Abstract
Binge eating (BE), a transdiagnostic feature that occurs across eating disorders and in the general population, carries significant health risks even in the absence of a full-syndrome diagnosis. The limited efficacy of current treatments for binge-type eating disorders highlights the need to better understand the mechanistic heterogeneity underlying BE to optimize treatment allocation, advance personalized medicine, and ultimately improve outcomes. We hypothesized considerable heterogeneity within three neurofunctional domains prevalent across compulsive behaviors and implicated in BE: approach-related behavior, executive function, and negative emotionality. We analyzed data from 612 participants (ages 18–59, 66% female) from the enhanced Nathan Kline Institute-Rockland Sample, including 461 controls and 151 individuals with BE behaviors. Using data-driven statistical modeling of comprehensive, multimodal measures across the three hypothesized domains, we identified subtypes of BE. Subtypes were validated using assessments of eating pathology, substance use, clinical diagnostics, and resting-state functional magnetic resonance imaging. Three distinct and stable subtypes emerged: a ‘Negative Emotionality’ subtype characterized by greater negative affect, emotion dysregulation and psychiatric comorbidity, an ‘Approach’ subtype with higher approach-related and impulsive behaviors, and a ‘Restrained’ subtype that was overcontrolled and harm avoidant. The Approach and Restrained subtypes further demonstrated unique neurobiological profiles, as determined by graph theory analysis of resting-state functional connectivity. All subtypes showed similar proportions of BE episodes meeting clinical-level threshold (≥4 objective binge episodes/month), and no differences in BMI, indicating functionally distinct expressions of BE, beyond clinical severity and diagnostic classification. This study is the first to explore the mechanistic heterogeneity of BE through a comprehensive multi-modal assessment across three neurofunctional domains in a single sample. Findings highlight the need for updated models of BE etiology that integrate approach/reward-related behaviors, impulsivity and overcontrolled behaviors, and negative emotionality, and suggest the potential of these functionally-derived subtypes to inform the development of personalized, targeted interventions.
Similar content being viewed by others
Introduction
Binge eating (BE) is a core transdiagnostic behavior prevalent across eating disorders (ED), including binge-eating disorder (BED), bulimia nervosa (BN), and anorexia nervosa-binge/purge type [1]. BE is characterized by consuming large amounts of food within a discrete period of time, while experiencing a sense of loss of control [1]. The prevalence of EDs marked by recurrent BE, herein binge-type eating disorders (BT-EDs), has increased [2]. BT-EDs are the most commonly diagnosed EDs with prominent global disease burden [3] and significant lifetime prevalence (0.6–2.8%) [2]. BE behaviors present substantial health risks, including psychiatric and physical comorbidity, escalated medication and healthcare utilization, and elevated risks of mortality and suicidality - irrespective of comorbid obesity [4,5,6,7,8]. Strikingly, 7–13% of community samples regularly engage in BE behaviors, indicating substantial prevalence in the general population [9]. This is crucial because BE behaviors are detrimental even in the absence of a full-syndrome ED diagnosis. That is, subclinical and low-frequency BE demonstrate similar consequences as full-syndrome BT-EDs [10,11,12,13] and are highly predictive of later diagnosed disorders [14, 15].
Standard treatments for BT-EDs demonstrate limited efficacy, with over 50% of individuals failing to achieve remission after treatment [16,17,18]. Longitudinal studies in BED patients showed that 3–48 months post-treatment, only about 50% abstained from BE [17], while 12 years post-treatment, 36% still retained the full-syndrome diagnosis [19]. Similarly, in a longitudinal study of BN inpatients, only 38 and 42% of patients achieved remission 11 and 21 years, respectively, post-treatment [20]. The inefficacy of these standard treatments is often attributed to the ‘heterogeneity problem,’ which suggests that distinct causal mechanisms underlie psychiatric disorders in different subsets of individuals [21]. Methods such as latent class or clustering approaches, when applied to psychiatric populations, aim to dissect this heterogeneity, stratifying individuals into homogenous subgroups, termed subtypes, maximizing similarity within each group and distinctiveness between them [22, 23]. Previous efforts within EDs have, largely, focused on delineating subtypes using ED symptomatology or a combination of eating and comorbid psychopathology [24,25,26,27]. While these methods show advantages over traditional diagnostic classification in terms of diagnostic stability [28] and improved prediction of treatment outcomes [26, 29], they remain inconclusive regarding the number and validity of subtypes [24, 25, 29]. Importantly, even though ‘symptom-based’ subtyping approaches can help refine current categorical diagnostics, relying solely on ED symptoms for subtyping may not be sufficient to inform clinical practice. To improve efficacy, shifting towards ‘mechanism-based’ subtyping, which identifies subtypes based on data reflecting the underlying functional processes of EDs, is essential. This approach has the potential to personalize clinical practice by identifying subtype-specific mechanisms that can be targeted differentially [23, 29,30,31].
Several theoretical models emphasize multifactorial mechanisms in BE [32], supported by a broad span of evidence [33,34,35,36,37,38,39,40,41,42]. This research supports three neurobehavioral or ‘functional’ domains underlying BE behaviors: a) approach behavior, b) executive functioning, and c) negative emotionality. These domains, present not only in BE but also across various disorders characterized by impulsive-compulsive behaviors [30, 31, 43,44,45,46], are functionally independent and associated with distinct brain networks [8, 34, 36,37,38,39,40,41,42,43,44,45]. The ‘Approach Behavior’ domain is characterized by sensation seeking, incentive motivation, and engagement in reward-related processes, with neural underpinnings in the brain’s reward and salience networks [44, 47]. These networks involve regions critical for appraising rewarding value and directing attention toward salient stimuli [44, 47]. Impairments within this domain – such as heightened reward-driven motivations and impulsive behaviors - contribute significantly to the pathology of BE, leading to excessive approach behaviors toward food as a primary reinforcer [36, 37].
The ‘Executive Function’ domain encompasses behaviors ranging from cognitive control, set shifting, inhibition to attention shifting [47] and is supported by the brain’s executive or frontoparietal network (FPN) which integrates distributed brain regions essential for cognitive and attentional control [48]. Both extremes within this domain, ranging from excessive restraint or overcontrol to disinhibition or undercontrol, contribute to the risk and maintenance mechanisms in BE [32, 34]. Undercontrol leads to difficulties inhibiting impulses to engage in rewarding but harmful behaviors, increasing the risk of losing control over eating and BE [32,33,34], while overcontrol, often involving dietary restraint, has been evidenced to increase the likelihood of subsequent binge episodes, reinforcing a harmful cycle of restriction and bingeing [15, 32, 34].
The ‘Negative Emotionality’ domain involves an increased sensitivity to negative emotions and punishments, often also manifesting as impulsive behavior during negative mood states [33, 47], with impairments observed in the reward, salience, FPN, and the default-mode network (DMN) [49]. Numerous studies suggest that negative affect often precedes BE, and individuals who engage in BE struggle to regulate negative emotions [35, 50]. This difficulty is a significant factor in the development and frequency of BE, serving to regulate or escape negative affective states [32, 35, 50]. Despite evidence for the significant role of approach behavior, executive function, and negative emotionality across BT-EDs, existing research and theoretical models have not fully integrated these neurobehavioral domains [51]. This hinders our understanding of how these domains interrelate and contribute to BE, impeding the development of comprehensive BE pathology models [51]. Examining these domains collectively could reveal distinct processes driving heterogeneity in BE, facilitating the development of precision medicine approaches. However, existing ED subtyping research often overlooks the full spectrum of underlying mechanisms, and none have explored the neurobiological correlates of the subtypes identified.
Numerous studies, frequently using samples of mixed ED diagnoses, have characterized subtypes based on various combinations of comorbid symptoms (e.g., anxiety, depression), diagnoses (e.g., mood, personality disorders), or personality traits, with most focusing on personality psychopathology (see review: 29). Despite differences in the number of identified subtypes (ranging from 2–6), a variation of three potential mechanistic clusters have emerged: a) ‘undercontrolled’, marked by impulsivity, novelty seeking, emotional dysregulation, and heightened negative emotions; b) ‘overcontrolled’, characterized by inhibition, avoidance, emotional dysregulation, internalizing behaviors and poor self-esteem; and c) ‘low psychopathology’, with minimal comorbidities [29]. Subtyping studies that use a comprehensive comorbid psychopathology approach within samples specifically with BT-EDs are rare [29, 52]. Research in BT-EDs has generally subtyped participants on the basis of a combination of negative affect (e.g., depressive symptoms) and ED-specific behaviors, such as dietary restraint [29]. This approach primarily identified two subtypes: ‘dietary’ subtypes, characterized by heightened dietary restraint, and ‘dietary-negative affect’ subtypes, marked by both dietary restraint and increased negative affect [29]. In summary, previous subtyping approaches in samples with mixed EDs or within BT-EDs each offer unique strengths and limitations. While mixed ED subtyping typically adopts a broader psychopathology approach, assessing heterogeneity across aspects of different functional domains (e.g., negative emotionality, executive functioning domain), BT-ED subtyping often focuses more narrowly on a single-domain – frequently assessing heterogeneity within the functional domain of negative emotionality [29]. Importantly, neither approach has considered non-food related approach behaviors (e.g., increased reward seeking independent of food) as a potentially relevant underlying mechanism. Additionally, both approaches share a common limitation: they generally rely on narrow assessments (e.g., a depression scale) to represent broad functional domains (e.g., negative emotionality) using a single measurement tool as a proxy [29]. As a result, these approaches oversimplify the complexities of the mechanisms underlying these disorders [29]. To comprehensively understand BE heterogeneity and inform effective treatments, a mechanism-based subtyping approach incorporating a broad, multi-modal assessment of neurobehavioral domains is necessary.
