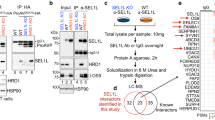
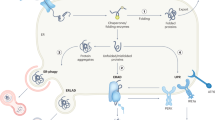
Abstract
Proteolytic cleavage of proglucagon by prohormone convertase 2 (PC2) is required for islet α cells to generate glucagon. However, the regulatory mechanisms underlying this process remain largely unclear. Here, we report that SEL1L-HRD1 endoplasmic reticulum (ER)-associated degradation (ERAD), a highly conserved protein quality control system responsible for clearing misfolded proteins from the ER, plays a key role in glucagon production by regulating turnover of the nascent proform of the PC2 enzyme (proPC2). Using a mouse model with SEL1L deletion in proglucagon-expressing cells, we observe a progressive decline in stimulated glucagon secretion and a reduction in pancreatic glucagon content. Mechanistically, we find that endogenous proPC2 is a substrate of SEL1L-HRD1 ERAD, and that degradation of misfolded proPC2 ensures the maturation of activation-competent proPC2 protein in the ER. Here, we identify ERAD as a regulator of PC2 biology and an essential mechanism for maintaining α cell function.
Similar content being viewed by others
Data availability
The materials and reagents used are either commercially available or are available upon request. All data and materials for the manuscript are described in Methods. Values for all datapoints in graphs and full blots are reported in the Source Data file. Source data are provided with this paper.
References
Unger, R. H. & Cherrington, A. D. Glucagonocentric restructuring of diabetes: a pathophysiologic and therapeutic makeover. J. Clin. Invest. 122, 4–12 (2012).
Finan, B., Capozzi, M. E. & Campbell, J. E. Repositioning glucagon action in the physiology and pharmacology of diabetes. Diabetes 69, 532–541 (2020).
Unger, R. H. & Orci, L. Physiology and pathophysiology of glucagon. Physiol. Rev. 56, 778–826 (1976).
Dinneen, S., Alzaid, A., Turk, D. & Rizza, R. Failure of glucagon suppression contributes to postprandial hyperglycaemia in IDDM. Diabetologia 38, 337–343 (1995).
Pettus, J. et al. Effect of a glucagon receptor antibody (REMD-477) in type 1 diabetes: a randomized controlled trial. Diab Obes. Metab. 20, 1302–1305 (2018).
Pettus, J. et al. Glucagon receptor antagonist volagidemab in type 1 diabetes: a 12-week, randomized, double-blind, phase 2 trial. Nat. Med 28, 2092–2099 (2022).
Wei, R. et al. Antagonistic glucagon receptor antibody promotes α-cell proliferation and increases β-cell mass in diabetic mice. Iscience 16, 326–339 (2019).
Hayashi, Y. et al. Mice deficient for glucagon gene-derived peptides display normoglycemia and hyperplasia of islet {alpha}-cells but not of intestinal L-cells. Mol. Endocrinol. 23, 1990–1999 (2009).
Gelling, R. W. et al. Lower blood glucose, hyperglucagonemia, and pancreatic alpha cell hyperplasia in glucagon receptor knockout mice. Proc. Natl. Acad. Sci. USA. 100, 1438–1443 (2003).
Svendsen, B. et al. Insulin secretion depends on intra-islet glucagon signaling. CellReports 25, 1127–1134.e1122 (2018).
Capozzi, M. E. et al. β cell tone is defined by proglucagon peptides through cAMP signaling. JCI Insight 4, 66 (2019).
El, K., Capozzi, M. E. & Campbell, J. E. Repositioning the Alpha Cell in Postprandial Metabolism. Endocrinology 161 https://doi.org/10.1210/endocr/bqaa169 (2020).
Sandoval, D. A. & D’Alessio, D. A. Physiology of proglucagon peptides: role of glucagon and GLP-1 in health and disease. Physiological Rev. 95, 513–548 (2015).
Muller, L., Cameron, A., Fortenberry, Y., Apletalina, E. V. & Lindberg, I. Processing and sorting of the prohormone convertase 2 propeptide. J. Biol. Chem. 275, 39213–39222 (2000).
Lamango, N. S., Apletalina, E., Liu, J. & Lindberg, I. The proteolytic maturation of prohormone convertase 2 (PC2) is a pH-driven process. Arch. Biochem Biophys. 362, 275–282 (1999).
