Abstract
Acute respiratory infections (ARI) are the most common infections in the general population and represent an important socio-economic burden. Characterizing ARIs in primary care in patients of all ages in terms of clinical presentation, and virological results. We conducted a prospective multicenter study in primary care: 36 French general practitioners (GPs) included patients from all ages presenting with symptoms of ARI, and performed a nasopharyngeal swab, which was analyzed by Multiplex RT-PCR. 685 patients of all ages were included in the cohort. We found associations between clinical diagnosis and respiratory viruses: influenza was associated with the diagnosis of flu-like syndrome (p < 0.001), HRV with rhinitis (p < 0.05), and RSV with bronchiolitis (p < 0.001) and bronchitis (p < 0.05). Respiratory distress was associated with RSV (p = 0.002), and a cough at the inclusion was significantly not associated with the influenza virus (p = 0.009). Antibiotic prescriptions were not associated with any specific virus. By day 7, persistent cough was significantly associated with active and passive smoking (respectively p = 0.01 and p < 0.001), influenza and RSV-positive samples (p < 0.05) and an age of less than 2 years (p < 0.01). With this prospective cohort performed in primary care including patients of all ages, we characterized viral respiratory infections, to better understand correlations between clinical data and virological results.
Similar content being viewed by others
Introduction
Acute Respiratory Infections (ARI) are the most frequent community acquired infections with 0 to 6 episodes per person per year1. The prevalence in the whole population remained unspecified although frequent and costly. It has been estimated that there are about 25 million cases of rhinopharyngitis and 10 million cases of bronchitis per year in France and have caused from 20% to 40% of hospitalisations in the young population2. In the US, the cost of common cold and influenza has been estimated at 40 billion dollars3,4. Although bacteria and viruses both cause ARI, respiratory viruses have been shown to be the main factor behind various upper and lower tract infections in children, because the younger you are, the more likely you are to contract viral infections1. In adults, upper respiratory tract infections (URTI) have been also recognized as frequent virus-related diseases5. However, the SARS-CoV2 pandemic has rekindled interest in the responsibility of viruses for lower respiratory tract infections, which have also emerged as major causes not limited to the influenza epidemic. Understanding, treating and preventing respiratory viral infections are therefore major challenges, as they cause a high level of morbidity (acute bronchiolitis, asthma exacerbation, Chronic Obstructive Pulmonary Disease (COPD) or subsequent bacterial pneumonia)6,7. By contrast, the difference between bacterial and viral infection might be difficult to define with the clinics, imaging and biological point-of-care test. We previously showed that a younger age was associated with viral codetections, and that the rate of negative samples (with no viral aetiology found) increased with age8. Thus, antibiotics are still prescribed inappropriately in general practice and emergency care9 with the known consequences’ on the bacterial resistance.
Given that general practitioners manage most of ARIs, knowledge of virus-associated ARIs remains a major clinical challenge in the community. Health authorities have developed various epidemiological surveys on specific viruses, limited to RSV, influenza and SARS-CoV2, all three of which are associated with severe illness and hospital admissions10,11. However, ambulatory studies with the identification of an extensive panel of viruses linked to the clinical features and outcome are limited, because they included only children or elderly12,13. We therefore conducted a prospective study in general practice, which assessed a panel of 15 viruses and 3 bacteria in patients of all ages visiting their GP for any ARIs. The aim of the study was to depict ARI in primary care according to patient age, clinical course after GP consultation and prescribing according to the viruses highlighted in nasopharyngeal swabs.