Our study aimed to address this gap by applying a data-driven, mechanism-based approach to identify latent subtypes of BE, using a comprehensive, multi-modal assessment that captures the three neurobehavioral domains - approach behavior, executive functioning, and negative emotionality - that are hypothesized to drive BE. Importantly, ED psychopathology and eating-related attitudes were excluded from the subtyping process. Instead, these measures were used to clinically validate and characterize the derived subtypes. Furthermore, we investigated the neurobiological underpinnings of these subtypes using whole-brain resting-state functional magnetic resonance imaging (rs-fMRI) graph theory analysis [53]. We hypothesized that BE behaviors would exhibit considerable variability, such that impairments in any one functional domain – approach behavior (e.g., elevated sensation seeking/risk-taking), executive functioning (e.g., poor cognitive control), or negative emotionality (e.g., increased negative affect/ internalizing and emotional dysregulation) – would be sufficient to drive BE [31]. We thus expected to identify three subtypes, each characterized by distinct functional domain impairments and corresponding rs-fMRI network alterations.
Methods
Participants
We analyzed data from the enhanced Nathan Kline Institute-Rockland Sample study conducted at the Nathan Kline Institute in New York between 2012 and 2016 [54]. This study was approved by the Nathan Kline Institute Institutional Review Board in accordance with the Declaration of Helsinki. Written informed consent and permission to share de-identified data were obtained from all study participants. A protocol filed with the University of Minnesota Institutional Review Board met criteria for exemption. After removing measures with > 10% missing data, 612 participants aged 18–59 had complete phenotypic data. Among them, individuals with BE behaviors (N = 151, 66% female) and Controls (N = 461, 66% female) were identified based on the Eating Disorder Examination Questionnaire’s (EDE-Q) [55] reported frequency of objective binge-episodes (OBE; BE > = 1 OBE; Controls: 0 OBE), including participants with a range of BE behaviors from low frequency to clinical (≥4 OBEs/month). We excluded outliers based on OBE frequency (N = 1). See Supplementary Table 1 for details on the full sample, and Table 1 for demographics by subtype.
Phenotypic measures
To encompass the entire phenotypic space, we refrained from selecting measures based on theoretical considerations and instead included all available assessments, spanning a diverse range across behavior, affect, clinical symptoms, and cognition. When summary scores encompassing the task construct of interest were absent, we used all item-level scores. Our primary behavioral analysis included 74 item-level and summary scores derived from 18 assessments (see Supplementary Table 2 for a comprehensive list). Notably, we excluded measures of ED psychopathology and eating behavior from this analysis and instead used these for internal validation of the behavioral results (see ‘Internal clinical validation measures’ below).
Factor analysis of phenotypic data
We conducted an exploratory factor analysis (EFA), involving all study participants (N = 612), to reduce our phenotypic dataset into underlying latent variables while maintaining a comprehensive representation of the entire phenotypic space. This analysis was very well powered (N > 600; participant/variable ratio = 8.0), based on sample size recommendations for EFA [56]. Monte Carlo permutation analysis (parallel analysis) was implemented to determine the number of factors to retain at a statistical significance of p < 0.05 [57], ensuring factor selection remains independent of distributional assumptions [58]. Subsequently, latent factors were extracted using maximum likelihood estimation with the expectation-maximization (EM) algorithm [59], which has been shown to be robust to distributional violations [58, 60,61,62]. To accommodate correlated factors, we utilized ‘direct oblimin’ rotation, essential for data reduction over a large variable space where factors are expected to be closely related but separable [59]. Both the parallel analysis and EFA were conducted in R using the ‘psych’ package [63]. To best operationally define our factors, factor loadings > 0.3 were retained [59]. Factor scores quantifying individual differences in underlying factors were obtained using the regression method. Functional domains describing the interrelationships of factors were empirically determined via factor score correlations and visualized using a spring-embedded graph [64]. This spring-embedded graph was computed in MATLAB (‘graph’ function) using minimal correlation weights, such that each factor was connected to at least one other factor.
Subtype identification
To identify whether distinct phenotypic subtypes exist within the BE group (N = 149), we implemented a latent profile analysis (LPA) using the EFA factor scores as input. LPA, a form of Gaussian-mixture modelling (GMM), is a commonly used model-based clustering approach which assumes the data is distributed according to a mixture of multivariate Gaussian distributions [65]. The LPA was performed in R (4.1.2) using the ‘mclust’ package [65]. The model was fitted by the EM algorithm and initialized by model-based hierarchical clustering based on scaled singular value decomposition [65]. LPA models with one to nine profiles were fit to determine the optimal number of clusters/subtypes. Bayesian Information Criterion (BIC) [66] and Integrated Complete-Data Likelihood (ICL) were used to assess model fit [66], with model selection determined by the lowest BIC and ICL [65]. Bootstrap Likelihood Ratio Tests (BLRTs) [66] were conducted to compare the fit between a k-profile and a k − 1-profile model. We additionally considered model parsimony and whether a solution included small profiles (<5% of the sample size) [67, 68]. According to simulations [69,70,71], this analysis had adequate statistical power (sample size: N = 149; 12 continuous input variables; N = 24 in the smallest profile; observed effect sizes for profile differentiation: partial-eta squared = 0.08–0.48). Briefly, the power was considered adequate due to subtype sample sizes being expected to be equal based on theoretical considerations [see [31]], the large number of (continuous) input variables included for profile differentiation, the smallest observed subtype sample size being N > 20, and robust group separation being demonstrated by the large effect sizes distinguishing the subtypes [69,70,71].
Model stability and validation
When utilizing a mixture model for subtyping, uncertainties arise regarding the optimal model choice (e.g., number of clusters/subtypes, certainty of profile assignments) and the estimation of model parameters. To address these concerns, we employed various diagnostic statistics and sensitivity analyses as summarized here and reported more fully in the Supplementary Methods. To validate the model, we employed: a) entropy, which assesses the model’s certainty in assigning individuals to clusters [67, 68], b) Average Posterior Probability, which gauges the accuracy of individual profile membership predictions [67, 68], and c) mixture model discriminant analysis, assessing the ability of a classifier to predict the obtained clusters under 10-fold cross-validation [65, 72]. All uncertainty metrics were evaluated against standard goodness-of-fit thresholds specific to each measure. We additionally validated the stability of our GMM under random restarts of the EM algorithm to avoid local maxima [65], and we quantified uncertainty of the resulting model parameters via resampling with the percentile bootstrap approach [65, 73].
Subtype phenotypic characterization
Control group assignment
To reduce bias and avoid skewed results in statistical comparisons due to unbalanced sample sizes, we matched a control group to the largest subtype. This control group, selected from the existing sample of controls, was matched for age, gender, and body mass index (BMI), using the ‘matchControls’ function from the R ‘e1071’ library [74]. We verified the control group was consistently well-matched across all identified subtypes, ensuring no significant differences in age, gender, or BMI.
Phenotypic profiles
To delineate phenotypic profiles, we characterized the subtypes using the EFA-derived factors. In the primary analysis each subtype was compared to the matched control group, while the secondary analysis compared the subtypes to each other. We utilized multivariate analysis of covariance (MANCOVA), covarying for age, gender, and BMI. All comparisons were corrected for multiple testing (adjusting for number of EFA factors) using a false discovery rate (FDR) threshold of p-FDR < 0.05.
Internal clinical validation measures
We additionally used measures of demographics and clinical characteristics, not incorporated in the subtyping analysis (LPA), to internally validate the subtyping solution. These measures included: a) the EDE-Q [55], capturing ED symptoms and pathology, b) the Three-Factor Eating Questionnaire (TFEQ) [75], examining cognitive and behavioral aspects of eating behavior, c) the Adult Self-Report (ASR) [76], measuring substance use frequency, and 4) the Structured Clinical Interview for DSM-IV [77], assessing subject clinical diagnoses. Further measurement details are provided in the Supplementary Methods. MANCOVAs and chi-squared analyses were applied where appropriate (two-sided), with FDR adjustment for multiple comparisons (corrected for number of measures) in post-hoc analyses.