Riahi, Y., Israeli, T., Cerasi, E. & Leibowitz, G. Effects of proinsulin misfolding on β-cell dynamics, differentiation and function in diabetes. Diab Obes. Metab. 20, 95–103 (2018).
Sokolowski, E. K. et al. Multi-omic human pancreatic islet endoplasmic reticulum and cytokine stress response mapping provides type 2 diabetes genetic insights. Cell Metab. 36, 2468–2488. e2467 (2024).
Eizirik, D. L., Cardozo, A. K. & Cnop, M. The role for endoplasmic reticulum stress in diabetes mellitus. Endocr. Rev. 29, 42–61 (2008).
Shrestha, N., Franco, E. D., Arvan, P. & Cnop, M. Pathological β-cell endoplasmic reticulum stress in type 2 diabetes: current evidence. Front. Endocrinol. 12, 650158 (2021).
Shrestha, N., Reinert, R. B. & Qi, L. Endoplasmic reticulum protein quality control in β cells. Semin. Cell Dev. Biol. 103, 59–67 (2020).
Christianson, J. C., Jarosch, E. & Sommer, T. Mechanisms of substrate processing during ER-associated protein degradation. Nat. Rev. Mol. Cell Biol. 24, 777–796 (2023).
Krshnan, L., van de Weijer, M. L. & Carvalho, P. Endoplasmic Reticulum-Associated Protein Degradation. Cold Spring Harb Perspect Biol 14 https://doi.org/10.1101/cshperspect.a041247 (2022).
Qi, L., Tsai, B. & Arvan, P. New Insights into the Physiological Role of Endoplasmic Reticulum-Associated Degradation. Trends cell Biol. 27, 430–440 (2017).
Sun, S. et al. Sel1L is indispensable for mammalian endoplasmic reticulum-associated degradation, endoplasmic reticulum homeostasis, and survival. Proc. Natl. Acad. Sci. 111, E582–E591 (2014).
Gardner, R. G. et al. Endoplasmic reticulum degradation requires lumen to cytosol signaling: Transmembrane control of Hrd1p by Hrd3p. J. Cell Biol. 151, 69–82 (2000).
Lilley, B. N. & Ploegh, H. L. Multiprotein complexes that link dislocation, ubiquitination, and extraction of misfolded proteins from the endoplasmic reticulum membrane. Proc. Natl. Acad. Sci. USA 102, 14296–14301 (2005).
Lin, L. L. et al. SEL1L-HRD1 interaction is required to form a functional HRD1 ERAD complex. Nat. Commun. 15, 1440 (2024).
Shi, G. et al. ER-associated degradation is required for vasopressin prohormone processing and systemic water homeostasis. J. Clin. Invest. 127, 556–564 (2017).
Kim, G. H. et al. Hypothalamic ER-associated degradation regulates POMC maturation, feeding, and age-associated obesity. J. Clin. Invest. 128, 1125–1140 (2018).
Shrestha, N. et al. Sel1L-Hrd1 ER-associated degradation maintains β cell identity via TGF-β signaling. J. Clin. Invest. 130, 3499–3510 (2020).
Shrestha, N. et al. Integration of ER protein quality control mechanisms defines beta cell function and ER architecture. J. Clin. Invest 133, e163584 (2023).
Hu, Y. et al. Endoplasmic reticulum-associated degradation (ERAD) has a critical role in supporting glucose-stimulated insulin secretion in pancreatic beta-cells. Diabetes 68, 733–746 (2019).
Wu, T. et al. HRD1, an Important Player in Pancreatic beta-Cell Failure and Therapeutic Target for Type 2 Diabetic Mice. Diabetes 69, 940–953 (2020).
Chambers, A. P. et al. The role of pancreatic preproglucagon in glucose homeostasis in mice. Cell Metab. 25, 927–934.e923 (2017).
Dean, E. D. et al. Interrupted glucagon signaling reveals hepatic α cell axis and role for L-glutamine in α cell proliferation. Cell Metab. 25, 1362–1373.e1365 (2017).
Kim, J. et al. Amino acid transporter slc38a5 controls glucagon receptor inhibition-induced pancreatic α cell hyperplasia in mice. Cell Metab. 25, 1348–1361.e1348 (2017).
