Abstract
Strigolactones (SLs) are a class of plant hormones that regulate numerous processes of growth and development. SL perception and signal activation involves interaction between F-box E3 ubiquitin ligase D3/MAX2 and DWARF14 (D14) α/β-hydrolase in a SL-dependent manner and targeting of D53/SMXL6/7/8 transcriptional repressors (SMXLs) for proteasome-mediated degradation. D3/MAX2 has been shown to exist in multiple conformational states in which the C-terminal helix (CTH) undergoes a closed-to-open dynamics and regulates D14 binding and SL perception. Despite the multiple modes of D3–D14 interactions found in vitro, the residues that regulate the conformational switch of D3/MAX2 CTH in targeting D53/SMXLs and the subsequent effect on SL signalling remain unclear. Here we elucidate the functional dynamics of ASK1–D3/MAX2 in SL signalling by leveraging conformational switch mutants in vitro and in plants. We report the crystal structure of a dislodged CTH of the ASK1–D3 mutant and demonstrate that disruptions in CTH plasticity via either CRISPR–Cas9 genome editing or expression of point mutation mutants result in impairment of SL signalling. We show that the conformational switch in ASK1–D3/MAX2 CTH directly regulates ubiquitin-mediated protein degradation. A dislodged conformation involved in D53/SMXLs SL-dependent recruitment and ubiquitination and an engaged conformation are required for the release of polyubiquitinated D53/SMXLs and subsequently D14 for proteasomal degradation. Finally, we uncovered an organic acid metabolite that can directly trigger the D3/MAX2 CTH conformational switch. Our findings unravel a new regulatory function of a SKP1–CUL1–F-box ubiquitin ligase in plant signalling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The atomic coordinates and all related data of ASK1–D3 mutant structure were deposited in the Protein Data Bank with accession code 7SA1 (PDB:7SA1). All other materials are available from the corresponding author upon request. Source data are provided with this paper.
References
Stirnberg, P., van De Sande, K. & Leyser, H. M. O. MAX1 and MAX2 control shoot lateral branching in Arabidopsis. Development 129, 1131–1141 (2002).
Stirnberg, P., Furner, I. J. & Ottoline Leyser, H. M. MAX2 participates in an SCF complex which acts locally at the node to suppress shoot branching. Plant J. 50, 80–94 (2007).
Umehara, M. et al. Inhibition of shoot branching by new terpenoid plant hormones. Nature 455, 195–200 (2008).
Gomez-Roldan, V. et al. Strigolactone inhibition of shoot branching. Nature 455, 189–194 (2008).
Cook, C. E., Whichard, L. P., Turner, B., Wall, M. E. & Egley, G. H. Germination of witchweed (Striga lutea Lour.): isolation and properties of a potent stimulant. Science 154, 1189–1190 (1966).
Akiyama, K., Matsuzaki, K. & Hayashi, H. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 435, 824–827 (2005).
Shabek, N. & Zheng, N. Plant ubiquitin ligases as signaling hubs. Nat. Struct. Mol. Biol. 21, 293–296 (2014).
Arite, T. et al. d14, a strigolactone-insensitive mutant of rice, shows an accelerated outgrowth of tillers. Plant Cell Physiol. 50, 1416–1424 (2009).
Hamiaux, C. et al. DAD2 is an α/β hydrolase likely to be involved in the perception of the plant branching hormone, strigolactone. Curr. Biol. 22, 2032–2036 (2012).
Ishikawa, S. et al. Suppression of tiller bud activity in tillering dwarf mutants of rice. Plant Cell Physiol. 46, 79–86 (2005).
Jiang, L. et al. DWARF 53 acts as a repressor of strigolactone signalling in rice. Nature 504, 401–405 (2013).
Zhou, F. et al. D14-SCF D3-dependent degradation of D53 regulates strigolactone signalling. Nature 504, 406–410 (2013).
Soundappan, I. et al. SMAX1-LIKE/D53 family members enable distinct MAX2-dependent responses to strigolactones and karrikins in Arabidopsis. Plant Cell 27, 3143–3159 (2015).
