Abstract
Reservoir pressure parameters (i.e., reservoir pressure [RP] and excess pressure [XSP]) independently predict cardiovascular events in adults, but this has not been investigated in children. This study aimed to determine (1) the association of reservoir pressure parameters with carotid intima-media thickness (carotid IMT), a preclinical vascular phenotype, and (2) whether a multivariable regression model with or without reservoir pressure parameters fits better for estimating carotid IMT in children. Study participants were 11–12-year-old children (n = 1231, 50% male) from the Child Health CheckPoint study, a cross-sectional substudy of the population-based Longitudinal Study of Australian Children. RP and XSP were obtained using brachial-cuff oscillometry (SphygmoCor XCEL, AtCor, Sydney). Carotid IMT was quantified by vascular ultrasonography. XSP was associated with carotid IMT after adjusting for confounders including age, sex, BMI z-score, heart rate, pubertal stage, moderate-to-vigorous physical activity, and mean arterial pressure (β = 0.93 µm, 95% CI 0.30–1.56 for XSP peak and β = 0.04 µm, 95% CI 0.01–0.08 for XSP integral). The results of the likelihood ratio test indicated a trend that the model with XSP and the above confounders fit better than a similar model without XSP for estimating carotid IMT. Our findings indicate that brachial-cuff device-measured XSP is associated with carotid IMT independent of conventional cardiovascular risk factors, including standard BP. This implies that a clinically convenient cuff approach could provide meaningful information for the early assessment of cardiovascular risk among children.
Similar content being viewed by others
Introduction
Cardiovascular events such as myocardial infarction and stroke remain the leading cause of death, and the development of cardiovascular risk begins in childhood [1,2,3]. Establishment of early detection and prevention strategies in childhood is effective because of the lifelong benefits of healthy behaviors (such as diet and physical activity patterns) [4]. High blood pressure (BP) is a leading modifiable risk factor for vascular damage and cardiovascular events [5, 6]. BP is standardly defined based on the peak (systolic BP), nadir (diastolic BP), and average mean arterial pressure (MAP) measures. However, standard BP parameters may overlook important risk information contained in the arterial BP waveform.
Several theoretical models have been proposed to explain the physiology of the BP waveform [7]. One such construct is the reservoir-excess pressure model, which proposes that the BP waveform comprises reservoir pressure (RP) and excess pressure (XSP) components [8]. RP is attributable to changes in arterial blood volume and is dependent on multiple factors, including left ventricular output and systemic arterial compliance. XSP is calculated as the difference between total BP and RP. In the ascending aorta, XSP is approximately equal to the pressure that would be produced by the left ventricle in the absence of any wave reflection [9, 10]; it has also been proposed to represent excess hydraulic work above the theoretical minimum level required to eject stroke volume from the left ventricle into the arterial system [11]. Peripheral XSP depends on local arterial properties, but its physiological interpretation is less clear. Recent clinical studies have shown that reservoir-excess pressure model-related parameters (i.e., RP, XSP and the systolic rate constant) predict cardiovascular events beyond conventional risk factors in adults [12,13,14]. These clinical studies used the noninvasive tonometry technique to measure RP parameters, but this method has not been adopted by clinicians for many reasons, including high operator dependence [15].
Operator-independent oscillometric cuff devices are routinely used for BP measurement. One such device (SphygmoCor XCEL, Atcor Medical Pty Ltd) has been developed in addition to standard cuff BP to measure the brachial artery BP waveform. Concordance of the XCEL cuff-based RP and XSP compared with invasive aortic measures of RP and XSP in adults has been previously published [16], which indicates that conventional cuff-based devices could offer a convenient, operator-independent method to estimate RP parameters (albeit with accuracy improvement needed [16]). We recently showed that brachial cuff-measured RP parameters were associated with preclinical phenotypes of cardiovascular risk among adults, including carotid intima-media thickness (carotid IMT) and pulse wave velocity (PWV) [17]. However, whether brachial-cuff RP parameters are independently associated with cardiovascular risk in children is unknown. Therefore, in a large population of Australian children, we aimed to determine the association between RP parameters and a widely used preclinical vascular structural phenotype (carotid IMT), which is predictive of cardiovascular events in adults. In addition, we examined whether adding RP parameters into the conventional risk model would improve the current assessment accuracy of carotid IMT.
