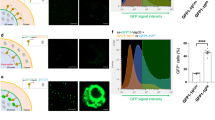
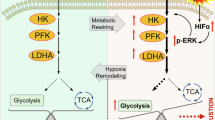
Abstract
Late endosomal secretion is an unconventional secretion mechanism that depends on the SNARE protein VAMP7. We previously showed that VAMP7 mediates the secretion of the ER protein Reticulon3. However, the functional relevance and molecular mechanism of this secretory pathway remain unclear. Here, we show that VAMP7 knockout cells exhibit impaired secretion of ER- and mitochondrial-derived proteins and signs of ER and mitochondrial stress. In addition, pharmacological induction of organellar stress enhances the VAMP7-dependent secretion. We assess the pathophysiological significance of this mechanism using a preclinical glioblastoma model. VAMP7 knockout glioblastoma cells implanted in male rat brain develop into more necrotic tumors with reduced macrophage infiltration compared to controls, suggesting that VAMP7-dependent late endosomal secretion contributes to the tumor microenvironment and affects macrophage infiltration. Together, our results support a model in which late endosomal secretion functions as an organelle quality-control and stress-communication mechanism, with particular relevance to cancer.
Similar content being viewed by others
Data availability
All data associated with this study can be found in the paper, the Supplementary materials, and the Source data file. Research materials are available upon request. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD057152. The RNA sequencing data have been deposited to the GEO repository with the dataset identifier GSE280209 https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE280209. The script for the Sphere plugin has been deposited in Zenodo under the following https://doi.org/10.5281/zenodo.17473585. Source data are provided with this paper.
References
Chiritoiu-Butnaru, M., Stewart, S. E., Zhang, M., Malhotra, V. & Villeneuve, J. Editorial: Unconventional protein secretion: From basic mechanisms to dysregulation in disease. Front. Cell Dev. Biol. 10, 1088002 (2022).
Dimou, E. & Nickel, W. Unconventional mechanisms of eukaryotic protein secretion. Curr. Biol. 28, R406–R410 (2018).
Rabouille, C. Pathways of unconventional protein secretion. Trends Cell Biol. 27, 230–240 (2017).
Chen, R.-H. et al. Beyond the secretory pathway: new insights into protein release. Traffic 26, e70022 (2025).
Galli, T. et al. A novel tetanus neurotoxin-insensitive vesicle-associated membrane protein in SNARE complexes of the apical plasma membrane of epithelial cells. Mol. Biol. Cell 9, 1437–1448 (1998).
Coco, S. et al. Subcellular localization of tetanus neurotoxin-insensitive vesicle-associated membrane protein (VAMP)/VAMP7 in neuronal cells: evidence for a novel membrane compartment. J. Neurosci. 19, 9803–9812 (1999).
Advani, R. J. et al. VAMP-7 mediates vesicular transport from endosomes to lysosomes. J. Cell Biol. 146, 765–776 (1999).
Pols, M. S. et al. hVps41 and VAMP7 function in direct TGN to late endosome transport of lysosomal membrane proteins. Nat. Commun. 4, 1361 (2013).
Filippini, F. et al. Secretion of VGF relies on the interplay between LRRK2 and post-Golgi v-SNAREs. Cell Rep. 42, 112221 (2023).
Filippini, F. et al. Longins: a new evolutionary conserved VAMP family sharing a novel SNARE domain. Trends Biochem. Sci. 26, 407–409 (2001).
Vivona, S. et al. The longin SNARE VAMP7/TI-VAMP adopts a closed conformation. J. Biol. Chem. 285, 17965–17973 (2010).
Martinez-Arca, S., Alberts, P., Zahraoui, A., Louvard, D. & Galli, T. Role of tetanus neurotoxin insensitive vesicle-associated membrane protein (TI-VAMP) in vesicular transport mediating neurite outgrowth. J. Cell Biol. 149, 889–900 (2000).
