Abstract
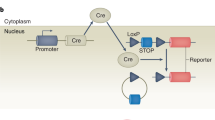
The Cre/loxP system is extensively utilized to pinpoint gene functions in specific cell types or developmental stages, typically without major disturbance to the host’s genome. However, we found that the random insertion of the Mrp8-cre transgene significantly promotes the host’s innate immune response. This effect is characterized by elevated susceptibility to cartilage antibody-induced arthritis, likely due to interference with genes near the insertion site. These findings underscore the potential biological disturbances caused by random transgene integration, and the necessity for stringent control strategies to avoid biased interpretations when using Cre-conditional strains.
Similar content being viewed by others
Introduction
Cartilage antibody-induced arthritis (CAIA) is a mouse model that mimics the effector phase of rheumatoid arthritis (RA). It utilizes arthritogenic antibody cocktails targeting joint proteins, typically inducing arthritis within days or even hours in susceptible strains [1]. This model bypasses adaptive immunity and directly activates the innate immunity, initiating inflammation by maximizing acute neutrophil expansion. Therefore, investigating neutrophil-specific gene expression patterns is essential to understand the onset of both mouse CAIA and human RA.
The Cre/loxP system has revolutionized medical research by enabling site-specific recombination, thus laying the groundwork for its widespread application [2]. This system allows for precise dissection of gene function within specific cells or developmental stages, effectively overcoming the limitations of conventional knockout strategies that disrupt genes universally. For instance, the Mrp8-cre/ires-eGFP transgenic mice (stock no: 021614), when crossed with mouse strains carrying loxP-flanked genes, can be utilized to manipulate these genes specifically expressed in granulocytes [3]. However, homozygous Mrp8-creTg/Tg mice are nonviable [4], indicating the inherent toxicity of the inserted allele and its significant impact on the host’s biological systems. Therefore, using these mice necessitates careful characterization of the altered phenotypes and the implementation of stringent controls.
Here, we report that a single allele of the Mrp8-cre/ires-eGFP transgene predisposes mice to CAIA, likely due to the interfered expressions of genes close to the insertion site. These findings underscore the need for rigorous control strategies when using conditional mouse strains.
Materials and methods
See Supplementary materials for details.
This study was performed according to the ethical permits (N35/16, 2660–2021) approved by the local animal ethics committee (Stockholm region) affiliated with the Swedish Board of Agriculture.
Results
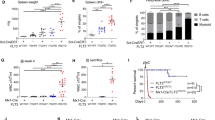
Our initial investigation aimed to elucidate the role of neutrophil-derived reactive oxygen species (ROS) in CAIA. To achieve this, we established mouse strains by crossing Ncf1Tn3/m1j mice with Ncf1m1j/m1j.Mrp8-creTg/0 mice. Offspring harboring both Ncf1Tn3 and Mrp8-creTg alleles exhibits restored ROS production exclusively in MRP8-expressing cells, mainly neutrophils, while littermates lacking either gene displayed uniformly low ROS levels across all cell subsets [5]. Following arthritogenic antibody cocktail transfer, Ncf1Tn3/m1j.Mrp8-creTg/0 mice (with ROS restored in neutrophils) displayed significantly more severe arthritis compared to Ncf1Tn3/m1j.Mrp8-cre0/0 or Ncf1m1j/m1j.Mrp8-cre0/0 littermates (Fig. 1a). These results seem in favor that neutrophil-derived ROS promotes arthritis development. However, there are two different variables within the experimental group that differ from the two control groups. First, Ncf1Tn3/m1j.Mrp8-creTg/0 mice have ROS production restored in neutrophils; Second, they harbor the Mrp8-cre/ires-eGFP transgene. Further investigation involving Ncf1m1j/m1j.Mrp8-creTg/0 mice revealed comparable arthritis severity to Ncf1Tn3/m1j.Mrp8-creTg/0 littermates (Fig. 1b). Conversely, littermates lacking the transgenic Mrp8-Cre allele exhibited milder arthritis (Fig. 1b). These results suggest that the presence of the Mrp8-cre transgene itself, rather than the restoration of ROS in neutrophils, predisposes mice to CAIA.