Subtype neurobiological characterization
Detailed information on the MRI acquisition, quality control, pre-processing, functional connectivity (FC) and graph theory analysis can be found in the Supplementary Methods. In short, after assessing data quality using the automated MRI Quality Control tool (MRIQC) [78], we preprocessed the anatomical and functional data (rs-fMRI) using fMRIPrep 20.2.1 [79] on the Minnesota Supercomputing Institute’s High Performance Computing cluster (www.msi.umn.edu). Next, we conducted the FC analysis in MATLAB (version 2021a; The MathWorks, Inc.), using the brain connectivity toolbox (https://sites.google.com/site/bctnet/) to compute graph theory metrics. The graph analysis focused on four key metrics that were computed for each brain region (or ‘node’): global functional integration (global efficiency), local functional integration/ global segregation (local efficiency), modularity (participation coefficient), and centrality (degree centrality). These metrics assess functional topology during resting-state. Global efficiency measures how efficiently brain regions are functionally coupled across the entire brain, reflecting a region’s capacity to effectively communicate and integrate information across the brain, while local efficiency measures functional coupling with neighboring brain regions, reflecting its capacity for locally integrated (but globally segregated) processing [80, 81]. The participation coefficient quantifies a region’s functional integration within its own (versus other) ‘module’, while degree centrality assesses a brain region’s overall functional connectedness, reflecting its functional importance and influence on information processing [80, 81]. For further details, see the Supplementary Methods. We conducted generalized linear models (GLMs), comparing each subtype to the matched control group for each of the four graph metrics, adjusting for age and BMI, and correcting for multiple comparisons (correcting for number of parcellated brain regions [N = 379]) at p-FDR < 0.05.
Results
Phenotypic results
Bartlett’s test of Sphericity (χ2 = 21,947.39, p < 0.001) and Kaiser–Meyer–Olkin test (KMO = 0.86) confirmed data suitability for EFA. Across all participants (N = 612), parallel analysis and EFA identified 12 significant latent factors (p < 0.05), reducing the initial 74-variable phenotypic space to a 12-dimensional phenotypic latent space, explaining 45% of the common variance (see Supplementary Fig. 2 for the scree plot). Model fit indices indicated good separation (RMSEA = 0.046, Tucker-Lewis Index = 0.82), and each factor met the minimum requirement of three salient loadings (>|0.30|) [59]. See Supplementary Table 3 and Supplementary Table 4 for details on the 12 factors, labels, and loadings. Subjects with outliers on factor scores (>3 SD from the mean) were excluded from further analysis (N = 1). The spring-embedded plot, derived from the underlying factor correlations, revealed the self-organization of the twelve latent factors into the three hypothesized functional domains (Fig. 1; see Supplementary Table 5 for factor correlations).
Visualization of how the latent factors, identified through exploratory factor analysis, empirically self-organized into the hypothesized three domains. Each node represents a latent factor, with spatial proximity reflecting the strength of correlations – strongly correlated nodes are plotted closer together. The emergent functional domains are color-coded in the spring-embedded network: Blue = Executive Functioning Domain, Red = Negative Emotionality Domain, Green = Approach Behavior Domain.
Subtyping results
Among the nine profiles assessed, the three-profile model exhibited the lowest BIC and ICL indicating superior fit (see Supplementary Table 6 for model fit indices). The significant BLRT supported the three-profile model’s goodness-of-fit relative to the two-profile model (Supplementary Table 6). Of note, BLRT p-values were significant for all models, except when comparing the seven-profile to the six-profile models, limiting the utility of this indicator in model selection (Supplementary Table 6). Given the convergence of BIC and ICL on the three-profile solution for optimal fit, and that this solution provided a sufficient sample size for each profile (>5% of sample), this model was selected for further validation.
Multiple model stability and validation analyses all strongly supported the robustness of the three-profile model (Supplementary Tables 7–10, Supplementary Fig. 3, and Supplementary Fig. 4). Overall, model entropy (0.82), average posterior probability (0.91–0.96), and cross-validated mixture model discriminant analysis (F1 score = 0.98) all indicated high quality classification and profile separation. Results held over repeated random initializations, identified a 3-profile solution with VEI variance-covariance parameterization as the optimal model. See Supplement for mixture model SEs and CIs, based on non-parametric bootstrap (Supplementary Table 10, Supplementary Fig. 4).
Subtype phenotypic & clinical characterization
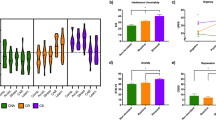
Based on the 3-profile solution, we found a) a ‘Negative Emotionality’ type (N = 24) with significantly higher internalizing, general psychiatric symptoms, negative affect, (lack-of) effortful control, and lower extraversion/sociability; b) an ‘Approach’ type (N = 75) with significantly higher unethical behavior/norm-breaking, extraversion/sociability, urgency and lower risk perception; c) and a ‘Restrained’ type (N = 50) with significantly higher risk perception, effortful control, extraversion/sociability, and lower unethical behavior/norm-breaking, urgency, internalizing and general psychiatric symptoms (See Fig. 2, and Table 1). Secondary analysis confirmed that subtype profiles were consistent in both between-subtype comparisons and comparisons with controls (Table 1, Fig. 3, and Supplementary Table 11). Results of MANCOVAs and chi-squared tests for these comparisons are detailed in Table 1, Fig. 3, and Supplementary Table 11. Each subtype also demonstrated distinct clinical profiles, generally consistent in comparisons both between subtypes and with controls. The ‘Negative Emotionality’ type exhibited significantly higher ED psychopathology (EDE-Q: Restraint, Eating, Shape and Weight Concern) (Fig. 3), and a greater prevalence of current internalizing disorders (SCID-IV) (Supplementary Table 11). The ‘Approach’ type reported significantly more frequent substance use (over past 6-months), particularly in days drunk (ASR), had the lowest dietary restraint levels (TFEQ), and a higher prevalence of past substance use disorders (Fig. 3, Supplementary Table 11). The ‘Restrained’ type exhibited significantly lower disinhibition (TFEQ) and higher levels of dietary restraint (TFEQ) (Fig. 3), with psychiatric disorder prevalence similar to matched controls (Supplementary Table 11). Among the identified subtypes, only the ‘Negative Emotionality’ type had a significantly higher proportion of OBEs meeting clinical-level threshold, while the ‘Approach’ and ‘Restrained’ types had similar proportions of both clinical and subclinical OBEs (Table 1). Additionally, no differences between subtypes were observed regarding purging behaviors (Table 1). Lastly, the ‘Approach’ type had a higher BMI compared to controls (Table 1), with no other demographic differences observed across subtypes or between subtypes and controls regarding age, gender, race, education or socioeconomic status (Table 1).
Heatmap illustrating the contribution of input variables to the three-profile subtyping solution. Each cell represents the relative impact or importance of a feature (x-axis) to defining the profile, with color indicating both the direction and strength of the contribution. Red indicates a strong, positive contribution (i.e., an increase in the feature significantly influences the profile); Blue indicates a strong, negative contribution (i.e., a decrease in the feature significantly influences the profile). Positive and negative contributions are weighted equally. Values are standardized to the [−1,1] range. Colored boxes around feature labels (x-axis) indicate relationship to phenotypic domain: Red = Negative Emotionality, Green = Approach Behaviors, Blue = Executive Function.
A–C: Subtype and control comparisons on internal clinical validation measures - assessments not included in subtype formation. All comparisons were corrected for multiple-comparisons (p-FDR < 0.05). Asterisk denotes statistical significance at *p-FDR < 0.05, **p-FDR < 0.01, ***p-FDR < 0.001, ****p-FDR < 0.0001.
Subtype functional connectivity profiles
No significant differences in framewise displacement were observed between groups (Table 1). Unique neurobiological patterns linked to BE were identified in two subtypes as described below (p-FDR < 0.05; Fig. 4, see Supplementary Table 12 and Supplementary Table 13 for subtype- and metric-specific details). After multiple comparison correction (p-FDR < 0.05), the ‘Negative Emotionality’ subtype (N = 24) did not exhibit significant differences compared to matched controls across FC metrics.
Subtype-specific neurobiological profiles, determined by graph theory analysis of resting-state functional connectivity (p-FDR < 0.05). A Approach Subtype: Cortical results. B Restrained Subtype: Cortical results. C Restrained Subtype: Subcortical results. Depicted results are collapsed across graph metrics. For subtype-specific results by graph metric, see Supplementary Table 12 and 13. Warm colors represent increased beta values, and cool colors represent decreased beta values.
