Abstract
Patients with type 2 diabetes (T2D) frequently have multiple cardiovascular, metabolic, and renal comorbidities, such as hypertension, dyslipidemia, hyperuricemia, chronic kidney disease, and heart failure. Accordingly, this patient population often requires polypharmacy, which is associated with an increased risk of drug-drug interactions, poor adherence, and even adverse outcomes. Accumulating evidence on newer hypoglycemic agents, such as glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors, has highlighted the marked improvements in cardiovascular and renal outcomes associated with the off-target benefits for relevant comorbidities, including hypertension. These classes of hypoglycemic agents are unique, as they achieve consistently modest but significant reductions in systolic and diastolic blood pressure (BP), an effect that has not been targeted and observed with conventional hypoglycemic agents. In addition to this BP-lowering effect, these agents also have multifaceted beneficial impacts on other cardiometabolic and renal parameters, which appear to be helpful for providing an important comprehensive therapeutic approach to improve the prognosis in patients with T2D. The clinical advantages of these agents may reduce the dose and number of concomitant medications used to treat T2D and related comorbidities. These positive spillover effects may also enhance the clinical use of agents to achieve better diabetes care. As a consequence, the clinical significance of these hypoglycemic agents now extends beyond their hypoglycemic effects, thereby providing a new-normal strategy to use in an evidence-based, patient-centric approach to diabetes care.
Similar content being viewed by others
Introduction
Hypertension is a major comorbidity in patients with type 2 diabetes (T2D) and contributes synergistically to an increased risk of cardiovascular events and mortality. At the same time, patients with T2D often have multiple cardiovascular, metabolic, and renal comorbidities, such as dyslipidemia, hyperuricemia, chronic kidney disease (CKD), and heart failure (HF). Several previous landmark clinical trials demonstrated that strict glucose-lowering therapy alone failed to sufficiently improve the prognosis of patients with T2D [1,2,3]. However, it is possible that some meaningful long-term legacy effects may have influenced the positive effects of maintaining strict glucose control [4]. It is therefore clinically important to initiate and continue comprehensive care that includes not only safe and appropriate glucose-lowering therapy but also has a positive effect on multimorbidity [5, 6]. This approach suggests that diabetes care should be shifted from a predominantly glucocentric approach to a patient-centric, holistic approach [7, 8]. However, a recent cohort study showed that the control of cardiovascular risk factors is still suboptimal in patients with T2D [9]. Furthermore, even when these risk factors are managed optimally, patients with T2D still have a higher risk of cardiovascular events than people without T2D [9]. These findings indicate that patients with T2D have residual risk factors in addition to classic risk factors, suggesting that more comprehensive interventions for these factors are needed to improve the prognosis of patients with T2D.
When performing simultaneous intervention for multiple risk factors and related diseases, physicians and patients often require polypharmacy in the daily clinical setting, which in turn makes it difficult to receive safe and sufficient benefits from individual medications. Polypharmacy, particularly in patients with T2D, is associated with worsened cardiometabolic status and adverse events, such as hypoglycemia and poor clinical outcomes [10, 11]. These adverse effects may be, at least in part, caused by potential drug-drug and drug-food interactions [12,13,14]. It is therefore necessary to minimize polypharmacy while balancing the benefits and harmful effects of medications [15].
Recent cardiovascular outcome trials (CVOTs) on new hypoglycemic agents assessed their safety and demonstrated that two classes, glucagon-like peptide-1 [GLP-1] receptor agonists and sodium-glucose cotransporter 2 [SGLT2] inhibitors, reduced the risk of cardiovascular and renal events in a broad range of the patient population [16,17,18,19,20]. The common features of these trials were that (i) the cardiovascular and renal benefits of the drugs were independent of their glucose-lowering effect, (ii) the drugs also had favorable effects on multiple diabetes-related comorbidities via hemodynamic and nonglycemic metabolic actions in a simultaneous and transient fashion, and (iii) blood pressure (BP) reduction, body weight loss, and renal protection were commonly observed, emphasizing the clinical benefits of these drugs [21,22,23,24]. Therefore, in addition to their approval as hypoglycemic agents, these drugs have multifaceted effects and would be expected to have a beneficial impact on diabetes-related comorbidities, such as hypertension, obesity, CKD, and HF.
This review article focuses on recent evidence and understanding of hypertension care in patients with T2D and examines the known effects of hypoglycemic agents on BP. In addition, based on the aforementioned background of diabetes care, we discuss the emerging roles of newer hypoglycemic agents with proven cardiovascular benefits and their potential effect on diabetes-related comorbidities and contribution to minimizing polypharmacy in patients with T2D.
