Abstract
Digital tools for pre-test education provision and decision support could assist the scalability of opportunistic genomic screening. We evaluated the utility of a digital platform, the Genetics Adviser (GA), for supporting parental decisions about screening for additional findings in the paediatric acute care context. Parents of children who had completed ultrarapid diagnostic genomic testing in the acute setting were offered opportunistic screening following hospital discharge. Interested participants were provided with optional access to GA and offered pre-test genetic counselling (GC). GC sessions were audio-recorded, transcribed verbatim, and participant/counsellor interactions qualitatively analysed to examine the impact of GA use on counselling sessions. Surveys were administered: prior to and after pre-test GC; 1 month after return of results. One hundred and sixty-seven families were offered genomic screening and given access to GA. Family engagement with GA was 52% (87/167) overall, with three-quarters (81/119) of those who attended genetic counselling having engaged with GA. GA use impacted genetic counselling: in consultations where not all parents used GA, more concerns were raised and more questions asked about topics included in GA; GCs also spent more time clarifying values or understanding. GA users correctly answered more knowledge questions at every survey time point. Eighty-three per cent of post-result survey respondents believed GA contained enough information for them to make decisions about opportunistic screening without additional genetic counselling. These findings demonstrate the utility of GA in supporting the scalability of opportunistic genomic screening.
Similar content being viewed by others
Introduction
The use of genomic sequencing offers the opportunity to expand testing to include genomic screening, supporting early detection and intervention for genetic disease. However, there are challenges in scaling genetics service delivery to include screening. Traditional clinical genetic service delivery models incorporate pre- and post-test genetic counselling (GC), a clinical encounter with a specialised health care practitioner [1]. Pre-test GC includes elements of education and personalised decision support to help individuals make preference-sensitive decisions about undergoing genetic testing [2]. However, this traditional clinical genetics model is resource-intensive [3] and may not be appropriate or applicable for genomic screening settings. Novel approaches to health service delivery are needed to support broader availability of genomic screening.
Digital applications, including decision support tools, are one approach that could facilitate broader access to genomics. In clinical genetics, decision support tools have a long history of use in augmenting GC, particularly in prenatal and screening settings [4,5,6]. This is consistent with the purpose of decision support tools, which is to support preference-sensitive decision making, where the options do not have inherently superior health outcomes; the ‘right’ decision is that which aligns with an individual’s values. The more recent shift to digital tools has allowed for additional levels of complexity and customisation in information delivery and decision support [7, 8]. In genomic medicine, digital tools are used to support: risk assessment; pre-test education and counselling; return of results; and management [9]. A systematic review of patient-facing digital tools for delivering genetic services found positive effects on patient outcomes, including knowledge, psychosocial well-being, behaviour changes, family communication, and level of engagement [10]. Digital tools also led to efficiencies for health care practitioners [10].
Decisions about genomic sequencing can vary in their complexity: clinical testing may involve sequencing more than one person (trio testing) and, if screening is offered in addition to diagnostic testing, multiple decisions may be involved. Decisional multiplicity is known to complicate information provision and decision-making [11, 12]. Digital decision support tools are well-suited to help address this issue through their ability to provide information relevant to multiple decisions that families can work through at their own pace. For families in complex decision-making contexts, decision support tools are intended to augment, not replace, GC. Empirical evidence has demonstrated many benefits to digital tool use [10], but their impact on complex clinical consultations remains under-investigated.
To address this gap and generate insights to inform optimal use of digital decision support tools in genomic screening contexts, we evaluated the utility of the Genetics Adviser (GA) digital platform in supporting parents’ decisions about screening for additional findings in the paediatric acute care context. Additional findings were offered via a two-step model; participants were offered opportunistic genomic screening after results were returned from diagnostic genomic sequencing [13, 14]. We assessed uptake and use of the GA platform, and the impact of GA on the content of GC sessions.
Methods
Study design
The protocol for this study [14] provides details of the study design, including the modifications made to GA for this study. Uptake of screening results has previously been reported [13]. This study focuses on the impact of GA on GC and participant understanding of opportunistic genomic screening.
