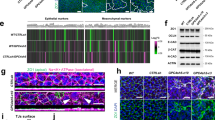
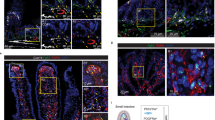
Abstract
The primitive gut tube of mammals initially forms as a simple cylinder consisting of the endoderm-derived, pseudostratified epithelium and the mesoderm-derived surrounding mesenchyme. During mid-gestation, a dramatic transformation occurs in which the epithelium is both restructured into its final cuboidal form and simultaneously folded and refolded to create intestinal villi and intervillus regions. Here, we show that the mesenchymal winged helix transcription factor Foxl1, itself induced by epithelial hedgehog signaling, controls villification by activating BMP and PDGFRα and the planar cell polarity factor Fat4 in epithelial-adjacent telocyte progenitors either directly or indirectly. In the absence of Foxl1-dependent mesenchymal signaling, villus formation and the separation of epithelial cells into mitotic intervillus and postmitotic villus are delayed, and the differentiation of secretory progenitors temporarily blocked. Thus, Foxl1 orchestrates key events during the epithelial transition of the fetal mammalian gut.
Similar content being viewed by others
Data availability
The single-cell RNA sequencing datasets generated in this study are available in the NCBI Gene Expression Omnibus (GEO) under accession number GSE302290. A source data file is provided. All other relevant data supporting the findings of this study are available from the corresponding author upon request. Source data are provided with this paper.
References
Helander, H. F. & Fandriks, L. Surface area of the digestive tract—revisited. Scand. J. Gastroenterol. 49, 681–689 (2014).
Shyer, A. E., Huycke, T. R., Lee, C., Mahadevan, L. & Tabin, C. J. Bending gradients: how the intestinal stem cell gets its home. Cell 161, 569–580 (2015).
Shyer, A. E. et al. Villification: how the gut gets its villi. Science 342, 212–218 (2013).
Walton, K. D., Mishkind, D., Riddle, M. R., Tabin, C. J. & Gumucio, D. L. Blueprint for an intestinal villus: Species-specific assembly required. Wiley Interdiscip. Rev. Dev. Biol. 7, e317 (2018).
Karlsson, L., Lindahl, P., Heath, J. K. & Betsholtz, C. Abnormal gastrointestinal development in PDGF-A and PDGFR-(alpha) deficient mice implicates a novel mesenchymal structure with putative instructive properties in villus morphogenesis. Development 127, 3457–3466 (2000).
Walton, K. D., Freddo, A. M., Wang, S. & Gumucio, D. L. Generation of intestinal surface: an absorbing tale. Development 143, 2261–2272 (2016).
Walton, K. D. et al. Hedgehog-responsive mesenchymal clusters direct patterning and emergence of intestinal villi. Proc. Natl. Acad. Sci. USA 109, 15817–15822 (2012).
Freddo, A. M. et al. Coordination of signaling and tissue mechanics during morphogenesis of murine intestinal villi: a role for mitotic cell rounding. Integr. Biol. 8, 918–928 (2016).
Grosse, A. S. et al. Cell dynamics in fetal intestinal epithelium: implications for intestinal growth and morphogenesis. Development 138, 4423–4432 (2011).
Walton, K. D. et al. Villification in the mouse: Bmp signals control intestinal villus patterning. Development 143, 427–436 (2016).
Huycke, T. R. et al. Patterning and folding of intestinal villi by active mesenchymal dewetting. Cell 187, 3072–3089 e3020 (2024).
Mathan, M., Moxey, P. C. & Trier, J. S. Morphogenesis of fetal rat duodenal villi. Am. J. Anat. 146, 73–92 (1976).
Guiu, J. et al. Tracing the origin of adult intestinal stem cells. Nature 570, 107–111 (2019).
Madison, B. B. et al. Epithelial hedgehog signals pattern the intestinal crypt-villus axis. Development 132, 279–289 (2005).
Kaestner, K. H., Silberg, D. G., Traber, P. G. & Schutz, G. The mesenchymal winged helix transcription factor Fkh6 is required for the control of gastrointestinal proliferation and differentiation. Genes Dev. 11, 1583–1595 (1997).
Kondo, A. & Kaestner, K. H. FoxL1(+) mesenchymal cells are a critical source of Wnt5a for midgut elongation during mouse embryonic intestinal development. Cells Dev. 165, 203662 (2021).
Madison, B. B., McKenna, L. B., Dolson, D., Epstein, D. J. & Kaestner, K. H. FoxF1 and FoxL1 link hedgehog signaling and the control of epithelial proliferation in the developing stomach and intestine. J. Biol. Chem. 284, 5936–5944 (2009).
Kolev, H. M. et al. A FoxL1-CreERT-2A-tdTomato Mouse Labels Subepithelial Telocytes. Cell Mol. Gastroenterol. Hepatol. 12, 1155–1158 e1154 (2021).
Katz, J. P. et al. Foxl1 null mice have abnormal intestinal epithelia, postnatal growth retardation, and defective intestinal glucose uptake. Am. J. Physiol. Gastrointest. Liver Physiol. 287, G856–G864 (2004).