Compared to matched controls, after multiple comparison correction (p-FDR < 0.05), the ‘Approach’ type (N = 75; Fig. 4, Supplementary Table 12) demonstrated increases in both local efficiency and degree centrality. Increased local efficiency was observed in regions of the DMN (ventral and dorsal posterior cingulate), Parietal Association (Premotor cortex), and Sensorimotor (Early somatosensory and motor cortex), while increased degree centrality was observed in regions of the Dorsal FPN (dorsolateral prefrontal [dlPFC], Superior parietal, Inferior parietal), Somatosensory (Posterior opercular), and Visual (Inferior parietal, Dorsal stream) networks. Additionally, a few regions demonstrated decreased global efficiency, including the dorsal ACC, dlPFC, and within the temporal-parietal-occipital junction, whereas increased global efficiency was observed in two regions of the visual cortex.
Compared to matched controls, after multiple comparison correction (p-FDR < 0.05), the ‘Restrained’ type (N = 50; Fig. 4, Supplementary Table 13) exhibited widespread changes, marked by reductions in global efficiency, local efficiency, and participation coefficient. Decreased global efficiency was observed in regions of the Salience (anterior cingulate cortex/medial prefrontal cortex), Ventral FPN (inferior frontal, inferior parietal), Dorsal FPN (posterior dlPFC) and Reward (inferior frontal) networks. Decreased local efficiency was demonstrated in regions of the Salience (anterior cingulate cortex/medial prefrontal cortex), Ventral FPN (inferior frontal), Dorsal FPN (dlPFC), Reward (orbital-polar frontal) networks, and sub-cortically (bilateral thalamus). Decreased participation coefficient was observed in regions of the Salience (anterior cingulate cortex/medial prefrontal cortex), Dorsal FPN (dlPFC), Ventral FPN (inferior frontal), Reward (orbital-polar frontal, inferior frontal) networks, and in subcortical regions (bilateral putamen, bilateral thalamus, and brain-stem).
Discussion
We applied a rigorous, data-driven, mechanism-based approach to characterize heterogeneity in the mechanisms underlying BE behavior using integrated multidimensional data across three hypothesized neurobehavioral domains: approach behavior, executive functioning, and negative emotionality. Three subtypes emerged, each demonstrating unique profiles across these domains, as well as distinct clinical characteristics and neurobiological functioning. Individuals in the ‘Negative Emotionality’ type exhibited greater negative affect, lack of effortful control (poorer emotion regulation) and increased psychiatric comorbidity (e.g., highest general psychiatric and internalizing behaviors). Individuals in the ‘Approach’ type exhibited higher approach-related and impulsive behaviors (e.g., highest unethical behavior/norm-breaking, urgency, and current substance use, lowest risk-perception) and increased brain connectivity in regions of the frontoparietal, somato-motor and visual networks. Individuals in the ‘Restrained’ type were characterized by overcontrolled, restrictive and harm-avoidant behaviors (e.g., highest effortful control and risk-perception, lowest unethical behavior/norm-breaking), along with decreased functional connectivity in regions of the salience, frontoparietal and reward networks and subcortical regions. Importantly, while previous research has emphasized the importance of approach behavior, executive functioning, and negative emotionality in BT-EDs [36, 37, 42, 51], this study represents the first to investigate heterogeneity in BE across a comprehensive and multi-modal assessment of these domains in a single sample, revealing complexities of BE pathology beyond clinical severity and diagnostic classification.
Our factor analysis provided strong empirical support for a phenotypic space of functioning which empirically self-organized into the three hypothesized functional domains of interest: negative emotionality, executive function, and approach behavior. Additionally, consistent with previous research in an independent dataset [58], our analysis revealed that two typically ‘cognitive’ factors, specifically ‘effortful control’ and ‘urgency’, did not self-organize into the executive function domain but were robustly associated with factors in the negative emotionality domain. This link between these typically ‘cognitive’ factors and negative emotionality aligns with the well-established relationship between affective processes and emotion regulation [82, 83]. Further examination of the ‘executive function’ and ‘negative emotionality’ domains reveals critical distinctions in their construction which may also contribute to this finding. The ‘executive function’ domain primarily consisted of factors that loaded on task-based measures of cognition, while the ‘effortful control’ and ‘urgency’ factors—linked to the negative emotionality domain—were derived from questionnaires on impulsivity, temperament, and personality. This distinction between data modalities is important, as questionnaires and task-based measures capture different constructs that are minimally correlated [84, 85]. Task-based measures probing cognition evaluate cognitive performance under standardized conditions, whereas questionnaires assess self-reported executive difficulties influenced by mood and context [84].
The ‘Negative Emotionality’ subtype that we identified was characterized by heightened affective disturbance and emotion dysregulation. Emotion/affect regulation is a central and empirically supported concept across several BE theories, which propose that BE functions to manage, moderate, and avoid aversive emotions and states [32]. Accordingly, individuals within this subtype may uniquely struggle with self-regulating negative affect, potentially driving BE as a form of negative reinforcement to escape or relieve aversive states [35, 50]. This ‘Negative Emotionality’ type shared characteristics with both the commonly identified ‘dietary-negative affect’ and ‘undercontrolled’ subtypes from prior ED research [29]. Similar to the ‘dietary-negative affect’ subtypes, our ‘Negative Emotionality’ type was marked by heightened negative affect and internalizing behaviors. Clinical validation measures further supported these similarities, demonstrating that our ‘Negative Emotionality’ subtype similarly exhibits elevated ED psychopathology (EDE-Q), increased rates of clinical-level OBEs, and a higher prevalence of current internalizing disorders compared with the other identified subtypes and controls. The ‘Negative Emotionality’ type also resembled the ‘undercontrolled’ subtype identified in prior research, which is characterized by emotion dysregulation, heightened negative emotions, and a lack of effortful control. However, an important distinction was that previously-defined ‘undercontrolled’ subtypes have also demonstrated approach-related dysregulation and novelty seeking, while our identified subtype was distinctly driven by dysregulation related to negative affect, lacking any approach-related deficits. Interestingly, our ‘Negative Emotionality’ subtype also paralleled a similar subtype identified in individuals with Substance Use Disorders, known as the ‘Relief’ subtype. This ‘Relief’ type is also characterized by heightened negative affect and emotion dysregulation, where negative affect is believed to drive alcohol or substance use as a means of ‘relief’ or coping mechanism [30, 86]. This underscores the crucial role of negative emotionality as a transdiagnostic construct underlying compulsive consumption of food, alcohol, and other substances, revealing shared mechanisms across disorders and its potential to differentially drive BE.
The ‘Approach’ subtype was characterized by high levels of impulsive, risk-taking, and externalizing behaviors. Adaptations (e.g., sensitization or habituation) in reward sensitivity may drive impulsive actions and poor decision-making, leading to maladaptive reward-driven behaviors that uniquely reinforce compulsive BE despite negative outcomes within this subtype [36, 37, 45]. This subtype also shared similarities with the ‘undercontrolled’ subtypes identified in prior ED research [29, 52], particularly in behaviors related to dysregulation and impulsivity within the context of approach behavior – such as heightened positive urgency and novelty-seeking. However, these similarities in dysregulation were not additionally related to negative emotions as observed in prior ED subtypes [29]. Unlike previous research, our findings distinguish two subtypes based on different aspects of dysregulation: one characterized by heightened negative affect, internalizing symptoms and emotion dysregulation (the ‘Negative Emotionality’ type), and another marked by uninhibited approach-related behaviors (the ‘Approach’ type). This characterization was further supported by clinical validation measures, which indicated the highest frequency of current substance use among this group, the lowest levels of dietary restraint, and a higher proportion of past substance use disorders among its members. We further found that this ‘Approach’ subtype closely resembled the ‘Reward’ type observed in individuals with Substance Use Disorder, where alcohol/drug-seeking behaviors are driven by underlying impairments in approach-related, sensation-seeking tendencies [30, 86]. Overall, these findings reaffirm the transdiagnostic nature of compulsive consumption and emphasize the role of approach-related mechanisms in differentially contributing to BE behaviors.
