors but also provides manufacturing simplicity compared to previous solvent-mediated integration methods. Finally, as a proof of concept for a standalone, portable device, a three-dimensional (3D) printed frame was integrated with a data processing unit, wireless module, and battery. The TMDC-drawn paper was integrated as a replaceable sensor and enabled real-time NO2 monitoring via a smartphone, demonstrating its practical utility as a portable and on-demand sensing platform.
Results and discussions
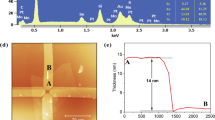
Figure 1a schematically outlines the fabrication process of a mechanically drawn TMDC-based gas sensor. We used WSe2 or MoS2 as a representative TMDC (Fig. 1b–d), and a multiwalled CNT pellet was prepared and drawn on the paper for comparison of the gas sensing performance. It is noted that the drawing process is reliable and reproducible, evidenced by the average mass of WSe2 after five times drawings on the paper, measured at 0.19 ± 0.031 mg (Table S1). This measured mass of WSe2 on paper is also well consistent with the estimated mass of 0.186 mg. TEM images of dispersed and dried WSe2 and MoS2 confirmed their layered structure (Fig. S1). As the sensor is manufactured simply by hand drawing without expensive equipment and complicated procedures, the process can be rapidly (within ~1 min) and cost-effectively (~$0.05 per sensor for materials) completed once the pellet is prepared. The pellet can be used multiple times for the fabrication of a number of sensors that exhibit reproducible sensing responses. In addition, owing to the flexibility of the paper substrate, the sensor can be folded and unfolded, and can be easily trimmed to form the desired shape (Fig. 1e). Thus, our approach is applicable for preparing flexible and disposable gas sensors at any location and time.
2D TMDCs mechanically drawn on a cellulose paper for sensitive and flexible gas sensors. a Schematic of the fabrication process of a TMDC-drawn, paper-based gas sensor. Photographs of the WSe2 and MoS2 pellets (b), silver electrodes drawn on the paper (c), and fabricated gas sensor with mechanically drawn WSe2 (d). e Photograph of the sensor that is folded in half, showing its high flexibility originated from the nature of the paper substrate. f–h SEM images of WSe2 drawn on a cellulose paper. WSe2 covers the surface of cellulose microfibers (f). Enlarged image shows WSe2 nanolayers with abundantly exposed edges (g). Cross-sectional view shows a ~1 μm-thick WSe2 sensing layer (h). The dashed line in (h) indicates the boundary between WSe2 and paper substrate
SEM images in Fig. 1f–h show the WSe2-drawn paper. WSe2 covers the surface of cellulose microfibers following the abrasion trajectory, and the boundary between the WSe2 layer and the paper substrate is clearly shown in Fig. 1f. Owing to the high roughness of the substrate, WSe2 was easily separated from the pellet and bound well on the paper surface. As shown in Fig. 1g, WSe2 nanolayers overlap each other to form an electrical pathway with abundantly exposed edges. It is noted that the edges of TMDCs can improve the chemical sensing responses, as they promote the adsorption of gas molecules13,14,15,16. We also harness this characteristic to achieve ultrahigh gas sensitivity in this work. The thickness of WSe2 after five consecutive drawings was found to be ~1 μm, as shown in the cross-sectional SEM image in Fig. 1h. In addition to WSe2, MoS2 nanolayers were formed by mechanical drawing on the paper substrate. They exhibited similar morphological features such as overlapped layers and exposed edges (Fig. S2). The electrical resistance of the sensor measured between the interdigitated electrodes drastically decreased with increasing the number of TMDC drawings (Figs. S3a and S4). The difference in the resistance between drawn WSe2 and MoS2 could originate from their different sizes (Fig. S5). In addition, Raman and XPS spectra confirm the successful formation of nanolayered TMDCs on the cellulose paper by mechanical drawing of the compressed pellet without any degradation and damage (Fig. S3b–f).
The transient sensing responses of the mechanically drawn WSe2-, MoS2-, and CNT-based sensors were investigated under exposure to 10 ppm NO2. All experiments were conducted in the air environment at room temperature, and the changes in the current were continuously recorded. It is noted that the number of TMDC drawings and the electrode gap were optimized to five times and 2 mm, respectively, to maximize the response (Figs. S6 and S7). In addition to WSe2 and MoS2, a multiwalled CNT-drawn sensor was prepared by the procedure identical to that of TMDCs, and its sensing performance was tested for comparison. These materials were selected to evaluate the gas sensing performance of two representative TMDCs, with CNTs included as a control material based on their prior use in mechanically drawn sensors. The TEM and SEM observations, Raman and XPS spectra, and I–V behavior with respect to the number of CNT drawings verified the successful integration of the CNTs on the surface of the cellulose paper (Fig. S8).