Shiota, C. et al. Gcg (CreERT2) knockin mice as a tool for genetic manipulation in pancreatic alpha cells. Diabetologia 13, 126–110 (2017).
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001).
Muller, L., Zhu, X. & Lindberg, I. Mechanism of the Facilitation of PC2 Maturation by 7B2: Involvement in ProPC2 Transport and Activation but Not Folding. J. Cell Biol. 139, 625–638 (1997).
Furuta, M. et al. Defective prohormone processing and altered pancreatic islet morphology in mice lacking active SPC2. Proc. Natl. Acad. Sci. 94, 6646–6651 (1997).
Lee, S.-N. & Lindberg, I. 7B2 prevents unfolding and aggregation of prohormone convertase 2. Endocrinology 149, 4116–4127 (2008).
Furuta, M. et al. Severe defect in proglucagon processing in islet A-cells of prohormone convertase 2 null mice. J. Biol. Chem. 276, 27197–27202 (2001).
Helwig, M. et al. Dynamic modulation of prohormone convertase 2 (PC2)-mediated precursor processing by 7B2 protein: preferential effect on glucagon synthesis. J. Biol. Chem. 286, 42504–42513 (2011).
Li, Q.-L. et al. Prohormone convertase 2 enzymatic activity and its regulation in neuro-endocrine cells and tissues. Regulatory Pept. 110, 197–205 (2003).
Zuber, C., Fan, J.-Y., Guhl, B. & Roth, J. Misfolded proinsulin accumulates in expanded pre-Golgi intermediates and endoplasmic reticulum subdomains in pancreatic beta cells of Akita mice. FASEB J. 18, 917–919 (2004).
Marchetti, P. et al. The endoplasmic reticulum in pancreatic beta cells of type 2 diabetes patients. Diabetologia 50, 2486–2494 (2007).
Jacquemyn, J., Cascalho, A. & Goodchild, R. E. The ins and outs of endoplasmic reticulum-controlled lipid biosynthesis. EMBO Rep. 18, 1905–1921 (2017).
Longuet, C. et al. Liver-specific disruption of the murine glucagon receptor produces α-cell hyperplasia: evidence for a circulating α-cell growth factor. Diabetes 62, 1196–1205 (2013).
Larger, E. et al. Pancreatic alpha-cell hyperplasia and hyperglucagonemia due to a glucagon receptor splice mutation. Endocrinol. Diab Metab. Case Rep. 2016, 16–0081 (2016).
Kostic, A. et al. A first-in-human pharmacodynamic and pharmacokinetic study of a fully human anti-glucagon receptor monoclonal antibody in normal healthy volunteers. Diab Obes. Metab. 20, 283–291 (2018).
Marroqui, L. et al. Pancreatic α cells are resistant to metabolic stress-induced apoptosis in type 2 diabetes. EBioMedicine 2, 378–385 (2015).
Leite, N. C. et al. Modeling type 1 diabetes in vitro using human pluripotent stem cells. Cell Rep. 32, 107894 (2020).
Bosi, E., Marchetti, P., Rutter, G. A. & Eizirik, D. L. Human alpha cell transcriptomic signatures of types 1 and 2 diabetes highlight disease-specific dysfunction pathways. Iscience 25, 105056 (2022).
Zhang, P. et al. The PERK eukaryotic initiation factor 2 alpha kinase is required for the development of the skeletal system, postnatal growth, and the function and viability of the pancreas. Mol. Cell. Biol. 22, 3864–3874 (2002).
Akiyama, M. et al. X-box binding protein 1 is essential for insulin regulation of pancreatic α-cell function. Diabetes 62, 2439–2449 (2013).
Bhattacharya, A. & Qi, L. ER-associated degradation in health and disease - from substrate to organism. J. Cell. Sci. 132 jcs232850 (2019).
Bozadjieva, N. et al. Loss of mTORC1 signaling alters pancreatic α cell mass and impairs glucagon secretion. J. Clin. Invest. 127, 4379–4393 (2017).
Shiota, C. et al. α-Cells are dispensable in postnatal morphogenesis and maturation of mouse pancreatic islets. Am. J. Physiol. Endocrinol. Metab. 305, E1030–E1040 (2013).
Kim, K.-S., Seeley, R. J. & Sandoval, D. A. Signalling from the periphery to the brain that regulates energy homeostasis. Nat. Rev. Neurosci. 19, 185–196 (2018).