Martinez-Arca, S. et al. A dual mechanism controlling the localization and function of exocytic v-SNAREs. Proc. Natl. Acad. Sci. USA 100, 9011–9016 (2003).
Alberts, P. et al. Cross talk between tetanus neurotoxin-insensitive vesicle-associated membrane protein-mediated transport and L1-mediated adhesion. Mol. Biol. Cell 14, 4207–4220 (2003).
Kuster, A. et al. The Q-soluble N-Ethylmaleimide-sensitive Factor Attachment Protein Receptor (Q-SNARE) SNAP-47 Regulates Trafficking of Selected Vesicle-associated Membrane Proteins (VAMPs). J. Biol. Chem. 290, 28056–28069 (2015).
D’Souza, Z., Pokrovskaya, I. & Lupashin, V. V. Syntaxin-5’s flexibility in SNARE pairing supports Golgi functions. Traffic 24, 355–379 (2023).
Rao, S. K., Huynh, C., Proux-Gillardeaux, V., Galli, T. & Andrews, N. W. Identification of SNAREs involved in synaptotagmin VII-regulated lysosomal exocytosis. J. Biol. Chem. 279, 20471–20479 (2004).
Proux-Gillardeaux, V., Raposo, G., Irinopoulou, T. & Galli, T. Expression of the Longin domain of TI-VAMP impairs lysosomal secretion and epithelial cell migration. Biol. Cell 99, 261–271 (2007).
Liu, C. et al. Identification of the SNARE complex that mediates the fusion of multivesicular bodies with the plasma membrane in exosome secretion. J. Extracell. Vesicles 12, e12356 (2023).
Verderio, C. et al. TI-VAMP/VAMP7 is the SNARE of secretory lysosomes contributing to ATP secretion from astrocytes. Biol. Cell 104, 213–228 (2012).
Carolina Barbosa, M. et al. A new role of RAB21 and VARP in autophagy and autophagic exocytosis of ATP. Autophagy Rep. 4, 2501365 (2025).
Martinez-Arca, S. et al. A common exocytotic mechanism mediates axonal and dendritic outgrowth. J. Neurosci. 21, 3830–3838 (2001).
Moreau, K., Ravikumar, B., Renna, M., Puri, C. & Rubinsztein, D. C. Autophagosome precursor maturation requires homotypic fusion. Cell 146, 303–317 (2011).
Aoyagi, K. et al. VAMP7 Regulates Autophagosome Formation by Supporting Atg9a Functions in Pancreatic β-Cells From Male Mice. Endocrinology 159, 3674–3688 (2018).
Takáts, S. et al. Autophagosomal Syntaxin17-dependent lysosomal degradation maintains neuronal function in Drosophila. J. Cell Biol. 201, 531–539 (2013).
McLelland, G.-L., Lee, S. A., McBride, H. M. & Fon, E. A. Syntaxin-17 delivers PINK1/parkin-dependent mitochondrial vesicles to the endolysosomal system. J. Cell Biol. 214, 275–291 (2016).
Wojnacki, J. et al. Role of VAMP7-Dependent Secretion of Reticulon 3 in Neurite Growth. Cell Rep. 33, 108536 (2020).
Wang, F. et al. ATG5 provides host protection acting as a switch in the atg8ylation cascade between autophagy and secretion. Dev. Cell 58, 866–884.e8 (2023).
Jerabkova-Roda, K. et al. Peripheral positioning of lysosomes supports melanoma aggressiveness. Nat. Commun. 16, 3375 (2025).
Domingues, N. et al. Oxidized cholesteryl ester induces exocytosis of dysfunctional lysosomes in lipidotic macrophages. Traffic 24, 284–307 (2023).
Fernandez-Mosquera, L. et al. Mitochondrial respiratory chain deficiency inhibits lysosomal hydrolysis. Autophagy 15, 1572–1591 (2019).
Ebner, M. et al. Nutrient-regulated control of lysosome function by signaling lipid conversion. Cell 186, 5328–5346.e26 (2023).