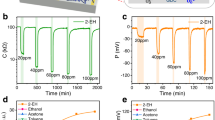
Figure 2a shows the responses of freshly drawn WSe2-, MoS2-, and CNT-based sensors when they were first exposed to 10 ppm NO2 at room temperature. The electrical resistances decreased for all tested sensors upon NO2 exposure. This p-type sensing behavior can be attributed to the increased hole concentration in the sensing materials, because NO2 molecules withdraw electrons when they are adsorbed on the surface6,11,58. Although O2 molecules in air can also be adsorbed onto the sensing material and withdraw electrons, the subsequent exposure to NO2 after equilibrium with background O2 results in a further decrease in sensor resistance. This response is attributed both to the reaction of NO2 with pre-adsorbed O2- ions, which extracts additional electrons from the sensing material, and to the direct electron capture by NO2 through competitive adsorption at active sites61,62. It is noted that NO2 exhibit a higher adsorption energy on TMDCs compared to O227,63, leading to a net increase in hole concentration and thus a further reduction in resistance. Under identical conditions, WSe2 showed an ultrahigh response of 1978.63%, which was 10.04 and 73.8 times higher than those of MoS2 (197.08%) and the CNT (26.81%), respectively. This exceptional response of WSe2 significantly exceeded the reported responses of other 2D-material-based flexible NO2 sensors34. Note that the response of MoS2 was also sufficiently high, and the CNT exhibited a comparable response to that reported previously45. The excellent NO2 reactivity of TMDCs may result from the high adsorbability of the abundantly exposed edge sites after the drawing process. This is further supported by the poor response (~34.48%) of the bulk WSe2 pellet to 10 ppm NO2 exposure, likely due to the limited availability of reactive sites (Fig. S9). This implies that, given the simple, fast, and low-cost preparation process as well as the high initial response, our sensor can be used as a high-performance disposable device. In addition, the incomplete desorption of NO2 can be improved by UV irradiation during recovery cycles, if necessary, while maintaining high responses (Fig. S10). This suggests that integrating the sensing material with a miniaturized light source could be an effective strategy to enhance recovery performance in portable gas sensors.
Transient sensing responses of WSe2-, MoS2-, and CNT-drawn papers under exposure to 10 ppm NO2 at room temperature (18 °C). a Responses of the sensors when they are first exposed to NO2 after the drawing is completed. b Responses of the sensors upon the second exposure to NO2 after the initial reaction. c Consecutive NO2 exposures and corresponding transient responses of the sensors. d Schematics of sensing principles of the WSe2-drawn paper during the first and second exposures to NO2 gas
When the sensors were exposed to NO2 again after their first reaction rather than disposed off, the responses became 73.34%, 44.84%, and 4.54% for the WSe2-, MoS2-, and CNT-drawn sensors, respectively (Fig. 2b). These were lower than the initial responses for all tested sensors; however, the responses of TMDCs were still higher or comparable to those of previously reported flexible and/or paper-based NO2 sensors34,55. In addition, the recovery rate, which is defined as the ratio of the change in response between exposure (ΔRexposure) and recovery (ΔRrecovery) cycles (i.e., ΔRrecovery/ΔRexposure), was significantly improved to 84%, 78.42%, and 71.12% for the WSe2, MoS2, and CNT sensors, respectively. As shown in Fig. 2c, all sensors respond sensitively and stably to subsequent repeated exposures to NO2 molecules. The reduced response compared to the initial reaction results from NO2 molecules remaining on the sensing layer from previous exposures, which occupy adsorption sites and limit further interaction (Fig. 2d). These occupied sites also enhance the recovery rate by preventing additional reactions, resulting in an average recovery of 94.28% for the WSe2-drawn sensor. In addition, the response time was decreased from 14.7 to 3.5 min after the initial cycle, suggesting surface stabilization and faster adsorption kinetics upon repeated NO2 exposure. The WSe2-drawn sensor still exhibits 1.6- and 13.9-times higher response than the MoS2- and CNT-drawn sensors, respectively. This tendency was also observed in the case of NH3 sensing (Fig. S11). This indicates that WSe2 mechanically drawn on the paper can be sensitively operable multiple times.
The different NO2 responses among WSe2, MoS2, and CNT may originate from the differences in the adsorption energy of the gas molecules. Previous studies reported that the adsorption energy of NO2 molecules to WSe264 could be higher than that of MoS265 and CNT66 indicating stronger binding. Moreover, the differences in electronegativity, which is a measure of the tendency of attracting electrons and can be defined as half of the sum of the ionization potential (IP) and electron affinity (EA) (i.e., (IP + EA)/2), drive the electron transfer between TMDCs and NO2 molecules. Considering the electronegativity values of NO2 molecule (5.53–7.34 eV)67,68, WSe2 (4.21–4.34 eV), and MoS2 (5.06–5.26 eV)35, it can be expected that NO2 molecules withdraw electrons from TMDCs once they interact with each other, as demonstrated in our experiments. Under such conditions, a larger difference in electronegativity between WSe2 and NO2 than that between MoS2 and NO2 can lead to a higher charge transfer and, thus, to a higher sensing response.
Figure 3a shows the sensing responses of the WSe2-drawn sensor to NO2 concentrations ranging from 0.1 to 1 ppm at various environmental temperatures from 18 to 150 °C. Increasing the NO2 concentration increases the number of molecules to be adsorbed on WSe2, thereby resulting in a higher charge transfer, and thus the response increases. Here, the response to each NO2 concentration was characterized as the difference between the values before and after the NO2 exposure (for 15 min) by considering the baseline drift. As the environmental temperature increased, the response decreased with exposure to identical NO2 concentrations, whereas the recovery rate improved with a suppressed signal drift. This is predominantly due to the accelerated desorption rate of the physisorbed gas molecules by increased thermal energy8,23. Based on the experimental results, the NO2 concentration- and temperature-dependent sensing responses were characterized, as shown in Fig. 3b. At each temperature, the sensing response as a function of the NO2 concentration can be described by the Langmuir adsorption model (dotted lines in Fig. 3b with R2 = 0.962–0.988 for all measured cases)69. This indicates that the response can be governed by the number of adsorbed molecules at the active sites of WSe2, whereas an increase in the temperature decreases the adsorption rate. In parallel, the sensitivity (i.e., response per ppm NO2) can also be found from linear fitting in the concentration range of 0.1–1 ppm (solid lines in Fig. 3b). The sensitivity at room temperature (18 °C) reached ~139.9% ppm−1 (R2 = 0.948) and decreased to ~133.85% (R2 = 0.980), ~119.7% (R2 = 0.965), and ~94.89 % ppm−1 (R2 = 0.983) at temperatures of 50, 100, and 150 °C, respectively. In addition, the limit of detection (LOD) at 18, 50, 100, and 150 °C are found to be 9.65, 3.36, 1.25, and 1.9 ppb, respectively (see Supporting Information). To the best of our knowledge, our sensor significantly outperforms the reported TMDC-based flexible NO2 sensors with increased sensitivity by several orders of magnitude and the lowest LOD (Table 1). This comparison (Table 1) highlights the superior performance of our sensor. The ultra-low LOD, especially at elevated temperatures, can be attributed to high sensitivity as well as suppressed noise by increased oxygen desorption in air70. It is also noteworthy that, even after the sensor was prepared using a pellet stored in an air environment for more than a year, its NO2 responses and sensitivity were not degraded (Fig. S12). In addition, the WSe2-drawn sensors exhibited stable responses after 14 days of storage in open air (Fig. S13). Considering the potential use of our sensor, the high response at room temperature and the excellent stability in the air are advantageous for the immediate and sensitive detection of harmful gases at anytime and anywhere by the mechanical drawing of TMDCs on the paper.