The ‘Restrained’ subtype was characterized by low approach behaviors and low negative emotionality, demonstrating risk-aversion and overcontrolled behaviors. This profile shared similarities with several previously identified subtypes in the ED literature [29], exhibiting inhibited and avoidant traits akin to the ‘overcontrolled’ ED subtypes, while also demonstrating characteristics of the ‘low psychopathology’ and ‘dietary’ subtypes [29], albeit with distinct differences. Despite having the lowest ED pathology (EDE-Q) and comorbid psychopathology among subtypes, this group demonstrated dietary restraint levels (TFEQ) comparable to the ‘Negative Emotionality’ type and a similar proportion of clinical-level OBEs as the ‘Approach’ type. The characteristic overcontrolled, restrained and harm-avoidant behaviors underlying this ‘Restrained’ type, identified even without BMI variations between groups, suggests a pathway to BE that is distinct from typical internalizing or externalizing pathways to compulsive behavior. This observation challenges notions of severity based primarily on the presence of comorbid psychopathology or obesity, highlighting the complex role of restraint in BE. Historically, restraint was among the first cognitive factors linked to BE development and maintenance [32], theorized to function as either a cause or consequence of BE behaviors. While recognized as a risk factor for (and/or a consequence of) BE, only a small subset of highly restrained individuals develop BE [87], indicating that restraint may increase susceptibility to BE and BT-EDs, especially in individuals with a genetic predisposition [87]. This interpretation could offer an explanation into the distinct features contributing to the risk and development of BE in individuals within this subtype. Interestingly, the concept of restraint is not uncommon in compulsive disorders, particularly substance addiction [88]. Unsuccessful attempts to cut down or abstain, despite wanting to, are prevalent in addiction and included in DSM substance use criteria, reflecting the unsuccessful restraint observed in BE [88]. Importantly, while the ‘Negative Emotionality’ and ‘Approach’ types in this study appeared to replicate the ‘Relief’ and ‘Reward’ phenotypes from the broader addiction literature [30, 86], this ‘Restrained’ subtype, to our knowledge, has not been similarly identified in addiction. This suggests that the ‘Restrained’ subtype represents a unique perspective and potential pathway for compulsive BE, where unsuccessful restraint and overcontrolled behaviors may serve as both a cause or consequence underlying compulsive behaviors [32, 88].
Our subtype-specific findings revealed distinct neurobiological features aligned with each subtype’s phenotypic profile, demonstrating distinct brain functional topology and information processing patterns per subtype. The ‘Approach’ subtype exhibited increased local efficiency in regions of the DMN, Sensorimotor, and Parietal Association networks. Heightened local efficiency reflects enhanced local information processing but reduced cross-talk globally between brain networks [89]. Degree Centrality was both increased and decreased in different regions of the FPN, and increased in Visual and Somatosensory networks. Such dysregulated functional organization in frontoparietal brain networks has been associated with cognitive deficits, impulsivity, biased attention, and impaired motor planning [37, 38, 49, 90,91,92,93,94,95], and also distorted self- and body-image perceptions central to ED pathology [39, 40, 92, 93]. Notably, aberrant connectivity in the sensorimotor and visual networks also contributes to impaired attentional processing, motor control, and body perception, as observed in BT-EDs [92,93,94, 96, 97]. Increased connectivity within these networks has also been linked to psychopathological symptoms in BN patients [92, 93]. Furthermore, the DMN, crucial for self-referential processing and interoception [49, 98], shows both significant hyper- and hypo-connectivity connectivity across BT-EDs, suggesting a role in maladaptive self-focused processing, emotional memories, and persistent preoccupation with food and body image [38, 40, 94, 97]. Enhanced segregation of processing in these networks may also reflect enhanced sensory and self-referential processing [89]. Interestingly, crucial regions in the Salience and FPN, including the dorsal ACC and dlPFC, also showed decreased global efficiency. This finding points to decreased global processing efficiency in networks critical for attentional control, and re-orienting salient stimuli [34, 45, 49, 98], aligning with the impulsive and compulsive characteristics observed in this subtype. Connectivity disruptions within the ACC and PFC, have also been associated with maladaptive response inhibition and reward processing, potentially underlying the loss-of-control and impulsive behaviors observed in BT-EDs [34, 37, 38, 40, 45] and with other compulsive behaviors [43, 45, 49]. Taken together, these neurobiological findings support prior research linking disruptions in these networks to maladaptive impulsivity, compulsivity, and ED-related pathology in BT-EDs and other impulsive-compulsive populations [37, 38, 45]. In particular, this subtype-specific neurobiological pattern may reflect a maladaptive prioritization of sensory and self-referential processing, limiting the brain’s ability to allocate attention, motor control, and goal-directed behaviors to other processes, ultimately reinforcing compulsive BE. This distinct pattern may serve as a unique marker for the characteristic externalizing, impulsive behaviors, and elevated ED pathology in this ‘Approach’ subtype, and potentially guide the development of targeted, brain-based interventions.
In contrast, the ‘Restrained’ subtype demonstrated a pattern of disrupted information processing, evidenced by the decreased global efficiency, local efficiency, and participation coefficient in brain regions associated with the FPN, salience and reward networks, and notably the bilateral putamen and thalamus - regions consistently implicated in BT-EDs [34, 37, 39,40,41, 92, 99]. These findings suggest a disruption in information flow and network integration crucial for approach behaviors and effortful control, aligning with prior research indicating reduced participation coefficient in these networks and subcortical regions in BN [100] and reduced fronto-striatal functional connectivity in BED [101] and across BT-EDs [38, 101]. Furthermore, reflecting the overcontrolled and restrained characteristics of this subtype, its neurobiological profile aligns with and extends previous research on the neural correlates of restrained eating [102,103,104,105,106,107]. While similar patterns are observed among highly restrained, yet healthy, ‘emotional eaters’ [105], fMRI research investigating restraint in clinically-relevant populations are rare, including only (to our knowledge) two studies [106, 107]. Consistent with our findings, these studies revealed reduced activation in impulse-control regions inversely correlated with dietary restraint in BED [106] and hypoconnectivity within the dlPFC and between the dlPFC and regions related to attention and reward valuation in individuals with high BN symptoms and restrained eating tendencies [107]. This pattern of dysconnectivity may potentially serve as a biomarker for individuals with risk-averse, highly restrained tendencies and BE, providing potential for neurobiologically targeted subtype-specific interventions. While previous research has identified neurobiological differences in BE and in BT-EDs, particularly in networks related to reward-related processing, executive control, and self-referential processing [37,38,39,40,41,42], definitive biomarkers remain elusive [37,38,39, 41, 42], likely due to a reliance on categorical diagnoses and standard case-control comparisons that obscure or lack sensitivity in identifying neurobiological impairments [21, 30, 108]. Our findings provide additional support for the notion that BT-EDs are characterized by disrupted communication between distributed brain regions and networks [96] and provide the first evidence within BT-EDs that a mechanism-based subtyping approach can reveal distinct neurobiological patterns aligned with each subtype’s unique phenotypic profile and BE behaviors. This highlights the potential for identifying subtype-specific biomarkers to inform the development of targeted interventions.
Our findings demonstrate the heterogeneous roles of negative emotionality, approach behaviors, and effortful control/urgency in BE within a single sample, underscoring the importance of assessing these domains in future research to inform more comprehensive models of BE etiology and treatments. They also demonstrate how different approach behaviors contribute to distinct pathways of BE (e.g., increases or decreases in unethical behaviors, risk-perception), supporting recent calls to integrate a range of reward-related processes alongside established factors like affect and cognition in BT-ED research [36, 37, 42, 51]. The involvement of these functional domains in BE supports the growing evidence that links BE to other conditions characterized by impulsive and compulsive behaviors, emphasizing their dimensional nature [30, 42,43,44,45,46, 51, 109]. Moreover, our inclusive sample encompassed a range of BE behaviors, from low frequency to clinical tendencies, recognizing the transdiagnostic nature of BE and the significance of subclinical and low-frequency presentations.
The potential of psychiatric subtyping ultimately lies in its ability to improve mechanistic understanding of pathophysiology and guide precision medicine. Tailoring interventions to address the most pertinent underlying mechanisms driving each subtype offers substantial clinical promise [29, 30, 110]. Our findings provide empirical evidence for significant variation in neurobehavioral impairments associated with BE highlighting opportunities for targeted, subtype-specific interventions. The ‘Negative Emotionality’ subtype, like previously identified ED subtypes with heightened affective disturbance, may benefit most from treatments focused on affect regulation and enhancing tolerance to negative emotions [29, 52], including psychotherapies and pharmacotherapies used in anxiety and depression [111,112,113]. Interventions addressing impulsivity, risk-perception, and addictive behaviors may be particularly effective for the ‘Approach’ subtype. Interventions focused on impulse control and urgency, especially those targeting approach behaviors and self-regulation, are likely to benefit this group [110, 112, 113]. For example, lisdexamfetamine, a medication also used for enhancing effortful control in ADHD, demonstrates efficacy in reducing BE frequency, trait-impulsive features of BE, and improving regulation of eating and goal-directed behaviors [112, 114]. Given this subtype’s neurobiological profile, brain-based interventions such as excitatory non-invasive brain stimulation (NIBS) targeting hypo-active salience network nodes have shown efficacy in reducing impulsivity, improving attention, and decreasing cue-induced cravings and BE behaviors [38, 110], potentially offering targeted benefits for this subtype. For the ‘Restrained’ subtype, interventions targeting cognitive flexibility and rigidity, rather than emotional targets, may provide an effective therapeutic approach [110, 112, 113]. Given this subtype’s neurobiological profile, inhibitory NIBS targeting the dlPFC and FPN, rather than excitatory, may be most effective in addressing overcontrolled and rigid behaviors [110]. Overall, we argue that therapies targeting mechanism-based skills, may prove effective in addressing the characteristic impairments associated with the identified BE subtypes.