Wang, L. et al. Strigolactone signaling in Arabidopsis regulates shoot development by targeting D53-like SMXL repressor proteins for ubiquitination and degradation. Plant Cell 27, 3128–3142 (2015).
Liang, Y., Ward, S., Li, P., Bennett, T. & Leyser, O. SMAX1-LIKE7 signals from the nucleus to regulate shoot development in Arabidopsis via partially EAR motif-independent mechanisms. Plant Cell 28, 1581–1601 (2016).
Wang, L. et al. Transcriptional regulation of strigolactone signalling in Arabidopsis. Nature 583, 277–281 (2020).
Tal, L., Anleu Gil, M. X., Guercio, A. M. & Shabek, N. Structural aspects of plant hormone signal perception and regulation by ubiquitin ligases. Plant Physiol. 182, 1537–1544 (2020).
Tan, X. et al. Mechanism of auxin perception by the TIR1 ubiquitin ligase. Nature 446, 640–645 (2007).
Sheard, L. B. et al. Jasmonate perception by inositol-phosphate-potentiated COI1-JAZ co-receptor. Nature 468, 400–405 (2010).
Carlsson, G. H., Hasse, D., Cardinale, F., Prandi, C. & Andersson, I. The elusive ligand complexes of the DWARF14 strigolactone receptor. J. Exp. Bot. 69, 2345–2354 (2018).
Yao, R. et al. DWARF14 is a non-canonical hormone receptor for strigolactone. Nature 536, 469–473 (2016).
Shabek, N. et al. Structural plasticity of D3–D14 ubiquitin ligase in strigolactone signalling. Nature 563, 652–656 (2018).
de Saint Germain, A. et al. An histidine covalent receptor and butenolide complex mediates strigolactone perception. Nat. Chem. Biol. 12, 787–794 (2016).
Seto, Y. et al. Strigolactone perception and deactivation by a hydrolase receptor DWARF14. Nat. Commun. 10, 191 (2019).
Stanga, J. P., Smith, S. M., Briggs, W. R. & Nelson, D. C. SUPPRESSOR OF MORE AXILLARY GROWTH2 1 controls seed germination and seedling development in Arabidopsis. Plant Physiol. 163, 318–330 (2013).
Waters, M. T. et al. Specialisation within the DWARF14 protein family confers distinct responses to karrikins and strigolactones in Arabidopsis. Development 139, 1285–1295 (2012).
Swarbreck, S. M., Guerringue, Y., Matthus, E., Jamieson, F. J. C. & Davies, J. M. Impairment in karrikin but not strigolactone sensing enhances root skewing in Arabidopsis thaliana. Plant J. 98, 607–621 (2019).
Stanga, J. P., Morffy, N. & Nelson, D. C. Functional redundancy in the control of seedling growth by the karrikin signaling pathway. Planta 243, 1397–1406 (2016).
Wang, L. et al. Strigolactone and karrikin signaling pathways elicit ubiquitination and proteolysis of SMXL2 to regulate hypocotyl elongation in Arabidopsis. Plant Cell 32, 2251–2270 (2020).
Marrink, S. J., Risselada, H. J., Yefimov, S., Tieleman, D. P. & de Vries, A. H. The MARTINI force field: coarse grained model for biomolecular simulations. J. Phys. Chem. B 111, 7812–7824 (2007).
Uusitalo, J. J., Ingólfsson, H. I., Akhshi, P., Tieleman, D. P. & Marrink, S. J. Martini coarse-grained force field: extension to DNA. J. Chem. Theory Comput. 11, 3932–3945 (2015).
Ueda, H. & Kusaba, M. Strigolactone regulates leaf senescence in concert with ethylene in Arabidopsis. Plant Physiol. 169, 138–147 (2015).
Bennett, T. et al. Strigolactone regulates shoot development through a core signalling pathway. Biol. Open 5, 1806–1820 (2016).
Jia, K. P., Luo, Q., He, S. B., Lu, X. D. & Yang, H. Q. Strigolactone-regulated hypocotyl elongation is dependent on cryptochrome and phytochrome signaling pathways in Arabidopsis. Mol. Plant 7, 528–540 (2014).