Participants
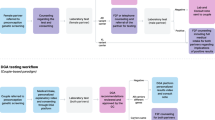
Participants were parents of a child who had had ultra-rapid trio/duo diagnostic genomic testing in the acute care setting at one of 17 hospitals across Australia through the Acute Care Genomics study [15]. In this study, the diagnostic test was focused on the analysis of genes associated with the child’s condition. Parents were offered up to three genomic screening tests after return of diagnostic results: for the child (for treatable and non-treatable childhood onset conditions); for each parent (for adult-onset conditions with publicly funded management or treatment available); for couple carrier screening (1283 genes [16]— individual carrier status not determined). Screening was offered between September 2020 and May 2023.
Genetic advisers use
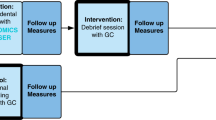
The GA platform [17, 18] pre-test modules (in English) were modified to support parental education, decision-making and consent about genomic screening for this study. Parents were provided access to GA prior to pre-test counselling; participants were not required to use it. GA was accessed via a web interface on the participant’s own device. Unique usernames were provided to each parent to allow tracking of individual users. Data was collected on login and duration of use of GA (captured for the most recent login). As the aim of our study was to explore the impact of GA use on decision-making about, and GC for, genomic screening, we determined a threshold for meaningful engagement with DA. We defined ‘use’ of GA as logging in and spending at least 5 min using the platform. We determined 5 min as sufficient time to orient to the platform and engage in an area of interest. Use of GA was defined at the individual level (did/did not use GA) and at the family level (concordant, discordant and no use of GA).
Counselling data collection and analysis
The focus for counselling data collection and analysis was the family, including family use of GA. All parents attended pre-test counselling with a health professional trained in GC for genomic screening, regardless of whether they had used GA or not. Counselling sessions were audio-recorded with consent, transcribed verbatim by a professional transcription service and analysed. Qualitative data were managed using NVivo 14. Data were structurally coded based on the speaker and content or prompting of their speech using a modification of a previously published method [19, 20] (Supplementary Fig. 1). GC information provision was coded according to whether information was ‘offered’ by the GC or ‘prompted’ by the participant; GC questions clarifying values or assessing understanding were coded separately. Participant speech was coded for three types of behaviour: ‘assertions’ of feelings or beliefs; ‘concerns’ regarding participation; and ‘questions’ about screening. ‘Questions’ were subdivided into categories based on whether the information had been ‘addressed’ in GA, if it was ‘novel’, or if it was based on a ‘misconception’. All information not directly related to genomic screening was coded as ‘other’. Time and occurrences are reported.
Coding was conducted by one independent reviewer (AJ) who did not participate in GC, with 10% of transcripts recoded by a second reviewer (CM) who was not informed of the purpose of the research or the different categories of participants prior. Inter-coder reliability was calculated with a Cohen's Kappa score. The primary reviewer was blinded as best as possible to GA use when coding; however, many participants referred to the use of GA within the transcript.
Verbal behaviours were compared based on participant use of GA with the analysis focusing on three groups of participants: concordant (all parent participants in the genetic counselling session used GA); discordant (one participant used and one did not use GA); and no use of GA. GC appointments with only one participant were categorised based on that person’s GA use and not couple use. Bivariate analyses of coded verbal behaviours were examined with two-sided t-tests, with equal or unequal variances as appropriate.
Separately, appointment transcripts were analysed using inductive content analysis [21]. One reviewer (AJ) first read through each transcript and selected data segments pertaining to the perception of GA or its influence on participation in genomic screening. These chunks of data were then coded by AJ for big picture categories, which were verified in discussion with a second member of the research team who did not participate in counselling (MM).
Participant survey data collection and analysis
The focus of analysis for participant surveys was the individual parent—their GA use and survey completion. Participants were asked to complete optional surveys after use of GA (where used) but prior to counselling (S1), after counselling (S2), and about one month after return of results (S3). Details of the content of surveys [14] and findings [13] have been published previously. The number of knowledge questions answered correctly for each respondent at each time point was determined by collapsing responses into correct/incorrect answers. Group means and standard deviations were used to assess knowledge between groups (individual GA users/non-users) and across time. The pre-counselling and post-counselling surveys asked seven True/False/Unsure knowledge statements and two multiple-choice knowledge questions. The post-result survey contained three True/False/Unsure questions; the number of knowledge questions correct post-results was multiplied by three to allow comparison across surveys. Validated measures included in the surveys (DCS [22], STAI [23, 24], DR [25], GOS [26], FACToR [27], Satisfaction with genetic counselling [28], TMSI [29]) were calculated in line with the authors’ instructions. DR is reported as per a previously published categorisation system [30]. Demographic data were used for univariate associations of GA uptake and knowledge, analysed with Pearson chi-squared tests, t-tests, or Wilcoxon rank-sum tests analysis as appropriate to the type of data. Quantitative data were analysed in Stata 18.0 [31] and R [32]. A p-value of less than 0.05 was considered statistically significant. Open-ended survey responses were inductively coded by one reviewer (AJ) and verified by a second member of the research team (MM).