The current study is only an initial step into the comprehensive investigation of mechanism-based subtypes of BE and is not without limitations. While clustering methods have limitations [23, 108], we took several steps to validate and ensure subtype stability. Nonetheless, external validation remains the gold standard for confirming cluster generalizability and replicability, necessitating independently collected datasets [115]. Additionally, while the power for the current study was adequate, replication will provide a more robust characterization of the recovered subtypes. For example, the smaller sample size of the ‘Negative Emotionality’ subtype (N = 24) likely impeded the detection of widespread, robust, and meaningful neurobiological findings for this group [116]. Notably, the cross-sectional nature of this sample limits our ability to determine whether these subtypes predate the onset of BE or emerge as a consequence of BE. Future research should utilize larger sample sizes and longitudinal, follow-up or post-treatment data to enhance the identification of subtype-specific neurobiological markers, as well as evaluate developmental trajectories, treatment efficacy and changes over time [23].
Conclusions
This study is the first to investigate the mechanistic heterogeneity of BE through a comprehensive, multi-modal assessment of three neurofunctional domains linked to compulsive behaviors in a single sample. Using a purely data-driven mechanism-based approach, three distinct subtypes of BE were identified, characterized by unique behavioral and neurobiological profiles, capturing functionally distinct expressions of BE pathology beyond clinical severity and diagnostic classifications. These findings underscore the need for updated BE etiology and maintenance models that integrate approach/reward, effortful control/impulsivity, and negative emotionality and their underlying neural correlates. With further validation, these findings could inform nosology and bolster the development of personalized behavioral, pharmacological, and neurobiological interventions tailored to individual mechanistic differences in BE and BT-EDs.
Data availability
Nathan Kline Institute-Rockland Sample data access and details are available on the study website (https://fcon_1000.projects.nitrc.org/indi/enhanced/access.html). Phenotypic data are protected by a Data Usage Agreement, which must be completed and approved by an institutional official before access is granted (https://fcon_1000.projects.nitrc.org/indi/enhanced/phenotypicdata.html). The neuroimaging data are available for download from an Amazon S3 Bucket: s3://fcp-indi/data/Projects/RocklandSample/RawDataBIDSLatest.
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
Galmiche M, Déchelotte P, Lambert G, Tavolacci MP. Prevalence of eating disorders over the 2000–2018 period: a systematic literature review. Am J Clin Nutr. 2019;109:1402–13.
Santomauro DF, Melen S, Mitchison D, Vos T, Whiteford H, Ferrari AJ. The hidden burden of eating disorders: an extension of estimates from the global burden of disease study 2019. Lancet Psychiatry. 2021;8:320–8.
Frank GKW, Berner LA, editors. Binge eating: a transdiagnostic psychopathology [Internet]. Cham: Springer International Publishing; 2020. http://link.springer.com/10.1007/978-3-030-43562-2.
Ágh T, Kovács G, Supina D, Pawaskar M, Herman BK, Vokó Z, et al. A systematic review of the health-related quality of life and economic burdens of anorexia nervosa, bulimia nervosa, and binge eating disorder. Eat Weight Disord. 2016;21:353–64.
Hambleton A, Pepin G, Le A, Maloney D, Aouad P, Barakat S, et al. Psychiatric and medical comorbidities of eating disorders: findings from a rapid review of the literature. J Eat Disord. 2022;10:132.
Mandelli L, Arminio A, Atti AR, Ronchi DD. Suicide attempts in eating disorder subtypes: a meta-analysis of the literature employing DSM-IV, DSM-5, or ICD-10 diagnostic criteria. Psychol Med. 2019;49:1237–49.
Boswell RG, Potenza MN, Grilo CM. The neurobiology of binge-eating disorder compared with obesity: implications for differential therapeutics. Clin Ther. 2021;43:50–69.
Burton AL, Abbott MJ. Processes and pathways to binge eating: development of an integrated cognitive and behavioural model of binge eating. J Eat Disord. 2019;7:18.
Crow SJ, Stewart Agras W, Halmi K, Mitchell JE, Kraemer HC. Full syndromal versus subthreshold anorexia nervosa, bulimia nervosa, and binge eating disorder: a multicenter study. Int J Eat Disord. 2002;32:309–18.
Withnell SJ, Kinnear A, Masson P, Bodell LP. How different are threshold and other specified feeding and eating disorders? Comparing severity and treatment outcome. Front Psychol. 2022;13:784512.
Johnson SN, Forbush KT, Swanson TJ, Christensen KA. An empirical evaluation of the diagnostic threshold between full-threshold and sub-threshold bulimia nervosa. Eat Behav. 2021;42:101540.
Dang AB, Giles S, Fuller-Tyszkiewicz M, Kiropoulos L, Krug I. A systematic review and meta-analysis on the DSM–5 severity ratings for eating disorders. Clin Psychol: Sci Pract. 2022;29:325–44.
Stice E, Marti CN, Shaw H, Jaconis M. An 8-year longitudinal study of the natural history of threshold, subthreshold, and partial eating disorders from a community sample of adolescents. J Abnorm Psychol. 2009;118:587–97.
Stice E, Gau JM, Rohde P, Shaw H. Risk factors that predict future onset of each DSM-5 eating disorder: predictive specificity in high-risk adolescent females. J Abnorm Psychol. 2017;126:38–51.
Treasure J, Leslie M, Chami R, Fernández-Aranda F. Are trans diagnostic models of eating disorders fit for purpose? A consideration of the evidence for food addiction. Eur Eat Disord Rev. 2018;26:83–91.
Linardon J. Rates of abstinence following psychological or behavioral treatments for binge-eating disorder: meta-analysis. Int J Eat Disord. 2018;51:785–97.
Linardon J, Wade TD. How many individuals achieve symptom abstinence following psychological treatments for bulimia nervosa? A meta-analytic review. Int J Eat Disord. 2018;51:287–94.
Fichter MM, Quadflieg N, Hedlund S. Long-term course of binge eating disorder and bulimia nervosa: relevance for nosology and diagnostic criteria. Int J Eat Disord. 2008;41:577–86.
Quadflieg N, Fichter MM. Long-term outcome of inpatients with bulimia nervosa—results from the Christina Barz study. Int J Eat Disord. 2019;52:834–45.
Feczko E, Miranda-Dominguez O, Marr M, Graham AM, Nigg JT, Fair DA. The heterogeneity problem: approaches to identify psychiatric subtypes. Trends Cogn Sci. 2019;23:584–601.
Van Dam NT, O’Connor D, Marcelle ET, Ho EJ, Cameron Craddock R, Tobe RH, et al. Data-driven phenotypic categorization for neurobiological analyses: beyond DSM-5 labels. Biol Psychiatry. 2017;81:484–94.
Brucar LR, Feczko E, Fair DA, Zilverstand A. Current approaches in computational psychiatry for the data-driven identification of brain-based subtypes. Biol Psychiatry. 2023;93:704–16.
Keel PK, Brown TA, Holland LA, Bodell LP. Empirical classification of eating disorders. Annu Rev Clin Psychol. 2012;8:381–404.
Striegel-Moore R, Wonderlich S, Walsh BT, Mitchell J, Hilbert A, Wilfley D, et al. editors. Developing an evidence-based classification of eating disorders: scientific findings for DSM-5. Arlington, VA: American Psychiatric Publishing, Inc; 2011.
Peterson CB, Crosby RD, Wonderlich SA, Mitchell JE, Crow SJ, Engel S. Predicting group cognitive-behavioral therapy outcome of binge eating disorder using empirical classification. Behav Res Ther. 2013;51:526–32.
Sysko R, Hildebrandt T, Wilson GT, Wilfley DE, Agras WS. Heterogeneity moderates treatment response among patients with binge eating disorder. J Consult Clin Psychol. 2010;78:681–90.
Peterson CB, Crow SJ, Swanson SA, Crosby RD, Wonderlich SA, Mitchell JE, et al. Examining the stability of DSM–IV and empirically derived eating disorder classification: Implications for DSM–5. J Consult Clin Psychol. 2011;79:777–83.
Wildes JE, Marcus MD. Alternative methods of classifying eating disorders: models incorporating comorbid psychopathology and associated features. Clin Psychol Rev. 2013;33:383–94.
Drossel G, Brucar LR, Rawls E, Hendrickson TJ, Zilverstand A. Subtypes in addiction and their neurobehavioral profiles across three functional domains. Transl Psychiatry. 2023;13:1–12.
Drossel G, Zilverstand A. Towards personalized medicine: subtyping using functional profiles. Neuropsychopharmacology. 2024;49:347–8.
