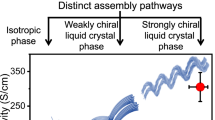
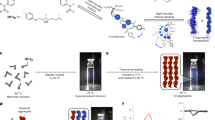
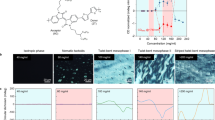
Abstract
Controlled chemical reactions in the condensed state are key steps in designing smart materials. Due to the absence of diffusion and sufficient molecular collision, the reaction efficiency in the solid or aggregated state is expectedly much lower than the solution phase. Herein we report the templating effects in self-assemblies undergoing multiple reactions using an aromatic cystine derivative. Disulfide bond cleavage into cysteine triggered by a reductant is quantitative in aggregates, leading to the molecular rearrangement. Coassembled with a guest pentafluoropyridine through π-hole/π interaction allows for a cascade two-step reaction including reduction and aromatic nucleophilic substitution. These reactions in the condensed, self-assembled state are efficient with significant impacts on the structures of nanoarchitectures. Interestingly, the chiral expression at macroscopic scale and corresponding chiroptical activities exhibit templating and inheritance effect thanks to the sacrificial templating role of pristine aggregates. We introduce new type efficient reactions in the self-assembled states, realizing flexible control by introducing guest, and unveil the chiral inheritance effect in topochemical evolutions.
Similar content being viewed by others
Data availability
All relevant data generated or analyzed during this study can be obtained from the corresponding author. The Supplementary Information is available in the online version of the paper. Source data are provided with this manuscript. Source data are provided with this paper.
References
Yashima, E. et al. Supramolecular helical systems: helical assemblies of small molecules, foldamers, and polymers with chiral amplification and their functions. Chem. Rev. 116, 13752–13990 (2016).
Jia, X. & Zhu, L. Photoexcitation-induced assembly: a bottom-up physical strategy for driving molecular motion and phase evolution. Acc. Chem. Res. 56, 655–666 (2023).
Kumar, J., Nakashima, T. & Kawai, T. Circularly polarized luminescence in chiral molecules and supramolecular assemblies. J. Phys. Chem. Lett. 6, 3445–3452 (2015).
Behera, P. et al. Electric field control of chirality. Sci. Adv. 8, eabj8030 (2022).
Li, J. et al. Programmable supramolecular chirality in non-equilibrium systems affording a multistate chiroptical switch. Nat. Commun. 14, 5030 (2023).
Xing, P. & Zhao, Y. Controlling supramolecular chirality in multicomponent self-assembled systems. Acc. Chem. Res. 51, 2324–2334 (2018).
Ren, X. et al. The dominant role of oxygen in modulating the chemical evolution pathways of tyrosine in peptides: dityrosine or melanin. Angew. Chem. Int. Ed. 58, 5872–5876 (2019).
Wang, Y. et al. Bioinspired amino acid based materials in bionanotechnology: from minimalistic building blocks and assembly mechanism to applications. ACS Nano 18, 1257–1288 (2024).
Yuan, W. et al. Cooperative supramolecular polymerization of styrylpyrenes for color-dependent circularly polarized luminescence and photocycloaddition. Nat. Commun. 14, 8022 (2023).
Wu, A. L. et al. Tunable chirality of self-assembled dipeptides mediated by bipyridine derivative. Angew. Chem. Int. Ed. 62, e202314368 (2023).
Zhao, C. et al. Handedness-inverted and stimuli-responsive circularly polarized luminescent nano/micromaterials through pathway-dependent chiral supramolecular polymorphism. Adv. Mater. 36, 2403329 (2024).
Li, J. J. et al. Self-regulating hydrogel with reversible optical activity in its gel-to-gel transformation. J. Am. Chem. Soc. 147, 17361–17371 (2025).
Chang, R., Yuan, C., Zhou, P., Xing, R. & Yan, X. Peptide self-assembly: from ordered to disordered. Acc. Chem. Res. 57, 289–301 (2024).
Zhang, H., Han, J., Jin, X. & Duan, P. Improving the overall properties of circularly polarized luminescent materials through arene-perfluoroarene interactions. Angew. Chem. Int. Ed. 60, 4575–4580 (2021).
Grossmann, L. et al. On-surface photopolymerization of two-dimensional polymers ordered on the mesoscale. Nat. Chem. 13, 730–736 (2021).
Zhong, H., Zhao, B. & Deng, J. Solvent-Dependent chirality transmission and amplification from cellulose derivative to achiral helical polymer for achieving full-color and white circularly polarized luminescence. Angew. Chem. Int. Ed. 64, e202418463 (2025).
Zhang, S., Chen, A., An, Y. & Li, Q. Arene-perfluoroarene interaction: properties, constructions, and applications in materials science. Matter 7, 3317–3350 (2024).
Peluso, P. & Chankvetadze, B. Recognition in the domain of molecular chirality: from noncovalent interactions to separation of enantiomers. Chem. Rev. 122, 13235–13400 (2022).