Song, C. et al. Strigo-D2—a bio-sensor for monitoring the spatio-temporal pattern of strigolactone signaling in intact plants. Plant Physiol. 188, 97–110 (2022).
Wang, Y. et al. Molecular basis for high ligand sensitivity and selectivity of strigolactone receptors in Striga. Plant Physiol. 185, 1411–1428 (2021).
Zhou, F. et al. D14-SCF D3-dependent degradation of D53 regulates strigolactone signalling. Nature 504, 406–410 (2013).
Khosla, A. et al. Structure–function analysis of SMAX1 reveals domains that mediate its karrikin-induced proteolysis and interaction with the receptor KAI2. Plant Cell 32, 2639–2659 (2020).
Chevalier, F. et al. Strigolactone promotes degradation of DWARF14, an α/β hydrolase essential for strigolactone signaling in Arabidopsis. Plant Cell 26, 1134–1150 (2014).
Hu, Q. et al. DWARF14, a receptor covalently linked with the active form of strigolactones, undergoes strigolactone-dependent degradation in rice. Front. Plant Sci. 8, 1935 (2017).
Hu, X. et al. Expanding the range of CRISPR/Cas9 genome editing in rice. Mol. Plant 9, 943–945 (2016).
Alessandri, R. et al. Pitfalls of the Martini model. J. Chem. Theory Comput. 15, 5448–5460 (2019).
Qian, S. B. et al. Engineering a ubiquitin ligase reveals conformational flexibility required for ubiquitin transfer. J. Biol. Chem. 284, 26797–26802 (2009).
Baek, K. et al. NEDD8 nucleates a multivalent cullin–RING–UBE2D ubiquitin ligation assembly. Nature 578, 461–466 (2020).
Wu, S. et al. CAND1 controls in vivo dynamics of the cullin 1-RING ubiquitin ligase repertoire. Nat. Commun. 4, 1642 (2013).
Rusnac, D.-V. & Zheng, N. in Cullin-RING Ligases and Protein Neddylation: Biology and Therapeutics (eds. Sun, Y. et al.) 9–31 (Springer, 2020).
Verdecia, M. A. et al. Conformational flexibility underlies ubiquitin ligation mediated by the WWP1 HECT domain E3 ligase. Mol. Cell 11, 249–259 (2003).
Russell, R. J. M., Ferguson, J. M. C., Hough, D. W., Danson, M. J. & Taylor, G. L. The crystal structure of citrate synthase from the hyperthermophilic archaeon Pyrococcus furiosus at 1.9 Å resolution. Biochemistry 36, 9983–9994 (1997).
Hicks, K. G. et al. Protein–metabolite interactomics reveals novel regulation of carbohydrate metabolism. Preprint at bioRxiv https://doi.org/10.1101/2021.08.28.458030 (2021).
Diether, M. & Sauer, U. Towards detecting regulatory protein–metabolite interactions. Curr. Opin. Microbiol. 39, 16–23 (2017).
Bennett, B. D. et al. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nat. Chem. Biol. 5, 593–599 (2009).
Ahn, E., Kumar, P., Mukha, D., Tzur, A. & Shlomi, T. Temporal fluxomics reveals oscillations in TCA cycle flux throughout the mammalian cell cycle. Mol. Syst. Biol. 13, 953 (2017).
Wittmann, C., Hans, M., van Winden, W. A., Ras, C. & Heijnen, J. J. Dynamics of intracellular metabolites of glycolysis and TCA cycle during cell-cycle-related oscillation in Saccharomyces cerevisiae. Biotechnol. Bioeng. 89, 839–847 (2005).
Lee, C. P. et al. The versatility of plant organic acid metabolism in leaves is underpinned by mitochondrial malate–citrate exchange. Plant Cell 33, 3700–3720 (2021).
Martínez-Reyes, I. & Chandel, N. S. Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun. 11, 102 (2020).
Quandt, E. M. et al. Fine-tuning citrate synthase flux potentiates and refines metabolic innovation in the Lenski evolution experiment. eLife 4, e09696 (2015).
Fürtauer, L., Küstner, L., Weckwerth, W., Heyer, A. G. & Nägele, T. Resolving subcellular plant metabolism. Plant J. 100, 438–455 (2019).