Management of hypertension in T2D
The incidence rates of comorbid hypertension in patients with diabetes and conversely, diabetes in patients with hypertension are both 2–3 times higher than that in patients without diabetes or hypertension. In patients with diabetes, systolic BP is an independent predictor of cardiovascular death [25], with an increase in systolic BP of 10 mmHg shown to be associated with increased risks of coronary artery disease (18%), ischemic stroke (29%), and hemorrhagic stroke (56%) [26]. In contrast, a decrease in systolic BP of 10 mmHg is associated with a substantial reduction in the risk of macro- and microvascular complications and mortality in patients with T2D [27]. However, whether intensive BP-lowering therapy results in a further reduction in the risk of such complications and mortality in patients with T2D has been controversial for a long period of time [28]. The United Kingdom Prospective Diabetes Study (UKPDS) 36 reported that the incidence rates of macro- and microvascular diseases were significantly lower in the group with strict systolic BP control (<120 mmHg) than in the group without strict control (≥160 mmHg) [29]. Similarly, the UKPDS 38 showed that compared to the non-strict BP control group (<180/105 mmHg), the strict BP control group (<150/85 mmHg) had 24% fewer diabetes-related endpoints, 32% fewer diabetes-related deaths, 44% fewer strokes, and 37% fewer microvascular complications [30]. In contrast, the Action to Control Cardiovascular Risk in Diabetes blood pressure trial in patients with T2D at high risk of cardiovascular events reported that there was no significant difference in major cardiovascular events or mortality in the strict systolic BP control group (<120 mmHg) compared to that observed in the standard control group (<140 mmHg) and that the group with strict BP control actually had an increase in adverse events [31]. That trial raised a clinical concern about strict BP lowering in patients with diabetes and resulted in a decrease in target BP levels. However, based on several clinical issues, including (i) study design, (ii) a secondary protocol of the original evaluation of strict glycemic control [32], (iii) a greater benefit of strict BP control on stroke [33, 34], and (iv) a difference in the incidence of stroke between Western and Japanese populations, the target BP level in patients with T2D is currently recommended to be <130/80 mmHg in the Japanese Society of Hypertension Guidelines for the Management of Hypertension [35]. In the Japan Diabetes Optimal Integrated Treatment Study for three Major Risk Factors of Cardiovascular Diseases (J-DOIT3) trial that assessed the effect of intensified multifactorial intervention on relevant risk factors, systolic BP decreased from 134 mmHg to 123 mmHg in the intensive therapy group and was associated with a 58% reduction in the risk of cerebrovascular events [6].
The first step in selecting antihypertensive agents in patients with T2D and hypertension is to evaluate the presence of microalbuminuria or proteinuria [35]. If these complications are present, an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB) should be considered first. In the absence of complications, ACE inhibitors, ARBs, calcium channel blockers (CCBs), or diuretics are selected. In particular, there is robust evidence that ACE inhibitors and ARBs have an inhibitory effect on macro- and microvascular complications [36,37,38]. On the other hand, if the antihypertensive effect of these single agents is insufficient, combination therapy with two or three agents is required. The Gauging Albuminuria Reduction With Lotrel in Diabetic Patients With Hypertension (GUARD) trial, which compared the effects of a CCB or diuretic in combination with an ACE inhibitor in patients with diabetic nephropathy, showed that the diuretic combination was effective for reducing proteinuria, while the CCB combination was effective for maintaining the estimated glomerular filtration rate [39].
Despite the importance of BP-lowering therapy and widespread recognition of target levels of BP, only approximately half of Western and Japanese patients with T2D achieve their BP targets [40, 41]. Our recent data obtained from randomized clinical trials on patients with T2D with/without previous cardiovascular diseases also showed that baseline mean systolic BP was still over 130 mm Hg [42, 43]. Thus, hypertension is well-known but residual risk that has not yet been fully overcome [44], and in clinical practice, there is a need for further implementation of individualized optimal antihypertensive care [45]. Of note, newer hypoglycemic agents, such as SGLT2 inhibitors and GLP-1 receptor agonists, have been uniquely and frequently proven to lower BP (details are shown later), suggesting that the combined use of conventional hypotensive agents and newer hypoglycemic agents may be helpful for further improving BP control and subsequent outcomes.
Hypoglycemic agents and their BP-lowering effects
In this section, we briefly summarize the effects of several hypoglycemic drugs on BP. Because the essential pharmacological action of each class of hypoglycemic agents is beyond the scope of this paper, this topic is not discussed in detail in this manuscript. More importantly, the modes of action of the drugs on BP remain to be fully understood.
First, the BP-lowering effect of most conventional hypoglycemic agents, such as metformin and sulfonylureas, has been investigated only rarely, and no obvious clinical effects on BP have been acknowledged. Thiazolidinediones (TZDs) are a class of insulin-sensitizing hypoglycemic agents that work by activating peroxisome proliferator-activated receptor-gamma. Although some studies have shown that the TZD pioglitazone reduced systolic and diastolic pressures [46], it is possible that this was an incidental effect of improved vascular function rather than a direct antihypertensive action. Given the overt anti-inflammatory and antiatherogenic actions of TZDs observed in several previous clinical trials [47,48,49], it remains uncertain whether the direct effect on BP explains the cardiovascular benefit of pioglitazone [50]. Alpha-glucosidase inhibitors (α-GIs) are a class of hypoglycemic agents that inhibit increases in postprandial blood glucose levels by delaying the absorption of carbohydrates in the small intestine. Although available data on the effect of α-GIs on BP are also limited, these agents are known to reduce systolic BP and lower the risk of hypertension developing in patients with T2D and/or prediabetes [51, 52]. This effect is thought to be due to the resultant increase in secretion of GLP-1 in the upper part of the small intestine [53]. Nonetheless, we acknowledge that the antihypertensive effects of these classes of drugs have not received a great deal of clinical attention within our daily practice.