Perreault, N., Katz, J. P., Sackett, S. D. & Kaestner, K. H. Foxl1 controls the Wnt/beta-catenin pathway by modulating the expression of proteoglycans in the gut. J. Biol. Chem. 276, 43328–43333 (2001).
Perreault, N., Sackett, S. D., Katz, J. P., Furth, E. E. & Kaestner, K. H. Foxl1 is a mesenchymal Modifier of Min in carcinogenesis of stomach and colon. Genes Dev. 19, 311–315 (2005).
Bergen, V., Lange, M., Peidli, S., Wolf, F. A. & Theis, F. J. Generalizing RNA velocity to transient cell states through dynamical modeling. Nat. Biotechnol. 38, 1408–1414 (2020).
He, X. C. et al. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat. Genet. 36, 1117–1121 (2004).
Wang, S., Cebrian, C., Schnell, S. & Gumucio, D. L. Radial WNT5A-Guided Post-mitotic Filopodial Pathfinding Is Critical for Midgut Tube Elongation. Dev. Cell 46, 173–188 e173 (2018).
Rao-Bhatia, A. et al. Hedgehog-activated Fat4 and PCP pathways mediate mesenchymal cell clustering and villus formation in gut development. Dev. Cell 52, 647–658 e646 (2020).
Gracz, A. D. et al. Sox4 promotes Atoh1-independent intestinal secretory differentiation toward tuft and enteroendocrine fates. Gastroenterology 155, 1508–1523 e1510 (2018).
Gregorieff, A. et al. The ets-domain transcription factor Spdef promotes maturation of goblet and paneth cells in the intestinal epithelium. Gastroenterology 137, 1333–1345 e1331–1333 (2009).
Katz, J. P. et al. The zinc-finger transcription factor Klf4 is required for terminal differentiation of goblet cells in the colon. Development 129, 2619–2628 (2002).
Motoyama, J. et al. Overlapping and non-overlapping Ptch2 expression with Shh during mouse embryogenesis. Mech. Dev. 78, 81–84 (1998).
Ramalho-Santos, M., Melton, D. A. & McMahon, A. P. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development 127, 2763–2772 (2000).
Hallikas, O. et al. Genome-wide prediction of mammalian enhancers based on analysis of transcription-factor binding affinity. Cell 124, 47–59 (2006).
Maimets, M. et al. Mesenchymal-epithelial crosstalk shapes intestinal regionalisation via Wnt and Shh signalling. Nat. Commun. 13, 715 (2022).
Shoshkes-Carmel, M. et al. Subepithelial telocytes are an important source of Wnts that supports intestinal crypts. Nature 557, 242–246 (2018).
Ormestad, M. et al. Foxf1 and Foxf2 control murine gut development by limiting mesenchymal Wnt signaling and promoting extracellular matrix production. Development 133, 833–843 (2006).
Braet, F., De Zanger, R. & Wisse, E. Drying cells for SEM, AFM and TEM by hexamethyldisilazane: a study on hepatic endothelial cells. J. Microsc. 186, 84–87 (1997).
Aoki, R. et al. Foxl1-expressing mesenchymal cells constitute the intestinal stem cell niche. Cell Mol. Gastroenterol. Hepatol. 2, 175–188 (2016).
Martin M. Cutadapt removes adapter sequences from high-throughput sequencing reads. 2011 17, 3 (2011).
Zheng, G. X. et al. Massively parallel digital transcriptional profiling of single cells. Nat. Commun. 8, 14049 (2017).
Young M. D., Behjati S. SoupX removes ambient RNA contamination from droplet-based single-cell RNA sequencing data. Gigascience 9, (2020).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587 e3529 (2021).
Guo, H. & Li, J. scSorter: assigning cells to known cell types according to marker genes. Genome Biol. 22, 69 (2021).
Kassambara, A. rstatix: Pipe-Friendly. Framework for Basic Statistical Tests, (2023).
La Manno, G. et al. RNA velocity of single cells. Nature 560, 494–498 (2018).
Acknowledgements
We thank members of the Kaestner lab for helpful discussions. This work was supported by NIH grants R37DK053839 and R01DK139049. We thank the UPenn Center for Molecular Studies in Digestive and Liver Diseases (P30 DK050306) for the use of the Molecular Pathology and Imaging Core (MPIC) for tissue processing, the UPenn Diabetes Research Center Functional Genomics Core (P30 DK019125) for help with data analysis, and Yuri Veklich from the Cell & Developmental Biology Microscopy Core in the UPenn Department of Cell Biology for the use of their scanning electron microscope and confocal imaging services.
Author information
Authors and Affiliations
Contributions
G.Z. and K.H.K.—Conceptualization and writing. G.Z., G.R., K.B., M.T., L.C. and D.L.—Methodology. G.Z. and J.S.—Data curation and visualization. K.H.K.—Supervision. K.H.K.—Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhu, G., Rozenberg, G., Lahori, D. et al. Villification of the intestinal epithelium is driven by Foxl1 through activation of PDGFRα and BMPs. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69791-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69791-5