Sulpice, R. & McKeown, P. C. Moving toward a comprehensive map of central plant metabolism. Annu. Rev. Plant Biol. 66, 187–210 (2015).
López-Bucio, J., de la Vega, O. M., Guevara-García, A. & Herrera-Estrella, L. Enhanced phosphorus uptake in transgenic tobacco plants that overproduce citrate. Nat. Biotechnol. 18, 450–453 (2000).
Brewer, P. B., Koltai, H. & Beveridge, C. A. Diverse roles of strigolactones in plant development. Mol. Plant 6, 18–28 (2013).
Liu, G. et al. Changes in the allocation of endogenous strigolactone improve plant biomass production on phosphate-poor soils. New Phytol. 217, 784–798 (2018).
Saeed, W., Naseem, S. & Ali, Z. Strigolactones biosynthesis and their role in abiotic stress resilience in plants: a critical review. Front. Plant Sci. 8, 1487 (2017).
Tahjib-Ul-Arif, M. et al. Citric acid-mediated abiotic stress tolerance in plants. Int. J. Mol. Sci. 22, 7235 (2021).
Besserer, A. et al. Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biol. 4, e226 (2006).
Kihara, T., Wada, T., Suzuki, Y., Hara, T. & Koyama, H. Alteration of citrate metabolism in cluster roots of white lupin. Plant Cell Physiol. 44, 901–908 (2003).
Proost, S. et al. PLAZA 3.0: an access point for plant comparative genomics. Nucleic Acids Res. 43, D974–D981 (2015).
Bailey, T. L. et al. MEME Suite: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009).
Geldner, N. et al. Rapid, combinatorial analysis of membrane compartments in intact plants with a multicolor marker set. Plant J. 59, 169–178 (2009).
Unger, T., Jacobovitch, Y., Dantes, A., Bernheim, R. & Peleg, Y. Applications of the Restriction Free (RF) cloning procedure for molecular manipulations and protein expression. J. Struct. Biol. 172, 34–44 (2010).
Gleave, A. P. A versatile binary vector system with a T-DNA organisational structure conducive to efficient integration of cloned DNA into the plant genome. Plant Mol. Biol. 20, 1203–1207 (1992).
Fauser, F., Schiml, S. & Puchta, H. Both CRISPR/Cas-based nucleases and nickases can be used efficiently for genome engineering in Arabidopsis thaliana. Plant J. 79, 348–359 (2014).
Swinnen, G., Jacobs, T., Pauwels, L. & Goossens, A. in Plant and Food Carotenoids: Methods and Protocols (eds. Rodríguez-Concepción, M. & Welsch, R.) 321–341 (Humana, 2020).
Swinnen, G. et al. Constitutive steroidal glycoalkaloid biosynthesis in tomato is regulated by the clade IIIe basic helix–loop–helix transcription factors MYC1 and MYC2. Preprint at bioRxiv https://doi.org/10.1101/2020.01.27.921833 (2020).
Porra, R. J., Thompson, W. A. & Kriedemann, P. E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta Bioenerg. 975, 384–394 (1989).
Inskeep, W. P. & Bloom, P. R. Extinction coefficients of chlorophyll a and b in N,N-dimethylformamide and 80% acetone 1. Plant Physiol. 77, 483–485 (1985).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Otwinowski, Z. & Minor, W. B. T.-M. in Methods in Enzymology Vol. 276, 307–326 (Academic Press, 1997).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Sanner, M. Python: a programming language for software integration and development. J. Mol. Graph. Model. 17 1, 57–61 (1999).
Trott, O. & Olson, A. J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461 (2010).
Kutzner, C. et al. More bang for your buck: Improved use of GPU nodes for GROMACS 2018. J. Comput. Chem. 40, 2418–2431 (2019).
Bordoli, L. et al. Protein structure homology modeling using SWISS-MODEL workspace. Nat. Protoc. 4, 1–13 (2009).