Dipeptidyl peptidase-4 (DPP-4) inhibitors are the most frequently prescribed class of hypoglycemic agents in Japan and act by inhibiting the degradation of incretin hormones. Several experimental and human studies have reported that DPP-4 inhibitors also exhibit anti-inflammatory and antiatherogenic effects [54,55,56,57,58,59]. Furthermore, DPP-4 inhibitors are thought to have a vasodilatory effect induced by inactivated peptides, such as neuropeptide Y, and enhanced endothelium-dependent nitric oxide-cyclic guanosine monophosphate signaling [60, 61]. However, the effects of DPP-4 inhibitors on BP have been inconsistent in patients with T2D, and to date, only limited clinical data showing a clinically apparent BP-lowering effect are available [62]. This may be attributed, at least in part, to their neutral impacts on cardiovascular events and mortality seen in previous CVOTs. In addition, several randomized clinical trials in Japanese patients with T2D showed neutral effects of DPP-4 inhibitors on BP compared to the standard therapy for T2D, even though those trials did not have the primary objective of evaluating changes in BP [43, 54, 55]. Therefore, DPP-4 inhibitors are also likely to have a limited impact on BP in our daily clinical practice.
Recent CVOTs and subsequent meta-analyses of GLP-1 receptor agonists have shown that they reduce the risk of major adverse cardiovascular events in patients with T2D [16], with the underlying mechanisms of these cardiovascular benefits likely to be multifactorial and complex [63]. Regarding their effect on BP, a meta-analysis of GLP-1 receptor agonists demonstrated that treatment had a beneficial impact on BP in overweight or obese patients with or without T2D, in contrast to DPP-4 inhibitors, with a mean reduction in systolic BP of −3.57 mmHg and diastolic BP of −1.38 mmHg [64]. These agents are generally assumed to have vasodilatory and antiatherosclerotic effects through an anti-inflammatory effect and improvement of endothelial function resulting from enhanced GLP-1 concentrations and subsequent activation of multiple signaling pathways [65]. In addition, a recent experimental study demonstrated that regulation of BP following administration of a GLP-1 receptor agonist was associated with modulation of Na+/H+ exchanger isoform 3-mediated sodium reabsorption at the proximal tubule and activation of intrarenal renin angiotensin signaling [66]. GLP-1 receptor agonists have therefore been shown to consistently have a beneficial impact on BP control in patients with T2D, as well as maintaining glycemic control and reducing body weight [67, 68]. However, the profound underlying mechanisms of their BP-lowering action and differences in the impact on BP between GLP-1 receptor agonists and DPP-4 inhibitors also remain uncertain [69].
Robust cardiovascular benefits have also been shown in recent CVOTs on SGLT2 inhibitors carried out in a broad range of patients with cardiovascular and renal diseases regardless of their diabetes status [17, 18, 20]. These effects are likely driven by multiple actions and mechanisms resulting from primary natriuresis and glycosuria following SGLT2 inhibition [21, 23]. Regarding their effect on BP, several meta-analyses have consistently shown a modest reduction in systolic BP of between −4.0 and −2.5 mmHg and diastolic BP of between −1.8 and −1.5 mmHg [70,71,72], suggesting a class effect of SGLT2 inhibitors. In the SGLT2 inhibitor and ARB Combination theRapy in pAtients with diabetes and uncontrolled nocturnal hypertension (SACRA) study, 12 weeks of SGLT2 inhibition reduced nighttime systolic BP by −6.3 mmHg and 24 h systolic BP by −7.7 mmHg relative to placebo in Japanese patients with T2D and uncontrolled nocturnal hypertension [73]. This day-and-night improvement in BP control could be a major contributor to the reduction in the cardiovascular and renal events seen in recent CVOTs on SGLT2 inhibitors [74]. In addition, this beneficial impact on BP control was similar between subgroups aged <75 or ≥75 years [75]. Interestingly, the reduction in systolic and diastolic BP in patients with T2D and hypertension treated with an SGLT2 inhibitor was apparent irrespective of background use of conventional antihypertensives [76]. Furthermore, a head-to-head clinical trial comparing the clinical effects of a GLP-1 receptor agonist and the SGLT2 inhibitor canagliflozin (300 mg daily) showed that they were superior to once-weekly semaglutide 1.0 mg for reducing systolic and diastolic BP [77]. Therefore, SGLT2 inhibitors are expected to provide an additive benefit in the management of hypertension in patients with T2D [78, 79], although currently they are not approved for treatment of this condition. The detailed mechanisms of the BP-lowering effect of SGLT2 inhibition are also not fully understood [80]. However, given the essential pharmacological action of SGLT2 inhibitors, it is reasonable to accept the biphasic effects in combination with the hemodynamic and multiple metabolic actions on cardiovascular and renal systems, including BP control [21]. In the early phase, immediate osmotic and gradual natriuretic diuresis and the resultant reduction in fluid volume were likely the most dominant factors for reducing BP [81]. In the later phase, subsequent beneficial modulations of vascular, renal, and nervous system functions are likely to contribute to continuous BP control [82, 83]. In addition, an experimental model showed that SGLT2 inhibition ameliorated arterial pressure lability via baroreflex sensitization and attenuated sympathetic nerve activity [84]. This suggested that SGLT2 inhibition may improve the quality and level of BP. Such favorable impacts of SGLT2 inhibition are attributable mainly to cardiovascular and renal benefits, especially in patients at risk of HF and CKD.
