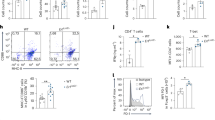
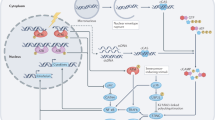
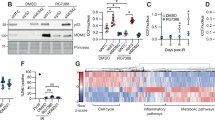
Abstract
Macrophage senescence drives inflammaging, a chronic, age-related inflammation. To date, the protective mechanisms against inflammaging are poorly defined. Here, we identify DNA-PK-mediated phosphorylation of murine STAT6 at serine 807 (Ser807) as a crucial post-translational modification for preventing macrophage senescence. Ser807 phosphorylation blocks STAT6 ubiquitination-mediated degradation and promotes STAT6 partnering with PU.1 to activate DNA repair genes. Macrophages lacking Ser807 phosphorylation exhibit DNA repair defects, undergo senescence, and fuel inflammaging. In vivo, the phosphor-null STAT6 mutant (STAT6(S807A)) accelerates macrophage senescence, tissue fibrosis, and systemic aging. Adoptive transfer of phosphomimetic STAT6(S807E)-expressing macrophages rescues accelerated aging. Importantly, phosphorylation of human STAT6 at the homologous residue (Ser817) is significantly reduced in the lungs of patients with chronic obstructive pulmonary disease (COPD), correlating with increased DNA damage and senescence. Thus, our findings reveal a DNA-PK-STAT6 axis enacting a non-canonical type 2 immunity via DNA repair to prevent macrophage senescence, presenting a therapeutic target for healthy aging.
Similar content being viewed by others
Data availability
This paper does not report original code. The original RNA-seq datasets of Thio-PMs is available from GEO (GSE278458, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE278458). The ChIP-seq dataset of macrophages (PU.1) are obtained from GSE38377 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the iProX partner repository with the dataset identifier PXD074331. All materials generated in this study, including stable cell lines and newly developed pSTAT6 antibodies for mouse and human, will be made available upon request. Source data are provided in this paper.
References
Takeuchi, O. & Akira, S. Pattern Recognition Receptors and Inflammation. Cell 140, 805–820 (2010).
Chen, G. Y. & Nuñez, G. Sterile inflammation: sensing and reacting to damage. Nat. Rev. Immunol. 10, 826–837 (2010).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: An expanding universe. Cell 186, 243–278 (2023).
Yousefzadeh, M. J. et al. An aged immune system drives senescence and ageing of solid organs. Nature 594, 100–105 (2021).
Franceschi, C. et al. Inflamm-aging -: An evolutionary perspective on immunosenescence. Ann. Ny. Acad. Sci. 908, 244–254 (2000).
Gulen, M. F. et al. cGAS-STING drives ageing-related inflammation and neurodegeneration. Nature 620, 374–380 (2023).
Minhas, P. S. et al. Restoring metabolism of myeloid cells reverses cognitive decline in ageing. Nature 590, 122–128 (2021).
Schneider, J. L. et al. The aging lung: Physiology, disease, and immunity. Cell 184, 1990–2019 (2021).
Santoro, A., Bientinesi, E. & Monti, D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity. Ageing Res. Rev. 71, 101422 (2021).
He, S. H. & Sharpless, N. E. Senescence in health and disease. Cell 169, 1000–1011 (2017).
Moiseeva, V. et al. Senescence atlas reveals an aged-like inflamed niche that blunts muscle regeneration. Nature 613, 169–178 (2023).
Liu, J.-Y. et al. Cells exhibiting strong p16 INK4a promoter activation in vivo display features of senescence. Proc. Natl. Acad. Sci. USA 116, 2603–2611 (2019).
Zhou, Z. et al. Type 2 cytokine signaling in macrophages protects from cellular senescence and organismal aging. Immunity 57, 513–527 (2024).
Franceschi, C., Garagnani, P., Vitale, G., Capri, M. & Salvioli, S. Inflammaging and ‘Garb-aging. Trends Endocrin. Met. 28, 199–212 (2017).
Park, M. D., Silvin, A., Ginhoux, F. & Merad, M. Macrophages in health and disease. Cell 185, 4259–4279 (2022).
Sturmlechner, I. et al. p21 produces a bioactive secretome that places stressed cells under immunosurveillance. Science 374, eabb3420 (2021).
Van Dyken, S. J. & Locksley, R. M. Interleukin-4- and Interleukin-13-Mediated Alternatively Activated Macrophages: Roles in Homeostasis and Disease. Annu. Rev. Immunol. 31, 317–343 (2013).
Czimmerer, Z. et al. The transcription factor STAT6 mediates direct repression of inflammatory enhancers and limits activation of alternatively polarized macrophages. Immunity 48, 75 (2018).
Piccolo, V. et al. Opposing macrophage polarization programs show extensive epigenomic and transcriptional cross-talk. Nat. Immunol. 18, 530–540 (2017).
Chen, H. H. et al. Activation of STAT6 by STING is critical for antiviral innate immunity. Cell 147, 436–446 (2011).