Gao, J. et al. Spatiotemporal-dependent confinement effect of bubble swarms enables a fractal hierarchical assembly with promoted chirality. J. Am. Chem. Soc. 146, 18104–18116 (2024).
Wang, Z., Xie, X., Hao, A. & Xing, P. Multiple-state control over supramolecular chirality through dynamic chemistry mediated molecular engineering. Angew. Chem. Int. Ed. 63, e202407182 (2024).
Sahoo, J. K., Pappas, C. G., Sasselli, I. R., Abul-Haija, Y. M. & Ulijn, R. V. Biocatalytic self-assembly cascades. Angew. Chem. Int. Ed. 56, 6828–6832 (2017).
Hao, W., Li, Y. & Liu, M. Endowing phosphor materials with long-afterglow circularly polarized phosphorescence via ball milling. Adv. Opt. Mater. 9, 2100452 (2021).
Wang, Z., Chu, C., Hao, A. & Xing, P. Dynamic borate esterification for evolved supramolecular chirality and chiral optics. Angew. Chem. Int. Ed. 64, e202504617 (2025).
Liu, Y., Guan, X.-R., Wang, D.-C., Stoddart, J. F. & Guo, Q.-H. Soluble and processable single-crystalline cationic polymers. J. Am. Chem. Soc. 145, 13223–13231 (2023).
Hu, F. et al. Highly efficient preparation of single-layer two-dimensional polymer obtained from single-crystal to single-crystal synthesis. J. Am. Chem. Soc. 143, 5636–5642 (2021).
Cao, M. et al. Self-assembly of amphiphilic peptides: effects of the single-chain-to-gemini structural transition and the side chain groups. Colloids Surf. A 6, 7419 (2015).
Wang, H., Wang, Y., Shen, B., Liu, X. & Lee, M. Substrate-driven transient self-assembly and spontaneous disassembly directed by chemical reaction with product release. J. Am. Chem. Soc. 141, 4182–4185 (2019).
Zhang, Q. et al. Stereodivergent chirality transfer by noncovalent control of disulfide bonds. J. Am. Chem. Soc. 144, 4376–4382 (2022).
Morimoto, M. et al. Advances in supramolecular host-mediated reactivity. Nat. Catal. 3, 969–984 (2020).
Raymond, D. M. & Nilsson, B. L. Multicomponent peptide assemblies. Chem. Soc. Rev. 47, 3659–3720 (2018).
Hema, K., Ravi, A., Raju, C. & Sureshan, K. M. Polymers with advanced structural and supramolecular features synthesized through topochemical polymerization. Chem. Sci. 12, 5361–5380 (2021).
Yang, C. et al. High-fidelity topochemical polymerization in single crystals, polycrystals, and solution aggregates. Nat. Commun. 16, 3498 (2025).
Wang, Z., Gai, Y., Hao, A. & Xing, P. Superhelical self-assembly of microcrystals from cyclodipeptides. Angew. Chem. Int. Ed. 64, e202501832 (2025).
Sarojini, V. & Glossop, H. Accessing the thiol toolbox: synthesis and structure–activity studies on fluoro-thiol conjugated antimicrobial peptides. Bioconjug. Chem. 34, 218–227 (2023).
Andrews, K. G., Piskorz, T. K., Horton, P. N. & Coles, S. J. Enzyme-like acyl transfer catalysis in a bifunctional organic cage. J. Am. Chem. Soc. 146, 17887–17897 (2024).
Hess, S. et al. Noncovalent peptide assembly enables crystalline, permutable, and reactive thiol frameworks. J. Am. Chem. Soc. 145, 19588–19600 (2023).
Qiu, X. et al. Regulating reversible untwisting and twisting motions in helical dynamic molecular crystals. J. Am. Chem. Soc. 147, 17772–17783 (2025).
Panja, S. & Adams, D. J. Stimuli-responsive dynamic transformations in supramolecular gels. Chem. Soc. Rev. 50, 5165–5200 (2021).
Ji, W. et al. Coassembly-induced transformation of dipeptide amyloid-like structures into stimuli-responsive supramolecular materials. ACS Nano 14, 7181–7190 (2020).
Dou, X. et al. Supramolecular hydrogels with tunable chirality for promising biomedical applications. Acc. Chem. Res. 53, 852–862 (2020).
Alfuth, J., Jeannin, O. & Fourmigué, M. Topochemical Single-crystal-to-single-crystal [2+2] photocycloadditions driven by chalcogen-bonding interactions. Angew. Chem. Int. Ed. 61, e202206249 (2022).
Liu, J. et al. One-pot ‘click’ synthesis of ring-in-rings complexes with customizable π-stacked dyads. J. Am. Chem. Soc. 147, 15838–15846 (2025).
Jiang, Q. et al. Assembly drives regioselective azide-alkyne cycloaddition reaction. Nat. Commun. 14, 3935 (2023).
