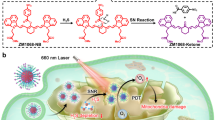
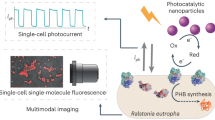
Abstract
Succinic acid is an important platform chemical traditionally produced via energy-intensive and environmentally unfriendly processes. Actinobacillus succinogenes offers a sustainable biosynthetic route, yet its productivity is constrained by limited intracellular electron transfer. Here, we develop a photoelectrocatalytic–microbial biohybrid system to overcome these metabolic bottlenecks. Adaptive laboratory evolution using gold nanoparticles establishes an enhanced charge-transfer pathway in Actinobacillus succinogenes, which is subsequently immobilized on a layer-by-layer NiO@PAA@NHS (NiO nanosheets coated with hydrogel of poly acrylic acid (PAA) grafted with N-Hydroxysuccinimide (NHS)) photoelectrode to construct a NiO@PAA@NHS/Au@ Actinobacillus succinogenes biohybrid. Under simulated solar illumination at −0.3 V vs. RHE, the system delivers a photocurrent density of 1.9 mA cm-2, a CO2 conversion efficiency of 67%, and a succinic acid production rate of 1.41 ± 0.04 g L-1 h-1 cm-2. This work demonstrates an effective strategy for coupling solar energy with microbial metabolism for scalable, carbon-neutral chemical production.
Similar content being viewed by others
Data availability
Data supporting the findings of this work are available within the paper and its Supplementary Information files. Source data are provided with this paper.
References
Kim, J. Y., Lee, J. A., Ahn, J. H. & Lee, S. Y. High-level succinic acid production by overexpressing a magnesium transporter in Mannheimia succiniciproducens. Proc. Natl. Acad. Sci. USA 121, e2407455121 (2024).
Cui, Z. et al. Reconfiguration of the reductive TCA cycle enables high-level succinic acid production by Yarrowia lipolytica. Nat. Commun. 14, 8480 (2023).
Kaur, R. & Chauhan, I. Biodegradable plastics: mechanisms of degradation and generated bio microplastic impact on soil health. Biodegradation 35, 863–892 (2024).
Kumar, V. et al. Recent advances in bio-based production of top platform chemical, succinic acid: an alternative to conventional chemistry. Biotechnol. Biofuels Bioprod. 17, 72 (2024).
Tran, V. G. An end-to-end pipeline for succinic acid production at an industrially relevant scale using Issatchenkia orientalis. Nat. Commun. 14, 6152 (2023).
Narisetty, V. et al. Technological advancements in valorization of second generation (2G) feedstocks for bio-based succinic acid production. Bioresour. Technol. 360, 127513 (2022).
Dickson, R. et al. Sustainable bio-succinic acid production: superstructure optimization, techno-economic, and lifecycle assessment. Energy Environ. Sci. 14, 3542–3558 (2021).
Guan, X., Yu, Y. & Zhang, M. Theoretical insights into the reaction mechanism of direct hydrogenation of maleic anhydride to produce 1,4-butanediol on the Cu–ZnO surface. ACS Catal 14, 6488–6502 (2024).
Lin, F. et al. Advances in succinic acid production: the enhancement of CO2 fixation for the carbon sequestration benefits. Front. Bioeng. Biotechnol. 12, 1392414 (2024).
Bello, S. et al. Renewable carbon opportunities in the production of succinic acid applying attributional and consequential modelling. Chem. Eng. J. 428, 132011 (2022).
Yang, Y. et al. Progress on production of succinic acid by Actinobacillus succinogenes-new opportunities for cheap biomass and waste gas utilization. J. Clean. Prod. 434, 140005 (2024).
Pinazo, J. M., Domine, M. E., Parvulescu, V. & Petru, F. Sustainability metrics for succinic acid production: a comparison between biomass-based and petrochemical routes. Catal. Today 239, 17–24 (2015).
Liang, G. et al. Designing a periplasmic photosynthetic biohybrid system for succinate and electric energy production. Chem. Eng. J. 477, 147152 (2023).
Ahn, J. H. et al. Enhanced succinic acid production by Mannheimia employing optimal malate dehydrogenase. Nat. Commun. 11, 1970 (2020).
Dessie, W. et al. Opportunities, challenges, and future perspectives of succinic acid production by Actinobacillus succinogenes. Appl. Microbiol. Biotechnol. 102, 9893–9910 (2018).