Clinical perspectives
Hypoglycemic agents essentially lower blood glucose levels but are not pure antihypertensives. Importantly, there is evidence from recent CVOTs that GLP-1 receptor agonists and SGLT2 inhibitors are associated with a reduction in the risk of adverse cardiovascular and renal events. Although the pharmacological actions of these classes of drugs are apparently different, they have several common and apparent benefits on cardiovascular risk factors, including a reduction in BP (Table 1). On the other hand, the precise mechanisms underlying the clinical benefits of these drugs remain uncertain [85], and the extent to which their impact on BP contributes to these benefits has also not been fully examined. Given their multifaceted derivative actions, it is reasonable to conclude that these agents have multiple and simultaneous benefits on relevant risk factors, rather than just one effect contributing to the improved prognosis. This possibility reminds us of the clinical significance of a comprehensive approach to diabetes care [5, 6]. At the same time, it is also important to consider how the effects of these drugs on each factor influence patient outcomes and daily clinical practice. In recent mediation analyses of some CVOTs on GLP-1 receptor agonists and SGLT2 inhibitors, the BP-lowering effect induced had only a small effect on the cardiovascular benefits of these drugs [86,87,88]. A recent meta-analysis also revealed that these drugs reduced cardiovascular events independent of their BP-lowering effect [89]. Therefore, it is also necessary to reconsider how cardiovascular benefits should be monitored following the initiation of medications with proven benefits [90].
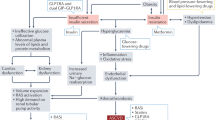
What are the clinical implications of the nonglycemic effects of hypoglycemic agents on diabetes care, such as lowering BP (Fig. 1)? In this paper, we reviewed the clinical characteristics and concerns of patients with T2D who frequently had multimorbidity and received polypharmacy [91]. When a drug has multifaceted off-target effects on relevant comorbidities, this could lead to a possible reduction in the dose and number of medications, thereby minimizing polypharmacy [92]. Furthermore, this approach could decrease the risk of drug-drug interactions and adverse side effects in patients receiving polypharmacy. Such influences may in part affect patients’ adherence and even outcomes, although this remains to be elucidated. In the Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients-Removing Excess Glucose (EMPA-REG OUTCOME) trial, the incidence of additional prescriptions for hypoglycemic and antihypertensive medications, including diuretics, was actually lower in the empagliflozin group than in the placebo group [93]. Therefore, in addition to the essential pharmacological effects, these various types of secondary benefits may be partially responsible for the improvement in outcomes observed in CVOTs on GLP-1 receptor agonists or SGLT2 inhibitors. This integrated approach using these agents will make it possible to select drugs that are truly necessary for individual patients and bring us closer to realizing evidence-based, patient-centric diabetes care.
Treating diabetes and multimorbidity with essential medication. In conventional-style care (red), patients with diabetes and multimorbidity often receive polypharmacy, with some of these drugs interacting unfavorably, leading to the development of adverse effects. In the new normal care proposed in this paper (blue), some key drugs (e.g., Drugs A, D, and F), including hypoglycemic agents that have potentially safe and multidisciplinary effects on relevant risk factors and comorbidities, have the potential to reduce the use and dosage of other drugs, leading to a possible reduction in polypharmacy and avoidance of undesirable drug-drug interactions and adverse effects. Importantly, physicians need to select essential drugs based on clinical evidence and the individual situation and reconsider the need for drugs that are administered chronically to patients. NAFLD nonalcoholic fatty liver disease, PAD peripheral artery disease
Conclusion
In the past decade, diabetes care has been changing rapidly and dynamically, with the clinical positioning of each hypoglycemic agent also having shifted progressively. Hypoglycemic agents proven recently to have cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, have highlighted the clinical importance of nonglycemic multifactorial benefits in daily diabetes care, including BP reduction. These classes of hypoglycemic agents may therefore be helpful for improving the quality of the entire clinical treatment strategy in diabetes care. Although these drugs are essentially hypoglycemic agents, they are also strong candidates for having additional beneficial impacts on relevant risk factors, including hypertension, and opening a new horizon for a better comprehensive therapeutic approach through a qualified reduction in polypharmacy in patients with T2D.
References
Duckworth W, Abraira C, Moritz T, Reda D, Emanuele N, Reaven PD, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360:129–39.
Gerstein HC, Miller ME, Byington RP, Goff DC Jr, Bigger JT, Buse JB, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358:2545–59.
Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358:2560–72.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med. 2003;348:383–93.
Ueki K, Sasako T, Okazaki Y, Kato M, Okahata S, Katsuyama H, et al. Effect of an intensified multifactorial intervention on cardiovascular outcomes and mortality in type 2 diabetes (J-DOIT3): an open-label, randomised controlled trial. Lancet Diabetes Endocrino. 2017;5:951–64.
Rodriguez-Gutierrez R, Gonzalez-Gonzalez JG, Zuñiga-Hernandez JA, McCoy RG. Benefits and harms of intensive glycemic control in patients with type 2 diabetes. BMJ. 2019;367:l5887.
Tanaka A, Node K. Evidence-based and tailored medication in type 2 diabetes: a pathway learned from clinical trials. Cardiovasc Diabetol. 2019;18:19.
Wright AK, Suarez-Ortegon MF, Read SH, Kontopantelis E, Buchan I, Emsley R, et al. Risk Factor Control and Cardiovascular Event Risk in People With Type 2 Diabetes in Primary and Secondary Prevention Settings. Circulation. 2020;142:1925–36.
Al-Musawe L, Martins AP, Raposo JF, Torre C. The association between polypharmacy and adverse health consequences in elderly type 2 diabetes mellitus patients; a systematic review and meta-analysis. Diabetes Res Clin Pr. 2019;155:107804.
van Oort S, Rutters F, Warlé-van Herwaarden MF, Schram MT, Stehouwer CD, Tack CJ, et al. Characteristics associated with polypharmacy in people with type 2 diabetes: the Dutch Diabetes Pearl cohort. Diabet Med. 2020;38:e14406.