Patients and methods
Cohort ECOVIR
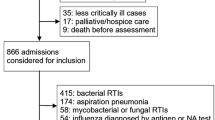
Details on study design, laboratory methods, and bioinformatics were previously published14. Project ECOVIR was a prospective, multicenter, non-interventional study developed in primary care in Normandy, France. ECOVIR was performed during two epidemic seasons (2018–2019 and 2019–2020). The second season was stopped in March 2020, and it was not possible to relaunch it because of the lockdown. Patients were included by 36 General Practitioners (GPs) during medical visits, in primary care. Inclusion criteria were: French speaking people, whatever their age, visiting their general practitioner with symptoms of ARI. Each patient was informed, then examined, and a nasopharyngeal swab was performed by GP, kept at +4 °C and collected at the office. Samples were submitted to Nx-TAG RPP Luminex® kit for virologic identification and following manufacturer’s recommendations in the virologic lab of Caen’s CHU. Fifteen viruses (RSV-A, RSV-B, Rhinovirus/Enterovirus, Metapneumovirus, Adenovirus, CoV-HKU1, CoV-NL63, CoV-229E, CoV-OC43, Influenza, Parainfluenza 1, 2, 3, 4, Bocavirus) and 3 intra-cellular bacteria (Chlamydophila pneumoniae, Legionella pneumophila, Mycoplasma pneumoniae) were screened. Clinical data was collected by the GP at the inclusion. Seven days after the inclusion, patients were called by residents and asked about their environment, demographic characteristics, and evolution of respiratory symptoms. Data was collected on Lime Survey® software.
Statistical analysis
Statistical analyses were performed with standard methods using RStudio software (version 1.1.456). Univariate, descriptive statistics and graphs were prepared using Excel® (Microsoft) and RStudio® softwares. All significance tests were 2-tailed (0.05). For normally distributed data, Student’s t test was used. To compare means of different groups, Anova’s test was implemented with RStudio software.
Results
Clinical diagnosis and age of the patients
685 patients were included. The median age of the patients was 32 years old 95%CI % [13; 54] with a range from 1 month to 94 years (Fig. 1A); 77% were adults. Women accounted for 405 patients (59%), 541 patients (79%) and 144 (21%) had URTI and lower respiratory tract infection (LRTI), respectively. Main GP diagnoses were rhinitis and pharyngitis in all age groups, but flu-like syndrome occurred in 32% of the children aged of 6–17 years (Fig. 1B).
Distribution of the age of the patients A, clinical diagnosis according to the age of patients B and viral diagnosis in Multiplex RT-PCR according to the age of patients C. General Practitioners could give several diagnoses for the same patient (e.g.: rhinitis and pharyngitis). Bronchiolitis: viral infection of the lower respiratory tract in infants less than 24 months old. Bronchitis: inflammation of the bronchial tubes caused by infectious agents. Pneumonia: lung inflammation caused by bacterial or viral infection. Flu syndrome: group of symptoms that are similar to those caused by the influenza (flu) virus. These include fever, chills, headache, muscle or body aches, cough, sore throat, runny nose, fatigue, nausea, vomiting and diarrhea.
Relationship between identified viruses and GP diagnosis
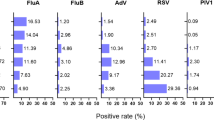
The age distribution of patients according to the viruses detected by PCR is illustrated in Fig. 1C. Virological results according to the age of patients are available8. RSV was mainly associated with bronchitis (24%) and pharyngitis (18%) (Fig. 2A). RSV accounts for 33% of all bronchiolitis cases in our cohort. HRV was mainly associated with pharyngitis (34%) and rhinitis (13%). Endemic coronaviruses were primarily associated with pharyngitis (38%), flu-like syndrome (16%) and rhinitis (13%). Influenza was associated with 44% of flu-like syndromes. In bivariate analysis (Fig. 2B), influenza was significantly associated with flu-like syndrome (p < 0.001), HRV with rhinitis (p < 0.05) and RSV with LRTI (acute bronchiolitis (p < 0.001) and bronchitis (p < 0.05)).
A Clinical initial diagnosis according to the viruses found in Multiplex RT-PCR. B Mosaic plot showing the correlation between viruses found in Multiplex PCR and clinical initial diagnosis. The area of the squares is proportional to the number of cases found (diagnosis and virus). The correlation between diagnosis and virus is represented by colors. If the diagnosis is strongly associated with a virus, the square is blue (for example flu-like syndrome and flu); if the diagnosis is strongly not associated with a virus, the square is red (for example flu-like syndrome and HRV). *: p < 0.05, **: p < 0.01.