Acknowledgements
N.S. is supported by National Science Foundation, NSF-CAREER (Award No. 2047396) and NSF-EAGER (Award No. 2028283). We thank the beamline staff for providing technical assistance at the Advanced Light Source (U.S. Department of Energy Office of Science User Facility under Contract No. DE-AC02-05CH11231, supported in part by the ALS-ENABLE program funded by the National Institutes of Health, National Institute of General Medical Sciences, grant P30 GM124169-01). A.B.'s laboratory is supported by National Science Foundation, NSF BTT EAGER (Award No. 1844705). L.T. is supported by BARD, the United States–Israel Binational Agricultural Research and Development Fund, Vaadia-BARD Postdoctoral Fellowship Award FI-559-2017.
Author information
Authors and Affiliations
Contributions
L.T. and N.S. conceived and designed the experiments. N.S. and L.T. conducted the protein purification, biochemical and crystallization experiments with the help of A.Y. and M.P. Structural and functional analyses were determined and performed by N.S., L.T. and M.P. N.S. and M.P. designed and performed molecular dynamics simulations. N.S. and L.T. designed, and L.T. generated the transgenic lines including phenotype characterizations. L.T., M.R. and A.B. designed and L.T. generated CRISPR–Cas9 Arabidopsis lines. N.S. and L.T. wrote the manuscript with the help from M.P., M.R. and A.B.
Corresponding author
Ethics declarations
Competing interests
N.S. has an equity interest in Oerth Bio and serves on the company’s Scientific Advisory Board. The work and data submitted here have no competing interests, nor other interests that might be perceived to influence the results and/or discussion reported in this paper. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks Tom Bennett, Shinjiro Yamaguchi and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Molecular dynamics simulation of ASK1-D3D720K and functionality analyses.
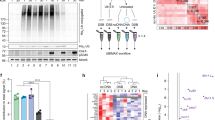
a, Molecular dynamics simulation of ASK1-D3 and ASK1-D3D720K. Root mean square deviation (RMSD, upper panels) and RMS fluctuation (RMSF, lower panels) of all atoms plotted as a function of time for ASK-D3 (brown) and ASK1-D3D720K (cyan) (left panels) and for the CTH of ASK-D3 and ASK1-D3D720K (right panels). b, Conformational clusters over time plotted with the cut-off of 0.25 nm. c, Pulldown of GST-D14 and His-D3 or His-D3D720K in the presence or absence of the synthetic SL, GR24. Proteins were resolved by SDS-PAGE and visualized via Western blot with anti-His and anti-GST antibodies. Asterisk denotes non-specific protein band. d, D14-YLG hydrolysis assay in the presence of either ASK1-D3, or ASK1-D3D720K. Colored lines represent non-linear regression curved fit based on duplications of the raw data points (shown in dots).
Extended Data Fig. 2 max2 complementation experiments.
a, Top panel shows 5 week old plants, SL deficiency phenotypes include changes in rosettes size and leaf shape. Bottom panel shows 8 week old plants, an SL deficiency phenotype of excessive branching is shown. Transgenic plants are in max2 background. b, Mean number of axillary rosettes branches (± SE). (WT (n = 7), max2 (n = 7), pUBQ:MAX2 (n = 8) and pUBQ:MAX2D693K (n = 11), One-way ANOVA and post hoc Tukey test, P < 0.001). Transgenic plants are in max2 background.
Extended Data Fig. 3 D-pocket perturbed MAX2 plants.
5-week-old plants show SL deficiency phenotypes including changes in rosettes size and leaf shape (upper panel). 8-week-old plants exhibit SL deficiency phenotype of excessive branching (lower panel). Transgenic plants are in WT Col-0 background.
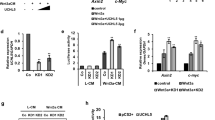
Extended Data Fig. 4 Ubiquitination and time scale degradation of GST-SMXL7D2.
a, Representative Western blot gel of a polyubiquitinated D53D2 levels measurement using anti-GST (α-GST), anti-Ubiquitin (α-Ubiquitin) and anti-His (a-His) antibodies in the presence of max2 total protein extract and His-D3 or His-D3D720K. b, Time scale of cell-free degradation of GST-SMXL7D2 in comparison to a sample incubated with protease inhibitor MG132. Loading control (LD) is shown by Ponceau stain.