a, b Mice were immunized with cartilage antibody cocktail Cab4 containing M2139, ACC1, 15A, L10D9 on day 0 (3 mg, intravenously (i.v.)), and boosted with lipopolysaccharides (LPS) on day 5 (25 μg, intraperitoneally (i.p.)). a Ncf1Tn3/m1j.Mrp8-creTg/0 mice exhibited more severe arthritis compared to the other two control groups (Two-way ANOVA followed by multiple comparisons, p < 0.005 from d10 to d14); b Mrp8-creTg/0 mice with or without ROS restored exhibited more severe arthritis compared to the other two control groups (Two-way ANOVA followed by multiple comparisons, p < 0.001 between NCF1 mutated mice with or without Mrp8-cre transgenic allele from d7 to d15); c Neutrophils expressed mildly lower of FCGR2B, and comparable levels of FCGR3 and and FCGR4 in Mrp8-creTg/0 mice compared to WT littermates; d Neutrophils internalized comparable levels of immune complex (IC) in Mrp8-creTg/0, WT, and Ncf1m1j/m1j mice; e Recombinant antibody R69-4 phosphorylated more phospholipase Cγ2 (PLCG2) in ROS sufficient neutrophils.
To determine whether this predisposition involves adaptive immune response apart from CAIA (which predominantly depends on innate immunity [6]), we employed a delayed-type hypersensitivity (DTH) model. DTH is a model for evaluating adaptive immune responses, primarily Th1 response [7]. After challenge, Ncf1m1j/m1j.Mrp8-cre0/0 mice exhibited comparable increase of ear thickness compared to Ncf1m1j/m1j.Mrp8-creTg/0 littermates (Fig. S1a). These results suggest that Mrp8-creTg/0 transgenic allele may not disturb adaptive immunity. Instead, its effect could be restricted in antibody-induced inflammation.
To investigate how the Mrp8-cre transgene disposes mice to CAIA, we measured the levels of FCGRs which are responsible for IgG-induced inflammation. The insertion of the Mrp8-cre transgene did not alter the expressions of any activating FCGRs on neutrophils but mildly lowered the expression of inhibitory FCGR2B (Fig. 1c, Fig. S1b). However, the internalization of IgG immune complexes (IC) by neutrophils was not affected (Fig. 1d). We then used a recombinant antibody, R69-4, known to phosphorylate neutrophil phospholipase Cγ2 (PLCG2) [8]. This phosphorylation mimics neutrophil activation upon IC stimulation through FCGRs. After a 10-min incubation, R69-4 phosphorylated more PLCG2 in ROS-sufficient neutrophils (both Ncf1Tn3/m1j.Mrp8-creTg/0 and Ncf1+/+.Mrp8-cre0/0 mice) (Fig. 1e), indicating that ROS, rather than the inserted Mrp8-cre transgene, plays a role in IC-mediated neutrophil activation.
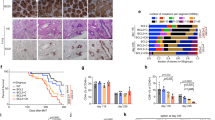
The above results suggest that Mrp8-cre insertion may not sensitize neutrophils upon IC stimulation at the priming stage. We then tracked neutrophil population during arthritis onset, and observed that neutrophil proportions (Fig. 2a) or absolute counts (Fig. S1c) in peripheral blood did not differ between Mrp8-creTg/0 and Mrp8-cre0/0 mice during either antibody phase or lipopolysaccharide (LPS) phase. Here it seems that Mrp8-cre transgene does not affect a typical pathophysiological procedure of antibody-induced arthritis.
a No significance was recorded regarding neutrophil proportion during arthritis development among the 4 genotypes (One-way ANOVA, p > 0.05); b No significance was recorded regarding PAI-1 expression on peripheral blood cells under steady state (Mann–Whitney U test, p > 0.05); c Mrp8-creTg/0 neutrophils exhibited compromised expression of PAI-1 upon stimulation compared to WT neutrophils (Mann–Whitney U test, p < 0.01); d No significance was recorded regarding PAI-1 expression on synovial lining macrophage between Mrp8-creTg/0 and WT neutrophils under steady state (Mann–Whitney U test, p > 0.05); Mrp8-creTg/0 synovial lining macrophages expressed significantly lower PAI-1 upon LPS stimulation compared to WT counterparts (Mann–Whitney U test, p < 0.01).