Clinical characteristics at the inclusion (Day 0)
Regarding respiratory symptoms at D0, cough was observed in 73% of patients, expectorations in 21%, dyspnea in 8% and respiratory distress in 9% (Fig. 3). In bivariate analysis, day cough was significantly associated with the influenza virus (p = 0.009). Night cough, expectorations and dyspnea were not significantly associated with any specific virus. RSV was significantly associated with respiratory distress (p = 0.002). Table 1 shows the treatments prescribed by the GPs, which differed according to age groups. In infants, nose washings were prescribed in 83% of cases, followed by antipyretics in 54%. Antibiotics were prescribed in 33% of the total cohort. They were significantly associated with the diagnoses of angina, bronchitis, pneumonia, and otitis (p < 0.05), and significantly not associated with the diagnoses of pharyngitis, rhinitis, and flu-like syndrome (p < 0.05). They have not been associated with a specific virus. Oral corticosteroids were prescribed for around 10% of patients, across all age groups, and they were significantly associated with the diagnoses of asthma, laryngitis, and sinusitis (p < 0.05).
Clinical outcome at Day 7 (D7)
633 patients (92%) responded to the follow-up call at D7. The GPs asked 12 patients (2%) to return within 48 hours for a second assessment. No patient needed to be referred at the emergency department or hospitalized. At D7, 61% of patients reported cough, 45% asthenia, 19% dyspnea and 3% fever (Table 2). In multivariate analysis, fever, asthenia, and dyspnea at D7 were not significantly associated with any specific virus. Persistent cough was significantly not associated with the 6–17 age group (p < 0.001). Fever, asthenia, and dyspnea were not significantly associated with patient age. Coughing at D7 was significantly associated with active and passive smoking (respectively p = 0.01 and p < 0.001), influenza and RSV-positive samples (p < 0.05) and an age of less than 2 years (p < 0.01).
Discussion
We report here the results of our clinical and biological, multicenter cohort, performed in primary care, in France, with 36 general practitioners, who included 685 of all ages, presenting with symptoms of ARI over two epidemical seasons. We found correlations between clinical diagnosis and respiratory viruses: influenza was associated with the diagnosis of flu-like syndrome (p < 0.001), HRV with rhinitis (p < 0.05), and RSV with bronchiolitis (p < 0.001) and bronchitis (p < 0.05). Respiratory distress was associated with RSV (p = 0.002), and a cough at the inclusion was significantly not associated with the influenza virus (p = 0.009). Antibiotic prescriptions were not associated with any specific virus. By day 7, persistent cough was significantly associated with active and passive smoking (respectively p = 0.01 and p < 0.001), influenza and RSV-positive samples (p < 0.05) and an age of less than 2 years (p < 0.01).
The main characteristic of our cohort, from primary care, is the important proportion of children (about 30%). The profile of our cohort is so unique, firstly because of the age of patients (11% were between 6 and 17 years old), and the majority were adults between 18 and 64 years old (57%): this is a real-life study. Most studies are performed at hospital, on patients at the extreme ages of life, or with comorbidities, and patients between 6 and 65 years old are rarely hospitalised15. Unfortunately, our study had to be interrupted early due to the containment of the SARS-CoV2 pandemic, which is why we did not include the entire epidemic season in the second season.
The viruses responsible for ARI belong to some fifteen major species with a preferential tropism for the respiratory tract. However, most of data is hospital-based, due to the high cost of virological diagnosis. Results from primary care are rare, so the knowledge of these viruses is limited in ambulatory medicine. Around 60% of samples performed in children presenting to their GP with symptoms of ARI between 2011 and 2013 in England were positive for at least one virus12. The type of sampling used in primary care also raises concerns about reproducibility, feasibility, and ease of use. Oropharyngeal sampling is simple and easily accessible, particularly in children, and is little feared by primary care practitioners13. Nasopharyngeal swabbing, not known before the SARS-CoV2 pandemic, is now easily performed by primary care practitioners.
We noticed a high level of antibiotic prescription, specifically in young children aged from 2 to 5 years old. Surprisingly, these age groups are characterized by highest level of positive viral tests. This highlights the real value of implementing qualitative virus detection tests in primary care, to guide GPs in their prescriptions. This could be POCT (point of care tests): these tests are designed to be performed by non-professional staff (patients themselves) or paramedical staff (nurses). The value of microbiological POCT, particularly in terms of antibiotic savings, is currently being evaluated16. A review published in 2022 estimated the interest of the POCT for the C-reactive protein in primary care in patients consulting for acute respiratory infections17: the POCT could decrease significantly the prescription of antibiotics, but an algorithm seems necessary. Combined rapid diagnosis tests exists, like influenza/RSV/SARS-CoV2 rapid tests18. Compared with the results obtained with specific RT-PCR, the positive and negative concordance percentages of these rapid tests, carried out in the GP’s cab, were high19.