Ethical approval
Ethical approval for this research was obtained from the main HREC, the Melbourne Health Human Research Ethics Committee, as part of the Australian Genomics Health Alliance protocol (HREC/16/MH/251). Informed consent to participate in this sub-study was obtained from all participants as required by the HREC.
Results
One hundred and sixty-seven families who were offered genomic screening were given access to a modified version of the GA patient platform [17, 18] (Fig. 1).
Engagement with GA
GA was used by 74 mothers and 54 fathers from 87 families, giving a family engagement of 52% (87/167) and individual engagement of 128/315 (41%). No participant characteristics (e.g. demographic and family features) were associated with engagement with GA (Supplementary Table 1). GA utilisation metrics showed individuals spent a median of 12 min (IQR 7 min to 19 min) using GA at their most recent login.
Perspectives of GA
Family and individual survey response rates have been published elsewhere [13]. To recap, response rates by individuals were: pre-counselling survey (S1) 58/209 (28%); post-counselling survey (S2) 98/223 (44%); post-result survey (S3) 50/209 (24%). The pre-counselling survey (S1) was returned a median of 1 day after it was sent out (IQR 0–3). Median time between pre-test counselling and return of the post-counselling survey (S2) was 16 days (IQR 4–29); median time between return of results and return of post-result survey (S3) was 26 days (IQR 8–55). Median time between post-test counselling and return of results surveys (S2 and S3) was 149 days (114–215 days).
Most GA users who completed the pre-counselling survey indicated they used GA for between 5–20 min (30/45, 67%). The majority of survey respondents who used GA rated the amount of information in GA as ‘about right’ (42/45, 93%). The videos and family example scenarios were identified as the most useful aspects of GA from responses to an open survey question. Respondents appreciated the information included, as well as the layout and structure; ‘it broke things down into easy-to-read language and helped with processing the steps to decision making’ (Family 23). Inductive content analysis of the genetic counselling transcripts supported the survey findings, with comments about the value of the case studies and that GA was ‘straightforward’ to use. One father did report that he had found it overwhelming and had relied on his wife to use it. Two other couples also reported that one member of the couple had reviewed on behalf of both of them. This analysis also identified three participants who specifically stated that GA had influenced their decision about participating in genomic screening: ‘I initially didn’t want to find out at all and then after actually watching the videos and doing the [GA] you sent through… [I] changed my mind on it’(Family 11).
The post-counselling survey asked GA users if they believed GA contained enough information for them to make decisions about each of the screening tests without the additional counselling provided: 83% (29/35) of respondents agreed it did. Six participants indicated they still required GC input after GA use to decide on genomic screening; 4 provided reasons included outstanding questions (n = 2), and complex medical situations (n = 2). Of note, among survey respondents, the only factor associated with increased use of GA (Supplementary Table 2) was a high score for information seeking in the validated measure Threatening Medical Situation Inventory [29], which assesses the tendency to seek or avoid information under stress. Decisional conflict [22] and State Trait Anxiety [23, 24] were not associated with GA use.
Analysis of genetic counselling sessions
One hundred and nineteen families of the 167 provided access to GA subsequently attended genetic counselling: 81 of these had at least one parent who had used GA and 38 families had no parent use GA. Six families that used GA did not go on to attend genetic counselling.
When comparing family genetic counselling appointment attendance by GA use for all 119 families, we noted only one family of 44 with concordant GA use required a repeat appointment (2%), compared to families with discordant or no GA use (11/75, 15%, χ2 = 4.698, p = 0.030).