Interestingly, Wang et al. [4] have demonstrated that the insertion of Mrp8-cre transgene disrupts two genes, Ap1s1 and Serpine1. The absence of Ap1s1 expression may underlie the preweaning lethality observed in homozygous Mrp8-creTg/Tg mice, as Ap1s1-/- mice fail to survive into adulthood [9] while Serpine1-/- mice do survive [10]. Instead, the lack of Serpine1 expression appears to be a more likely cause of the heightened susceptibility to CAIA compared to the lack of Ap1s1, because plasminogen activator inhibitor-1 (PAI-1), encoded by Serpine1, can indirectly inhibit the activity of matrix metalloproteinases [11] which dominates cartilage degradation. Furthermore, it has been shown that administration of PAI-1 suppresses septic arthritis by lowering plasmin-induced inflammatory responses and subsequent cartilage destruction [12].
Under steady state, we recorded significantly lower intracellular AP1S1 expression in Mrp8-creTg/0 neutrophils (Fig. S1d), but upon stimulation by LPS, Mrp8-creTg/0 neutrophils expressed equivalent levels of AP1S1 compared to Mrp8-creTg/0 neutrophils (Fig. S1d). In contrast, PAI-1 expression did not significantly differ between the two genotypes under steady state on any cell types from peripheral blood (Fig. 2b). However, once stimulated by LPS, Mrp8-creTg/0 neutrophils expressed significantly lower PAI-1 compared to WT littermates (Fig. 2c, Fig. S1e). Due to the universal disruption of Serpine1 caused by Mrp8-cre insertion, irrespective of the transcription of Mrp8 promoter in only granulocyte lineage, we proceeded to check PAI-1 expression in other cell types related to arthritis development, such as synovial lining macrophages. These macrophages (CD11b+CD68+CX3CR1+) expressed comparable PAI-1 in Mrp8-creTg/0 mice under steady-state conditions, but the difference became significant upon stimulation by LPS (Fig. 2d). Taken together, these results suggest that PAI-1 and AP1S1 expressions are affected by the insertion of Mrp8-cre transgene in different scenarios even in a heterozygous state, likely affecting all cell types.
Discussion
In this study, we demonstrate that the Mrp8-cre transgene promotes CAIA in the corresponding transgenic mice. This effect might be due to the disruption of Serpine1 and Ap1s1 sequences where Mrp8-cre is inserted [4], leading to insufficient expressions of the two proteins in different scenarios. The mechanism of PAI-1 and AP1S1 on antibody-induced inflammation warrants further investigation.
Although precise genome editing technologies have expanded the application for creating genetically modified mice, transgenic mouse strains with random insertions of genes are still widely used. However, the random insertion of Cre genes could have unexpected impact on the host. Therefore, the integration site, potential disruption of regulatory elements, copy number variation, and chromosomal destabilization need to be carefully controlled. Mrp8-cre/ires-eGFP transgenic mice are a valuable tool for investigating functions of specific genes in granulocytes. However, the random insertion of the Mrp8-cre transgene has been shown to cause a 44-kb deletion of the host genome [4], and largely affect innate immunity, as demonstrated in this study. The disruption of Serpine1 within this 44-kb sequence, leading to insufficient PAI-1 production, might be responsible for the increased susceptibility. PAI-1 principally inhibits plasminogen, and we have previously shown that functional plasminogen is needed for the development of collagen-induced arthritis [13]. Moreover, PAI-1 possesses pleiotropic functions in atherosclerosis, inflammation, fibrosis, and even cancer [14]. It is especially critical in neutrophil biology, including efferocytosis, activation, and influx, etc. [15,16,17]. In Mrp8-cre mice, the production of PAI-1 is not only compromised in neutrophils, but also in other cells like macrophages, further complicating the biology of this mouse strain.