Neyland MKH, Shank LM, Lavender JM. Theoretical development and maintenance models of binge eating. In: Frank GKW, Berner LA, editors. Binge eating: a transdiagnostic psychopathology. Cham: Springer International Publishing; 2020. p. 69–82.
Vainik U, García-García I, Dagher A. Uncontrolled eating: a unifying heritable trait linked with obesity, overeating, personality and the brain. Eur J Neurosci. 2019;50:2430–45.
Steward T, Berner LA. The neurobiological basis of executive function alterations in binge eating populations. In: Frank GKW, Berner LA, editors. Binge eating: a transdiagnostic psychopathology. Cham: Springer International Publishing; 2020. p. 137–52.
Dingemans A, Danner U, Parks M. Emotion regulation in binge eating disorder: a review. Nutrients. 2017;9:1274.
Leenaerts N, Jongen D, Ceccarini J, Van Oudenhove L, Vrieze E. The neurobiological reward system and binge eating: a critical systematic review of neuroimaging studies. Int J Eat Disord. 2022;55:1421–58.
Kessler RM, Hutson PH, Herman BK, Potenza MN. The neurobiological basis of binge-eating disorder. Neurosci Biobehav Rev. 2016;63:223–38.
Donnelly B, Touyz S, Hay P, Burton A, Russell J, Caterson I. Neuroimaging in bulimia nervosa and binge eating disorder: a systematic review. J Eat Disord. 2018;6:3.
Steward T, Menchon JM, Jiménez-Murcia S, Soriano-Mas C, Fernandez-Aranda F. Neural network alterations across eating disorders: a narrative review of fMRI studies. Curr Neuropharmacol. 2018;16:1150–63.
Mele G, Alfano V, Cotugno A, Longarzo M. A broad-spectrum review on multimodal neuroimaging in bulimia nervosa and binge eating disorder. Appetite. 2020;151:104712.
Hartogsveld B, Quaedflieg CWEM, van Ruitenbeek P, Smeets T. Volume and connectivity differences in brain networks associated with cognitive constructs of binge eating. eNeuro. 2022;9:ENEURO.0080-21.2021 https://www.eneuro.org/content/9/1/ENEURO.0080-21.2021.
Wonderlich JA, Bershad M, Steinglass JE. Exploring neural mechanisms related to cognitive control, reward, and affect in eating disorders: a narrative review of FMRI studies. Neuropsychiatr Dis Treat. 2021;17:2053–62.
Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3:760–73.
Michaud A, Vainik U, Garcia-Garcia I, Dagher A. Overlapping neural endophenotypes in addiction and obesity. Front Endocrinol. 2017;8:127.
Dawe S, Loxton NJ. The role of impulsivity in the development of substance use and eating disorders. Neurosci Biobehav Rev. 2004;28:343–51.
Moore CF, Sabino V, Koob GF, Cottone P. Pathological overeating: emerging evidence for a compulsivity construct. Neuropsychopharmacology. 2017;42:1375–89.
Belcher AM, Volkow ND, Moeller FG, Ferré S. Personality traits and vulnerability or resilience to substance use disorders. Trends Cogn Sci. 2014;18:211–7.
Dosenbach NUF, Fair DA, Cohen AL, Schlaggar BL, Petersen SE. A dual-networks architecture of top-down control. Trends Cogn Sci. 2008;12:99–105.
Zilverstand A, Huang AS, Alia-Klein N, Goldstein RZ. Neuroimaging impaired response inhibition and salience attribution in human drug addiction: a systematic review. Neuron. 2018;98:886–903.
Lavender JM, Wonderlich SA, Engel SG, Gordon KH, Kaye WH, Mitchell JE. Dimensions of emotion dysregulation in anorexia nervosa and bulimia nervosa: a conceptual review of the empirical literature. Clin Psychol Rev. 2015;40:111–22.
Schaefer LM, Forester G, Dvorak RD, Steinglass J, Wonderlich SA. Integrating aspects of affect, reward, and cognition to develop more comprehensive models of binge-eating pathology. Int J Eat Disord. 2023;56:1502–10.
Haynos AF, Pearson CM, Utzinger LM, Wonderlich SA, Crosby RD, Mitchell JE, et al. Empirically derived personality subtyping for predicting clinical symptoms and treatment response in bulimia nervosa. Int J Eat Disord. 2017;50:506–14.
Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10:186–98.
Nooner KB, Colcombe SJ, Tobe RH, Mennes M, Benedict MM, Moreno AL, et al. The NKI-rockland sample: a model for accelerating the pace of discovery science in psychiatry. Front Neurosci. 2012;6:152.
Fairburn CG, Beglin SJ. Assessment of eating disorders: interview or self-report questionnaire? Int J Eat Disord. 1994;16:363–70.
Kyriazos TA. Applied psychometrics: sample size and sample power considerations in factor analysis (EFA, CFA) and SEM in general. Psychology. 2018;9:2207–30.
Glorfeld LW. An improvement on Horn’s parallel analysis methodology for selecting the correct number of factors to retain. Educ Psychol Meas. 1995;55:377–93.
Rawls E, Kummerfeld E, Zilverstand A. An integrated multimodal model of alcohol use disorder generated by data-driven causal discovery analysis. Commun Biol. 2021;4:1–12.
Costello A, Osborne J. Best practices in exploratory factor analysis: four recommendations for getting the most from your analysis. Practical Assessment, Research, and Evaluation. 2005;10:1–9.
Muthen B, Kaplan D. A comparison of some methodologies for the factor analysis of non-normal likert variables: a note on the size of the model. Br J Math Stat Psychol. 1992;45:19–30.
Fuller EL, Hemmerle WJ. Robustness of the maximum-likelihood estimation procedure in factor analysis. Psychometrika. 1966;31:255–66.
Benson J, Fleishman JA. The robustness of maximum likelihood and distribution-free estimators to non-normality in confirmatory factor analysis. Qual Quant. 1994;28:117–36.
Revelle WR psych: procedures for personality and psychological research. 2017.
Huckins JF, Adeyemo B, Miezin FM, Power JD, Gordon EM, Laumann TO, et al. Reward-related regions form a preferentially coupled system at rest. Hum Brain Mapp. 2019;40:361–76.
Scrucca L, Fraley C, Murphy TB, Raftery AE. Model-based clustering, classification, and density estimation using mclust in R. New York: Chapman and Hall/CRC; 2023. p. 268 p.
McLachlan GJ, Rathnayake S. On the number of components in a gaussian mixture model. Wires Data Min Knowl Discov. 2014;4:341–55.
Spurk D, Hirschi A, Wang M, Valero D, Kauffeld S. Latent profile analysis: a review and “how to” guide of its application within vocational behavior research. J Vocat Behav. 2020;120:103445.
Nylund-Gibson K, Choi AY. Ten frequently asked questions about latent class analysis. Transl Issues Psychol Sci. 2018;4:440–61.
Dalmaijer ES, Nord CL, Astle DE. Statistical power for cluster analysis. BMC Bioinformatics. 2022;23:205.
Tein JY, Coxe S, Cham H. Statistical power to detect the correct number of classes in latent profile analysis. Struct Equ Modeling. 2013;20:640–57.
Nylund KL, Asparouhov T, Muthén BO. Deciding on the number of classes in latent class analysis and growth mixture modeling: a monte carlo simulation study. Struct Equ Modeling. 2007;14:535–69.
Hastie T, Friedman J, Tibshirani R. The elements of statistical learning. New York, NY: Springer; 2009. (Springer Series in Statistics)
Grün B, Leisch F Bootstrapping finite mixture models. 2004.
Meyer D, Dimitriadou E, Hornik K, Weingessel A, Leisch F, C++-code) CCC (libsvm, et al. e1071: misc functions of the department of statistics, probability theory group (Formerly: E1071). TU Wien. 2021.
Stunkard AJ, Messick S. The three-factor eating questionnaire to measure dietary restraint, disinhibition and hunger. J Psychosom Res. 1985;29:71–83.
Achenbach TM, Rescorla LA. Manual for the ASEBA adult forms and profiles. Burlington, VT. Burlington, VT.: University of Vermont, Research Center for Children, Youth and Families; 2003.
First MB. Structured clinical interview for DSM-IV axis I disorders (SCID-I), clinician version (administration booklet). Washington, DC: American Psychiatric Publishing, Inc; 1997.
Esteban O, Birman D, Schaer M, Koyejo OO, Poldrack RA, Gorgolewski KJ. MRIQC: advancing the automatic prediction of image quality in MRI from unseen sites. PLoS ONE. 2017;12:e0184661.
Esteban O, Markiewicz CJ, Blair RW, Moodie CA, Isik AI, Erramuzpe A, et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat Methods. 2019;16:111–6.
Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. Neuroimage. 2010;52:1059–69.
van den Heuvel MP, Sporns O. Network hubs in the human brain. Trends Cogn Sci. 2013;17:683–96.
