Abstract
Membrane distillation (MD) faces critical challenges at the industrial scale, including poor permeate flux and membrane fouling. To address these issues and efficiently treat highly saline water, this study presents a nature-inspired approach to fabricating a robust multilayer Janus membrane using a gecko-inspired adhesion mechanism. The proposed membrane was fabricated using a layer-by-layer co-deposition method, combining a surface-roughened PVDF flat-sheet membrane prepared via phase inversion with an electrosprayed hydrophobic PVDF-HFP interfacial layer modified with CuO nanoparticles and an electrospun hydrophilic PEI fiber bottom layer. The hydrophobic top layer exhibited a water contact angle of 131.5°, followed by a superhydrophobic interfacial layer and the bottom/support layer with a contact angle of 41.4°, enabling superior directional wettability. The Janus membrane achieved an impressive water gap membrane distillation (WGMD) flux of 37.16 kg m⁻² h⁻¹ with a high salt rejection rate of 99.99% over 24 h. Furthermore, the membrane demonstrated long-term stability and excellent resistance to fouling and delamination in harsh saline environments, maintaining performance over 60 h of continuous MD operation. This work highlights the potential of bio-inspired engineering in developing efficient and durable membranes, offering a promising pathway for advancing MD technology for industrial-scale desalination.

Similar content being viewed by others
Introduction
Rapid development and population increase have made water scarcity a major worldwide concern. It is now crucial to address the purification of seawater and hypersaline water, particularly since conventional separation methods have serious drawbacks, such as scaling and fouling brought about by impurities in the feed stream1. The ease in fabrication and implementation with promising value is among the motivational factors of utilizing polymeric membranes in water treatment and separation technologies2. Various traditional processes such as reverse osmosis (RO), thermal distillation make use of these membranes in purification technology3. Given its inherent benefits over conventional techniques, membrane distillation (MD), an innovative separation technology, has attracted a lot of attention. MD produces high-purity water under comparatively modest processing conditions. Furthermore, MD uses renewable energy sources, which makes it an affordable and environmentally friendly substitute4,5. MD is a non-isothermal technique where a porous structured hydrophobic membrane is used to separate the feed and the permeate. The feed is usually heated to an elevated temperature creating vapor that penetrates through the porous structure of the membrane to form a condensed permeate on the cold side, driven by vapor pressure gradient between the two non-isotherms6. MD uses less energy and has a smaller chance of corrosion than other distillation methods since it operates at moderate feed temperatures, usually below 90 °C. Additionally, by providing higher rejection, MD is positioned as a feasible desalination and wastewater treatment method7,8. However, problems like low permeate flux and the early failure of hydrophobic membranes in rejecting brine continue to hinder the extensive industrial application of MD9,10. Membrane fouling, which develops when pollutants adhere to hydrophobic surfaces like polyvinylidene fluoride (PVDF) membranes, is a significant problem in MD, leading to wetting and decreased performance11,12. By incorporating nanoparticles, surface roughening, chemical or physical modifications, recent research has concentrated on improving membrane hydrophobicity and fouling resistance1,13,14,15,16,17.
Mass transfer over the membrane is still a crucial component even with improvements in fouling mitigation and scalability. Because of increased mass transfer resistance, increasing membrane thickness to improve mechanical and thermal stability frequently reduces vapor flux18,19,20. Through a novel innovation in MD, these compromises are avoided using multiple-layer Janus membranes, which are composed of a hydrophilic reinforcement layer that draws water closer to the permeate or acts as a barrier to contaminants, improving saline rejection and/or increasing permeate flux21. These novel multiple-layer membranes function better than their single-layered equivalents in promising water treatment applications22,23. Various Janus membranes are fabricated by depositing the hydrophilic layer on top of a hydrophobic substrate consisting of any of the polymeric materials (Polyvinylidene fluoride (PVDF), Poly tetrafluoroethylene (PTFE), Polypropylene (PP), Polysulfone (Psf)) or their composites24,25,26.
Delamination offered a greater issue despite the numerous publications on the fabrication of Janus membranes employed in high salinity treatment, which happened due to differences in the physicochemical characteristics of layers at the interface27. Layer-by-layer coating method has been widely explored in coating a hydrophilic layer on top of a hydrophobic layer, however, the difference in shrinkage coefficient and weak interaction between one polymer and the other was the primary driving factor for delamination27,28,29. To maximize the interaction between these layers for strong adhesion, for instance, connecting the layers via covalent bonding may cause issues with heat transfer, which poses challenges to the interfacial mass transfer and results in low water flux30. When an appropriate selection is made, an interaction between the middle layer and the surface layers will increase the adhesion between the two asymmetric layers of the Janus membrane. This was well reviewed and explained elsewhere in the literature18,31,32,33. Regardless of layer delamination, the overall thickness of the multilayer membrane also increases the mass transfer resistance with a detrimental effects of temperature polarization18. Zuo et al. developed dual layer membrane involving PVDF and PEI for VMD applications through co-extrusion technique which involves dope solution manipulations and incorporation of Al2O3 nanoparticles to reduce the vapor permeate resistance across the membrane interface and enhance the mechanical strength of the membrane. Overall, the fabricated membrane achieved a high flux of 45.8 kgm−2 h−1 at 70 oC and 3.5 wt.% feed temperature and concentrations, respectively. However, the interfacial chemistry between the layers were not well investigated in the extruded hollow fiber membrane34. Hence, careful material selection and employing robust fabrication techniques have shown to enhance MD performance of Janus membranes with multi-layer structures.
