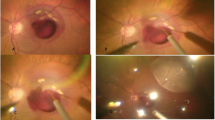
Abstract
Objective
To determine whether subconjunctival or intrastromal administration of anti-VEGF agents is more effective on suture-induced corneal neovascularization (CoNV) in rabbits.
Methods
CoNV was induced in 48 eyes of 24 New Zealand white rabbits by using an 8/0 silk suture. On the 7th day after suturing, the rabbits were divided into four treatment groups as follows: six rabbits received subconjunctival bevacizumab (group 1), six rabbits received subconjunctival aflibercept (group 2), six rabbits received intrastromal bevacizumab (group 3) and six rabbits received intrastromal aflibercept (group 4). On the 7th and 14th days after suturing, the CoNV area was calculated by standardised analysis of photographs using the Image-J program. On the 14th day after suturing, all rabbits were sacrificed and then corneal tissue was harvested for the analysis of vascular endothelial growth factor (VEGF)-A, VEGF-B and placental growth factor (PIGF) levels.
Results
On the 7th day after suturing, CoNV areas were 17.10 ± 2.98, 18.88 ± 3.78, 17.36 ± 4.52, 18.57 ± 4.16 and 17.31 ± 2.81 mm2 in the groups 1–4 and control group, respectively. On the 7th day after intervention and removal of suture, CoNV areas were 4.85 ± 1.99, 6.66 ± 1.73, 2.83 ± 1.08, 2.63 ± 1.16 and 11.93 ± 2.64 mm2 in the group 1–4 and control group, respectively. CoNV area was reduced by 88.1% and 82.5% in eyes receiving intrastromal aflibercept and bevacizumab, respectively (both p < 0.001), and by 64.5% and 69.9% in eyes receiving subconjunctival aflibercept and bevacizumab, respectively (both p = 0.001).
Conclusion
Intrastromal anti-VEGF therapy regressed CoNV more effectively than subconjunctival therapy regardless of the type of anti-VEGF agent.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Chang JH, Gabison EE, Kato T, Azar DT. Corneal neovascularization. Curr Opin Ophthalmol. 2001;12:242–9.
Bachmann B, Taylor RS, Cursiefen C. Corneal neovascularization as a risk factor for graft failure and rejection after keratoplasty: an evidence-based meta-analysis. Ophthalmology. 2010;117:1300–5.e7.
Sener E, Yuksel N, Yildiz DK, Yilmaz B, Ozdemir O, Caglar Y, et al. The impact of subconjuctivally injected EGF and VEGF inhibitors on experimental corneal neovascularization in rat model. Curr Eye Res. 2011;36:1005–13.
Akar EE, Oner V, Kucukerdonmez C, Aydin Akova Y. Comparison of subconjunctivally injected bevacizumab, ranibizumab, and pegaptanib for inhibition of corneal neovascularization in a rat model. Int J Ophthalmol. 2013;6:136–40.
Park YR, Chung SK. Inhibitory effect of topical aflibercept on corneal neovascularization in rabbits. Cornea. 2015;34:1303–7.
Fogli S, Del ReM, Rofi E, Posarelli C, Figus M, Danesi R. Clinical pharmacology of intravitreal anti-VEGF drugs. Eye. 2018;32:1010–20.
Cai S, Yang Q, Li X, Zhang Y. The efficacy and safety of aflibercept and conbercept in diabetic macular edema. Drug Des Devel Ther. 2018;12:3471–83.
Gal-Or O, Livny E, Sella R, Nisgav Y, Weinberger D, Livnat T, et al. Efficacy of Subconjunctival aflibercept versus bevacizumab for prevention of corneal neovascularization in a rat model. Cornea. 2016;35:991–6.
Sella R, Gal-Or O, Livny E, Dachbash M, Nisgav Y, Weinberger D, et al. Efficacy of topical aflibercept versus topical bevacizumab for the prevention of corneal neovascularization in a rat model. Exp Eye Res. 2016;146:224–32.
Dastjerdi MH, Sadrai Z, Saban DR, Zhang Q, Dana R. Corneal penetration of topical and subconjunctival bevacizumab. Invest Ophthalmol Vis Sci. 2011;52:8718–23.
Kim SW, Ha BJ, Kim EK, Tchah H, Kim TI. The effect of topical bevacizumab on corneal neovascularization. Ophthalmology. 2008;115:e33–38.
Rocher N, Behar-Cohen F, Pournaras JA, Naud MC, Jeanny JC, Jonet L, et al. Effects of rat anti-VEGF antibody in a rat model of corneal graft rejection by topical and subconjunctival routes. Mol Vis. 2011;17:104–12.
Bhatti N, Qidwai U, Hussain M, Kazi A. Efficacy of sub-conjunctival and topical bevacizumab in high-risk corneal transplant survival. J Pak Med Assoc. 2013;63:1256–9.
Sharif Z, Sharif W. Corneal neovascularization: updates on pathophysiology, investigations & management. Rom J Ophthalmol. 2019;63:15–22.
Gupta AA, Mammo DA, Page MA. Intrastromal bevacizumab in the management of corneal neovascularization: a retrospective review. Graefes Arch Clin Exp Ophthalmol. 2020;258:167–73.
Sarah B, Ibtissam H, Mohammed B, Hasna S, Abdeljalil M. Intrastromal injection of bevacizumab in the management of corneal neovascularization: about 25 eyes. J Ophthalmol. 2016;2016:6084270.