Methods
Study participants
Participants were from the Child Health CheckPoint study, a cross-sectional substudy of the Longitudinal Study of Australian Children (LSAC) birth cohort [18]. CheckPoint focused on physical health and biomarkers and was performed between the LSAC sixth and seventh waves when the children were 11–12 years old. Recruitment to LSAC applied a two-stage sampling design by (1) randomly selecting ten percent of all Australian postcodes (stratified by state and urban/rural domicile) and (2) selecting children from the Medicare database. The LSAC birth cohort recruited 8928 healthy infants who were aged 0–1 years in 2014, of whom 57.2% responded for wave 1 in 2004 and a further 73.7% were retained for wave 6 in 2014. CheckPoint participants were recruited from wave 6, and 1874 children attended the CheckPoint assessment. CheckPoint study population and health assessment protocols have been published [18]. The study protocol was approved by the Royal Children’s Hospital Melbourne Human Research Ethics Committee (33225D) and Australian Institute of Family Studies Ethics Committee and performed in accordance with the 1975 Declaration of Helsinki. All participants provided written consent.
Study procedures
CheckPoint visited each Australian state between February 2015 and March 2016. A total of 15 assessment centers were set up nationwide, and a home visit was provided for those participants (n = 365, 20%) who were unable to attend the assessment center. Measurement of carotid IMT and blood sampling, from which total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, and triglycerides were measured, were only performed at assessment centers because these interventions were not feasible at home visits. All other measurements were obtained at assessment centers and home visits.
Participants’ characteristics
Anthropometry was measured with the participants in light clothing and without shoes. Standing height was measured in duplicate using a portable stadiometer (Invicta stadiometer IP0955). If the first two measurements differed by ≥0.5 cm, a third measurement was taken. Height was determined by the mean of all measurements. Weight was measured using a four-limb bioelectrical impedance analysis scale (InBody230, Biospace Co., Ltd Seoul, South Korea). Body mass index z-score (BMI z-score) was derived based on a reference population for age and sex using both Centers for Disease Control and UK 1990 reference values [19]. Heart rate was obtained using SphygmoCor XCEL during the BP measurement. Pubertal stage was self-reported and categorized using the Pubertal Development Scale [20]. Ambulatory physical activity was assessed using a wrist-worn accelerometer (GENE Activ Original, Cambs, UK) that participants wore on the nondominant wrist for 8 full days. Every 60 s epoch of waking wear time was classified as sedentary, light and moderate-to-vigorous physical activity using the Phillips cut points [21]. Hypertension was defined as ≥95th percentile for age, sex, and height of systolic and/or diastolic BP [22]. Pulse pressure (PP) was defined as the difference between systolic BP and diastolic BP. MAP was estimated as the sum of diastolic BP and 1/3 × (systolic BP − diastolic BP). Measurement of other sample characteristics is outlined in the published protocol [18]. Aortic stiffness was measured by carotid-femoral PWV in triplicate using SphygmoCor XCEL [17].
Carotid intima-media thickness
Carotid IMT was determined by B-mode ultrasound (Vivid I Bt06 with 10 MHz L-RS vascular probe, GE Healthcare, Chicago, IL, USA). Trained technicians measured ultrasound images over 5–10 cardiac cycles (tracked using three-lead ECG) at 10 mm proximal to the carotid bulb on the right common carotid artery in the supine position. Carotid IMT was measured at end-diastole, corresponding to the r-wave of a contemporaneous ECG. The images were analyzed using a Carotid Analyzer (Medical Imaging Applications, Coralville, IA, USA) for semiautomated border detection. The mean thickness in micrometers of 3–5 frames of the one carotid IMT measurement over the 5- to 10 mm section was measured as the carotid IMT, and an average of three carotid IMT measurements was used in the analysis. The analysis of carotid IMT was performed by six trained raters. The within-observer and between-observer coefficients of variation were 6.5% and 9.5% for mean carotid IMT values, respectively. The inter- and intraoperator reliability of measurements were comparable to the results reported in other studies [23]. The assessors of the carotid IMT were blinded to the values of the RP parameters.