Some diagnoses established by the GPs were associated with respiratory viruses. Influenza viruses were significantly associated with flu-syndrome, RSV was significantly associated with bronchitis and bronchiolitis, and HRV was associated with rhinitis. So, correlations between clinical and virological diagnoses exist. These findings suggest that clinical observations alone may not reliably predict the presence of a bacterial infection that would warrant antibiotic treatment. Further research is needed to identify clinical predictors of bacterial infection in patients with ARI. This information could be promoted to the GPs, to limit antibiotics prescriptions, particularly in children20. Use of antibiotics in children do modify their respiratory microbiota, with an impact on their respiratory development21. Interestingly, RSV is responsible for other diagnosis than bronchiolitis, and is not responsible for all cases of bronchiolitis. New therapies are now available to prevent the infections due to RSV: some are designed for the infants22,23, and other designed for pregnant women24 or the elderly25. The burden of RSV in the elderly has been assessed, and active prevention is necessary26. We should keep in mind that RSV can induce respiratory distress in all ages, and its prevention should be a priority for the public health policies.
Conclusion
Our study, performed in primary care before the pandemics, implicating 36 GPIs, is unique in terms of population, and virological results. We found correlations between clinical data and respiratory viruses. Antibiotic prescriptions were not associated with any specific virus, and corticosteroids were prescribed according to international recommendations. This highlights good practices of our primary care system.
Data Availability
No datasets were generated or analysed during the current study.
Abbreviations
- ARI:
-
acute respiratory infections
- CoV:
-
coronavirus
- Flu:
-
influenza virus
- HRV:
-
human rhinovirus
- GP:
-
general practitioner
- GPI:
-
general practitioner investigator
- RSV:
-
respiratory syncytial virus
- RT-PCR:
-
reverse transcription polymerase chain reaction
References
Monto, A. S., Malosh, R. E., Petrie, J. G., Thompson, M. G. & Ohmit, S. E. Frequency of acute respiratory illnesses and circulation of respiratory viruses in households with children over 3 surveillance seasons. J. Infect. Dis. 210, 1792–1799 (2014). https://academic-oup-com.gate2.inist.fr/jid/article/210/11/1792/2908499.
GBD 2015 Disease and Injury Incidence and Prevalence Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Lond. Engl. 388, 1545–1602 (2016).
Molinari, N. A. M. et al. The annual impact of seasonal influenza in the US: measuring disease burden and costs. Vaccine 25, 5086–5096 (2007).
Fendrick, A. M., Monto, A. S., Nightengale, B. & Sarnes, M. The economic burden of non-influenza-related viral respiratory tract infection in the United States. Arch. Intern. Med. 163, 487–494 (2003).
Thomas, M., Bomar, P. A. Upper Respiratory Tract Infection. StatPearls [Internet]. StatPearls Publishing: Treasure Island (FL), 2024). http://www.ncbi.nlm.nih.gov/books/NBK532961/.
de Blic, J., Brouard, J., Vabret, A. & Deschildre, A. Interactions micro-organismes et voies aériennes distales: spécificités pédiatriques. Rev. Mal. Respir. 34, 134–146 (2017). http://linkinghub.elsevier.com/retrieve/pii/S0761842517300311.
Delmas, M. C. et al. Augmentation de la prévalence de l’asthme chez le jeune enfant en France. Rev. Mal. Respir. 34, 525–534 (2017). http://linkinghub.elsevier.com/retrieve/pii/S0761842516301991.
Petat, H. et al. Positive and negative viral associations in patients with acute respiratory tract infections in primary care: the ECOVIR study. Front. Public. Health 11, 1269805 (2023).
Tonkin-Crine, S. K. et al. Clinician-targeted interventions to influence antibiotic prescribing behaviour for acute respiratory infections in primary care: an overview of systematic reviews. Cochrane Database Syst. Rev. 9, CD012252 (2017).