Dobrică EC, Găman MA, Cozma MA, Bratu OG, Pantea Stoian A, Diaconu CC. Polypharmacy in Type 2 Diabetes Mellitus: Insights from an Internal Medicine Department. Med (Kaunas, Lith). 2019;55:436.
Al-Musawe L, Torre C, Guerreiro JP, Rodrigues AT, Raposo JF, Mota-Filipe H, et al. Polypharmacy, potentially serious clinically relevant drug-drug interactions, and inappropriate medicines in elderly people with type 2 diabetes and their impact on quality of life. Pharm Res Perspect. 2020;8:e00621.
Saito T, Ohmura H, Nojiri S, Daida H. Impact of sitagliptin combination therapy and hypoglycemia in Japanese patients with type 2 diabetes: a multi-center retrospective observational cohort study. J Pharm Health Care Sci. 2020;6:13.
Lipska KJ, Krumholz H, Soones T, Lee SJ. Polypharmacy in the Aging Patient: a Review of Glycemic Control in Older Adults With Type 2 Diabetes. JAMA. 2016;315:1034–45.
Kristensen SL, Rørth R, Jhund PS, Docherty KF, Sattar N, Preiss D, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrino. 2019;7:776–85.
Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31–39.
McGuire DK, Shih WJ, Cosentino F, Charbonnel B, Cherney DZI, Dagogo-Jack S, et al. Association of SGLT2 Inhibitors With Cardiovascular and Kidney Outcomes in Patients With Type 2 Diabetes: a Meta-analysis. JAMA Cardiol. 2021;6:148–58.
Palmer SC, Tendal B, Mustafa RA, Vandvik PO, Li S, Hao Q, et al. Sodium-glucose cotransporter protein-2 (SGLT-2) inhibitors and glucagon-like peptide-1 (GLP-1) receptor agonists for type 2 diabetes: systematic review and network meta-analysis of randomised controlled trials. BMJ. 2021;372:m4573.
Giugliano D, Longo M, Scappaticcio L, Caruso P, Esposito K. Sodium-glucose transporter-2 inhibitors for prevention and treatment of cardiorenal complications of type 2 diabetes. Cardiovasc Diabetol. 2021;20:17.
Tanaka A, Node K. Emerging roles of sodium-glucose cotransporter 2 inhibitors in cardiology. J Cardiol. 2017;69:501–7.
Tanaka A, Node K. Clinical application of glucagon-like peptide-1 receptor agonists in cardiovascular disease: lessons from recent clinical cardiovascular outcomes trials. Cardiovasc Diabetol. 2018;17:85.
Heerspink HJ, Perkins BA, Fitchett DH, Husain M, Cherney DZ. Sodium Glucose Cotransporter 2 Inhibitors in the Treatment of Diabetes Mellitus: Cardiovascular and Kidney Effects, Potential Mechanisms, and Clinical Applications. Circulation. 2016;134:752–72.
Ni L, Yuan C, Chen G, Zhang C, Wu X. SGLT2i: beyond the glucose-lowering effect. Cardiovasc Diabetol. 2020;19:98.
Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care. 1993;16:434–44.
Kengne AP, Patel A, Barzi F, Jamrozik K, Lam TH, Ueshima H, et al. Systolic blood pressure, diabetes and the risk of cardiovascular diseases in the Asia-Pacific region. J Hypertens. 2007;25:1205–13.
Emdin CA, Rahimi K, Neal B, Callender T, Perkovic V, Patel A. Blood pressure lowering in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2015;313:603–15.
Briasoulis A, Al Dhaybi O, Bakris GL. SGLT2 Inhibitors and Mechanisms of Hypertension. Curr Cardiol Rep. 2018;20:1.
Adler AI, Stratton IM, Neil HA, Yudkin JS, Matthews DR, Cull CA, et al. Association of systolic blood pressure with macrovascular and microvascular complications of type 2 diabetes (UKPDS 36): prospective observational study. BMJ. 2000;321:412–9.
UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ. 1998;317:703–13.
Cushman WC, Evans GW, Byington RP, Goff DC Jr, Grimm RH Jr, Cutler JA, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362:1575–85.
Margolis KL, O’Connor PJ, Morgan TM, Buse JB, Cohen RM, Cushman WC, et al. Outcomes of combined cardiovascular risk factor management strategies in type 2 diabetes: the ACCORD randomized trial. Diabetes Care. 2014;37:1721–8.
Bangalore S, Kumar S, Lobach I, Messerli FH. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation. 2011;123:2799–810.
Tsujimoto T, Kajio H. Benefits of Intensive Blood Pressure Treatment in Patients With Type 2 Diabetes Mellitus Receiving Standard but Not Intensive Glycemic Control. Hypertension. 2018;72:323–30.
Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2019). Hypertens Res. 2019;42:1235–481.
Heart Outcomes Prevention Evaluation Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet. 2000;355:253–9.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861–9.
Kurokawa K, Chan JC, Cooper ME, Keane WF, Shahinfar S, Zhang Z. Renin angiotensin aldosterone system blockade and renal disease in patients with type 2 diabetes: a subanalysis of Japanese patients from the RENAAL study. Clin Exp Nephrol. 2006;10:193–200.
Bakris GL, Toto RD, McCullough PA, Rocha R, Purkayastha D, Davis P. Effects of different ACE inhibitor combinations on albuminuria: results of the GUARD study. Kidney Int. 2008;73:1303–9.