Torres, M. et al. Purkinje cell-specific deficiency in SEL1L-hrd1 endoplasmic reticulum-associated degradation causes progressive cerebellar ataxia in mice. JCI Insight https://doi.org/10.1172/jci.insight.174725 (2024).
Zhu, X. & Lindberg, I. 7B2 facilitates the maturation of proPC2 in neuroendocrine cells and is required for the expression of enzymatic activity. J. Cell Biol. 129, 1641–1650 (1995).
Westphal, C. H. et al. The neuroendocrine protein 7B2 is required for peptide hormone processing in vivo and provides a novel mechanism for pituitary cushing’s disease. Cell 96, 689–700 (1999).
Leak, T. S. et al. Association of the proprotein convertase subtilisin/kexin-type 2 (PCSK2) gene with type 2 diabetes in an African American population. Mol. Genet Metab. 92, 145–150 (2007).
Zheng, X. et al. Association of type 2 diabetes susceptibility genes (TCF7L2, SLC30A8, PCSK1 and PCSK2) and proinsulin conversion in a Chinese population. Mol. Biol. Rep. 39, 17–23 (2012).
Chang, T. J. et al. Genetic polymorphisms of PCSK2 are associated with glucose homeostasis and progression to type 2 diabetes in a Chinese population. Sci. Rep. 5, 14380 (2015).
Winters, A. et al. Functional analysis of PCSK2 coding variants: A founder effect in the Old Order Amish population. Diab Res. Clin. Pract. 131, 82–90 (2017).
Campbell, S. A. et al. Human islets contain a subpopulation of glucagon-like peptide-1 secreting α cells that is increased in type 2 diabetes. Mol. Metab., 39, 101014 (2020).
Holter, M. M., Saikia, M. & Cummings, B. P. Alpha-cell paracrine signaling in the regulation of beta-cell insulin secretion. Front. Endocrinol. 13, 934775 (2022).
Teitelman, G. A controversy regarding the identity of the enzyme that mediates glucagon-like peptide 1 synthesis in human alpha cells. J. Histochem. Cytochem. 72, 545–550 (2024).
Rouille, Y., Bianchi, M., Irminger, J. C. & Halban, P. A. Role of the prohormone convertase PC2 in the processing of proglucagon to glucagon. Febs Lett. 413, 119–123 (1997).
Thorel, F. et al. Normal glucagon signaling and β-cell function after near-total α-cell ablation in adult mice. Diabetes 60, 2872–2882 (2011).
Bonnevie-Nielsen, V. Experimental diets affect pancreatic insulin and glucagon differently in male and female mice. Metabolism 29, 386–391 (1980).
Jo, S. et al. Sex differences in pancreatic β-cell physiology and glucose homeostasis in C57BL/6J Mice. J. Endocr. Soc. 7, bvad099 (2023).
Ling, Z. et al. Glucagon-like peptide 1 receptor signaling influences topography of islet cells in mice. Virchows Arch. 438, 382–387 (2001).
Wei, X. et al. Proteomic screens of SEL1L-HRD1 ER-associated degradation substrates reveal its role in glycosylphosphatidylinositol-anchored protein biogenesis. Nat. Commun. 15, 659 (2024).
Wang, H. H. et al. Hypomorphic variants of SEL1L-HRD1 ER-associated degradation are associated with neurodevelopmental disorders. J. Clin. Investig. 134, e170054 (2023).
Kaestner, K. H., Powers, A. C., Naji, A., Consortium, H. & Atkinson, M. A. NIH initiative to improve understanding of the pancreas, islet, and autoimmunity in type 1 diabetes: the human pancreas analysis program (HPAP). Diabetes 68, 1394–1402 (2019).
Shapira, S. N., Naji, A., Atkinson, M. A., Powers, A. C. & Kaestner, K. H. Understanding islet dysfunction in type 2 diabetes through multidimensional pancreatic phenotyping: the human pancreas analysis program. Cell Metab. 34, 1906–1913 (2022).
Brissova, M. et al. The integrated islet distribution program answers the call for improved human islet phenotyping and reporting of human islet characteristics in research articles. Diabetes 68, 1363–1365 (2019).