Johnson, D. E., Ostrowski, P., Jaumouillé, V. & Grinstein, S. The position of lysosomes within the cell determines their luminal pH. J. Cell Biol. 212, 677–692 (2016).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Hetz, C., Zhang, K. & Kaufman, R. J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 21, 421–438 (2020).
Lei, Y. et al. Molecular mechanism of ATF6 in unfolded protein response and its role in disease. Heliyon 10, e25937 (2024).
Costa-Mattioli, M. & Walter, P. The integrated stress response: From mechanism to disease. Science 368, 6489 (2020).
Xu, F. et al. COPII mitigates ER stress by promoting formation of ER whorls. Cell Res 31, 141–156 (2021).
Aoyagi, K. et al. VAMP7 Regulates Autophagy to Maintain Mitochondrial Homeostasis and to Control Insulin Secretion in Pancreatic β-Cells. Diabetes 65, 1648–1659 (2016).
Stephenson, L. M. et al. Identification of Atg5-dependent transcriptional changes and increases in mitochondrial mass in Atg5-deficient T lymphocytes. Autophagy 5, 625–635 (2009).
Plitzko, B. & Loesgen, S. Measurement of oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) in culture cells for assessment of the energy metabolism. Bio Protoc 8, 1–13 (2018).
Chaudhry, A., Shi, R. & Luciani, D. S. A pipeline for multidimensional confocal analysis of mitochondrial morphology, function, and dynamics in pancreatic β-cells. Am. J. Physiol. Endocrinol. Metab. 318, E87–E101 (2020).
Chen, Y.-J., Williams, J. M., Arvan, P. & Tsai, B. Reticulon protects the integrity of the ER membrane during ER escape of large macromolecular protein complexes. J. Cell Biol. 219, e201908182 (2020).
Grumati, P. et al. Full length RTN3 regulates turnover of tubular endoplasmic reticulum via selective autophagy. eLife 6, e25555 (2017).
Di Scala, F. et al. Tissue specificity and regulation of the N-terminal diversity of reticulon 3. Biochem. J. 385, 125–134 (2005).
Kitani, S., Berenstein, E., Mergenhagen, S., Tempst, P. & Siraganian, R. P. A cell surface glycoprotein of rat basophilic leukemia cells close to the high affinity IgE receptor (Fc epsilon RI). Similarity to human melanoma differentiation antigen ME491. J. Biol. Chem. 266, 1903–ME1909 (1991).
Van Deun, J. et al. The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J. Extracell. Vesicles 3, v3.24858 (2014).
Pols, M. S. & Klumperman, J. Trafficking and function of the tetraspanin CD63. Exp. Cell Res. 315, 1584–CD1592 (2009).
Freeze, H. H. & Kranz, C. Endoglycosidase and glycoamidase release of N-linked glycans. Curr. Protoc. Mol. Biol. Chapter 17, Unit 17.13A (2010).
Cao, L. et al. Global site-specific analysis of glycoprotein N-glycan processing. Nat. Protoc. 13, 1196–1212 (2018).
Burgo, A. et al. A molecular network for the transport of the TI-VAMP/VAMP7 vesicles from cell center to periphery. Dev. Cell 23, 166–180 (2012).
Chaineau, M., Danglot, L. & Galli, T. Multiple roles of the vesicular-SNARE TI-VAMP in post-Golgi and endosomal trafficking. FEBS Lett. 583, 3817–3826 (2009).
Burgo, A. et al. Role of Varp, a Rab21 exchange factor and TI-VAMP/VAMP7 partner, in neurite growth. EMBO Rep. 10, 1117–1124 (2009).
Nüchel, J. et al. An mTORC1-GRASP55 signaling axis controls unconventional secretion to reshape the extracellular proteome upon stress. Mol. Cell 81, 3275–3293.e12 (2021).