Kobayashi M, Yamazaki K, Hirao K, Oishi M, Kanatsuka A, Yamauchi M, et al. The status of diabetes control and antidiabetic drug therapy in Japan-a cross-sectional survey of 17,000 patients with diabetes mellitus (JDDM 1). Diabetes Res Clin Pr. 2006;73:198–204.
Pagidipati NJ, Navar AM, Pieper KS, Green JB, Bethel MA, Armstrong PW, et al. Secondary Prevention of Cardiovascular Disease in Patients With Type 2 Diabetes Mellitus: international Insights From the TECOS Trial (Trial Evaluating Cardiovascular Outcomes With Sitagliptin). Circulation. 2017;136:1193–203.
Tanaka A, Shimabukuro M, Machii N, Teragawa H, Okada Y, Shima KR, et al. Effect of Empagliflozin on Endothelial Function in Patients With Type 2 Diabetes and Cardiovascular Disease: results from the Multicenter, Randomized, Placebo-Controlled, Double-Blind EMBLEM Trial. Diabetes Care. 2019;42:e159–e161.
Oyama J, Murohara T, Kitakaze M, Ishizu T, Sato Y, Kitagawa K, et al. The Effect of Sitagliptin on Carotid Artery Atherosclerosis in Type 2 Diabetes: the PROLOGUE Randomized Controlled Trial. PLoS Med. 2016;13:e1002051.
Navar AM, Gallup DS, Lokhnygina Y, Green JB, McGuire DK, Armstrong PW, et al. Hypertension Control in Adults With Diabetes Mellitus and Recurrent Cardiovascular Events: Global Results From the Trial Evaluating Cardiovascular Outcomes With Sitagliptin. Hypertension. 2017;70:907–14.
Shen Y, Dai Y, Wang XQ, Zhang RY, Lu L, Ding FH, et al. Searching for optimal blood pressure targets in type 2 diabetic patients with coronary artery disease. Cardiovasc Diabetol. 2019;18:160.
Clarke GD, Solis-Herrera C, Molina-Wilkins M, Martinez S, Merovci A, Cersosimo E, et al. Pioglitazone Improves Left Ventricular Diastolic Function in Subjects With Diabetes. Diabetes Care. 2017;40:1530–6.
Saremi A, Schwenke DC, Buchanan TA, Hodis HN, Mack WJ, Banerji M, et al. Pioglitazone slows progression of atherosclerosis in prediabetes independent of changes in cardiovascular risk factors. Arterioscler Thromb Vasc Biol. 2013;33:393–9.
Nissen SE, Nicholls SJ, Wolski K, Nesto R, Kupfer S, Perez A, et al. Comparison of pioglitazone vs glimepiride on progression of coronary atherosclerosis in patients with type 2 diabetes: the PERISCOPE randomized controlled trial. JAMA. 2008;299:1561–73.
Mazzone T, Meyer PM, Feinstein SB, Davidson MH, Kondos GT, D’Agostino RB Sr, et al. Effect of pioglitazone compared with glimepiride on carotid intima-media thickness in type 2 diabetes: a randomized trial. JAMA. 2006;296:2572–81.
Dormandy JA, Charbonnel B, Eckland DJ, Erdmann E, Massi-Benedetti M, Moules IK, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet. 2005;366:1279–89.
Hanefeld M, Cagatay M, Petrowitsch T, Neuser D, Petzinna D, Rupp M. Acarbose reduces the risk for myocardial infarction in type 2 diabetic patients: meta-analysis of seven long-term studies. Eur Heart J. 2004;25:10–16.
Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M. Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: the STOP-NIDDM trial. JAMA. 2003;290:486–94.
McCarty MF, DiNicolantonio JJ. Acarbose, lente carbohydrate, and prebiotics promote metabolic health and longevity by stimulating intestinal production of GLP-1. Open Heart. 2015;2:e000205.
Mita T, Katakami N, Yoshii H, Onuma T, Kaneto H, Osonoi T, et al. Alogliptin, a Dipeptidyl Peptidase 4 Inhibitor, Prevents the Progression of Carotid Atherosclerosis in Patients With Type 2 Diabetes: the Study of Preventive Effects of Alogliptin on Diabetic Atherosclerosis (SPEAD-A). Diabetes Care. 2016;39:139–48.
Mita T, Katakami N, Shiraiwa T, Yoshii H, Onuma T, Kuribayashi N, et al. Sitagliptin Attenuates the Progression of Carotid Intima-Media Thickening in Insulin-Treated Patients With Type 2 Diabetes: the Sitagliptin Preventive Study of Intima-Media Thickness Evaluation (SPIKE): A Randomized Controlled Trial. Diabetes Care. 2016;39:455–64.
Tanaka A, Yoshida H, Nanasato M, Oyama JI, Ishizu T, Ajioka M, et al. Sitagliptin on carotid intima-media thickness in type 2 diabetes patients receiving primary or secondary prevention of cardiovascular disease: a subgroup analysis of the PROLOGUE study. Int J Cardiol. 2018;271:331–5.
Akita K, Isoda K, Shimada K, Daida H. Dipeptidyl-peptidase-4 inhibitor, alogliptin, attenuates arterial inflammation and neointimal formation after injury in low-density lipoprotein (LDL) receptor-deficient mice. J Am Heart Assoc. 2015;4:e001469.