NO2 sensing performance of the WSe2-drawn paper. a Transient sensing responses of the WSe2 sensor with respect to NO2 concentrations from 0.1 to 1 ppm and environmental temperatures from 18 to 150 ºC in air. b Sensitivity of the sensor, which is defined as the response per gas concentration, with respect to the environmental temperature. Filled objects are experimental data, dotted lines are curve fits from the Langmuir adsorption model, and solid lines are linear fits. c Transient sensing responses to 1 ppm NO2 under different humidity from 1 to 80% RH at room temperature (18 °C). d Responses of the sensor to 1 ppm NO2 in different RH levels at room temperature (18 °C). The setting temperature for RH control was also room temperature (18–20 °C). e Responses of the sensor under exposure to 5 ppm of six species of VOCs, CO, NH3, and NO2 at room temperature (18–20 °C)
In addition, we investigated the effect of humidity on the response of WSe2-drawn sensor. As shown in Fig. 3c, the sensor was exposed to 1 ppm NO2 under different relative humidity (RH) levels from 1 to 80% at room temperature. The sensing current immediately increased upon exposure to NO2 regardless of RH, but the response exhibited a humidity dependence, as characterized in Fig. 3d. Specifically, the response increased from 71.23 to 162.09% within the 1–50% RH range, then decreased to 21.49% at 80% RH. This behavior would result from the adsorption of water molecules on the WSe2 surface, competing with oxygen adsorption. It was reported that water molecules above Se vacancy have higher adsorption energy than oxygen, but they transfer fewer electrons from WSe2 than oxygen does71. Consequently, as RH increases, more water adsorption reduces the hole concentration in WSe2, leading to a decrease in the initial current (I0) (Fig. S14). It is noted that change in current (ΔI) upon NO2 exposure remains stable up to 70% RH, likely due to the higher adsorption energy and more charge transfer between NO2 and WSe2 compared to water molecules27. This contributes to the increased NO2 response as humidity rises up to 50% RH. Beyond 50% RH, water molecules could accumulate to form a thin liquid layer on the surface, creating additional conductive pathways across the WSe2 flakes and thus increasing the initial current. This enhances NO2 sensing by allowing more WSe2 to participate in the response, leading to an increase in ΔI at 80% RH (Fig. S14). However, elevated humidity would reduce NO2 concentration below 1 ppm due to the high solubility of NO2 in water. Additionally, excessive water molecules occupy active sites on WSe2, impeding NO2 adsorption and finally decreasing the response. Despite the reduced response in highly humid conditions, the sensor still shows high response, and the effect of humidity can be compensated if needed. On the other hand, it is expected that sensitivity can be further improved by optimizing the humidity level. Moreover, the recovery time also depends on RH, as shown in Fig. S15. It is defined as the time required for the response to return to 10% of its peak after NO2 is removed. The recovery time decreases with increasing RH up to 50%, likely due to competitive adsorption by H2O molecules and the formation of nitric acid-related species that facilitate NO2 desorption72,73,74. However, above 50% RH, water accumulation on the sensing layer hinders desorption and increases baseline current, leading to slower recovery. To evaluate the robustness of the sensor under varying environmental conditions, we exposed the WSe₂-drawn sensor to 1 ppm NO2 while sequentially changing temperature and humidity. As shown in Fig. S16, the sensor exhibited stable and repeatable responses across different conditions, including variations in RH (1% to 50%) and temperature (RT to 50 °C), demonstrating its durability under fluctuating environments.
Our sensor is applicable for the selective detection of NO2 over diverse species of chemical vapors, such as VOCs (acetone, ethanol, p-xylene, toluene, benzene, and ethylbenzene), CO, and NH3, as shown in Fig. 3e. All sensing experiments were conducted under identical conditions (5 ppm concentration for all target gases in the air environment at room temperature), except for the gas species exposed to the WSe2-drawn sensor. Each vapor results in a different sensing response (Fig. S17), and the response to NO2 is ~38.51–489.76 times higher than that to other gases, which can be due to the different adsorption energy and charge transfer between WSe2 and the gas molecules27. While VOCs and CO primarily interact through weak dipole scattering or limited reactions with pre-adsorbed O2⁻ ions75,76,77,78,79, NO2 can additionally interact directly with the WSe2 surface, enabling more efficient electron withdrawal61,62. NH3 also participates in charge transfer but shows a weaker interaction than NO2 due to its lower adsorption energy27,80. In addition, as the tested gases, except NO2, are known to act as reducing gases that donate electrons to sensing materials once they are adsorbed, the number of holes in WSe2 can decrease, leading to an increase in the resistance of the sensor (i.e., a decrease in ΔI/I0) upon exposure. For instance, the interaction of NH3 with O2- adsorption sites on the WSe2 induces charge transfer which donates electrons to the p-type sensing material81, thereby increasing the resistance of the sensor. The results indicate that our sensor is able to selectively detect NO2 molecules with distinctively high responses against other interfering vapors.