Li, J., Yu, S., Rao, M. & Cheng, B. Tumor-derived extracellular vesicles: key drivers of immunomodulation in breast cancer. Front. Immunol. 16, 1548535 (2025).
Cubillos-Ruiz, J. R., Bettigole, S. E. & Glimcher, L. H. Tumorigenic and immunosuppressive effects of endoplasmic reticulum stress in cancer. Cell 168, 692–706 (2017).
Ghazi, B. et al. Decoding the secret of extracellular vesicles in the immune tumor microenvironment of the glioblastoma: on the border of kingdoms. Front. Immunol. 15, 1423232 (2024).
Lhomond, S. et al. Dual IRE1 RNase functions dictate glioblastoma development. EMBO Mol. Med. 10, e7929 (2018).
Aas, A. T., Brun, A., Blennow, C. & Strömblad, S. & Salford, L. G. The RG2 rat glioma model. J. Neurooncol 23, 175–183 (1995).
Bayona, C., Ranđelović, T. & Ochoa, I. Tumor Microenvironment in Glioblastoma: The Central Role of the Hypoxic-Necrotic Core. Cancer Lett. 218216. https://doi.org/10.1016/j.canlet.2025.218216 (2025).
Marsh, T. et al. Autophagic Degradation of NBR1 Restricts Metastatic Outgrowth during Mammary Tumor Progression. Dev. Cell 52, 591–604.e6 (2020).
Wang, J.-B. et al. Targeting mitochondrial glutaminase activity inhibits oncogenic transformation. Cancer Cell 18, 207–219 (2010).
Loi, M., Raimondi, A., Morone, D. & Molinari, M. ESCRT-III-driven piecemeal micro-ER-phagy remodels the ER during recovery from ER stress. Nat. Commun. 10, 5058 (2019).
Flannery, A. R., Czibener, C. & Andrews, N. W. Palmitoylation-dependent association with CD63 targets the Ca2+ sensor synaptotagmin VII to lysosomes. J. Cell Biol. 191, 599–613 (2010).
Hesketh, G. G. et al. The GATOR-Rag GTPase pathway inhibits mTORC1 activation by lysosome-derived amino acids. Science 370, 351–356 (2020).
Kimura, T. et al. Cellular and molecular mechanism for secretory autophagy. Autophagy 13, 1084–1085 (2017).
Palmulli, R. et al. CD63 sorts cholesterol into endosomes for storage and distribution via exosomes. Nat. Cell Biol. 26, 1093–1109 (2024).
Maruyama, R., Kamoshida, Y., Shimizu, M., Inoue, J. & Sato, R. ATF6α stimulates cholesterogenic gene expression and de novo cholesterol synthesis. Biosci. Biotechnol. Biochem. 77, 1734–1738 (2013).
Zeng, L. et al. ATF6 modulates SREBP2-mediated lipogenesis. EMBO J. 23, 950–958 (2004).
Fusakio, M. E. et al. Transcription factor ATF4 directs basal and stress-induced gene expression in the unfolded protein response and cholesterol metabolism in the liver. Mol. Biol. Cell 27, 1536–1551 (2016).
D’Acunzo, P. et al. Mitovesicles are a novel population of extracellular vesicles of mitochondrial origin altered in Down syndrome. Sci. Adv. 7, eabe5085 (2021).
Song, M. S. et al. Tubular ER structures shaped by ER-phagy receptors engage in stress-induced Golgi bypass. Dev. Cell https://doi.org/10.1016/j.devcel.2025.01.011 (2025).
Choi, H. et al. Secretome translation shaped by lysosomes and lunapark-marked ER junctions. Nature https://doi.org/10.1038/s41586-025-09718-0 (2025).
D’Esposito, M. et al. A synaptobrevin-like gene in the Xq28 pseudoautosomal region undergoes X inactivation. Nat. Genet. 13, 227–229 (1996).