Kim YC, Grossniklaus HE, Edelhauser HF, Prausnitz MR. Intrastromal delivery of bevacizumab using microneedles to treat corneal neovascularization. Invest Ophthalmol Vis Sci. 2014;55:7376–86.
Schneider CA, Rasband WS, Eliceiri KW. NIH image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5.
Kim RY, Chung SK, Kim MS, Ra H. Effects of combined photodynamic therapy and topical bevacizumab treatment on corneal neovascularization in rabbits. Cornea. 2016;35:1615–20.
Kim JW, Chung SK. The effect of topical infliximab on corneal neovascularization in rabbits. Cornea. 2013;32:185–90.
Kim EK, Kong SJ, Chung SK. Comparative study of ranibizumab and bevacizumab on corneal neovascularization in rabbits. Cornea. 2014;33:60–64.
Byun YS, Chung SK. The effect of methotrexate on corneal neovascularization in rabbits. Cornea. 2011;30:442–6.
Lee HS, Chung SK. The effect of subconjunctival suramin on corneal neovascularization in rabbits. Cornea. 2010;29:86–92.
Jovanovic V, Nikolic L. The effect of topical doxycycline on corneal neovascularization. Curr Eye Res. 2014;39:142–8.
Murata M, Shimizu S, Horiuchi S, Taira M. Inhibitory effect of triamcinolone acetonide on corneal neovascularization. Graefes Arch Clin Exp Ophthalmol. 2006;244:205–9.
You IC, Kang IS, Lee SH, Yoon KC. Therapeutic effect of subconjunctival injection of bevacizumab in the treatment of corneal neovascularization. Acta Ophthalmol. 2009;87:653–8.
Lakshmipathy M, Susvar P, Popet K, Rajagopal R. Subconjunctival bevacizumab and argon laser photocoagulation for preexisting neovascularization following deep lamellar anterior keratoplasty. Indian J Ophthalmol. 2019;67:1193–4.
Petsoglou C, Balaggan KS, Dart JK, Bunce C, Xing W, Ali RR, et al. Subconjunctival bevacizumab induces regression of corneal neovascularisation: a pilot randomised placebo-controlled double-masked trial. Br J Ophthalmol. 2013;97:28–32.
Papadopoulos Z. Aflibercept: a review of its effect on the treatment of exudative age-related macular degeneration. Eur J Ophthalmol. 2019;29:368–78.
Yu L, Wu X, Cheng Z, Lee CV, LeCouter J, Campa C, et al. Interaction between bevacizumab and murine VEGF-A: a reassessment. Invest Ophthalmol Vis Sci. 2008;49:522–7.
Ozdemir O, Altintas O, Altintas L, Ozkan B, Akdag C, Yuksel N. Comparison of the effects of subconjunctival and topical anti-VEGF therapy (bevacizumab) on experimental corneal neovascularization. Arq Bras Oftalmol. 2014;77:209–13.
Lopes GJA, Casella AMB, Oguido AP, Matsuo T. Effects of topical and subconjunctival use of bevacizumab on corneal neovascularization in rabbits’ eyes. Arq Bras Oftalmol. 2017;80:252–6.
Yeung SN, Lichtinger A, Kim P, Amiran MD, Slomovic AR. Combined use of subconjunctival and intracorneal bevacizumab injection for corneal neovascularization. Cornea. 2011;30:1110–4.
Lichtinger A, Yeung SN, Kim P, Amiran MD, Elbaz U, Slomovic AR. Corneal endothelial safety following subconjunctival and intrastromal injection of bevacizumab for corneal neovascularization. Int Ophthalmol. 2014;34:597–601.
Cursiefen C, Hofmann-Rummelt C, Kuchle M, Schlotzer-Schrehardt U. Pericyte recruitment in human corneal angiogenesis: an ultrastructural study with clinicopathological correlation. Br J Ophthalmol. 2003;87:101–6.
Lin CT, Hu FR, Kuo KT, Chen YM, Chu HS, Lin YH, et al. The different effects of early and late bevacizumab (Avastin) injection on inhibiting corneal neovascularization and conjunctivalization in rabbit limbal insufficiency. Invest Ophthalmol Vis Sci. 2010;51:6277–85.
Chen WL, Chen YM, Chu HS, Lin CT, Chow LP, Chen CT, et al. Mechanisms controlling the effects of bevacizumab (avastin) on the inhibition of early but not late formed corneal neovascularization. PLoS ONE. 2014;9:e94205.
Author information
Authors and Affiliations
Contributions
RKU, SC and AYU designed and conducted the study. NSY and NB made substantial contributions to acquisition of biochemical data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercialor financial relationships that could be construed as a potential conflict of interest.
Ethics approval
The Animal Experiments Local Ethics Committee at the University of Abant Izzet Baysal approved the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ucgul, R.K., Celebi, S., Yilmaz, N.S. et al. Intrastromal versus subconjunctival anti-VEGF agents for treatment of corneal neovascularization: a rabbit study. Eye 35, 3123–3130 (2021). https://doi.org/10.1038/s41433-020-01347-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-020-01347-3
This article is cited by
-
Subconjunctival aflibercept inhibits corneal angiogenesis and VEGFR-3+CD11b+ cells
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)