Schumacher, B., Pothof, J., Vijg, J. & Hoeijmakers, J. H. J. The central role of DNA damage in the ageing process. Nature 592, 695–703 (2021).
Blackford, A. N. & Jackson, S. P. ATM, ATR, and DNA-PK: The trinity at the heart of the DNA damage response. Mol. Cell 66, 801–817 (2017).
Ferguson, B. J., Mansur, D. S., Peters, N. E., Ren, H. W. & Smith, G. L. DNA-PK is a DNA sensor for IRF-3-dependent innate immunity. Elife 1, e00047 (2012).
Burleigh, K. et al. Human DNA-PK activates a STING-independent DNA sensing pathway. Sci. Immunol. 5, eaba4219 (2020).
Wang, Y. et al. Cytoplasmic DNA sensing by KU complex in aged CD4+ T cell potentiates T cell activation and aging-related autoimmune inflammation. Immunity 54, 632–647.e9 (2021).
Stark, G. R. & Darnell, J. E. The JAK-STAT pathway at twenty. Immunity 36, 503–514 (2012).
Miller, K. N. et al. Cytoplasmic DNA: sources, sensing, and role in aging and disease. Cell 184, 5506–5526 (2021).
Yue, X. Q., Bai, C. J., Xie, D. F., Ma, T. & Zhou, P. K. DNA-PKcs: A multi-faceted player in DNA damage response. Front. Genet. 11, 607428 (2020).
Wang, C. W. et al. GPS 5.0: An Update on the prediction of kinase-specific phosphorylation sites in proteins. Genom. Proteom. Bioinf. 18, 72–80 (2020).
Huston, E. et al. EPAC and PKA allow cAMP dual control over DNA-PK nuclear translocation. Proc. Natl. Acad. Sci. USA 105, 12791–12796 (2008).
Sahoo, A., Alekseev, A., Obertas, L. & Nurieva, R. Grail controls Th2 cell development by targeting STAT6 for degradation. Nat. Commun. 5, 4732 (2014).
Qiao, G. L. et al. E3 Ubiquitin ligase Cbl-b suppresses proallergic T cell development and allergic airway inflammation. Cell Rep. 6, 709–723 (2014).
Wang, C. W. et al. GPS-Uber: a hybrid-learning framework for prediction of general and E3-specific lysine ubiquitination sites. Brief. Bioinform. 23, bbab574 (2022).
Yu, K. et al. qPTM: an updated database for PTM dynamics in human, mouse, rat and yeast. Nucleic Acids Res. 51, D479–D487 (2023).
Shi, J. H. et al. TRAF3/STAT6 axis regulates macrophage polarization and tumor progression. Cell Death Differ. 30, 2005–2016 (2023).
Hanson, E. M., Dickensheets, H., Qu, C. K., Donnelly, R. P. & Keegan, A. D. Regulation of the dephosphorylation of Stat6 -: Participation of TYR-713 in the interleukin-4 receptor α, the tyrosine phosphatase SHP-1, and the proteasome. J. Biol. Chem. 278, 3903–3911 (2003).
Lu, X. Q. et al. PTP1B is a negative regulator of interleukin 4-induced STAT6 signaling. Blood 112, 4098–4108 (2008).
Lu, X. Q. et al. T-cell protein tyrosine phosphatase, distinctively expressed in activated-B-cell-like diffuse large B-cell lymphomas, is the nuclear phosphatase of STAT6. Mol. Cell Biol. 27, 2166–2179 (2007).
Shen, W. K. et al. AnimaITFDB 4.0: a comprehensive animal transcription factor database updated with variation and expression annotations. Nucleic Acids Res. 51, D39–D45 (2023).
Rosenbauer, F. & Tenen, D. G. Transcription factors in myeloid development: balancing differentiation with transformation. Nat. Rev. Immunol. 7, 105–117 (2007).
Li, J. et al. Neurotensin is an anti-thermogenic peptide produced by lymphatic endothelial cells. Cell Metab. 33, 1449–1465.e6 (2021).
Aegerter, H., Lambrecht, B. N. & Jakubzick, C. Biology of lung macrophages in health and disease. Immunity 55, 1564–1580 (2022).
Mertins, P. et al. Proteogenomics connects somatic mutations to signalling in breast cancer. Nature 534, 55–62 (2016).
Aguet, F. et al. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science 369, 1318–1330 (2020).
Christenson, S. A., Smith, B. M., Bafadhel, M. & Putcha, N. Chronic obstructive pulmonary disease. Lancet 399, 2227–2242 (2022).
Prieto LI. et al. Senescent alveolar macrophages promote early-stage lung tumorigenesis. Cancer Cell 41, 1261–1275 (2023).
Haston, S. et al. Clearance of senescent macrophages ameliorates tumorigenesis in KRAS-driven lung cancer. Cancer Cell 41, 1242–1260 (2023).