Genetic counselling sessions were recorded for 78 families (n = 78/119, 66%). In two families, parents attended counselling separately, giving a total of 80 counselling transcripts. Sixteen genetic counsellors and one clinical geneticist were involved in appointments. The codebook used to analyse transcripts identified verbal behaviours by speaker (Table 1). Inter-coder comparison resulted in a weighted Cohen-Kappa score of 0.90. For transcript analysis, GA use was classified as ‘concordant’ (all parents used GA; n = 29), ‘discordant’ (one parent used and one did not; n = 22), and ‘not used’ (n = 29). Impact of GA use on counselling was compared for concordant v discordant/not used families, as clinically any effect of GA use on counselling would only be apparent with concordant use; with discordant use, GCs would still have a ‘non user’ in the consultation and so have to counsel accordingly.
Impact of GA use on genetic counselling
Participants were scheduled pre-test counselling appointments of standard length for the service at which they were seen. Table 2 shows the content of counselling sessions, comparing those where all parents had used GA, with non-users and discordant users. Looking at speech related to screening, 20% less time was spent discussing screening in families with concordant GA use compared to discordant and no use (p = 0.037). Counsellors spent more time clarifying values or understanding in appointments with families that had not (both) used GA (p = 0.037). Participants who had not all used GA raised more concerns during their appointments (p = 0.024) and asked more ‘addressed’ (p = 0.038) questions than families with concordant GA use. Counsellors also spent more time providing ‘prompted’ information to those who had not (both) used GA (p = 0.003).
Knowledge and decisional conflict
The use of GA significantly impacted individual participant knowledge, with GA users answering more knowledge questions correctly at every time point (Table 3a). Genetic counselling did not impact knowledge (Table 3b). Other factors that were positively associated with higher post-counselling knowledge scores were female gender (p = 0.004) and younger age (p = 0.002). Following counselling, decisional conflict [22] did not differ between GA users and non-users (χ2 = 3.603, p = 0.165) (Supplementary Table 2).
Discussion
As applications of genomic screening grow, alternative service delivery models are required to prevent resource pressures from becoming a limiting factor in offering screening. In a clinical setting, augmenting pre-test genetic counselling with digital tools is a potential solution; however, the impact of decision tools on complex clinical consultations is under-explored in the genomic screening setting. This study reports results of an approach focused on clinician-patient communication to examine the impact of a decision support tool in active decision-making related to opportunistic genomic screening during genetic counselling appointments. Our results show use of the web-based digital platform GA improved patient understanding of genomic screening and impacted subsequent genetic counselling, reducing the amount of consult time spent discussing genomic screening and the number of families requesting more than one pre-test genetic counselling session. These findings demonstrate the utility of GA in supporting education and decision support for genomic screening and suggest that such digital tools have the potential to help scale genomic screening.
One of the goals of digital tool use is to support clinical service efficiencies. We noted efficiencies in the number of pre-test counselling appointments, with only one family that used GA requiring more than one pre-test counselling appointment. We also noted less time spent discussing screening in consultations with GA users. Our study design does not enable us to draw causal conclusions between GA use and these findings, yet we did not find any differences in participant characteristics associated with GA use. In terms of time efficiencies, previous studies designed to compare consultation time found reductions in consultation time among digital tool users [10, 33]. In our study, families in complex medical situations who had already completed genomic testing were scheduled for a standard pre-test counselling session to discuss genomic screening. Many families were seen by the genetic counsellor who had provided diagnostic test counselling. Overall consult time did not differ based on GA use. We noted that about half the consultation time was spent on matters other than screening, which may suggest families using the time to meet unmet needs, and/or counsellors taking the opportunity to provide other support pertinent to the family. In this complex cohort already familiar with genomic testing, counselling for opportunistic screening required only 20–25 min (on average) of the scheduled consultation time. Although evaluation in other cohorts, including those less familiar with genomic testing, is needed, this result suggests that focused genetic counselling about opportunistic genomic screening requires less time than pre-test genetic counselling and thus could inform future service design.
Our study explored decision tool use to augment genetic counselling—it was not intended to replace it. Some studies have suggested that decision tools may replace genetic counselling, but caution may be needed in complex clinical populations. In particular, care must be taken not to exacerbate existing inequalities. Whilst digital solutions offer potential for optimising the use of healthcare professionals [34], and improve patient-centred care for families [17], they also have the potential to widen inequality in disadvantaged populations [35]. Service designs must continue to co-design and evaluate for all, including those with low digital literacy or poor access to digital infrastructure [36]. One benefit of digital tools is to support education and decision support, which allows genetic counselling time to be spent on more nuanced, patient-centred discussions of values and decision-making factors. Although some of our participants reported that they felt equipped to make their decision about genomic screening without genetic counsellor input, others valued the genetic counselling support. Allowing participants to opt out of pre-test counselling may be feasible when participant needs are met by the digital platform, reserving genetic counselling resources for patients who are most in need. A challenge will be in determining which patients require genetic counselling and ensuring those who opt out of genetic counselling are sufficiently informed and supported.