Pateraki, C. et al. Actinobacillus succinogenes: advances on succinic acid production and prospects for development of integrated biorefineries. Biochem. Eng. J. 112, 285–303 (2016).
Mokwatlo, S. C., Nicol, W. & Brink, H. G. Internal mass transfer considerations in biofilms of succinic acid producing Actinobacillus succinogenes. Chem. Eng. J. 407, 127220 (2021).
Chen, C. & Zheng, P. New insights into the biosynthesis of succinic acid by actinobacillus succinogenes with the help of its engineered strains. Fermentation 9, 1026 (2023).
Bradfield, M. F. A. & Nicol, W. The pentose phosphate pathway leads to enhanced succinic acid flux in biofilms of wild-type Actinobacillus succinogenes. Appl. Microbio. Biotechnol. 100, 9641–9652 (2016).
McKinlay, J. B., Shachar-Hill, Y., Zeikusa, J. G. & Vieille, C. Determining Actinobacillus succinogenes metabolic pathways and fluxes by NMR and GC-MS analyses of 13C-labeled metabolic product isotopomers. Metab. Eng. 9, 177–192 (2007).
McKinlay, J. B. & Vieille, C. 13C-metabolic flux analysis of Actinobacillus succinogenes fermentative metabolism at different NaHCO3 and H2 concentrations. Metab. Eng. 10, 55–68 (2008).
McKinlay, J. B. et al. A genomic perspective on the potential of Actinobacillus succinogenes for industrial succinate production. BMC Genomics 11, 680 (2010).
Gong, F. & Li, Y. Fixing carbon, unnaturally. Science 354, 830–831 (2016).
Steffens, L. et al. High CO2 levels drive the TCA cycle backwards towards autotrophy. Nature 592, 784–788 (2021).
Hu, G. et al. Light-driven CO2 sequestration in Escherichia coli to achieve theoretical yield of chemicals. Nat. Catal. 4, 395–406 (2021).
Guo, M. et al. Abiotic-biotic interfaces and electron transfer pathways in nanomaterial-microorganism biohybrids for efficient CO2 conversion. J. Environ. Chem. Eng. 12, 112794 (2024).
Zhan, C. et al. Reprogramming methanol utilization pathways to convert Saccharomyces cerevisiae to a synthetic methylotroph. Nat. Catal. 6, 435–450 (2023).
Mitchell, J. H., Freedman, A. H., Delaney, J. A. & Girguis, P. R. Co-expression analysis reveals distinct alliances around two carbon fixation pathways in hydrothermal vent symbionts. Nat. Microbiol. 9, 1526–1539 (2024).
Chen, H., Dong, F. & Minteer, S. D. The progress and outlook of bioelectrocatalysis for the production of chemicals, fuels and materials. Nat. Catal. 3, 225–244 (2020).
Wang, R. et al. Nanomaterials facilitating microbial extracellular electron transfer at interfaces. Adv. Mater. 33, 2004051 (2021).
Cestellos-Blanco, S., Zhang, H., Kim, J. M., Shen, Y. X. & Yang, P. Photosynthetic semiconductor biohybrids for solar-driven biocatalysis. Nat. Catal. 3, 245–255 (2020).
Han, H. X. et al. Reversing electron transfer chain for light-driven hydrogen production in biotic–abiotic hybrid systems. J. Am. Chem. Soc. 144, 6434–6441 (2022).
Yu, Y. Y. et al. Single cell electron collectors for highly efficient wiring-up electronic abiotic/biotic interfaces. Nat. Commun. 11, 4087 (2020).
Chen, X. et al. 3D-printed hierarchical pillar array electrodes for high-performance semi-artificial photosynthesis. Nat. Mater. 21, 811–818 (2022).
Fang, X., Kalathil, S., Divitini, G., Wang, Q. & Reisner, E. A three-dimensional hybrid electrode with electroactive microbes for efficient electrogenesis and chemical synthesis. Proc. Natl. Acad. Sci. USA. 117, 5074–5080 (2020).
Luo, H. et al. Chemical reaction-mediated covalent localization of bacteria. Nat. Commun. 13, 7808 (2022).
Ming, Z. et al. Living bacterial hydrogels for accelerated infected wound healing. Adv. Sci. 8, 2102545 (2021).
Tang, T. C. et al. Hydrogel-based biocontainment of bacteria for continuous sensing and computation. Nat. Chem. Biol. 17, 724–731 (2021).