Mittelbrunn, M. & Kroemer, G. Hallmarks of T cell aging. Nat. Immunol. 22, 687–698 (2021).
Desdín-Micó, G. et al. T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science 368, 1371–1376 (2020).
Kawamoto, S. et al. Bacterial induction of B cell senescence promotes age-related changes in the gut microbiota. Nat. Cell Biol. 25, 865–876 (2023).
Linehan, E. et al. Aging impairs peritoneal but not bone marrow-derived macrophage phagocytosis. Aging Cell 13, 699–708 (2014).
Blacher, E. et al. Aging disrupts circadian gene regulation and function in macrophages. Nat. Immunol. 23, 229–236 (2022).
Pluvinage, J. V. et al. CD22 blockade restores homeostatic microglial phagocytosis in ageing brains. Nature 568, 187–192 (2019).
Li, Z. G. et al. Aging-Impaired Filamentous Actin Polymerization Signaling Reduces Alveolar Macrophage Phagocytosis of Bacteria. J. Immunol. 199, 3176–3186 (2017).
Zhao, H. et al. Identifying specific functional roles for senescence across cell types. Cell 187, 7314–7334.e7321 (2024).
Amor, C. et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583, 127–132 (2020).
Yang, D. et al. NKG2D-CAR T cells eliminate senescent cells in aged mice and nonhuman primates. Sci. Transl. Med. 15, eadd1951 (2023).
Amor, C. et al. Prophylactic and long-lasting efficacy of senolytic CAR T cells against age-related metabolic dysfunction. Nat. Aging 4, 336–349 (2024).
Xu, J., Zeng, Q. Y., Li, S. Q., Su, Q. L. & Fan, H. Inflammation mechanism and research progress of COPD. Front. Immunol. 15, 1404615 (2024).
Bhatt, S. P. et al. Dupilumab for chronic obstructive pulmonary disease with type 2 inflammation: a pooled analysis of two phase 3, randomised, double-blind, placebo-controlled trials. Lancet Resp. Med. 13, 234–243 (2025).
Bhatt, S. P. et al. Dupilumab for COPD with Blood Eosinophil Evidence of Type 2 Inflammation. N. Engl. J. Med. 390, 2274–2283 (2024).
Bernaerts, E. & Vandooren, J. Protocol to generate human monocyte-derived macrophages in the absence of exogenous proteases and protease inhibitors to study proteolysis. STAR Protoc. 6, 103826 (2025).
Huebner, R. H. et al. Standardized quantification of pulmonary fibrosis in histological samples. Biotechniques 44, 507–514 (2008).
Qin, W. et al. Dynamic mapping of proteome trafficking within and between living cells by TransitID. Cell 186, 3307–3324 (2023).
Arnoult, N. et al. Regulation of DNA repair pathway choice in S and G2 phases by the NHEJ inhibitor CYREN. Nature 549, 548–552 (2017).
Duan, Y. J. et al. The clustered, regularly interspaced, short palindromic repeats-associated endonuclease 9 (CRISPR/Cas9)-created T309G mutation enhances vitreous-induced expression of MDM2 and proliferation and survival of cells. J. Biol. Chem. 291, 16339–16347 (2016).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Sanson, K. R. et al. Optimized libraries for CRISPR-Cas9 genetic screens with multiple modalities. Nat. Commun. 9, 5416 (2018).
Evangelou, K. et al. Robust, universal biomarker assay to detect senescent cells in biological specimens. Aging Cell 16, 192–197 (2017).
Ostuni, R. et al. Latent enhancers activated by stimulation in differentiated cells. Cell 152, 157–171 (2013).
Tarhan, L. et al. Single cellportal: an interactive home for single-cell genomics data. Preprint at https://doi.org/10.1101/2023.07.13.548886 (2023).
Acknowledgements
We thank members of the Qiu laboratory for comments on the manuscript. We are grateful to the Flow Cytometry Core of the National Center for Protein Sciences and the Core Facilities of Life Sciences at Peking University for assistance with flow cytometry and confocal microscopy. We also thank Ms. Meng Han in the Proteomics Facility at Tsinghua University Technology Center for Protein Sciences for mass spectrometry analysis of protein modification. This work was supported by grants from the National Key R&D Program of the Ministry of Science and Technology of the People’s Republic of China (2024YFA1802103, 2021YFA0804801 and 2018YFA0800702) to Y.Q.
Author information
Authors and Affiliations
Contributions
Z.Z. designed and performed the main experiments with assistance from X.L., Y.W., L.L., C.W., Z.Z., Y.S., D.W., and Y.Q. discussed and interpreted the results from the study; Z.Z., D.W., and Y.Q. conceived, supervised, and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks Christina Camell, Conor Finlay and Bruno Guigas for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, Z., Li, X., Wang, Y. et al. DNA-PK-mediated phosphorylation of STAT6 establishes a non-canonical type 2 immunity axis to prevent macrophage senescence. Nat Commun (2026). https://doi.org/10.1038/s41467-026-69996-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-69996-8