Extended Data Fig. 5 functional analysis of ASK1-D3-citrate complex dynamics.
a, D14-YLG hydrolysis assay in the presence of either ASK1-D3, or ASK1-D3D720K, and succinate (Suc, left) or citrate (Cit, right) as indicated. Colored lines represent non-linear regression curved fit based on duplications of the raw data points (shown in dots). b, Molecular docking, and fitting scores of the different ligands from ASK1-D3D720K crystallization conditions are shown in the table with DG values. Closeup view of the 2Fo-Fc (1.0 s, left panel) map and Fo-Fc (3 s, right panel) show fitting of citrate into Fo-Fc electron density and its interaction with residues in the D-Pocket. 2-D interaction plot was generated using LigPlot+ and shows citrate interaction network with the D-pocket (black dash lines represent hydrogen bonds, arches represent Van der Waals interactions). c, Representative Western blot gel of a polyubiquitinated D53D2 levels measurement using anti-GST (α-GST), anti-Ubiquitin (α -Ubiquitin) and anti-His (α -His) antibodies, in the presence of WT total protein extract with citrate or succinate d, Cell-free degradation of GST-AUX/IAA17D2 detected by Western blot using anti-GST (α -GST) antibody. Numbers under the blots are proportions of protein remained that were quantified and compared to T = 0. The experiment was repeated three times.
Extended Data Fig. 6 Genome edited MAX2 Arabidopsis lines.
a, Schematic representation of MAX2 protein variants generated by CRISPR/Cas9. MAX2 gene diagram is shown with focus on MAX2 coding sequence for the CTH amino acids. gRNAs are colored in grey with PAM sequence colored yellow. All WT sequences are shown in blue. DNA deletions as well as protein sequence changes are shown in red. b, Mean leaf blade length and width in the 7th leaf of WT, max2, CRISPR/CAS9 edited MAX2 mutants and kai2. measured at proliferative arrest (± SE). WT (n = 15), max2 (n = 14), MAX2∆CTH (n = 23), MAX2NH (n = 25), MAX2KSLTET (n = 29), MAX2NID (n = 23) and kai2 (n = 21); bars with the same letter are not significantly different from one another (One-way ANOVA and post hoc Tukey, P < 0.05). c, Mean of primary floral stem thickness of WT, max2 and MAX2 genome edited CRISPR/CAS9 lines. (± SE). n = WT (n = 17), max2 (n = 10), MAX2∆CTH (n = 28), MAX2NH (n = 27), MAX2KSLTET (n = 28) and MAX2NID (n = 20); bars with the same letter are not significantly different from one another (One-way ANOVA and post hoc Tukey, P < 0.05). d-e, Structural simulations of AtMAX2 mutants reveal salt bridges between H691 (b) or K690 (c) of CTH and E149 of ASK1.
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2 and Table 1
Source data
Source Data Fig. 1
Unprocessed western blots and gels.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Unprocessed western blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Unprocessed western blots and gels.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Unprocessed western blots.
Source Data Extended Data Fig. 6
Statistical source data.
Rights and permissions
About this article
Cite this article
Tal, L., Palayam, M., Ron, M. et al. A conformational switch in the SCF-D3/MAX2 ubiquitin ligase facilitates strigolactone signalling. Nat. Plants 8, 561–573 (2022). https://doi.org/10.1038/s41477-022-01145-7
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41477-022-01145-7
This article is cited by
-
Cotton GhMAX2 promotes single-celled fiber elongation by releasing the GhS1FA-mediated inhibition of fatty acid biosynthesis
Plant Cell Reports (2025)
-
Strigolactone and karrikin receptors regulate phytohormone biosynthetic and catabolic processes
Plant Cell Reports (2025)
-
Structural insights into strigolactone catabolism by carboxylesterases reveal a conserved conformational regulation
Nature Communications (2024)
-
Strigolactone and abscisic acid synthesis and signaling pathways are enhanced in the wheat oligo-tillering mutant ot1
Molecular Breeding (2024)
-
Genome-wide identification and analysis of the SUPPRESSOR of MAX2 1-LIKE gene family and its interaction with DWARF14 in poplar
BMC Plant Biology (2023)