Tannour-Louet, M. et al. Increased gene copy number of VAMP7 disrupts human male urogenital development through altered estrogen action. Nat. Med. 20, 715–724 (2014).
Molino, D. et al. Role of tetanus neurotoxin insensitive vesicle-associated membrane protein in membrane domains transport and homeostasis. Cell. Logist. 5, e1025182 (2015).
Obacz, J. et al. Endoplasmic reticulum proteostasis in glioblastoma-From molecular mechanisms to therapeutic perspectives. Sci. Signal. 10, 420 (2017).
Wculek, S. K., Dunphy, G., Heras-Murillo, I., Mastrangelo, A. & Sancho, D. Metabolism of tissue macrophages in homeostasis and pathology. Cell. Mol. Immunol. 19, 384–408 (2022).
Da Graça, J., Delevoye, C. & Morel, E. Morphodynamical adaptation of the endolysosomal system to stress. FEBS J. 292, 248–260 (2025).
Wang, G. et al. Biomechanical Control of Lysosomal Secretion Via the VAMP7 Hub: A Tug-of-War between VARP and LRRK1. iScience 4, 127–143 (2018).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Konstantakos, V., Nentidis, A., Krithara, A. & Paliouras, G. CRISPR-Cas9 gRNA efficiency prediction: an overview of predictive tools and the role of deep learning. Nucleic Acids Res 50, 3616–3637 (2022).
Demichev, V., Messner, C. B., Vernardis, S. I., Lilley, K. S. & Ralser, M. DIA-NN: neural networks and interference correction enable deep proteome coverage in high throughput. Nat. Methods 17, 41–44 (2020).
Tyanova, S. et al. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 13, 731–740 (2016).
Perez-Riverol, Y. et al. The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res 50, D543–D552 (2022).
Kim, H.-N. et al. Estrogens decrease osteoclast number by attenuating mitochondria oxidative phosphorylation and ATP production in early osteoclast precursors. Sci. Rep. 10, 11933 (2020).
Domingues, N. et al. Connexin43 promotes exocytosis of damaged lysosomes through actin remodelling. EMBO J. 43, 3627–3649 (2024).
Yambire, K. F. et al. Impaired lysosomal acidification triggers iron deficiency and inflammation in vivo. eLife 8, e51031 (2019).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
de Chaumont, F. et al. Icy: an open bioimage informatics platform for extended reproducible research. Nat. Methods 9, 690–696 (2012).
Bolte, S. & Cordelières, F. P. A guided tour into subcellular colocalization analysis in light microscopy. J. Microsc. 224, 213–232 (2006).
Delevoye, C. et al. BLOC-1 Brings Together the Actin and Microtubule Cytoskeletons to Generate Recycling Endosomes. Curr. Biol. 26, 1–13 (2016).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet j. 17, 10 (2011).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Anders, S., Pyl, P. T. & Huber, W. HTSeq — a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Ge, S. X., Son, E. W. & Yao, R. iDEP: an integrated web application for differential expression and pathway analysis of RNA-Seq data. BMC Bioinforma. 19, 534 (2018).
Tang, D. et al. SRplot: A free online platform for data visualization and graphing. PLoS ONE 18, e0294236 (2023).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Le Reste, P.-J. et al. Integration of Raman spectra with transcriptome data in glioblastoma multiforme defines tumour subtypes and predicts patient outcome. J. Cell. Mol. Med. 25, 10846–10856 (2021).
Etcheverry, A. et al. DNA methylation in glioblastoma: impact on gene expression and clinical outcome. BMC Genomics 11, 701 (2010).
Xiang, Y., Zhang, C.-Q. & Huang, K. Predicting glioblastoma prognosis networks using weighted gene co-expression network analysis on TCGA data. BMC Bioinforma. 13, S12 (2012).
Bowman, R. L., Wang, Q., Carro, A., Verhaak, R. G. W. & Squatrito, M. GlioVis data portal for visualization and analysis of brain tumor expression datasets. Neuro Oncol. 19, 139–141 (2017).
