Abstract
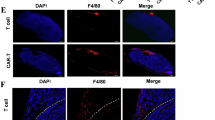
Tumor-associated macrophages are a key component that contributes to the immunosuppressive microenvironment in human cancers. However, therapeutic targeting of macrophages has been a challenge in clinic due to the limited understanding of their heterogeneous subpopulations and distinct functions. Here, we identify a clinically relevant CD19+ subpopulation of macrophages that is enriched in many types of cancer, particularly in hepatocellular carcinoma (HCC). The CD19+ macrophages exhibit increased levels of programmed cell death 1 ligand 1 (PD-L1) and CD73, enhanced mitochondrial oxidation, and compromised phagocytosis, indicating their immunosuppressive functions. Targeting CD19+ macrophages with anti-CD19 chimeric antigen receptor T (CAR-T) cells inhibited HCC tumor growth. We identify Paired Box 5 (PAX5) as a primary driver of up-regulated mitochondrial biogenesis in CD19+ macrophages, which depletes cytoplasmic Ca2+, leading to lysosomal deficiency and consequent accumulation of CD73 and PD-L1. Inhibiting CD73 or mitochondrial oxidation enhanced the efficacy of immune checkpoint blockade therapy in treating HCC, suggesting great promise for CD19+ macrophage-targeting therapeutics.
Similar content being viewed by others
Data availability
The remaining data are available within the Article, Supplementary Information or Source Data file. Source data are provided with this paper. Any additional information required to reanalyze the data reported in this paper is always available from the lead contact upon request (Qi Zhang: qi.zhang@zju.edu.cn). For external requests for additional data or materials required to reproduce the results presented in this manuscript, the requester must submit a formal application stating the research purpose, intended use of the data, and commitment to abide by relevant data protection and ethical guidelines. Human sequencing data were deposited in Genome Sequence Archive (GSA) (https://ngdc.cncb.ac.cn/gsa-human/browse/HRA008143). All processed sequencing data are deposited in the Science Data Bank database, available via https://cstr.cn/31253.11.sciencedb.11557 or CSTR:31253.11.sciencedb.11557. Source data are provided with this paper.
References
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Forner, A., Llovet, J. M. & Bruix, J. Hepatocellular carcinoma. Lancet 379, 1245–1255 (2012).
Gordan, J. D. et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline. J. Clin. Oncol. 38, 4317–4345 (2020).
Vogel, A., Meyer, T., Sapisochin, G., Salem, R. & Saborowski, A. Hepatocellular carcinoma. Lancet 400, 1345–1362 (2022).
Marra, F. & Tacke, F. Roles for chemokines in liver disease. Gastroenterology 147, 577–594 (2014).
Zhang, Q. et al. Integrated multiomic analysis reveals comprehensive tumour heterogeneity and novel immunophenotypic classification in hepatocellular carcinomas. Gut 68, 2019–2031 (2019).
Kalathil, S., Lugade, A. A., Miller, A., Iyer, R. & Thanavala, Y. Higher frequencies of GARP(+)CTLA-4(+)Foxp3(+) T regulatory cells and myeloid-derived suppressor cells in hepatocellular carcinoma patients are associated with impaired T-cell functionality. Cancer Res. 73, 2435–2444 (2013).
Shen, K. Y., Zhu, Y., Xie, S. Z. & Qin, L. X. Immunosuppressive tumor microenvironment and immunotherapy of hepatocellular carcinoma: current status and prospectives. J. Hematol. Oncol. 17, 25 (2024).
Noy, R. & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 24, 541–550 (2018).
Murray, P. J. Macrophage polarization. Annu. Rev. Physiol. 79, 541–566 (2017).
Mantovani, A., Sozzani, S., Locati, M., Allavena, P. & Sica, A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trend Immunol. 23, 549–555 (2002).
DeNardo, D. G. & Ruffell, B. Macrophages as regulators of tumour immunity and immunotherapy. Nat. Rev. Immunol. 19, 369–382 (2019).
Wu, Q. et al. Blocking triggering receptor expressed on myeloid cells-1-positive tumor-associated macrophages induced by hypoxia reverses immunosuppression and anti-programmed cell death ligand 1 resistance in liver cancer. Hepatology 70, 198–214 (2019).
Gordon, S. R. et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 545, 495–499 (2017).
Kim, H. J. et al. Blood monocyte-derived CD169(+) macrophages contribute to antitumor immunity against glioblastoma. Nat. Commun. 13, 6211 (2022).
Wang, R. et al. A novel phenotype of B cells associated with enhanced phagocytic capability and chemotactic function after ischemic stroke. Neural Regen. Res. 18, 2413–2423 (2023).
Kitamura, D., Roes, J., Kühn, R. & Rajewsky, K. A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin mu chain gene. Nature 350, 423–426 (1991).
Allard, B., Allard, D., Buisseret, L. & Stagg, J. The adenosine pathway in immuno-oncology. Nat. Rev. Clin. Oncol. 17, 611–629 (2020).
Junger, W. G. Immune cell regulation by autocrine purinergic signalling. Nat. Rev. Immunol. 11, 201–212 (2011).
Antonioli, L., Pacher, P., Vizi, E. S. & Haskó, G. CD39 and CD73 in immunity and inflammation. Trend Mol. Med. 19, 355–367 (2013).
Zhang, B. CD73: a novel target for cancer immunotherapy. Cancer Res. 70, 6407–6411 (2010).
Vijayan, D., Young, A., Teng, M. W. L. & Smyth, M. J. Targeting immunosuppressive adenosine in cancer. Nat. Rev. Cancer 17, 765 (2017).