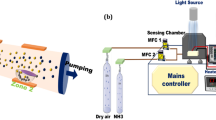
The WSe2-drawn sensor is mechanically flexible, as demonstrated by reversible resistance change under concave and convex bending from the flat state (0°) to a bending angle of 180° and 1000 cycles of 60° bending (Fig. S18a, b). In addition, the sensor maintained comparable NO2 responses under folding, crumpling, and bending (976%, 989%, and 1047%, respectively) compared to the flat state (1006%), indicating stable sensing performance even under severe deformation (Fig. S18c). Due to its high sensitivity, thinness, flexibility, and low-cost fabrication, the TMDC-drawn paper is suitable as a replaceable sensor that can be readily prepared and mounted as needed. As a proof of concept, we demonstrated a TMDC-drawn paper-based portable wireless device that can sensitively monitor gas exposure in real time via a smartphone. The device frame was manufactured by 3D printing, and a microcontroller unit, Bluetooth module, voltage dividing circuit, and battery were assembled within the frame, as illustrated in Fig. 4a. Here, the WSe2-drawn sensor was mounted face-down in the device, thus making contact with the electrodes while suspending over the flowing channel to maximize gas exposure. The sensor can be easily mounted and replaced, and the sensing signal (change in voltage) is monitored by a custom-made Android application installed on the smartphone (Fig. 4b). The electrical configuration is outlined in Fig. S19. As shown in Fig. 4c, upon sequential exposure to 1 and 5 ppm NO2, the voltage across the sensor decreased correspondingly due to a decrease in resistance by increasing the number of holes in WSe2 upon the absorption of NO2, as expected (see Movie S1 for the operation of wireless gas sensing). The calculated current responses were 21.11 and 77.44% for 1 and 5 ppm NO2 exposure, respectively. It is worth noting that the WSe2-based sensor in the device was exposed to NO2 for only tens of seconds, and thus, the response can be further increased by extending the exposure time. After calibration of the sensor, the users can identify the exposed gas concentration in real time. This successful demonstration indicates that our sensor can potentially be a part of a ubiquitous detector with ultrahigh sensitivity.
Portable, wireless gas sensing device based on the TMDC-drawn paper. a A microcontroller unit, Bluetooth module, battery, and WSe2-drawn sensor are integrated into a 3D printed frame. b Photographs of the sensing device before and after assembly of a top cover. Scale bars are 3 cm. c Screen captured image from the smartphone showing the wirelessly monitored NO2 sensing response at room temperature (18–20 °C)
Conclusion
We demonstrated a paper-based ultrasensitive gas sensor using mechanically drawn 2D TMDCs, such as WSe2 and MoS2 nanolayers, as the sensing material. Mechanical drawing of TMDCs on paper exposes abundant edges sites, leading to an exceptional response of 1978.63% toward 10 ppm NO2, a sensitivity of 139.9% ppm−1 in the NO2 concentration range of 0.1–1 ppm, and a low detection limit of 1.25 ppb at 100 °C. The performance surpasses that of previously reported 2D-material-based flexible NO2 sensors. Importantly, the sensing response of WSe2 to NO2 was considerably higher than to other tested chemical species, confirming its selective detection capability. The sensitivity and selectivity of the TMDC-drawn sensor could be further improved by additional functionalization or formation of heterostructures. In addition, the inherent flexibility of the paper substrate enables reversible 180° folding (bending radius of ~80 μm) and stable sensing under heavy crumpling without compromising the response. Given the simple, rapid, and cost-effective fabrication, this approach offers a viable route for disposable and replaceable sensor in a 3D printed portable wireless device for real-time smartphone-based monitoring. This indicates the potential for highly sensitive, ubiquitous environmental monitoring system. If required, sensor arrays could also be fabricated in parallel using automated multiple-drawing systems. Moreover, the method can be extended to other nanomaterials for detecting different target analytes or applied to optoelectronic devices such as wavelength-selective photodetectors based on excitonic transition of TMDCs.
Materials and methods
Preparation of TMDC-drawn sensors
The WSe2 and MoS2 powders were purchased from Alfa Aesar and used as received. The multiwalled CNT powder was purchased from Graphene Supermarket and also used as received. Nanomaterials (200 mg) were placed inside a pressing die (FTIR+XRF pellet press, Specac), and a hydraulic compressive pressure of 300 MPa was applied for 3 min to form a pellet. The pellet having a thickness of ~3 mm and diameter of ~13 mm was used for all tested nanomaterials. For the substrate, copy papers (Double A) were trimmed to the required size. The electrodes were hand-drawn using a commercial silver pen (Circuit Scribe). The drawn silver ink was dried in air at room temperature for 30 s, and the measured sheet resistance of the drawn silver electrode was ~3.75 Ω sq−1. Next, the TMDC pellet was also drawn by hand with an applied force of ~3–7 N over the drawn silver electrodes on the paper for at least five times until typical resistance values of ~0.3–0.5 GΩ for WSe2 and ~4–8 GΩ for MoS2 were obtained. The area where the nanomaterials were drawn was ~20 mm2 for all cases. If required, electrical wires were bonded to the sensor using silver paste (Elcoat P-100, CANS) or probe tips were contacted for measurements and characterization.