Reservoir pressure parameters
Standard cuff BP and brachial volumetric waveforms were measured using an oscillometric device (SphygmoCor XCEL, AtCor Medical Pty Ltd., West Ryde, Australia). These measures were obtained in triplicate in the supine position after 7 min of rest. Six participants did not complete the measurement of brachial BP or brachial volumetric waveforms. The brachial volumetric waveforms were ensemble averaged by the device software and automatically calibrated (scaled) with brachial systolic and diastolic BP to derive the brachial BP waveforms. A quality check of brachial BP waveforms was performed based on average pulse height (>80 unit), pulse height variation (≤5%), diastolic variation (≤5%), shape deviation (≤4%), operator index (default evaluated and reported by SphygmoCor XCEL, ≥75), and systolic BP between 50 and 200 mmHg. The waveforms in 268 participants did not meet the criteria and were excluded. A total of 1231 participants remained, and the first valid brachial BP waveform in each participant was calibrated with the average of three brachial systolic and diastolic BPs for the derivation of RP parameters.
RP parameters were calculated using the pressure-only approach according to Eq. (1), and this was undertaken using a customized MATLAB program (Mathworks, Inc, Natick, MA) [6].
where P is the measured total pressure, Preservoir is RP, and P∞ is the arterial asymptotic pressure. Sc and Dc are the systolic and diastolic rate constants, relating to the speed of the upslope and downslope on the BP waveform, respectively [8]. The XSP integral is defined as the total pressure minus the RP integral. An example BP waveform showing the RP and XSP components is shown in Fig. 1. An assumption of the RP algorithm is that waveforms exhibit exponential pressure decay during diastole. However, the waveforms were ensemble averaged without consideration of cardiac duration, and this resulted in an additional small upslope after the nadir of the BP waveform in diastole over a few measurements. These measurements generated nonphysiological values of P∞ (i.e., that were greater than diastolic BP). Waveform modification was conducted to solve this problem by removing the small upslope occurring at the end of diastole and then reapplying the algorithm to derive RP parameters.
Statistical analysis
Continuous data are presented as the mean (standard deviation), and categorical data are presented as percentages. Exposures were RP peak, RP integral, XSP peak, and XSP integral. The outcome was carotid IMT. Uni- and multivariable regression analyses were used to explore whether the exposures were associated with the outcome independent of confounders. Conventional cardiovascular risk factors in adolescents were considered potential confounders. Only the conventional risk factors that were correlated with both exposures and outcome (r > 0.1) or considered physiologically important (i.e., heart rate) were included in the adjusted models. Altogether, age, sex, BMI z-score, heart rate, pubertal stage, moderate-to-vigorous physical activity and the standard BP measurements (i.e., systolic BP, diastolic BP, PP, or MAP entered separately, or PP and MAP entered simultaneously) were analyzed in the adjusted models. Partial coefficients of determination (partial R2) are presented as the percentage variance in outcome explained by each risk factor. Mediation analysis was performed by decomposing the total effect of XSP into direct and indirect parts using structural equation models [24] to determine whether the relationship between XSP and carotid IMT was mediated through PWV (i.e., whether the indirect effect was statistically significant). The likelihood ratio test was used to compare the fit of multivariable regression models (including conventional BP parameters) with and without RP parameters for estimating the association with carotid IMT. Data were analyzed using Stata 15.0 (StataCorp LP, TX, USA).
Results
Participant characteristics
Figure 2 presents the participant flow from LSAC wave 6 onwards, and Table 1 shows the participant characteristics. Participants were 11–12 years old, and half were girls. There were 181 participants whose carotid IMT was greater than the 90th percentile (>558 μm). The majority were in the early or middle pubertal stage. Participants spent only 3.8% of awake time on moderate-to-vigorous physical activity. Participants were from a relatively socioeconomically advantaged background because the disadvantage index score [25] was higher than the national mean of 1000 and SD of 100.