Experimentally excluding the possible effects of putative phenotypes is challenging. In earlier studies using K/BxN serum transfer arthritis model, the presence of the Mrp8-cre transgenic allele alone did not significantly promote arthritis development, although a trend of increased paw swelling was recorded with only three Mrp8-cre mice included [18]. With the presently used CAIA model, the impact of the Mrp8-cre transgenic allele becomes discernible and significant (Fig. 1b). Moreover, the random insertion disrupts both Serpine1 and Ap1s1 genes, which unexpectedly has an effect even in a heterozygous state, highlighting the role of these two genes in impacting the development of antibody-induced arthritis. However, without specific Serpine1 and/or Ap1s1 knockout models, the causal relationship between these genes and immune regulation cannot be definitively established from our platform. Either Serpine1+/- or Ap1s1+/- mice showing similar phenotype compared to Mrp8-creTg/0 mice could enhance the likelihood of this putative causal relationship. Additionally, most of the experimental mice in this study exhibit low ROS levels due to NCF1 deficiency, which could result in different phenotypes under ROS-sufficient conditions.
In other strains carrying Cre recombinase, concerns have been raised in various contexts. For example, the LysM-Cre mouse line, where the Cre allele is inserted into the translational start site of the endogenous Lysozyme M (LysM) gene, was initially considered a suitable tool for manipulating myeloid cells [19]. However, it was later reported that most of the cells in the brain undergoing LysM-Cre-mediated recombination were neurons rather than myeloid cells [20]. Although the Cx3cr1-CreERT2 strain is recommended for such cases [21], it has been noted that only a subset of resident macrophages expresses CX3CR1, limiting its utility for targeting all monocyte/macrophage lineage cells [22]. In contrast to these heterogeneous cell populations, mouse neutrophils predominantly express Ly6G, making it an ideal target for neutrophil-specific gene manipulation. The Ly6G-Cre mouse line, where Cre is inserted into the first exon of Ly6g, appears not to interfere with other genes or neutrophil function [23], and thus may be preferred over Mrp8-Cre mice for neutrophil-targeted studies. Even so, it remains critical to mandate proper control strategy when Cre transgenes are included in experimental settings. Littermates carrying Cre allele(s) but without loxP sites should always be included to balance the potential variability.
In conclusion, our observations underscore the vital importance of implementing proper control strategies in conditional animal research, such as using littermate control [24]. When mouse strains are genetically modified by random integration of transgenes, the inserted loci should be more carefully controlled.
Data availability
All data supporting the findings in this study are available from the corresponding author upon reasonable request.
References
Xu Z, Moreno-Giró À, Zhao D, Krämer A, Pandey RK, Xu B, et al. Fcgr2b and Fcgr3 are the major genetic factors for cartilage antibody-induced arthritis, overriding the effect of Hc encoding complement C5. Eur J Immunol. 2024;54:2350659.
Sauer B, Henderson N. Site-specific DNA recombination in mammalian cells by the Cre recombinase of bacteriophage P1. Proc Natl Acad Sci. 1988;85:5166–70.
Passegué E, Wagner EF, Weissman IL. JunB deficiency leads to a myeloproliferative disorder arising from hematopoietic stem cells. Cell. 2004;119:431–43.
Wang G, Zhang C, Kambara H, Dambrot C, Xie X, Zhao L, et al. Identification of the transgene integration site and host genome changes in MRP8-Cre/ires-EGFP transgenic mice by targeted locus amplification. Front Immunol. 2022;13:875991.
Luo H, Urbonaviciute V, Saei AA, Lyu H, Gaetani M, Végvári Á, et al. NCF1-dependent production of ROS protects against lupus by regulating plasmacytoid dendritic cell development and functions. JCI Insight. 2023;8:e164875.
Nandakumar KS, Bäcklund J, Vestberg M, Holmdahl R. Collagen type II (CII)-specific antibodies induce arthritis in the absence of T or B cells but the arthritis progression is enhanced by CII-reactive T cells. Arthritis Res Ther. 2004;6:R544.
Allen IC. Delayed-type hypersensitivity models in mice. Methods Mol Biol. 2013;1031:101–7.
Xu Z, Xu B, Lundström SL, Moreno-Giró À, Zhao D, Martin M, et al. A subset of type-II collagen-binding antibodies prevents experimental arthritis by inhibiting FCGR3 signaling in neutrophils. Nat Commun. 2023;14:5949.
Koscielny G, Yaikhom G, Iyer V, Meehan TF, Morgan H, Atienza-Herrero J, et al. The International Mouse Phenotyping Consortium Web Portal, a unified point of access for knockout mice and related phenotyping data. Nucleic Acids Res. 2014;42:D802–9.