Genomic screening applications are increasingly being trialled and implemented, including opportunistic screening for additional findings, couples-based carrier screening [16], population-based screening for actionable conditions [37] and genomic newborn screening [38, 39]. Workforce capacity has been identified as a key issue facing national genomics initiatives [40]. It must be recognised that genomic screening creates new needs for clinical follow-up—all those with positive genomic screening findings require clinical follow-up to identify any associated symptoms. Quantification of the impact of this will be important to support decision-making in publicly funded healthcare systems about the introduction of screening initiatives. Alongside this, new paradigms for patient education and decision support are needed. Integrating a digital tool to supplement GC time can help to optimise the use of the genetic counselling workforce and is likely to benefit the scalability of genomic screening. Indeed, ongoing population genomic screening initiatives have employed digital enrolment and consent [37]. Given the complexity of our cohort, as well as the two-step model - meaning that patients had already received clinical support related to their decision to have genomic testing for the primary diagnostic indication - evaluations are needed in other cohorts, such as population screening, to examine how genetic counselling practice would be impacted by using digital tools in this setting. Additionally, while not explored in our current study, digital platforms can also support the return of results [10], further promoting the feasibility of population genomic screening.
Our study findings must be considered in light of several limitations. Whilst GA is now available in multiple languages, at the time of this study, GA was only available in English, which limited its accessibility. At the time of the study, the platform also only retained login data for the most recent login, so we did not have time data for earlier logins for individuals who accessed GA multiple times. However, most participants self-reported using GA for 5–20 min, which is consistent with the data from the platform. We did not collect data on reasons for declining to use GA. Digital tools can also be used to support the return of results; however, this was not tested in the current study and is an area that warrants future research. Our genetic counselling transcript analysis was designed to classify genetic counsellor and parent participation in counselling about genomic screening; we did not make any attempt to analyse the quality of those interactions. As such, we can make no comment on the value of ‘other’ conversation in the consultation. Our initial review of transcripts did not reveal obvious differences between GA users and non-users in counselling topics discussed, but we note our content analysis was not designed to explore detailed differences in consultations between GA users and non-users. We also note that counsellors and families were aware that sessions were being recorded and this may have impacted the consultation. Finally, our population was medically complex, and findings cannot necessarily be extrapolated to the general population.
As opportunistic genomic screening is increasingly offered in clinical populations, approaches are needed that allow clinicians to focus on decisions around diagnostic testing, while providing alternative supports for screening decisions. One approach is the augmentation of genetic counselling with digital tools. We found that digital tools can effectively support information delivery; transferring standardised information provision to the tool can allow genetic counsellors to allocate time to providing more individualised care tailored to each patient’s needs, which will help to promote value-congruent decisions. These findings demonstrate the utility of GA in supporting opportunistic genomic screening and suggest that digital tools have the potential to support the scalability of genomic screening.
Data availability
The data that support the findings of this study are available on request from the corresponding author (MM). The data are not publicly available due to containing information that could compromise research participant privacy.
References
Schmidlen T, Sturm AC, Hovick S, Scheinfeldt L, Scott Roberts J, Morr L, et al. Operationalizing the reciprocal engagement model of genetic counseling practice: a framework for the scalable delivery of genomic counseling and testing. J Genet Couns. 2018;27:1111–29.
Birch P, Adam S, Bansback N, Coe RR, Hicklin J, Lehman A, et al. DECIDE: a decision support tool to facilitate parents’ choices regarding genome-wide sequencing. J Genet Couns. 2016;25:1298–308.
Pacyna JE, Radecki Breitkopf C, Jenkins SM, Sutton EJ, Horrow C, Kullo IJ, et al. Should pretest genetic counselling be required for patients pursuing genomic sequencing? Results from a survey of participants in a large genomic implementation study. J Med Genet. 2019;56:317–24.
King E, Halliday J, Archibald AD, Delatycki M, Barlow-Stewart K, Newson AJ, et al. Development and use of the Australian reproductive genetic carrier screening decision aid. Eur J Hum Genet. 2022;30:194–202.