Associations between reservoir pressure parameters and carotid IMT
The effects of systolic BP, diastolic BP, PP, MAP, and both PP and MAP on the associations between RP parameters and carotid IMT were similar for the BP variable (Supplementary Table 1). Moreover, MAP best represents the arterial distending pressure [26], and thus, the results were principally presented using MAP as the BP parameter in models.
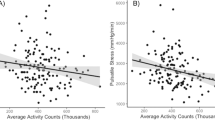
Table 2 shows the associations between RP parameters and carotid IMT in the uni- and multivariable regression models. XSP (both peak and integral), but not RP (neither peak nor integral), was associated with carotid IMT in univariable models, and this persisted after adjusting for confounders (age, sex, BMI z-score, heart rate, pubertal stage, moderate-to-vigorous physical activity, and MAP). Although the overall adjusted model R2 for the association between XSP and carotid IMT in the multivariable regression models was small, XSP (both peak and integral) was the strongest contributor to the total explainable variance in carotid IMT compared with the other risk factors, followed by age and sex, as indicated by partial R2. Mediation analysis separately evaluated the direct effect of XSP on carotid IMT as well as the indirect effect through PWV, which was nonsignificant (β = 0.01 µm, 95% CI −0.04–0.05, p = 0.86 for XSP peak and β = −0.01 µm, 95% CI −0.01–0.01, p = 0.86 for XSP integral).
Table 3 compares the model fit of the multivariable regression models with and without RP parameters for estimating the association with carotid IMT. Compared to the conventional cardiovascular risk assessment models including BP parameters (except for inclusion of PP or both PP and MAP), the model additionally incorporating XSP (both peak and integral) fit better for estimating carotid IMT.
Discussion
This is the first study to investigate the potential clinical utility of RP parameters in children. Our principal findings were that (1) brachial-cuff XSP was associated with carotid IMT above and beyond conventional cardiovascular risk factors, including standard BP, (2) brachial-cuff XSP was the strongest contributor to the total explainable variance in carotid IMT compared with conventional cardiovascular risk factors, and (3) in general, the model with brachial-cuff XSP and conventional cardiovascular risk factors was better for estimating the association with carotid IMT than a similar model without brachial-cuff XSP. These findings indicate that information provided by cuff-measured XSP may improve cardiovascular risk assessment in children.
Although the brachial volumetric BP waveform is measured at subdiastolic (low) BP, which is likely to dampen the actual waveform features [16], cuff device-measured RP parameters have been previously found to be related to preatherosclerosis and aortic stiffness in adults [17], and this current work found associations with the preclinical vascular phenotype in children. These findings are consistent with the results from the CAFÉ study, which applied tonometry to measure RP parameters and observed an association between brachial-cuff XSP and carotid IMT [12], suggesting that the clinical relevance of RP parameters is apparent irrespective of the pressure waveform measurement technique (tonometry or oscillometry) or measurement site (brachial or radial artery).
Similar to previous findings in adults [12, 17], XSP but not RP was associated with carotid IMT. XSP has been proposed as an index of generalized circulatory dysfunction, and although a link with impaired endothelial function has been proposed [12], this has not yet been demonstrated. On the other hand, RP is relatively constant in magnitude throughout the large arterial system, is related to total arterial compliance and is therefore less sensitive to local arterial properties, such as at the carotid artery [8, 27, 28]. Thus, the lack of association between RP and carotid preatherosclerosis was not unexpected. In contrast to our findings in adults, where XSP was the third strongest correlate of carotid IMT following age and sex [17], XSP was the strongest correlate of carotid IMT in children. This may be due to the narrow age range in the present study (11–12 years old) and that sex differences in the relationships with arterial physiology may not become apparent until approximately the age of 15 years [23]. The pubertal stage was negatively associated with carotid IMT among children, contrary to the finding of a recent study [29]. The reason for the inconsistency is unclear but may be due to the different sample sizes and population characteristics, as a prior study was performed in 55 children (9.2–14.8 years) with chronic kidney disease [29]. The multivariable regression model with XSP fit better than a similar model without XSP for estimating the association with carotid IMT, indicated by likelihood ratio. This indicates a future potential for incorporating XSP into assessment models to improve the assessment of risk related to preclinical vascular phenotype in children. However, the underlying reason for the association between brachial-cuff XSP and carotid IMT and the clinical significance of brachial-cuff XSP in predicting cardiovascular disease is unclear and requires further investigation.