Kong, Y., Ebrahimpour, P., Liu, Y., Yang, C. & Alonso, L. C. Pancreatic islet embedding for paraffin sections. J. Vis. Exp. https://doi.org/10.3791/57931 (2018).
Dai, C. et al. Islet-enriched gene expression and glucose-induced insulin secretion in human and mouse islets. Diabetologia 55, 707–718 (2012).
Brissova, M. et al. Intraislet endothelial cells contribute to revascularization of transplanted pancreatic islets. Diabetes 53, 1318–1325 (2004).
Reinert, R. B. et al. Vascular endothelial growth factor coordinates islet innervation via vascular scaffolding. Development 141, 1480–1491 (2014).
Scopsi, L., Gullo, M., Rilke, F., Martin, S. & Steiner, D. F. Proprotein convertases (PC1/PC3 and PC2) in normal and neoplastic human tissues: their use as markers of neuroendocrine differentiation. J. Clin. Endocrinol. Metab. 80, 294–301 (1995).
Zhou, A., Martin, S., Lipkind, G., LaMendola, J. & Steiner, D. F. Regulatory roles of the P domain of the subtilisin-like prohormone convertases. J. Biol. Chem. 273, 11107–11114 (1998).
Blazquez, M., Docherty, K. & Shennan, K. I. Association of prohormone convertase 3 with membrane lipid rafts. J. Mol. Endocrinol. 27, 107–116 (2001).
Ji, Y. et al. Toll-like receptors TLR2 and TLR4 block the replication of pancreatic β cells in diet-induced obesity. Nat. Immunol. 20, 677–686 (2019).
Okita, N. et al. Modified Western blotting for insulin and other diabetes-associated peptide hormones. Sci. Rep. 7, 6949 (2017).
Sha, H. et al. The IRE1alpha-XBP1 pathway of the unfolded protein response is required for adipogenesis. Cell Metab. 9, 556–564 (2009).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Acknowledgements
We thank Dr. Ernesto Bernal-Mizrachi for sharing αTC(1-6) cells; Dr. Yewei Ji, Dr. Leena Haataja, Jianing Zhang, Elise Corden, and Steve Lentz for experimental support; and all other members of the Qi and Arvan laboratories for comments and technical assistance. This work was completed with the assistance of NIH-supported core facilities, including the Microscopy, Imaging and Cellular Physiology Core (MICPC) and Islet Isolation Laboratory supported by the National Institute of Diabetes, Digestive, and Kidney (NIDDK)-funded Michigan Diabetes Research Center (P30DK020572 and Shared Instrument Grant S10OD28612-01-A1), the University of Michigan Vision Research Center Morphology and Imaging Core supported by the National Eye Institute (P30EY007003), the Tissue and Molecular Pathology Shared Resource supported by the National Cancer Institute (P30CA04659229), and the University of Michigan Biomedical Research Core Facilities Microscopy Core. This manuscript used human islets acquired from the University of Pennsylvania Islet Transplant Center in collaboration with the Human Pancreas Analysis Program (HPAP-RRID:SCR_016202), a Human Islet Research Network (RRID:SCR_014393) consortium (UC4DK112217), and the Integrated Islet Distribution Program (IIDP) (RRID:SCR_014387) through City of Hope (UC4DK098085), supported by Beckman Research Center grant 10028044 (to A.N.). We thank the human islet donors and their families for their generous contribution. This work was supported by R24DK110973 (to P.A.), R01DK11174 and 2-SRA-2018-539-A-B (to P.A. and L.Q.), R01DK121995 (to D.A.S), R01DK137794 and R35GM130292 (to L.Q.), and 5T32DK007245 and K08DK129719 (to R.B.R.).
Author information
Authors and Affiliations
Contributions
W.Z., L.P., X.C., A.C.R., R.R., B.P., X.W., L.L.L., M.T., H.H., B.G., and R.B.R. performed experiments that were designed by W.Z., L.P., L.Q., and R.B.R.; N.S. made the initial crosses of knockout mice and provided experimental support and helpful discussion; C.L., A.N., P.A., D.S., and I.L. provided key reagents and helpful discussion; L.Q. and R.B.R. supervised the project and wrote the manuscript. All other authors edited and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xi Chen, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhu, W., Pan, L., Cui, X. et al. SEL1L-HRD1 ER-associated degradation facilitates prohormone convertase 2 maturation and glucagon production in islet α cells. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69928-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69928-6