Alcedo, K. P., Bowser, J. L. & Snider, N. T. The elegant complexity of mammalian ecto-5’-nucleotidase (CD73). Trend Cell Biol. 31, 829–842 (2021).
Depoil, D. et al. CD19 is essential for B cell activation by promoting B cell receptor-antigen microcluster formation in response to membrane-bound ligand. Nat. Immunol. 9, 63–72 (2008).
Tedder, T. F. CD19: a promising B cell target for rheumatoid arthritis. Nat. Rev. Rheumatol. 5, 572–577 (2009).
Chung, E. Y. et al. CD19 is a major B cell receptor-independent activator of MYC-driven B-lymphomagenesis. J. Clin. Investig. 122, 2257–2266 (2012).
Finck, B. N. Kelly DP. PGC-1 coactivators: inducible regulators of energy metabolism in health and disease. J. Clin. Investig. 116, 615–622 (2006).
Cuervo, A. M., Mann, L., Bonten, E. J., d’Azzo, A. & Dice, J. F. Cathepsin A regulates chaperone-mediated autophagy through cleavage of the lysosomal receptor. EMBO J. 22, 47–59 (2003).
Settembre, C. et al. TFEB links autophagy to lysosomal biogenesis. Science 332, 1429–1433 (2011).
Roczniak-Ferguson, A. et al. The transcription factor TFEB links mTORC1 signaling to transcriptional control of lysosome homeostasis. Sci. Signal 5, ra42 (2012).
Medina, D. L. et al. Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB. Nat. Cell Biol. 17, 288–299 (2015).
Rooijen, N. & Sanders, A. Liposome mediated depletion of macrophages: mechanism of action, preparation of liposomes and applications. J. Immunol. Method 174, 83–93 (1994).
Moreno, S. G. Depleting macrophages in vivo with clodronate-liposomes. Method Mol. Biol. 1784, 259–262 (2018).
Xie, H., Ye, M., Feng, R. & Graf, T. Stepwise reprogramming of B cells into macrophages. Cell 117, 663–676 (2004).
Chen, C. et al. Cancer co-opts differentiation of B-cell precursors into macrophage-like cells. Nat. Commun. 13, 5376 (2022).
Hodawadekar, S. et al. B-Lymphoma cells with epigenetic silencing of Pax5 trans-differentiate into macrophages, but not other hematopoietic lineages. Exp. Cell Res. 313, 331–340 (2007).
Gabrilovich, D. Myeloid-derived suppressor cells. Cancer Immunol. Res. 5, 3–8 (2017).
Veglia, F., Sanseviero, E. & Gabrilovich, D. Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity. Nat. Rev. Immunol. 21, 485–498 (2021).
Ma, X. L. et al. CD73 promotes hepatocellular carcinoma progression and metastasis via activating PI3K/AKT signaling by inducing Rap1-mediated membrane localization of P110β and predicts poor prognosis. J. Hematol. Oncol. 12, 37 (2019).
Supernat, A. et al. CD73 expression as a potential marker of good prognosis in breast carcinoma. Appl. Immunohistochem. Mol. Morphol. 20, 103–107 (2012).
Beavis, P. A., Stagg, J., Darcy, P. K. & Smyth, M. J. CD73: a potent suppressor of antitumor immune responses. Trend Immunol. 33, 231–237 (2012).
Eichin, D., Laurila, J. P., Jalkanen, S. & Salmi, M. CD73 activity is dispensable for the polarization of M2 macrophages. PloS One 10, e0134721 (2015).
Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
The Lancet O CAR T-cell therapy for solid tumours. Lancet Oncol. 22, 893 (2021).
Albelda, S. M. CAR T cell therapy for patients with solid tumours: key lessons to learn and unlearn. Nat. Rev. Clin. Oncol. 21, 47–66 (2024).
Ashton, T. M., McKenna, W. G., Kunz-Schughart, L. A. & Higgins, G. S. Oxidative phosphorylation as an emerging target in cancer therapy. Clin. Cancer Res. 24, 2482–2490 (2018).
Yap, T. A. et al. Complex I inhibitor of oxidative phosphorylation in advanced solid tumors and acute myeloid leukemia: phase I trials. Nat. Med. 29, 115–126 (2023).
Acknowledgements
This work was supported by National Key Research & Development Program (No. 2020YFA0804300), National Natural Science Foundation of China (Nos. 82071865, 82403723, 81871320, 32321002, 82188102, 92359304), Zhejiang Provincial Natural Science Funds (Nos. LR20H160002, HDMD22H319373), Zhejiang Provincial Medical and Health Technology Project (No. WKJ-ZJ-2403), and Zhejiang Provincial Traditional Chinese Medicine Science and Technology Project (No. GZY-ZJ-KJ-23025). We thank Jianfeng Wang from the Zhejiang Provincial Key Laboratory of Pancreatic Disease for sample collection. Dr. Qi Zhang also gratefully acknowledges the support of K.C. Wong Education Foundation.
Author information
Authors and Affiliations
Contributions
T.L. and Q.Z. conceived the project. Q.Z., J.W. and J.S. designed the experiments. Q.Z., J.W., W.C., Y.Z., JQ.Y., JW.Y., J.T., M.Y., Z.H., J.W., H.D., and YQ.Z. performed most of the experiments under the supervision of T.L., X.B., X.L. and P.X. R.Z., Y.L., X.L., and J.H. performed the bioinformatic analysis. Q.Z. and J.W. wrote the manuscript and the other authors made critical revisions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Luca Di Tommaso, Yang Zhao and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, J., Cao, W., Huang, J. et al. Tumor-associated CD19+ macrophages induce immunosuppressive microenvironment in hepatocellular carcinoma. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69638-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69638-z