The strengths of this study include the comprehensive and high-quality measurement of variables in the CheckPoint examinations, which enabled adjustment for well-known conventional risk factors in the statistical models. We also acknowledge several limitations. First, the CheckPoint sample is underrepresented by families in a disadvantaged socioeconomic position [17], and thus, the results may have limited generalizability. Second, although the value of childhood carotid IMT for predicting adulthood cardiovascular disease has not been determined, carotid IMT has been consistently shown to be related to elevated levels of risk factors in children [30], which is predictive of cardiovascular events in adults [31]. Third, RP parameters were calculated using the pressure-only approach (no flow). This is a simplified method of pressure-flow approach involving additional assumptions but has been shown to be largely equivalent to the pressure-flow method [28]. Fourth, the accuracy of cuff-based methods to estimate RP parameters has not been tested according to standardized criteria in children. The XCEL cuff-based device was recently shown to overestimate central and peripheral BP compared with invasive measurements in children [32] but underestimate central BP in adults [33]. It is hard to gauge how this variable BP accuracy may have influenced RP findings in the current study, but it may have weakened the true level of observed associations. Finally, the 1/3 form-factor used to calculate MAP might produce inaccurate estimation of the true intra-arterial MAP [34]. In any case, the association between XSP and carotid IMT remained after adjusting for other conventional BP parameters (i.e., systolic BP, diastolic BP, and PP).
In conclusion, this study is the first to report the association between XSP and carotid IMT separate from conventional cardiovascular risk factors, including standard BP, among 11–12-year-old children. In addition, incorporating XSP into the current assessment model could help more accurately predict cardiovascular risk in children, but the full extent of clinical significance is yet to be determined. Thus, more investigations are required to confirm our observations in children.
References
Roth GA, Johnson C, Abajobir A, Abd-Allah F, Abera SF, Abyu G, et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J Am Coll Cardiol. 2017;70:1–25.
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R, Prospective Studies C. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360:1903–13.
Kannel WB. Role of blood pressure in cardiovascular disease: the Framingham Study. Angiology. 1975;26:1–14.
Williams CL, Hayman LL, Daniels SR, Robinson TN, Steinberger J, Paridon S, Bazzarre T. Cardiovascular health in childhood: a statement for health professionals from the committee on atherosclerosis, hypertension, and obesity in the young (AHOY) of the council on cardiovascular disease in the young, American Heart Association. Circulation. 2002;106:143–60.
Bonafini S, Giontella A, Tagetti A, Montagnana M, Benati M, Danese E, et al. Markers of subclinical vascular damages associate with indices of adiposity and blood pressure in obese children. Hypertens Res. 2019;42:400–10.
White WB. Hypertension: pathophysiology, diagnosis, and management. JAMA. 1991;265:1462.
Segers P, O’Rourke MF, Parker K, Westerhof N, Hughes A. Towards a consensus on the understanding and analysis of the pulse waveform: results from the 2016 Workshop on Arterial Hemodynamics: past, present and future. Artery Res. 2017;18:75–80.
Wang JJ, O’Brien AB, Shrive NG, Parker KH, Tyberg JV. Time-domain representation of ventricular-arterial coupling as a windkessel and wave system. Am J Physiol Heart Circ Physiol. 2003;284:H1358–68.
Westerhof N, Westerhof BE. The reservoir wave paradigm discussion. J Hypertens. 2015;33:458–60.
Mynard JP, Kondiboyina A, Kowalski R, Cheung MMH, Smolich JJ. Measurement, analysis and interpretation of pressure/flow waves in blood vessels. Front Physiol. 2020;11:1085. https://doi.org/10.3389/fphys.2020.01085.
Parker KH, Alastruey J, Stan GB. Arterial reservoir-excess pressure and ventricular work. Med Biol Eng Comput. 2012;50:419–24.
