Abstract
Background
To describe the association between maternal hypertension (chronic and gestational, MH) and mortality in very preterm singletons and twins, focusing on how estimates depend on gestational age (GA) and size at birth.
Methods
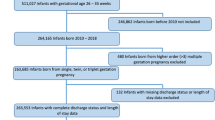
We estimated relative risks of in-hospital death in 12,320 singletons (MH: 22.4%) and 4381 twins (MH: 10.6%) born at 23–29 weeks in the Italian Neonatal Network (89 hospitals, 2008–2016).
Results
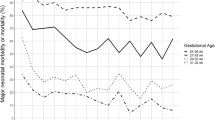
Babies with MH had higher GA and were more frequently small-for-gestational age (SGA), especially singletons. In crude analyses, MH was associated with lower mortality in singletons. In multivariable analyses, the effects of GA and size differed between twins and singletons with and without MH. The best-fitting models included continuous birth weight (rather than SGA) and were stratified by GA. In these models, MH was associated with lower mortality in singletons—but not twins—born after week 25.
Conclusions
In this cohort of very preterm infants, the association between MH and mortality differed between singletons and twins and across strata of GA at birth. These estimates cannot be interpreted causally, but suggest that, from a descriptive/predictive standpoint, singletons with MH born after week 25 have lower mortality than singletons born to women without MH.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Baud, O. et al. The relationships between antenatal management, the cause of delivery and neonatal outcome in a large cohort of very preterm singleton infants. BJOG 107, 877–884 (2000).
Chen, X. K., Wen, S. W., Smith, G., Yang, Q. & Walker, M. New-onset hypertension in late pregnancy is associated with lower fetal and infant mortality in preterm twins. Hypertens. Pregnancy 25, 205–215 (2006).
Chen, X. K., Wen, S. W., Smith, G., Yang, Q. & Walker, M. Pregnancy-induced hypertension is associated with lower infant mortality in preterm singletons. BJOG 113, 544–551 (2006).
Delorme, P. et al. Cause of preterm birth as a prognostic factor for mortality. Obstet. Gynecol. 127, 40–48 (2016).
Evans, N. et al. Prenatal predictors of mortality in very preterm infants cared for in the Australian and New Zealand Neonatal Network. Arch. Dis. Child. Fetal Neonatal Ed. 92, F34–F40 (2007).
Foix-L’helias, L., Baud, O., Lenclen, R., Kaminski, M. & Lacaze-Masmonteil, T. Benefit of antenatal glucocorticoids according to the cause of very premature birth. Arch. Dis. Child. Fetal Neonatal Ed. 90, F46–F48 (2005).
Gagliardi, L., Rusconi, F., Bellù, R., Zanini, R. & Italian Neonatal, N. Association of maternal hypertension and chorioamnionitis with preterm outcomes. Pediatrics 134, e154–e161 (2014).
Gemmell, L. et al. Hypertensive disorders of pregnancy and outcomes of preterm infants of 24 to 28 weeks’ gestation. J. Perinatol. 36, 1067–1072 (2016).
Johanzon, M., Odesjo, H., Jacobsson, B., Sandberg, K. & Wennerholm, U. B. Extreme preterm birth: onset of delivery and its effect on infant survival and morbidity. Obstet. Gynecol. 111, 42–50 (2008).
Luo, Z. C. et al. Effect on neonatal outcomes in gestational hypertension in twin compared with singleton pregnancies. Obstet. Gynecol. 108, 1138–1144 (2006).
McBride, C. A., Bernstein, I. M., Badger, G. J., Horbar, J. D. & Soll, R. F. The effect of maternal hypertension on mortality in infants 22, 29 weeks gestation. Pregnancy Hypertens. 5, 362–366 (2015).
McElrath, T. F. et al. Pregnancy disorders that lead to delivery before the 28th week of gestation: an epidemiologic approach to classification. Am. J. Epidemiol. 168, 980–989 (2008).
Tokumasu, H., Tokumasu, S. & Kawakami, K. Impact of pre-eclampsia in extremely premature infants: population-based study. Pediatr. Int. 58, 578–583 (2016).
Luo, Q. G., Zhang, J. Y., Cheng, W. W., Audibert, F. & Luo, Z. C. Is gestational hypertension protective against perinatal mortality in twin pregnancies? PLoS ONE 9, e94865 (2014).
Gagliardi, L. et al. Pregnancy disorders leading to very preterm birth influence neonatal outcomes: results of the population-based ACTION cohort study. Pediatr. Res. 73, 794–801 (2013).
Ananth, C. V. & Schisterman, E. F. Confounding, causality and confusion: the role of intermediate variables in interpreting observational studies in obstetrics. Am. J. Obstet. Gynecol. 217, 167–175 (2017).
Basso, O. & Wilcox, A. J. Might rare factors account for most of the mortality of preterm babies? Epidemiology 22, 320–327 (2011).