Liu, J. & Suzuki, S. Real-Time detection of flu season onset: a novel approach to flu surveillance. Int. J. Env. Res. Public. Health 19, 3681 (2022).
Pedroza-Uribe, I. M. et al. Beyond SARS-CoV-2: epidemiological surveillance of respiratory viruses in Jalisco, Mexico. Front. Public. Health 11, 1292614 (2023).
McGeoch, L. J. et al. Prognostic value of upper respiratory tract microbes in children presenting to primary care with respiratory infections: a prospective cohort study. PLOS ONE 17, e0268131 (2022). https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0268131.
Thornton, H. V. et al. Throat swabs in children with respiratory tract infection: associations with clinical presentation and potential targets for point-of-care testing. Fam. Pract. 34, 407–415 (2017).
Petat, H. et al. The design and implementation of the ECOVIR project: a primary health care surveillance system to strengthen co-detection of respiratory viruses in normandy. Methods Protoc. 5, 98 (2022). https://www.mdpi.com/2409-9279/5/6/98.
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Lond. Engl. 392, 1789–1858 (2018).
Khalid, T. Y. et al. Novel multi-virus rapid respiratory microbiological point-of-care testing in primary care: a mixed-methods feasibility evaluation. Fam. Pract. 38, 598–605 (2021).
Smedemark, S. A. et al. Biomarkers as point-of-care tests to guide prescription of antibiotics in people with acute respiratory infections in primary care. Cochrane Database Syst. Rev. 10, CD010130 (2022).
Bruning, A. H. L. et al. Rapid tests for influenza, respiratory syncytial virus, and other respiratory viruses: a systematic review and meta-analysis. Clin. Infect. Dis. Publ. Infect. Dis. Soc. Am. 65, 1026–1032 (2017).
Zuurbier, R. P. et al. Performance assessment of a rapid molecular respiratory syncytial virus point-of-care test: a prospective community study in older adults. J. Infect. Dis. 226, S63–S70 (2022).
Kamikawa, J., Granato, C. F. H. & Bellei, N. Viral aetiology of common colds of outpatient children at primary care level and the use of antibiotics. Mem. Inst. Oswaldo Cruz 110, 884–889 (2015).
Henares, D. et al. Differential nasopharyngeal microbiota composition in children according to respiratory health status. Microb. Genomics 7, 000661 (2021).
Venkatesan, P. Nirsevimab: a promising therapy for RSV. Lancet Microbe 3, e335 (2022).
Simões, E. A. F. et al. Efficacy of nirsevimab against respiratory syncytial virus lower respiratory tract infections in preterm and term infants, and pharmacokinetic extrapolation to infants with congenital heart disease and chronic lung disease: a pooled analysis of randomised controlled trials. Lancet Child. Adolesc. Health 7, 180–189 (2023).
Gong, X. et al. Clinical research on RSV prevention in children and pregnant women: progress and perspectives. Front. Immunol. 14, 1329426 (2023).
Walsh, E. E. et al. Efficacy and safety of a bivalent RSV prefusion F vaccine in older adults. N. Engl. J. Med. 388, 1465–1477 (2023).
Savic, M., Penders, Y., Shi, T., Branche, A. & Pirçon, J. Y. Respiratory syncytial virus disease burden in adults aged 60 years and older in high-income countries: a systematic literature review and meta-analysis. Influenza Other Respir. Viruses 17, e13031 (2023).
Acknowledgements
We would like to thank Mrs Naomi Stone for the reviewing of English writing. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
HP, MS, CM, MAR and AV conceptualised and designed the study. HP, SC, MS, AR, XH and FLB were responsible for data collection.HP, CM, AV and MAR were responsible for analysis and interpretation of the results. HP wrote the initial draft. All authors critically reviewed its content, approved the final version for submission and provided consent for publication. Each author takes responsibility for the accuracy and integrity of the work presented in this article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
We obtained the agreement of the “Est II” protection committee (study reference 18/10/10/63004) in January 2019. An information document was given to each patient included (non-opposition document).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Petat, H., Schuers, M., Le Bas, F. et al. Characterizing acute respiratory infections in primary care for better management of viral infections. npj Prim. Care Respir. Med. 35, 28 (2025). https://doi.org/10.1038/s41533-025-00434-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41533-025-00434-w