Inzlicht M, Bartholow BD, Hirsh JB. Emotional foundations of cognitive control. Trends Cogn Sci. 2015;19:126–32.
Okon-Singer H, Hendler T, Pessoa L, Shackman AJ. The neurobiology of emotion-cognition interactions: fundamental questions and strategies for future research. Front Hum Neurosci. 2015;9:58.
Toplak ME, West RF, Stanovich KE. Practitioner review: do performance-based measures and ratings of executive function assess the same construct? J Child Psychol Psychiatry. 2013;54:131–43.
Friedman NP, Banich MT. Questionnaires and task-based measures assess different aspects of self-regulation: both are needed. Proc Natl Acad Sci USA. 2019;116:24396–7.
Mann K, Roos CR, Hoffmann S, Nakovics H, Leménager T, Heinz A, et al. Precision medicine in alcohol dependence: a controlled trial testing pharmacotherapy response among reward and relief drinking phenotypes. Neuropsychopharmacology. 2018;43:891–9.
Racine SE, Burt SA, Iacono WG, McGue M, Klump KL. Dietary restraint moderates genetic risk for binge eating. J Abnorm Psychol. 2011;120:119–28.
Schulte EM, Grilo CM, Gearhardt AN. Shared and unique mechanisms underlying binge eating disorder and addictive disorders. Clin Psychol Rev. 2016;44:125–39.
Sporns O, Chialvo DR, Kaiser M, Hilgetag CC. Organization, development and function of complex brain networks. Trends Cogn Sci. 2004;8:418–25.
Fedota JR, Stein EA. Resting-state functional connectivity and nicotine addiction: prospects for biomarker development. Ann N Y Acad Sci. 2015;1349:64–82.
Robbins TW, Gillan CM, Smith DG, de Wit S, Ersche KD. Neurocognitive endophenotypes of impulsivity and compulsivity: towards dimensional psychiatry. Trends Cogn Sci. 2012;16:81–91.
Wang J, Wu G, Wang M, Li W, Wang Y, Ren X, et al. Exploring the thalamus: a crucial hub for brain function and communication in patients with bulimia nervosa. J Eat Disord. 2023;11:207.
Lavagnino L, Amianto F, D’Agata F, Huang Z, Mortara P, Abbate-Daga G, et al. Reduced resting-state functional connectivity of the somatosensory cortex predicts psychopathological symptoms in women with bulimia nervosa. Front Behav Neurosci. 2014;8:270.
Stopyra MA, Simon JJ, Skunde M, Walther S, Bendszus M, Herzog W, et al. Altered functional connectivity in binge eating disorder and bulimia nervosa: a resting-state fMRI study. Brain Behav. 2019;9:e01207.
Morris LS, Baek K, Tait R, Elliott R, Ersche KD, Flechais R, et al. Naltrexone ameliorates functional network abnormalities in alcohol‐dependent individuals. Addict Biol. 2017;23:425.
Wang L, Kong QM, Li K, Li XN, Zeng YW, Chen C, et al. Altered intrinsic functional brain architecture in female patients with bulimia nervosa. J Psychiatry Neurosci. 2017;42:414–23.
Chen X, Ai C, Liu Z, Wang G. Neuroimaging studies of resting-state functional magnetic resonance imaging in eating disorders. BMC Med Imaging. 2024;24:265.
Menon V. Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn Sci. 2011;15:483–506.
Kung PH, Soriano-Mas C, Steward T. The influence of the subcortex and brain stem on overeating: how advances in functional neuroimaging can be applied to expand neurobiological models to beyond the cortex. Rev Endocr Metab Disord. 2022;23:719–31.
Lan Z, Zhu LL, Wu YK, Yang JJ, Li JT, Zeng YW, et al. Aberrant modular segregation of brain networks in female patients with bulimia nervosa. Int J Eat Disord. 2023;56:1353–64.
Haynos AF, Camchong J, Pearson CM, Lavender JM, Mueller BA, Peterson CB, et al. Resting state hypoconnectivity of reward networks in binge eating disorder. Cereb Cortex. 2021;31:2494–504.
Burger KS, Stice E. Relation of dietary restraint scores to activation of reward-related brain regions in response to food intake, anticipated intake, and food pictures. Neuroimage. 2011;55:233–9.
Dong D, Wang Y, Jackson T, Chen S, Wang Y, Zhou F, et al. Impulse control and restrained eating among young women: evidence for compensatory cortical activation during a chocolate-specific delayed discounting task. Appetite. 2016;105:477–86.
Zhang Y, Wang S, Wei L, Jackson T, Gao X, Xiao M, et al. Resting state differences between successful and unsuccessful restrained eaters. Brain Imaging Behav. 2021;15:906–16.
Wood SMW, Schembre SM, He Q, Engelmann JM, Ames SL, Bechara A. Emotional eating and routine restraint scores are associated with activity in brain regions involved in urge and self-control. Physiol Behav. 2016;165:405–12.
Balodis IM, Molina ND, Kober H, Worhunsky PD, White MA, Sinha R, et al. Divergent neural substrates of inhibitory control in binge eating disorder relative to other manifestations of obesity. Obesity. 2013;21:367–77.
Chen S, Dong D, Jackson T, Su Y, Chen H. Altered frontal inter-hemispheric resting state functional connectivity is associated with bulimic symptoms among restrained eaters. Neuropsychologia. 2016;81:22–30.
Marquand AF, Wolfers T, Mennes M, Buitelaar J, Beckmann CF. Beyond lumping and splitting: a review of computational approaches for stratifying psychiatric disorders. Biol Psychiatry Cogn Neurosci Neuroimaging. 2016;1:433–47.
Den Ouden L, Suo C, Albertella L, Greenwood LM, Lee RSC, Fontenelle LF, et al. Transdiagnostic phenotypes of compulsive behavior and associations with psychological, cognitive, and neurobiological affective processing. Transl Psychiatry. 2022;12:1–11.
Dunlop KA, Woodside B, Downar J. Targeting neural endophenotypes of eating disorders with non-invasive brain stimulation. Front Neurosci. 2016;10:30 https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2016.00030/full.
Sloan E, Hall K, Moulding R, Bryce S, Mildred H, Staiger PK. Emotion regulation as a transdiagnostic treatment construct across anxiety, depression, substance, eating and borderline personality disorders: a systematic review. Clin Psychol Rev. 2017;57:141–63.
Rodan SC, Bryant E, Le A, Maloney D, Touyz S, McGregor IS, et al. Pharmacotherapy, alternative and adjunctive therapies for eating disorders: findings from a rapid review. J Eat Disord. 2023;11:112.
Pisetsky EM, Schaefer LM, Wonderlich SA, Peterson CB. Emerging psychological treatments in eating disorders. Psychiatr Clin North Am. 2019;42:219–29.
Brucar LR, Hyder A, Birmingham CL. Normalizing the neural correlates of binge-eating disorder with lisdexamfetamine: a case report and literature review. Open J Psychiatry. 2018;8:142–51.
Ramspek CL, Jager KJ, Dekker FW, Zoccali C, van Diepen M. External validation of prognostic models: what, why, how, when and where? Clin Kidney J. 2021;14:49–58.
Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, et al. Reproducible brain-wide association studies require thousands of individuals. Nature. 2022;603:654–60.
Acknowledgements
This works was supported by the National Institute on Alcohol Abuse and Alcoholism (RO1AA029406 to AZ).
Author information
Authors and Affiliations
Contributions
Leyla Brucar: Conceptualization, Methodology, Validation, Formal Analysis, Data Curation, Writing – Original Draft, Visualization. Eric Rawls: Methodology, Validation, Writing – Review & Editing. Ann Haynos: Conceptualization, Writing – Review & Editing. Carol Peterson: Conceptualization, Writing – Review & Editing. Anna Zilverstand: Conceptualization, Methodology, Resources, Writing – Review & Editing, Visualization, Supervision, Project Administration, Funding Acquisition.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Nathan Kline Institute Institutional Review Board in accordance with the Declaration of Helsinki. Written informed consent and permission to share de-identified data were obtained from all study participants. For the present study, after data use permission was obtained, a protocol filed with the University of Minnesota Institutional Review Board met criteria for exemption (STUDY00007003, initial approval 06-27-2019).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Brucar, L.R., Rawls, E., Haynos, A.F. et al. Mechanism-based subtyping in binge eating: understanding neurobehavioral heterogeneity across negative emotionality, approach behavior, and executive function. Transl Psychiatry 15, 193 (2025). https://doi.org/10.1038/s41398-025-03408-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03408-1
This article is cited by
-
Subtypes of cocaine use disorder and their neurobehavioral profiles
Translational Psychiatry (2025)
-
Sex difference in neural substrates linking negative body image and disordered eating behaviors
Brain Structure and Function (2025)