Bajou K, Herkenne S, Thijssen VL, D’Amico S, Nguyen N-Q-N, Bouché A, et al. PAI-1 mediates the antiangiogenic and profibrinolytic effects of 16K prolactin. Nat Med. 2014;20:741–7.
Lee E, Vaughan DE, Parikh SH, Grodzinsky AJ, Libby P, Lark MW, et al. Regulation of matrix metalloproteinases and plasminogen activator inhibitor-1 synthesis by plasminogen in cultured human vascular smooth muscle cells. Circ Res. 1996;78:44–9.
Sultana S, Dey R, Bishayi B. Role of plasminogen activator inhibitor—1 (PAI-1) in regulating the pathogenesis of S. aureus arthritis via plasminogen pathway. Immunol Lett. 2019;209:53–66.
Li J, Guo Y, Holmdahl R, Ny T. Contrasting roles of plasminogen deficiency in different rheumatoid arthritis models. Arthritis Rheum. 2005;52:2541–8.
Iwaki T, Urano T, Umemura K. PAI-1, progress in understanding the clinical problem and its aetiology. Br J Haematol. 2012;157:291–8.
Kwak SH, Wang XQ, He Q, Fang WF, Mitra S, Bdeir K, et al. Plasminogen activator inhibitor-1 potentiates LPS-induced neutrophil activation through a JNK-mediated pathway. Thromb Haemost. 2006;95:829–35.
Park Y-J, Liu G, Lorne EF, Zhao X, Wang J, Tsuruta Y, et al. PAI-1 inhibits neutrophil efferocytosis. Proc Natl Acad Sci. 2008;105:11784–9.
Roelofs JJTH, Teske GJD, Bonta PI, de Vries CJM, Meijers JCM, Weening JJ, et al. Plasminogen activator inhibitor-1 regulates neutrophil influx during acute pyelonephritis. Kidney Int. 2009;75:52–59.
Arandjelovic S, Perry JSA, Lucas CD, Penberthy KK, Kim T-H, Zhou M, et al. A noncanonical role for the engulfment gene ELMO1 in neutrophils that promotes inflammatory arthritis. Nat Immunol. 2019;20:141–51.
Clausen BE, Burkhardt C, Reith W, Renkawitz R, Förster I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 1999;8:265–77.
Orthgiess J, Gericke M, Immig K, Schulz A, Hirrlinger J, Bechmann I, et al. Neurons exhibit Lyz2 promoter activity in vivo: Implications for using LysM-Cre mice in myeloid cell research. Eur J Immunol. 2016;46:1529–32.
Blank T, Prinz M. CatacLysMic specificity when targeting myeloid cells? Eur J Immunol. 2016;46:1340–2.
Culemann S, Grüneboom A, Nicolás-Ávila JÁ, Weidner D, Lämmle KF, Rothe T, et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nature. 2019;572:670–5.
Hasenberg A, Hasenberg M, Männ L, Neumann F, Borkenstein L, Stecher M, et al. Catchup: a mouse model for imaging-based tracking and modulation of neutrophil granulocytes. Nat Methods. 2015;12:445–52.
Holmdahl R, Malissen B. The need for littermate controls. Eur J Immunol. 2012;42:45–47.
Acknowledgements
This work was supported by the Knut and Alice Wallenberg Foundation (2019.0059), the Leo Foundation (LF-OC-22-001023), Vetenskapsrådet (2019-01209), the Karolinska Institutet Research Foundation (2024-02534), and Karolinska Institutet´s Foundation for Rheumatology Research (2024-03940). The authors thank KMA technicians in taking care of the animals.
Funding
Open access funding provided by Karolinska Institute.
Author information
Authors and Affiliations
Contributions
ZX and RH designed the study, ZX, LRC, AMG, and RKP performed the experiments and analyzed the data. ZX drafted, and all authors revised and approved the manuscript. RH supervised all experiments and interpreted the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xu, Z., Romero‐Castillo, L., Moreno-Giró, À. et al. The need for Cre-loci controls in conditional mouse experiments: Mrp8-cre transgene predisposes mice to antibody-induced arthritis. Genes Immun 26, 169–172 (2025). https://doi.org/10.1038/s41435-024-00313-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41435-024-00313-3