Nagle C, Gunn J, Bell R, Lewis S, Meiser B, Metcalfe S, et al. Use of a decision aid for prenatal testing of fetal abnormalities to improve women’s informed decision making: a cluster randomised controlled trial [ISRCTN22532458]. BJOG. 2008;115:339–47.
Halliday JL, Muller C, Charles T, Norris F, Kennedy J, Lewis S, et al. Offering pregnant women different levels of genetic information from prenatal chromosome microarray: a prospective study. Eur J Hum Genet. 2018;26:485–94.
Griffith JM, Sorenson JR, Bowling JM, Jennings-Grant T. Assessment of an interactive computer-based patient prenatal genetic screening and testing education tool. Health Educ Behav. 2005;32:613–26.
Alden DL, Friend J, Schapira M, Stiggelbout A. Cultural targeting and tailoring of shared decision making technology: a theoretical framework for improving the effectiveness of patient decision aids in culturally diverse groups. Soc Sci Med. 2014;105:1–8.
Bombard Y, Hayeems RZ. How digital tools can advance quality and equity in genomic medicine. Nat Rev Genet. 2020;21:505–6.
Lee W, Shickh S, Assamad D, Luca S, Clausen M, Somerville C, et al. Patient-facing digital tools for delivering genetic services: a systematic review. J Med Genet. 2023;60:1–10.
Ballard LM, Horton RH, Dheensa S, Fenwick A, Lucassen AM. Exploring broad consent in the context of the 100,000 genomes project: a mixed methods study. Eur J Hum Genet. 2020;28:732–41.
Sanderson SC, Lewis C, Patch C, Hill M, Bitner-Glindzicz M, Chitty LS. Opening the ‘black box’ of informed consent appointments for genome sequencing: a multisite observational study. Genet Med. 2019;21:1083–91.
Martyn M, Lee L, Jan A, Tytherleigh R, Lynch F, Mighton C, et al. Offering complex genomic screening in acute pediatric settings: family decision-making and outcomes. Genet Med. 2025;27:101327.
Bouffler SE, Lee L, Lynch F, Martyn M, Lynch E, Macciocca I, et al. Two-step offer and return of multiple types of additional genomic findings to families after ultrarapid trio genomic testing in the acute care setting: a study protocol. BMJ Open. 2023;13:e072999.
Lunke S, Bouffler SE, Patel CV, Sandaradura SA, Wilson M, Pinner J, et al. Integrated multi-omics for rapid rare disease diagnosis on a national scale. Nat Med. 2023;29:1681–91.
Kirk EP, Delatycki MB, Archibald AD, Tutty E, Caruana J, Halliday JL, et al. Nationwide, couple-based genetic carrier screening. N Engl J Med. 2024;391:1877–89.
Shickh S, Rafferty SA, Clausen M, Kodida R, Mighton C, Panchal S, et al. The role of digital tools in the delivery of genomic medicine: enhancing patient-centered care. Genet Med. 2021;23:1086–94.
Clausen M, Krishnapillai S, Hirjikaka D, Kodida R, Shickh S, Reble E, et al. Genetics adviser: the development and usability testing of a new patient digital health application to support clinical genomic testing. Genet Med Open. 2024;2:101814.
Gordon HS, Street RL, Kelly PA, Souchek J, Wray NP. Physician-patient communication following invasive procedures: an analysis of post-angiogram consultations. Soc Sci Med. 2005;61:1015–25.
Street RL, Millay B. Analyzing patient participation in medical encounters. Health Commun. 2001;13:61–73.
Vears DF, Gillam L. Inductive content analysis: a guide for beginning qualitative researchers. Focus Health Prof Educ Multi Prof J. 2022;23:111–27.
O’Connor. A.M. User Manual - Decisional Conflict Scale. Ottawa: Ottawa Hospital Research Institute, 1993. Available from http://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf.
Marteau TM, Bekker H. The development of a six-item short-form of the state scale of the Spielberger State—Trait Anxiety Inventory (STAI). Br J Clin Psychol. 1992;31:301–6.
Spielberger CD. State-Trait Anxiety Inventory for Adults (STAI-AD), 1983. Available from: https://doi.org/10.1037/t06496-000.
O’Connor. A.M. User manual - Decision Regret Scale. Ottawa: Ottawa Hospital Research Institute. 1996. Available from http://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf.