Matsubara J, Sugiyama S, Sugamura K, Nakamura T, Fujiwara Y, Akiyama E, et al. A dipeptidyl peptidase-4 inhibitor, des-fluoro-sitagliptin, improves endothelial function and reduces atherosclerotic lesion formation in apolipoprotein E-deficient mice. J Am Coll Cardiol. 2012;59:265–76.
Ta NN, Schuyler CA, Li Y, Lopes-Virella MF, Huang Y. DPP-4 (CD26) inhibitor alogliptin inhibits atherosclerosis in diabetic apolipoprotein E-deficient mice. J Cardiovasc Pharm. 2011;58:157–66.
Jackson EK, Mi Z, Tofovic SP, Gillespie DG. Effect of dipeptidyl peptidase 4 inhibition on arterial blood pressure is context dependent. Hypertension. 2015;65:238–49.
Ban K, Noyan-Ashraf MH, Hoefer J, Bolz SS, Drucker DJ, Husain M. Cardioprotective and vasodilatory actions of glucagon-like peptide 1 receptor are mediated through both glucagon-like peptide 1 receptor-dependent and -independent pathways. Circulation. 2008;117:2340–50.
Zhang X, Zhao Q. Effects of dipeptidyl peptidase-4 inhibitors on blood pressure in patients with type 2 diabetes: a systematic review and meta-analysis. J Hypertens. 2016;34:167–75.
Nauck MA, Meier JJ, Cavender MA, Abd El Aziz M, Drucker DJ. Cardiovascular Actions and Clinical Outcomes With Glucagon-Like Peptide-1 Receptor Agonists and Dipeptidyl Peptidase-4 Inhibitors. Circulation. 2017;136:849–70.
Vilsbøll T, Christensen M, Junker AE, Knop FK, Gluud LL. Effects of glucagon-like peptide-1 receptor agonists on weight loss: systematic review and meta-analyses of randomised controlled trials. BMJ. 2012;344:d7771.
Drucker DJ. The Cardiovascular Biology of Glucagon-like Peptide-1. Cell Metab. 2016;24:15–30.
Martins FL, Bailey MA, Girardi ACC. Endogenous Activation of Glucagon-Like Peptide-1 Receptor Contributes to Blood Pressure Control: Role of Proximal Tubule Na(+)/H(+) Exchanger Isoform 3, Renal Angiotensin II, and Insulin Sensitivity. Hypertension. 2020;76:839–48.
Sun F, Wu S, Guo S, Yu K, Yang Z, Li L. Impact of GLP-1 receptor agonists on blood pressure, heart rate and hypertension among patients with type 2 diabetes: a systematic review and network meta-analysis. Diabetes Res Clin Pr. 2015;110:26–37.
Wang B, Zhong J, Lin H, Zhao Z, Yan Z, He H, et al. Blood pressure-lowering effects of GLP-1 receptor agonists exenatide and liraglutide: a meta-analysis of clinical trials. Diabetes Obes Metab. 2013;15:737–49.
Holst JJ, Deacon CF, Vilsbøll T, Krarup T, Madsbad S. Glucagon-like peptide-1, glucose homeostasis and diabetes. Trends Mol Med. 2008;14:161–8.
Baker WL, Smyth LR, Riche DM, Bourret EM, Chamberlin KW, White WB. Effects of sodium-glucose co-transporter 2 inhibitors on blood pressure: a systematic review and meta-analysis. J Am Soc Hypertens. 2014;8:262–.e9.
Mazidi M, Rezaie P, Gao HK, Kengne AP. Effect of Sodium-Glucose Cotransport-2 Inhibitors on Blood Pressure in People With Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of 43 Randomized Control Trials With 22 528 Patients. J Am Heart Assoc. 2017;6:e004007.
Baker WL, Buckley LF, Kelly MS, Bucheit JD, Parod ED, Brown R, et al. Effects of Sodium-Glucose Cotransporter 2 Inhibitors on 24-Hour Ambulatory Blood Pressure: a Systematic Review and Meta-Analysis. J Am Heart Assoc. 2017;6:e005686.
Kario K, Okada K, Kato M, Nishizawa M, Yoshida T, Asano T, et al. 24-Hour Blood Pressure-Lowering Effect of an SGLT-2 Inhibitor in Patients with Diabetes and Uncontrolled Nocturnal Hypertension: results from the Randomized, Placebo-Controlled SACRA Study. Circulation. 2018;139:2089–97.
Kario K, Ferdinand KC, O’Keefe JH. Control of 24-hour blood pressure with SGLT2 inhibitors to prevent cardiovascular disease. Prog Cardiovasc Dis. 2020;63:249–62.
Okada K, Hoshide S, Kato M, Kanegae H, Ishibashi S, Kario K. Safety and efficacy of empagliflozin in elderly Japanese patients with type 2 diabetes mellitus: a post hoc analysis of data from the SACRA study. J Clin Hypertens (Greenwich). 2020. https://doi.org/10.1111/jch.14131. Epub ahead of print.
Mancia G, Cannon CP, Tikkanen I, Zeller C, Ley L, Woerle HJ, et al. Impact of Empagliflozin on Blood Pressure in Patients With Type 2 Diabetes Mellitus and Hypertension by Background Antihypertensive Medication. Hypertension. 2016;68:1355–64.
Lingvay I, Catarig AM, Frias JP, Kumar H, Lausvig NL, le Roux CW, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7:834–44.
Sanidas EA, Papadopoulos DP, Hatziagelaki E, Grassos C, Velliou M, Barbetseas J. Sodium Glucose Cotransporter 2 (SGLT2) Inhibitors Across the Spectrum of Hypertension. Am J Hypertens. 2020;33:207–13.