Davies JE, Lacy P, Tillin T, Collier D, Cruickshank JK, Francis DP, et al. Excess pressure integral predicts cardiovascular events independent of other risk factors in the conduit artery functional evaluation substudy of Anglo-Scandinavian cardiac outcomes. Trial Hypertens. 2014;64:60–8.
Hametner B, Wassertheurer S, Hughes AD, Parker KH, Weber T, Eber B. Reservoir and excess pressures predict cardiovascular events in high-risk patients. Int J Cardiol. 2014;171:31–6.
Cheng HM, Chuang SY, Wang JJ, Shih YT, Wang HN, Huang CJ, et al. Prognostic significance of mechanical biomarkers derived from pulse wave analysis for predicting long-term cardiovascular mortality in two population-based cohorts. Int J Cardiol. 2016;215:388–95.
McEniery CM, Cockcroft JR, Roman MJ, Franklin SS, Wilkinson IB. Central blood pressure: current evidence and clinical importance. Eur Heart J. 2014;35:1719–25.
Peng X, Schultz MG, Picone DS, Dwyer N, Black JA, Roberts-Thomson P, Sharman JE. Non-invasive measurement of reservoir pressure parameters from brachial-cuff blood pressure waveforms. J Clin Hypertens. 2018;20:1703–11.
Peng X, Wake M, Schultz MG, Burgner DP, Otahal P, Mynard JP, Ellul S, et al. Association of brachial-cuff excess pressure with carotid intima-media thickness in Australian adults: a cross-sectional study. J Hypertens. 2020;38:723–30.
Clifford SA, Davies S, Wake M. Child Health CheckPoint Team. Child Health CheckPoint: cohort summary and methodology of a physical health and biospecimen module for the Longitudinal Study of Australian Children. BMJ Open. 2019;9(Suppl 3):3–22. https://doi.org/10.1136/bmjopen-2017-020261.
Kuczmarski RJ, Ogden CL, Guo SS, Grummer-Strawn LM, Flegal KM, Mei Z, et al. 2000 CDC Growth Charts for the United States: methods and development. Vital Health Stat 11. 2002:1–190.
Bond L, Clements J, Bertalli N, Evans-Whipp T, McMorris BJ, Patton GC, et al. A comparison of self-reported puberty using the pubertal development scale and the sexual maturation scale in a school-based epidemiologic survey. J Adolesc. 2006;29:709–20.
Phillips LR, Parfitt G, Rowlands AV. Calibration of the GENEA accelerometer for assessment of physical activity intensity in children. J Sci Med Sport. 2013;16:124–8.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et al. Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents. Pediatrics. 2017;140:e20171904. https://doi.org/10.1542/peds.2017-1904.
Doyon A, Kracht D, Bayazit AK, Deveci M, Duzova A, Krmar RT, et al. Carotid artery intima-media thickness and distensibility in children and adolescents: reference values and role of body dimensions. Hypertens. 2013;62:550–6.
Sobel ME. Direct and indirect effects in linear structural equation models. Sociol Methods Res. 1987;16:155–76.
Australian Bureau of Statistics. Census of population and housing: socio-economic indexes for areas (SEIFA), Australia (cat. no. 2033.0.55.001). 2016. https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/2033.0.55.001~2016~Main%20Features~IRSD~19.
Townsend RR, Wilkinson IB, Schiffrin EL, Avolio AP, Chirinos JA, Cockcroft JR, et al. Recommendations for improving and standardizing vascular research on arterial stiffness: a scientific statement from the American Heart Association. Hypertens. 2015;66:698–722.
Peng X, Schultz MG, Picone DS, Black JA, Dwyer N, Roberts-Thomson P, et al. Arterial reservoir characteristics and central-to-peripheral blood pressure amplification in the human upper limb. J Hypertens. 2017;35:1825–31.
Aguado-Sierra J, Davies JE, Hadjiloizou N, Francis D, Mayet J, Hughes AD, Parker KH. Reservoir-wave separation and wave intensity analysis applied to carotid arteries: a hybrid 1D model to understand haemodynamics. Conf Proc IEEE Eng Med Biol Soc. 2008;2008:1381–4.