Grant PE, Pampaka M, Payne K, Clarke A, McAllister M. Developing a short-form of the genetic counselling outcome scale: the genomics outcome scale. Eur J Med Genet. 2019;62:324–34.
Li M, Bennette CS, Amendola LM, Ragan Hart M, Heagerty P, Comstock B, et al. The feelings about genomic testing results (FACToR) questionnaire: development and preliminary validation. J Genet Couns. 2019;28:477–90.
Zellerino B, Milligan SA, Brooks R, Freedenberg DL, Collingridge DS, Williams MS. Development, testing, and validation of a patient satisfaction questionnaire for use in the clinical genetics setting. Am J Med Genet C Semin Med Genet. 2009;151C:191–9.
Van Zuuren FJ, De Groot KI, Mulder NL, Peter M. Coping with medical threat: an evaluation of the Threatening Medical Situations Inventory (TMSI). Personal Individ Differ. 1996;21:21–31.
Sheehan J, Sherman KA, Lam T, Boyages J. Association of information satisfaction, psychological distress and monitoring coping style with post-decision regret following breast reconstruction. Psychooncology. 2007;16:342–51.
StataCorp. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC; 2017.
Posit team (2025). RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA. http://www.posit.co/.
Bombard Y, Clausen M, Shickh S, Mighton C, Casalino S, Kim THM, et al. Effectiveness of the Genomics ADvISER decision aid for the selection of secondary findings from genomic sequencing: a randomized clinical trial. Genet Med. 2020;22:727–35.
Orton M, Agarwal S, Muhoza P, Vasudevan L, Vu A. Strengthening delivery of health services using digital devices. Glob Health Sci Pract. 2018;6:S61–71.
Zhang J, Gallifant J, Pierce RL, Fordham A, Teo J, Celi L, et al. Quantifying digital health inequality across a national healthcare system. BMJ Health Care Inform. [cited 2025;30. Available from: https://informatics.bmj.com/content/30/1/e100809.
Assamad D, Majeed S, Aguda V, Grewal S, Butkowsky C, Clausen M, et al. Digital health tools in genomics: advancing diversity, equity, and inclusion. Public Health Genom. 2023;26:194–200.
Foss KS, O’Daniel JM, Berg JS, Powell SN, Cadigan RJ, Kuczynski KJ, et al. The rise of population genomic screening: characteristics of current programs and the need for evidence regarding optimal implementation. J Pers Med. 2022;12:692.
Lunke S, Bouffler SE, Downie L, Caruana J, Amor DJ, Archibald A, et al. Prospective cohort study of genomic newborn screening: BabyScreen+ pilot study protocol. BMJ Open. 2024;14:e081426.
Ziegler A, Koval-Burt, Dm K, Sf S, AB, Kg L, et al. Expanded newborn screening using genome sequencing for early actionable conditions. JAMA. [cited 2025 May 29];333. Available from: https://pubmed.ncbi.nlm.nih.gov/39446378/.
The expanding global genomics landscape: Converging priorities from national genomics programs. [cited 2025 May 28]. Available from: https://pubmed.ncbi.nlm.nih.gov/40068686/.
Acknowledgements
The authors would like to thank the study participants for their time and insights into their experiences. The authors thank Rigan Tytherleigh for support with the submission of the manuscript.
Funding
This work was supported by the Australian Government’s Medical Research Future Fund grant number GHFM76747 and the Royal Children’s Hospital Foundation grant number 2020-1259. The research conducted at the Murdoch Children’s Research Institute was supported by the Victorian Government’s Operational Infrastructure Support Program. Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Conceptualisation: ZS, LD, CG and MM; Data curation: CM and AJ; Formal analysis: AJ and CM. Funding acquisition: ZS and CG; Investigation: SB; Methodology: AJ, SB and LL; Project administration: SB.; Resources: MC, YB and CG; Software: MC; Supervision: ZS and MM; Validation: CM; Visualization: AJ. Writing—original draft: AJ, CM, MM, SB and ZS. Writing—review and editing: All authors.
Corresponding author
Ethics declarations
Competing interests
Yvonne Bombard and Marc Clausen report ownership in Genetics Adviser, Inc. All other authors declare no potential competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mighton, C., Jan, A., Lee, L. et al. Impact of a digital platform on genetic counselling encounters in the screening context. Eur J Hum Genet (2026). https://doi.org/10.1038/s41431-026-02029-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41431-026-02029-6