Acknowledgements
We thank all members of the Galli team for their assistance and discussions. Work in our group was funded by grants from the French National Research Agency (MetDePaDi ANR-16-CE16-0012, GlioUPS ANR-23-CE16-0035-01), Institut National du Cancer (PLBIO 2018-149), Fondation pour la Recherche Médicale (FRM) Labellisation to TG and postdoctoral fellowship (SPF202110014120) to SV, Fondation de France (grant #00096652), FRM grant (MND202310019903), and Fondation Bettencourt Schueller (Coup d’Elan) to TG. We would like to thank the NeurImag Imaging core Facility team (part of IPNP, Inserm U. 1266 and Université Paris Cité) and member of the national infrastructure France-BioImaging supported by the French National Research Agency (ANR-10-INBS-04) for their technical and scientific support. Our lab is part of the DIM C-BRAINS, funded by the Conseil Régional d’Ile-de-France. NR acknowledges Four Diamonds Research Foundation, FCT 2022.09311.PTDC, 2022.04407.PTDC, FCT ERC-Portugal. This project also received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement MIA-Portugal No 857524 and the Comissão de Coordenação e Desenvolvimento Regional do Centro - CCDRC through the Centro2020 Programme. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. We thank the Leducq establishment for funding the Leica SP8 Confocal/STED 3DX system and Sésame Région Ile-de-France for funding the Zeiss 880 Confocal/Airyscan system. The Seahorse XF analyzer was funded by the DIM Cerveau et Pensée 2015 (Neuroflux) grant to DZ. We thank the BIOSIT H2P2 platform for immunohistochemistry, in particular Gevorg Ghukasyan (https://biosit.univ-rennes1.fr/). Sequencing was performed by the GenomEast platform, a member of the ‘France Génomique’ consortium (ANR-10-INBS-0009). This work was supported by the Agence Nationale de la Recherche ANR “MOBIDIC” (ANR-23-CE14-0041-02) to C.D. This project was also supported by funds from the Fondation pour la Recherche Médicale (FRM, EQU202403018041 and MAT202211016240), INCa PLBio2020, Olicogcyte Bretagne, INSERM (International Research Project – TUPRIC) to EC. TG and NR received the Mariano Gago Prize from the French Academy of Sciences for the project that led to this publication. We would like to extend special thanks to Sébastien Nola for contributing the IDH1/2 data. Illustrations were created with BioRender.
Author information
Authors and Affiliations
Contributions
T.G. and S.V. designed the study and prepared the original draft of the manuscript. S.V. performed and analyzed most of the experiments. P.D. performed and analyzed mitochondrial and autophagy experiments in NRK cells. Q.L. assisted with the manuscript revision and conducted studies on the impact of glycosylation on CD63 interactions. R.P. performed all the IHC for the tumors. L.T. performed the injections into the rat brains and carried out post-procedure care. J.L. and I.C.G. performed and analyzed the proteomic studies. B.C. assisted in the animal experiments. J.B.M. and D.Z. carried out and analyzed the respirometry studies in RG2 cells. J.W. generated the KOs in the N.R.K.; C.K. analyzed the RNA sequencing data. PB developed a plugin for analyzing CD63 distribution. S.F. and N.D. assisted PD in data analysis. L.D. performed the super-resolution STED imaging. C.D. performed the EM imaging and assisted in the EM data analysis. E.C. performed the human cohort analysis, supervised the animal experiments and their data analysis, and edited the manuscript. N.R. supervised the mitochondrial studies, edited the manuscript, and generated funding. T.G. conceptualized the study, managed the project, supervised the data analysis and writing of the manuscript, and generated funding. All authors reviewed the results and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vats, S., Dionisio, P., Lemercier, Q. et al. VAMP7-dependent late endosomal secretion of ER and mitochondrial proteins impacts the tumor microenvironment and macrophage engagement. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69900-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69900-4