Lopes R, Morais MB, Oliveira FLC, Brecheret AP, Abreu A, Andrade MC. Evaluation of carotid intima-media thickness and factors associated with cardiovascular disease in children and adolescents with chronic kidney disease. J Pediatr. 2019;95:696–704.
Juonala M, Magnussen CG, Venn A, Dwyer T, Burns TL, Davis PH, et al. Influence of age on associations between childhood risk factors and carotid intima-media thickness in adulthood: the Cardiovascular Risk in Young Finns Study, the Childhood Determinants of Adult Health Study, the Bogalusa Heart Study, and the Muscatine Study for the International Childhood Cardiovascular Cohort (i3C) Consortium. Circulation. 2010;122:2514–20.
Polak JF, Pencina MJ, Pencina KM, O’Donnell CJ, Wolf PA, D’Agostino RB Sr. Carotid-wall intima-media thickness and cardiovascular events. N Engl J Med. 2011;365:213–21.
Mynard JP, Goldsmith G, Springall G, Eastaugh L, Lane GK, Zannino D, et al. Central aortic blood pressure estimation in children and adolescents: results of the KidCoreBP study. J Hypertens. 2020;38:821–8.
Schultz MG, Picone DS, Armstrong MK, Black JA, Dwyer N, Roberts-Thomson P, Sharman JE. Validation Study to determine the accuracy of central blood pressure measurement using the sphygmocor Xcel cuff device. Hypertension. 2020;76:244–50.
Picone DS, Schultz MG, Peng X, Black JA, Dwyer N, Roberts-Thomson P, et al. Intra-arterial analysis of the best calibration methods to estimate aortic blood pressure. J Hypertens. 2019;37:307–15.
Funding
To date, this work has been supported by the National Health and Medical Research Council of Australia (1041352, 1109355), the Royal Children’s Hospital Foundation (2014-241), the Murdoch Children’s Research Institute, the University of Melbourne, the National Heart Foundation of Australia (100660), and the Financial Markets Foundation for Children (2014-055). The funding bodies played did not play any role in the study. The following authors were supported by the National Health and Medical Research Council of Australia: Early Career Fellowship (1104731) to MGS, R.D. Wright Career Development Fellowship (1143510) to JM, Senior Research Fellowships (1046518) to MW and (1064629, 1175744) to DPB, and Career Development Fellowship (reference 409940) to JS. MW was also supported by Cure Kids, New Zealand, and DB and JM were also supported by the Future Leader Fellowship of the National Heart Foundation of Australia (100369 and 101866). MJ was supported by the Federal Research Grant of Finland to Turku University Hospital, Finnish Cardiovascular Foundation. This article uses unit record data from the LSAC. The study is conducted in partnership between the Department of Social Services (DSS), the Australian Institute of Family Studies (AIFS), and the Australian Bureau of Statistics (ABS). The findings and views reported in this article are those of the author and should not be attributed to DSS, AIFS, or the ABS. We are also grateful to Mr. Xun Yang, HeFei University of Technology, China for providing some technical assistance.
Author information
Authors and Affiliations
Contributions
XP contributed to data collection, analysis, and interpretation of data and drafting of paper; DSP, MGS, and JES: project conception and study design, interpretation of data, and critical revision of paper; MW: Health Design Leader LSAC, principal investigator Child Health CheckPoint, project conception and critical revision of paper; MC, DPB, and MJ: project conception and critical revision of paper; GC and SE: statistical assistance and critical revision of paper; JM: suggestion of data analysis and critical revision of paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Peng, X., Picone, D.S., Schultz, M.G. et al. Brachial-cuff excess pressure is associated with carotid intima-media thickness among Australian children: a cross-sectional population study. Hypertens Res 44, 541–549 (2021). https://doi.org/10.1038/s41440-020-00576-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-020-00576-z



 ) with example reservoir pressure parameters. The reservoir pressure (
) with example reservoir pressure parameters. The reservoir pressure ( ) and excess pressures (
) and excess pressures ( ) are expressed in both the peak and integral, where the peak refers to the highest value and integral refers to the area under the curve
) are expressed in both the peak and integral, where the peak refers to the highest value and integral refers to the area under the curve