Sensor characterization
The surface morphology of the sensors was observed by field-emission scanning electron microscopy (SEM; S-4800, Hitachi). Field-emission transmission electron microscopy (TEM; H-7600, Hitachi) was used to inspect WSe2 and MoS2. For TEM inspection, the TMDC powders were dispersed in N,N-dimethylformamide at a concentration of ~0.1 mg mL−1 by short bath sonication. Next, the dispersion was dropped, dried onto a TEM grid, and inspected. Raman spectra were recorded using a confocal Raman microscope (HEDA, NOST) using a 532 nm laser with a spot diameter of ~1 μm in air at room temperature. The laser power was kept low to minimize overheating. X-ray photoelectron spectroscopy (XPS) spectra were obtained using a K-Alpha XPS system (Thermo Scientific) with a monochromatic Al Kα X-ray source. The XPS spectra were deconvoluted into Lorentzian curves for fitting. The current–voltage curves of the sensors were measured using a sourcemeter (2634B, Keithley) in air at room temperature. For the mechanical bending tests, a custom-built, automated translational stage was used, and the resistance change of the sensor was recorded using a digital multimeter (2000, Keithley). The resistance change is defined as ΔR/R0, where ΔR is the difference between the resistance before and after the bending and R0 is the initial resistance.
Gas sensing experiments
A custom-built chamber integrated with a gas inlet/outlet, a temperature controller, a humidity controller, a quartz window, and an electrical feedthrough was used. Pure air was introduced into the chamber before and after the sensors were exposed to the target gas. Air-balanced dry 10 ppm NO2, NH3, CO, and VOCs (acetone, ethanol, p-xylene, toluene, benzene, and ethylbenzene) were used as sensing gases. All gases (99.99% purity) were purchased from a gas supplier (MS Gas) and used as received. A constant flow rate of 500 sccm was maintained during all experiments using mass flow-rate controllers. To investigate the effect of humidity, the relative humidity inside the sensing chamber was controlled by adjusting the airflow rate supplied to a bubbler at room temperature (18–20 °C), while the humidity near the sensor was continuously monitored using a humidity sensor. It is noted that, except for the data presented in Fig. 3c, d, all target gases were exposed to the sensor under dry conditions. A constant bias voltage of 10 V was applied to the sensor, and the change in current was simultaneously measured using a sourcemeter (2634B, Keithley). The response is defined as ΔI/I0, where ΔI is the difference between the currents before and after the target gas exposure and I0 is the initial current. A time interval between the first, second, and subsequent measurements in Fig. 2 was ~24 h, respectively. For UV irradiation, a UV lamp (UVLMS-38, Analytik Jena) was placed over the chamber and operated during the sensing.
Demonstration of a wireless sensing device
An external frame composed of polylactic acid was manufactured using a 3D printer (DP200, Sindoh). A microcontroller unit (Arduino Pro Mini), Bluetooth module (HC06), 9 V battery, and voltage divider with a reference resistor of 100 MΩ were assembled inside the frame. Copper tapes were used to make electrical contact with the WSe2-drawn sensor. A custom-made Android application was installed on the smartphone, which recorded voltage changes for real time monitoring of the sensor signals. Similar to the gas-sensing experiments, air-balanced NO2 flowed inside the device at a total flow rate of 500 sccm at room temperature.
References
Meng, Z., Stolz, R. M., Mendecki, L. & Mirica, K. A. Electrically-transduced chemical sensors based on two-dimensional nanomaterials. Chem. Rev. 119, 478–598 (2019).
Jariwala, D., Sangwan, V. K., Lauhon, L. J., Marks, T. J. & Hersam, M. C. Emerging device applications for semiconducting two-dimensional transition metal dichalcogenides. ACS Nano 8, 1102–1120 (2014).
Li, H. et al. Fabrication of single- and multilayer MoS2 film-based field-effect transistors for sensing NO at room temperature. Small 8, 63–67 (2012).
Perkins, F. K. et al. Chemical vapor sensing with monolayer MoS2. Nano Lett. 13, 668–673 (2013).
Lee, K., Gatensby, R., McEvoy, N., Hallam, T. & Duesberg, G. S. High-performance sensors based on molybdenum disulfide thin films. Adv. Mater. 25, 6699–6702 (2013).
Liu, B. et al. High-performance chemical sensing using schottky-contacted chemical vapor deposition grown monolayer MoS2 transistors. ACS Nano 8, 5304–5314 (2014).
Kim, J.-S., Yoo, H.-W., Choi, H. O. & Jung, H.-T. Tunable volatile organic compounds sensor by using thiolated ligand conjugation on MoS2. Nano Lett. 14, 5941–5947 (2014).
Cho, B. et al. Bifunctional sensing characteristics of chemical vapor deposition synthesized atomic-layered MoS2. ACS Appl. Mater. Interfaces 7, 2952–2959 (2015).
Cho, S.-Y. et al. Superior chemical sensing performance of black phosphorus: comparison with MoS2 and graphene. Adv. Mater. 28, 7020–7028 (2016).
Ko, K. Y. et al. Recovery improvement for large-area tungsten diselenide gas sensors. ACS Appl. Mater. Interfaces 10, 23910–23917 (2018).
Guo, R. et al. Ultrasensitive room temperature NO2 sensors based on liquid phase exfoliated WSe2 nanosheets. Sens. Actuators B: Chem. 300, 127013 (2019).
Late, D. J. et al. Sensing behavior of atomically thin-layered MoS2 transistors. ACS Nano 7, 4879–4891 (2013).