Ji, J. et al. The more the slower: self-inhibition in supramolecular chirality induction, memory, erasure, and reversion. J. Am. Chem. Soc. 144, 1455–1463 (2022).
Zhang, L., Wang, H., Li, S. & Liu, M. Supramolecular chiroptical switches. Chem. Soc. Rev. 49, 9095–9120 (2020).
Sun, H. et al. Single-luminophore molecular engineering for rapidly phototunable solid-state luminescence. Angew. Chem. Int. Ed. 64, e202413827 (2024).
Wang, F., Gan, F., Shen, C. & Qiu, H. Amplifiable symmetry breaking in aggregates of vibrating helical molecules. J. Am. Chem. Soc. 142, 16167–16172 (2020).
Wu, B. et al. Controlling ultra-large optical asymmetry in amorphous molecular aggregations. Angew. Chem. Int. Ed. 60, 3672–3678 (2021).
Feng, X., Wang, X., Redshaw, C. & Tang, B. Z. Aggregation behaviour of pyrene-based luminescent materials, from molecular design and optical properties to application. Chem. Soc. Rev. 52, 6715–6753 (2023).
Balamut, B., Hughes, R. P. & Aprahamian, I. Tuning the properties of hydrazone/isosorbide-based switchable chiral dopants. J. Am. Chem. Soc. 146, 24561–24569 (2024).
Wang, H., Bisoyi, H. K., Urbas, A. M., Bunning, T. J. & Li, Q. Reversible circularly polarized reflection in a self-organized helical superstructure enabled by a visible-light-driven axially chiral molecular switch. J. Am. Chem. Soc. 141, 8078–8082 (2019).
Nie, F. & Yan, D. Bio-sourced flexible supramolecular glasses for dynamic and full-color phosphorescence. Nat. Commun. 15, 9491 (2024).
Garain, S., Shoyama, K., Ginder, L.-M., Sárosi, M. & Würthner, F. The delayed box: biphenyl bisimide cyclophane, a supramolecular nano-environment for the efficient generation of delayed fluorescence. J. Am. Chem. Soc. 146, 22056–22063 (2024).
Itoh, T. et al. Halogen bond effect for single-crystal-to-single-crystal transformation: topochemical polymerization of substituted quinodimethane. Macromolecules 48, 5450–5455 (2015).
Li, L., Jiang, P., Zhang, X. & Li, Y. Sign inversion of circularly polarized luminescence in cholesteric liquid crystals induced by mercury ions through binaphthyl dopants’ conjugation control. Angew. Chem. Int. Ed. 64, e202417149 (2025).
Fu, R. et al. A chiral emissive conjugated corral for high-affinity and highly enantioselective recognition in water. Angew. Chem. Int. Ed. 62, e202315990 (2023).
Ong, W. & Swager, T. Dynamic self-correcting nucleophilic aromatic substitution. Nat. Chem. 10, 1023–1030 (2018).
Sato, Y. et al. Shape- and size-tunable synthesis of covalent organic cages through Rh-catalyzed regioselective [2+2+2] cycloaddition. Angew. Chem. Int. Ed. 62, e202304041 (2023).
Dayanara, N. L., Froelich, J., Roomea, P. & Perrin, D. M. Chemoselective, regioselective, and positionally selective fluorogenic stapling of unprotected peptides for cellular uptake and direct cell imaging. Chem. Sci. 16, 584–595 (2025).
Chrzastek, A., Thanasi, I., Irving, J., Chudasama, V. & Baker, J. Dual reactivity disulfide bridging reagents; enabling new approaches to antibody fragment bioconjugation. Chem. Sci. 13, 11533–11539 (2022).
Zhou, P. et al. Steering phase-separated droplets to control fibrillar network evolution of supramolecular peptide hydrogels. Matter 6, 1945–1963 (2023).
Kai, U., Sumida, R., Tanaka, Y. & Yoshizawa, M. Discrimination of perfluorinated arenes/alkanes by modulable polyaromatic capsules. J. Am. Chem. Soc. 147, 10640–10646 (2025).
Huang, Z. et al. Host-enhanced phenyl-perfluorophenyl polar−π interactions. J. Am. Chem. Soc. 142, 7356–7361 (2020).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Acknowledgements
This works is also supported by the National Natural Science Foundation of China (Nos.22171165, 22371170, 22571184) and National Natural Science Foundation of Shandong Province (ZR2025QA15).
Author information
Authors and Affiliations
Contributions
Z.W. and C.C. contributed equally to this work. Z.W. and P.X. proposed the project. Z.W. performed morphology characterization and DFT simulations; C.C. carried out compound synthesis, spectroscopic measurements and related experiments. A.H. and P.X. supervised the research. P.X. wrote the manuscript. All authors discussed the results and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Chun-Sen Liu and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Z., Chu, C., Hao, A. et al. Chiral inheritance effect in the reactive cystine-based coassembly system. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69945-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69945-5