Snowden, J. M. & Basso, O. Causal inference in studies of preterm babies: a simulation study. BJOG 125, 686–692 (2018).
Wilcox, A. J., Weinberg, C. R. & Basso, O. On the pitfalls of adjusting for gestational age at birth. Am. J. Epidemiol. 174, 1062–1068 (2011).
Williams, T. C., Bach, C. C., Matthiesen, N. B., Henriksen, T. B. & Gagliardi, L. Directed acyclic graphs: a tool for causal studies in paediatrics. Pediatr. Res. 84, 487–493 (2018).
Campbell, D. M. & MacGillivray, I. Preeclampsia in twin pregnancies: incidence and outcome. Hypertens. Pregnancy 18, 197–207 (1999).
Chen, K., Wen, S. W., Smith, G. N., Yang, Q. & Walker, M. C. New-onset hypertension in late pregnancy and fetal growth: different associations between singletons and twins. Hypertens. Pregnancy 26, 259–272 (2007).
Ferrazzani, S. et al. Is gestational hypertension beneficial in twin pregnancies? Pregnancy Hypertens. 5, 171–176 (2015).
Sibai, B. M. et al. Hypertensive disorders in twin versus singleton gestations. Am. J. Obstet. Gynecol. 182, 938–942 (2000).
Hutcheon, J. A., Kuret, V., Joseph, K. S., Sabr, Y. & Lim, K. Immortal time bias in the study of stillbirth risk factors: the example of gestational diabetes. Epidemiology 24, 787–790 (2013).
Fenton, T. R. & Kim, J. H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 13, 59 (2013).
Hanley, J. A. & McNeil, B. J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 143, 29–36 (1982).
Lemeshow, S. & Hosmer, D. W. Jr A review of goodness of fit statistics for use in the development of logistic regression models. Am. J. Epidemiol. 115, 92–106 (1982).
Greenwood, C., Yudkin, P., Sellers, S., Impey, L. & Doyle, P. Why is there a modifying effect of gestational age on risk factors for cerebral palsy? Arch. Dis. Child. Fetal Neonatal Ed. 90, F141–F146 (2005).
VanderWeele, T. J., Mumford, S. L. & Schisterman, E. F. Conditioning on intermediates in perinatal. Epidemiology 23, 1–9 (2012).
Basso, O. & Wilcox, A. J. Intersecting birth weight-specific mortality curves: solving the riddle. Am. J. Epidemiol. 169, 787–797 (2009).
Basso, O., Wilcox, A. J. & Weinberg, C. R. Birth weight and mortality: causality or confounding? Am. J. Epidemiol. 164, 303–311 (2006).
Hernandez-Diaz, S., Schisterman, E. F. & Hernan, M. A. The birth weight “paradox” uncovered? Am. J. Epidemiol. 164, 1115–1120 (2006).
Whitcomb, B. W., Schisterman, E. F., Perkins, N. J. & Platt, R. W. Quantification of collider-stratification bias and the birthweight paradox. Paediatr. Perinat. Epidemiol. 23, 394–402 (2009).
Martin, L. J. et al. Country-specific vs. common birthweight-for-gestational age references to identify small for gestational age infants born at 24-28 weeks: an international study. Paediatr. Perinat. Epidemiol. 30, 450–461 (2016).
McBride, C. A., Bernstein, I. M., Badger, G. J. & Soll, R. F. Maternal hypertension and mortality in small for gestational age 22- to 29-week infants. Reprod. Sci. 25, 276–280 (2017). 1933719117711260.
Richardson, D. K., Corcoran, J. D., Escobar, G. J. & Lee, S. K. SNAP-II and SNAPPE-II: simplified newborn illness severity and mortality risk scores. J. Pediatr. 138, 92–100 (2001).
Zupancic, J. A. et al. Revalidation of the score for neonatal acute physiology in the Vermont Oxford Network. Pediatrics 119, e156–e163 (2007).
Gagliardi, L., Lucchini, R., Bellù, R. & Zanini, R. Antenatal corticosteroid prophylaxis in singleton and multiple pregnancies. Paediatr. Perinat. Epidemiol. 31, 394–401 (2017).
Acknowledgements
Members of the Italian Neonatal Network are listed in Supplemental File S2. The Italian Neonatal Network received non-restricted support from Chiesi Pharmaceuticals, Parma, Italy, for registration of centers to the Vermont -Oxford Network. Chiesi Pharmaceuticals had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author contributions
Both authors have participated in the concept and design; analysis and interpretation of data; and the drafting or revising of the manuscript. Both have approved the manuscript as submitted. No honorarium, grant, or other form of payment was given to anyone to produce the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Gagliardi, L., Basso, O. Maternal hypertension and survival in singletons and twins born at 23–29 weeks: not just one answer…. Pediatr Res 85, 697–702 (2019). https://doi.org/10.1038/s41390-019-0337-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-019-0337-4