Cho, S.-Y. et al. Highly enhanced gas adsorption properties in vertically aligned MoS2 layers. ACS Nano 9, 9314–9321 (2015).
Cha, J.-H., Choi, S.-J., Yu, S. & Kim, I.-D. 2D WS2-edge functionalized multi-channel carbon nanofibers: effect of WS2 edge-abundant structure on room temperature NO2 sensing. J. Mater. Chem. A 5, 8725–8732 (2017).
Deokar, G. et al. MoS2-carbon nanotube hybrid material growth and gas sensing. Adv. Mater. Interfaces 4, 1700801 (2017).
Shim, Y.-S. et al. Synthesis of numerous edge sites in MoS2 via SiO2 nanorods platform for highly sensitive gas sensor. ACS Appl. Mater. Interfaces 10, 31594–31602 (2018).
Kuru, C. et al. MoS2 Nanosheet-Pd nanoparticle composite for highly sensitive room temperature detection of hydrogen. Adv. Sci. 2, 1500004 (2015).
Sarkar, D. et al. Functionalization of transition metal dichalcogenides with metallic nanoparticles: implications for doping and gas-sensing. Nano Lett. 15, 2852–2862 (2015).
Ko, K. Y. et al. Improvement of gas-sensing performance of large-area tungsten disulfide nanosheets by surface functionalization. ACS Nano 10, 9287–9296 (2016).
Cho, S.-Y., Koh, H.-J., Yoo, H.-W., Kim, J.-S. & Jung, H.-T. Tunable volatile-organic-compound sensor by using Au nanoparticle incorporation on MoS2. ACS Sens 2, 183–189 (2017).
Lee, E., Yoon, Y. S. & Kim, D.-J. Two-dimensional transition metal dichalcogenides and metal oxide hybrids for gas sensing. ACS Sens 3, 2045–2060 (2018).
Li, Q., Meng, J. & Li, Z. Recent progress on Schottky sensors based on two-dimensional transition metal dichalcogenides. J. Mater. Chem. A 10, 8107–8128 (2022).
Kumar, R., Goel, N. & Kumar, M. UV-activated MoS2 based fast and reversible NO2 sensor at room temperature. ACS Sens 2, 1744–1752 (2017).
Pham, T., Li, G., Bekyarova, E., Itkis, M. E. & Mulchandani, A. MoS2-based optoelectronic gas sensor with sub-parts-per-billion limit of NO2 gas detection. ACS Nano 13, 3196–3205 (2019).
Zhao, S., Xue, J. & Kang, W. Gas adsorption on MoS2 monolayer from first-principles calculations. Chem. Phys. Lett. 595–596, 35–42 (2014).
Zhou, C., Yang, W. & Zhu, H. Mechanism of charge transfer and its impacts on Fermi-level pinning for gas molecules adsorbed on monolayer WS2. J. Chem. Phys. 142, 214704 (2015).
Wang, T. et al. Tunable donor and acceptor impurity states in a WSe2 monolayer by adsorption of common gas molecules. RSC Adv. 6, 82793–82800 (2016).
He, Q. et al. Fabrication of flexible MoS2 thin-film transistor arrays for practical gas-sensing applications. Small 8, 2994–2999 (2012).
Cho, B. et al. Chemical sensing of 2D graphene/MoS2 heterostructure device. ACS Appl. Mater. Interfaces 7, 16775–16780 (2015).
Kuru, C. et al. High-performance flexible hydrogen sensor made of WS2 nanosheet-Pd nanoparticle composite film. Nanotechnology 27, 195501 (2016).
Guo, H. et al. Transparent, flexible, and stretchable WS2 based humidity sensors for electronic skin. Nanoscale 9, 6246–6253 (2017).
Guo, J. et al. Piezotronic effect enhanced flexible humidity sensing of monolayer MoS2. ACS Appl. Mater. Interfaces 10, 8110–8116 (2018).
Zhao, Y. et al. Low-temperature synthesis of 2D MoS2 on a plastic substrate for a flexible gas sensor. Nanoscale 10, 9338–9345 (2018).
Kumar, R., Goel, N., Hojamberdiev, M. & Kumar, M. Transition metal dichalcogenides-based flexible gas sensors. Sens. Actuators A: Phys. 303, 111875 (2020).
Choi, J., Zhang, H., Du, H. & Choi, J. H. Understanding solvent effects on the properties of two- dimensional transition metal dichalcogenides. ACS Appl. Mater. Interfaces 8, 8864–8869 (2016).
Nutting, D. et al. Heterostructures formed through abraded van der Waals materials. Nat. Commun. 11, 3047 (2020).
Mazaheri, A., Lee, M., van der Zant, H. S. J., Frisenda, R. & Castellanos-Gomez, A. MoS2-on-paper optoelectronics: drawing photodetectors with van der Waals semiconductors beyond graphite. Nanoscale 12, 19068–19074 (2020).
Lee, M., Mazaheri, A., van der Zant, H. S. J., Frisenda, R. & Castellanos-Gomez, A. Drawing WS2 thermal sensors on paper substrates. Nanoscale 12, 22091–22096 (2020).
Nouri, B., Castellanos-Gomez, A. & Ghasemi, F. Paper-based flexible supercapacitors with drawn van der Waals materials. J. Alloy. Compd. 959, 170554 (2023).
Matatagui, D. et al. Eco-friendly disposable WS2 paper sensor for sub-ppm NO2 detection at room temperature. Nanomaterials 12, 1213 (2022).
Mahmoodi, E. et al. Paper-based broadband flexible photodetectors with van der Waals materials. Sci. Rep. 12, 12585 (2022).