Kim, J., Cestellos-Blanco, S., Shen, Y., Cai, R. & Yang, P. Enhancing biohybrid CO2 to multicarbon reduction via adapted whole-cell catalysts. Nano Lett 22, 5503–5509 (2022).
Zhang, H. et al. Bacteria photosensitized by intracellular gold nanoclusters for solar fuel production. Nat. Nanotechnol. 13, 900–905 (2018).
Chen, Z. & Rozhkova, E. A. Intracellular gold nanoclusters boost energy conversion. Nat. Nanotechnol. 13, 880–881 (2018).
Hu, Q. et al. Ultrafast electron transfer in au–cyanobacteria hybrid for solar to chemical production. ACS Energy Lett. 8, 677–684 (2023).
Correa-Llantén, D. N., Muñoz-Ibacache, S. A., Castro, M. E., Muñoz, P. A. & Blamey, J. M. Gold nanoparticles synthesized by Geobacillus sp. strain ID17 a thermophilic bacterium isolated from Deception Island, Antarctica. Microb. Cell. Fact. 12, 75 (2013).
Dhanasekar, N. N., Shirke, A. & Sakthivel, N. Bioreduction of gold ions from anisotropic to isotropic nanostructures by NADPH-dependent reductase from Bipolaris oryzae. ChemistrySelect 5, 11522–11529 (2020).
Chen, X., Yuk, H., Wu, J., Nabzdyk, C. S. & Zhao, X. Instant tough bioadhesive with triggerable benign detachment. Proc. Natl. Acad. Sci. USA. 117, 15497–15503 (2020).
Alberts, E. M. et al. Toward bioinspired polymer adhesives: activation assisted via HOBt for grafting of dopamine onto poly(acrylic acid). R. Soc. Open Sci 9, 211637 (2022).
Faghihzadeh, F., Anaya, N. M., Schifman, L. A. & Oyanedel-Craver, V. Fourier transform infrared spectroscopy to assess molecular-level changes in microorganisms exposed to nanoparticles. Nanotechnol. Environ. Eng. 1, 1 (2016).
Yang, J. et al. Novel modification of collagen: realizing desired water solubility and thermostability in a conflict-free way. ACS Omega 5, 5772–5780 (2020).
Khalil, M. et al. Suppressing the competing hydrogen evolution reaction in CO2 electroreduction: a review. Renew. Sustain. Energy Rev. 206, 114869 (2024).
Ren, L. et al. Construction of high selectivity and antifouling nanofiltration membrane via incorporating macrocyclic molecules into active layer. J. Membr. Sci. 597, 117641 (2020).
Pateraki, C. et al. Transcriptional regulation in key metabolic pathways of Actinobacillus succinogenes in the presence of electricity. Bioelectrochemistry 151, 108376 (2023).
Safarian, S. et al. Structure of a bd oxidase indicates similar mechanisms for membrane-integrated oxygen reductases. Science 352, 583–586 (2016).
Zhang, Y., Feng, T., Zhou, X. & Zhang, Z. Photoelectrocatalytic-microbial biohybrid for nitrogen reduction. Adv. Mater. 36, 2407239 (2024).
Huang, Q., Jiang, F., Wang, L. & Yang, C. Design of photobioreactors for mass cultivation of photosynthetic organisms. Engineering 3, 318–329 (2017).
Peter, A. P. et al. Continuous cultivation of microalgae in photobioreactors as a source of renewable energy: current status and future challenges. Renew. Sustain. Energy Rev. 154, 111852 (2022).
Wang, Y., Tahir, N., Cao, W., Zhang, Q. & Lee, D. J. Grid columnar flat panel photobioreactor with immobilized photosynthetic bacteria for continuous photofermentative hydrogen production. Bioresour. Technol. 291, 121806 (2019).
Palamae, S. et al. Production of renewable biohydrogen by Rhodobacter sphaeroides S10: a comparison of photobioreactors. J. Clean. Prod. 181, 318–328 (2018).
Acknowledgements
This research was supported by National Natural Science Foundation of China (No. 2274041, Z. Z.).
Author information
Authors and Affiliations
Contributions
T. F., X. Z., and Z. Z. designed the experiments. T. F., Y. Z., and X. Z. carried out the experiments. X.Z. conducted the characterizations. All authors wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Santiago Rodriguez Jimenez, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Feng, T., Zhou, X., Zhang, Y. et al. Photoelectrocatalytic-microbial biohybrid for succinic acid synthesis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69962-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69962-4