Herat LY, Matthews VB, Magno AL, Kiuchi MG, Carnagarin R, Schlaich MP. An evaluation of empagliflozin and it’s applicability to hypertension as a therapeutic option. Expert Opin Pharmacother. 2020;21:1157–66.
Maliha G, Townsend RR. SGLT2 inhibitors: their potential reduction in blood pressure. J Am Soc Hypertens. 2015;9:48–53.
Kawasoe S, Maruguchi Y, Kajiya S, Uenomachi H, Miyata M, Kawasoe M, et al. Mechanism of the blood pressure-lowering effect of sodium-glucose cotransporter 2 inhibitors in obese patients with type 2 diabetes. BMC Pharm Toxicol. 2017;18:23.
Scheen AJ. Effect of SGLT2 Inhibitors on the Sympathetic Nervous System and Blood Pressure. Curr Cardiol Rep. 2019;21:70.
Lytvyn Y, Bjornstad P, Udell JA, Lovshin JA, Cherney DZI. Sodium Glucose Cotransporter-2 Inhibition in Heart Failure: potential Mechanisms, Clinical Applications, and Summary of Clinical Trials. Circulation. 2017;136:1643–58.
Yoshikawa T, Kishi T, Shinohara K, Takesue K, Shibata R, Sonoda N, et al. Arterial pressure lability is improved by sodium-glucose cotransporter 2 inhibitor in streptozotocin-induced diabetic rats. Hypertens Res. 2017;40:646–51.
Lee MMY, Petrie MC, McMurray JJV, Sattar N. How Do SGLT2 (Sodium-Glucose Cotransporter 2) Inhibitors and GLP-1 (Glucagon-Like Peptide-1) Receptor Agonists Reduce Cardiovascular Outcomes?: Completed and Ongoing Mechanistic Trials. Arterioscler Thromb Vasc Biol. 2020;40:506–22.
Januzzi JL Jr, Xu J, Li J, Shaw W, Oh R, Pfeifer M, et al. Effects of Canagliflozin on Amino-Terminal Pro-B-Type Natriuretic Peptide: Implications for Cardiovascular Risk Reduction. J Am Coll Cardiol. 2020;76:2076–85.
Buse JB, Bain SC, Mann JFE, Nauck MA, Nissen SE, Pocock S, et al. Cardiovascular Risk Reduction With Liraglutide: an Exploratory Mediation Analysis of the LEADER Trial. Diabetes Care. 2020;43:1546–52.
Inzucchi SE, Zinman B, Fitchett D, Wanner C, Ferrannini E, Schumacher M, et al. How Does Empagliflozin Reduce Cardiovascular Mortality? Insights From a Mediation Analysis of the EMPA-REG OUTCOME Trial. Diabetes Care. 2018;41:356–63.
Qiu M, Ding LL, Zhang M, Lin JH, Wei XB, Huang H. GLP-1RAs and SGLT2is Reduce Cardiovascular Events Independent of Reductions of Systolic Blood Pressure and Body Weight: a Meta-Analysis with Meta-Regression. Diabetes Ther. 2020;11:2429–40.
Tanaka A, Node K. How should we monitor the cardiovascular benefit of sodium-glucose cotransporter 2 inhibition? Cardiovasc Diabetol. 2020;19:206.
Horii T, Iwasawa M, Kabeya Y, Atuda K. Polypharmacy and oral antidiabetic treatment for type 2 diabetes characterised by drug class and patient characteristics: a Japanese database analysis. Sci Rep. 2019;9:12992.
Li J, Fagbote CO, Zhuo M, Hawley CE, Paik JM. Sodium-glucose cotransporter 2 inhibitors for diabetic kidney disease: a primer for deprescribing. Clin Kidney J. 2019;12:620–8.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373:2117–28.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AT has received honoraria from Boehringer Ingelheim and research funding from GlaxoSmithKline. KN has received research grants from Asahi Kasei, Astellas, Bayer, Boehringer Ingelheim, Mitsubishi Tanabe, Teijin, and Terumo; scholarships from Astellas, Bayer, Bristol-Myers Squibb, Daiichi Sankyo, Daiichi Sankyo Healthcare, Takeda, and Teijin; and personal fees from Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Daiichi Sankyo Healthcare, Eli Lilly, Kowa, Mitsubishi Tanabe, MSD, Novartis, Ono, Takeda, and Teijin.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanaka, A., Node, K. Hypertension in diabetes care: emerging roles of recent hypoglycemic agents. Hypertens Res 44, 897–905 (2021). https://doi.org/10.1038/s41440-021-00665-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-021-00665-7
Keywords
This article is cited by
-
Blood Pressure-Lowering Effects of SGLT2 Inhibitors and GLP-1 Receptor Agonists
Current Hypertension Reports (2025)
-
2023 update and perspectives
Hypertension Research (2024)
-
Who benefits from the blood pressure-lowering effects of SGLT2 inhibitors in patients with type 2 diabetes mellitus and chronic kidney disease? — Obese or non-obese?
Hypertension Research (2024)
-
Associations of metabolic disorders with hypertension and cardiovascular disease: recent findings and therapeutic perspectives
Hypertension Research (2024)
-
Pretreatment body mass index affects achievement of target blood pressure with sodium-glucose cotransporter 2 inhibitors in patients with type 2 diabetes mellitus and chronic kidney disease
Hypertension Research (2024)