Kharchich, F. Z., Castellanos-Gomez, A. & Frisenda, R. Electrical properties of disordered films of van der Waals semiconductor WS2 on paper. Nanoscale 16, 8968–8974 (2024).
Ersu, G. et al. Low-cost and biodegradable thermoelectric devices based on van der Waals semiconductors on paper substrates. Energy Environ. Mater. 7, e12488 (2024).
Mirica, K. A., Weis, J. G., Schnorr, J. M., Esser, B. & Swager, T. M. Mechanical drawing of gas sensors on paper. Angew. Chem. Int. Ed. 51, 10740–10745 (2012).
Mirica, K. A., Azzarelli, J. M., Weis, J. G., Schnorr, J. M. & Swager, T. M. Rapid prototyping of carbon- based chemiresistive gas sensors on paper. Proc. Natl. Acad. Sci. USA 110, E3265–E3270 (2013).
Azzarelli, J. M., Mirica, K. A., Ravnsbæk, J. B. & Swager, T. M. Wireless gas detection with a smartphone via RF communication. Proc. Natl. Acad. Sci. USA 111, 18162–18166 (2014).
Frazier, K. M., Mirica, K. A., Walish, J. J. & Swager, T. M. Fully-drawn carbon-based chemical sensors on organic and inorganic surfaces. Lab Chip 14, 4059–4066 (2014).
Liao, X. et al. Flexible and highly sensitive strain sensors fabricated by pencil drawn for wearable monitor. Adv. Funct. Mater. 25, 2395–2401 (2015).
Zhang, J. et al. Pencil-trace on printed silver interdigitated electrodes for paper-based NO2 gas sensors. Appl. Phys. Lett. 106, 143101 (2015).
Zhao, H. et al. Drawn on paper: a reproducible humidity sensitive device by handwriting. ACS Appl. Mater. Interfaces 9, 28002–28009 (2017).
Park, J.-H., Park, M.-J. & Lee, J.-S. Dry writing of highly conductive electrodes on papers by using silver nanoparticle-graphene hybrid pencils. Nanoscale 9, 555–561 (2017).
Yu, Y. & Zhang, J. Pencil-drawing assembly to prepare graphite/MWNT hybrids for high performance integrated paper supercapacitors. J. Mater. Chem. A 5, 4719–4725 (2017).
Tobjörk, D. & Österbacka, R. Paper electronics. Adv. Mater. 23, 1935–1961 (2011).
Mahadeva, S. K., Walus, K. & Stoeber, B. Paper as a platform for sensing applications and other devices: a review. ACS Appl. Mater. Interfaces 7, 8345–8362 (2015).
Tai, H., Duan, Z., Wang, Y., Wang, S. & Jiang, Y. Paper-based sensors for gas, humidity, and strain detections: a review. ACS Appl. Mater. Interfaces 12, 31037–31053 (2020).
Yang, G., Lee, C., Kim, J., Ren, F. & Pearton, S. J. Flexible graphene-based chemical sensors on paper substrates. Phys. Chem. Chem. Phys. 15, 1798–1801 (2013).
Liu, H. et al. Physically flexible, rapid-response gas sensor based on colloidal quantum dot solids. Adv. Mater. 26, 2718–2724 (2014).
Lee, W. S. & Choi, J. Hybrid integration of carbon nanotubes and transition metal dichalcogenides on cellulose paper for highly sensitive and extremely deformable chemical sensors. ACS Appl. Mater. Interfaces 11, 19363–19371 (2019).
Kumar, S., Kaushik, S., Pratap, R. & Raghavan, S. Graphene on paper: a simple, low-cost chemical sensing platform. ACS Appl. Mater. Interfaces 7, 2189–2194 (2015).
Koga, H. et al. Paper-based disposable molecular sensor constructed from oxide nanowires, cellulose nanofibers, and pencil-drawn electrodes. ACS Appl. Mater. Interfaces 11, 15044–15050 (2019).
Moumen, A. et al. Energy-efficient NO2 sensors based on two-dimensional layered 2H-WS2 nanosheets. Sens. Actuators B: Chem. 406, 135379 (2024).
Tang, H. et al. Ultra-high sensitive NO2 gas sensor based on tunable polarity transport in CVD-WS2/IGZO p‑N heterojunction. ACS Appl. Mater. Interfaces 11, 40850–40859 (2019).
Nayeri, M., Moradinasab, M. & Fathipour, M. The transport and optical sensing properties of MoS2, MoSe2, WS2 and WSe2 semiconducting transition metal dichalcogenides. Semicond. Sci. Technol. 33, 025002 (2018).
Lu, Z., Zhai, Y., Liang, Q. & Wu, W. Promoting sensitivity and selectivity of NO2 gas sensor based on metal (Pt, Re, Ta)-doped monolayer WSe2: a DFT study. Chem. Phys. Lett. 755, 137737 (2020).
Xiao, Z., Wu, W., Wu, X. & Zhang, Y. Adsorption of NO2 on monolayer MoS2 doped with Fe, Co, and Ni, Cu: a computational investigation. Chem. Phys. Lett. 755, 137768 (2020).
Santucci, S. et al. NO2 and CO gas adsorption on carbon nanotubes: experiment and theory. J. Chem. Phys. 119, 10904 (2003).
Ervin, K. M., Ho, J. & Lineberger, W. C. Ultraviolet photoelectron spectrum of NO2-. J. Phys. Chem. 92, 5405–5412 (1988).
Clemmer, D. E. & Armentrout, P. B. Direct determination of the adiabatic ionization energy of NO2 as measured by guided ion-beam mass spectrometry. J. Chem. Phys. 97, 2451–2458 (1992).
Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 40, 1361–1403 (1918).
Shin, W. et al. Low-frequency noise in gas sensors: a review. Sens. Actuators B: Chem. 383, 133551 (2023).
Hoffman, A. N. et al. Atmospheric and long-term aging effects on the electrical properties of variable thickness WSe2 transistors. ACS Appl. Mater. Interfaces 10, 36540–36548 (2018).
Park, J.-Y. & Lee, Y.-N. Solubility and decomposition kinetics of nitrous acid in aqueous solution. J. Phys. Chem. 92, 6294–6302 (1988).
Yan, W. et al. Effects of ambient humidity and temperature on the NO2 sensing characteristics of WS2/graphene aerogel. Appl. Surf. Sci. 450, 372–379 (2018).
Dubowski, Y. et al. Interactions of gaseous nitric acid with surfaces of environmental interest. Phys. Chem. Chem. Phys. 6, 3879–3888 (2004).
Cho, S.-Y. et al. High-resolution p-type metal oxide semiconductor nanowire array as an ultrasensitive sensor for volatile organic compounds. Nano Lett. 16, 4508–4515 (2016).
Chen, W. Y., Yen, C.-C., Xue, S., Wang, H. & Stanciu, L. A. Surface functionalization of layered molybdenum disulfide for the selective detection of volatile organic compounds at room temperature. ACS Appl. Mater. Interfaces 11, 34135–34143 (2019).
Chang, J. F., Kuo, H. H., Leu, I. C. & Hon, M. H. The effect of thickness and operation temperature on ZnO:Al thin film CO gas sensor. Sens. Actuators B Chem. 84, 258–264 (2002).
Meng, J. et al. Triboelectric nanogenerator enhanced Schottky nanowire sensor for highly sensitive ethanol detection. Nano Lett. 20, 4968–4974 (2020).
Shaik, R., Kampara, R. K., Kumar, A., Sharma, C. S. & Kumar, M. Metal oxide nanofibers based chemiresistive H2S gas sensors. Coord. Chem. Rev. 471, 214752 (2022).
Shingange, K., Tshabalala, Z. P., Ntwaeaborwa, O. M., Motaung, D. E. & Mhlongo, G. H. High selective NH3 gas sensor based on Au loaded ZnO nanostructures prepared using microwave-assisted method. J. Colloid Interface Sci. 479, 127–138 (2016).
Sharma, I., Kumar, K. N. & Choi, J. Highly sensitive chemiresistive detection of NH3 by formation of WS2 nanosheets and SnO2 quantum dot heterostructures. Sens. Actuators B Chem. 375, 132899 (2023).
Yang, C. et al. Flexible NO2 sensors based on WSe2 nanosheets with bifunctional selectivity and superior sensitivity under UV activation. Sens. Actuators B: Chem. 333, 129571 (2021).
Yan, W. et al. Size-tunable flowerlike MoS2 nanospheres combined with laser-induced graphene electrodes for NO2 sensing. ACS Appl. Nano Mater. 3, 2545–2553 (2020).
Cho, B. et al. Metal decoration effects on the gas-sensing properties of 2D hybrid-structures on flexible substrates. Sensors 15, 24903–24913 (2015).
Guo, S. et al. Development of a cloud-based epidermal MoSe2 device for hazardous gas sensing. Adv. Funct. Mater. 29, 1900138 (2019).
Rathi, K., Kumar, A. N. & Pal, K. Fabrication of flexible La-MoS2 Hybrid-heterostructure Based Sensor for NO2 gas sensing at room temperature. Nanotechnology 31, 3955049 (2020).
Guo, J., Wen, R., Zhai, J. & Wang, Z. L. Enhanced NO2 gas sensing of a single-layer MoS2 by photogating and piezo-phototronic effects. Sci. Bull. 64, 128–135 (2019).
Kim, S. et al. Flexible chemical sensors based on hybrid layer consisting of molybdenum disulphide nanosheets and carbon nanotubes. Carbon 129, 607–612 (2018).
Jung, M. W., Kang, S. M., Nam, K.-H., An, K.-S. & Ku, B.-C. Highly transparent and flexible NO2 gas sensor film based on MoS2/RGO composites using soft lithographic patterning. Appl. Surf. Sci. 456, 7–12 (2018).
Cho, B. et al. Two-dimensional atomic-layered alloy junctions for high-performance wearable chemical sensor. ACS Appl. Mater. Interfaces 8, 19635–19642 (2016).
Ko, K. Y. et al. High-performance gas sensor using a large-area WS2XSe2-2X alloy for low-power operation wearable applications. ACS Appl. Mater. Interfaces 10, 34163–34171 (2018).
Quan, W. et al. Fully flexible MXene-based gas sensor on paper for highly sensitive room-temperature nitrogen dioxide detection. ACS Sens. 8, 103–113 (2023).
Hou, S. et al. Synergistic CNFs/CoS2/MoS2 flexible films with unprecedented selectivity for NO gas at room temperature. ACS Appl. Mater. Interfaces 12, 29778–29786 (2020).
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT, and Future Planning (RS-2023-00222166).
Author information
Authors and Affiliations
Contributions
Leilei Wang: Data curation, Formal analysis, Investigation, Visualization, Writing—original draft. Jungwook Choi: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Supervision, Writing—original draft, Writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, L., Choi, J. Mechanical drawing of two-dimensional transition metal dichalcogenides on paper for ultrasensitive gas sensing devices. Microsyst Nanoeng 12, 2 (2026). https://doi.org/10.1038/s41378-025-01045-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41378-025-01045-8