The incorporation of nanoparticles in membranes for MD applications has been greatly explored35,36. Copper oxide (CuO) nanoparticles have been reported in literature for enhancing the degree of roughness, crystallinity and bulk properties of polymeric membranes resulting in improvement in overall efficiency of membrane performance and fouling resistance. CuO nanoparticles were also found to improve the mechanical stability of membranes, especially when uniformly dispersed which was due to the strong interaction between the nanoparticles and the polymeric matrix37. Polyetherimide (PEI) as a hydrophilic polymer has been explored in various water treatment applications for which is purposely due to its excellent chemical, thermal and mechanical stability38,39. Hence, the application of PEI in membrane engineering and CuO inclusion in the multilayer membrane can be promising and worthy of exploring.
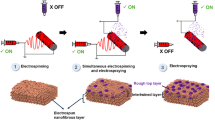
Herein, we report a novel interfacial engineering method for creating multilayer Janus flat-sheet membranes for the treatment of hypersaline water. For precise control over membrane thickness, the fabrication method combines non-solvent induced phase separation (NIPS), electrospraying, and electrospinning techniques. The primary driving force is the necessity for an ultrathin flat sheet membrane with high hydrophobicity and uniform porous structure to facilitate optimal interactions between the materials during layer-by-layer co-deposition. In this work, a multilayer Janus membrane using polyetherimide (PEI) fibers as support and surface-roughened thin PVDF membrane active layer was developed, drawing inspiration from the adhesion mechanisms of gecko feet, which are characterized by microstructures that depend on van der Waals forces. The interfacial layer was incorporated by electrospraying a bifunctional nanocomposite comprising polyvinylidene fluoride-co-hexafluoropropylene (PVDF-HFP) and CuO. By means of fiber interlocking processes, this layer improved adhesion between the PEI and PVDF layers and added gecko-like qualities including superhydrophobicity, and strong physical interaction. In water-gap membrane distillation (WGMD), the fabricated Janus membrane’s performance was assessed under highly saline water and organic foulant feed solution investigating its delamination, antiscaling and antifouling ability.
Results
Membrane structure analysis, surface chemistry and wettability
The objective of this study as stated earlier was to integrate an interfacial layer that physically joins a Janus membrane’s active hydrophobic layer and hydrophilic support layer. To optimize separation performance, the membrane interface is engineered to reduce mass transfer resistance and increase layer-to-layer adhesion. Although research on interfacial interactions in Janus membranes for MD applications is still in its early stages, most studies have concentrated on adhesion processes inspired by mussels40. These investigations, however, frequently fall short in providing thorough explanations of interfacial interactions.
Despite showing promising results for Janus membranes in MD, gecko-inspired adhesion mechanisms are still mostly unexplored because of the difficulty in fabrication, material selection and elucidating adhesion mechanisms. In the past, capillary action was thought to be responsible for Gecko foot adhesion, and the Young-Dupré equation was linked to adhesion energy (W)41. According to Young’s equation, the water contact angle (WCA) and adhesion energy are inversely related, with higher θ values denoting less adhesion. This theory was later disproved, though, because while diverse surfaces like glass and Gecko feet have different adhesion despite their wettability, identical surfaces show equal wettability. This disparity was explained by additional research, which revealed that Gecko adhesion is unaffected by surface chemistry or WCA. Rather, it relies on surface area and van der Waals interactions. Gecko adhesion strength is determined by the surface area of interaction rather than surface chemistry or wettability, according to a hydrophobic and hydrophilic polarizable physical surface model42. Motivated by these results, this study developed an interfacial layer with “Gecko-state” wettability by simulating the hydrophobic nature and high surface roughness of Gecko feet. A composite solution was electro-sprayed onto the PVDF membrane (M1) skin surface to deposit interfacial layer to improve interaction with the hydrophilic PEI support.
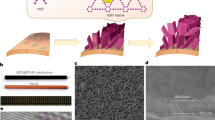
The morphology of the membrane fabricated via NIPS technique is dependent on several factors, such as the casting substrate, the concentration of the polymer, the mixing thermodynamics, and the precipitation kinetics43. The PVDF membrane (M1) developed in this work displayed rough textures (Fig. 1) due to sandpaper as casting substrate. The substrate’s abrasive effect produced basins, which resulted in a highly porous surface. Phase exchange processes are known to be accelerated by such surface features because they increase the surface area available for solvent-nonsolvent exchange, forming more troughs and locations for water ingress13,44. The cross-sectional image of M1 membrane as shown in Fig. 1c showed thin sheet with dense skin layer and finger-like characteristics from the NIPS process45. This structure forms due to thermodynamic instabilities at the solvent-nonsolvent interface near the substrate during immersion in the coagulation bath. Rapid solvent diffusion into the nonsolvent phase initiates pore nucleation, while interactions between the pore-former (LiCl) and the nonsolvent influence pore interconnectivity and the resulting finger-like structure46. A uniform thin unsupported PVDF membrane with rough and porous surface depicting water contact angle of 131.5° (Fig. 1d) was obtained with the help of sandpaper casting substrate, suitable for secondary layer deposition.
Subsequent electro-spraying and electrospinning techniques were employed to develop the Janus multilayer membranes. As a result, the other side of the membrane surface M1 (skin side) has undergone various morphological changes as depicted in Fig. 2. Typically, after electrospraying the surface of the flat sheet membrane exhibited pronounced roughness, characterized by granular and spherical features with uniform particle distribution (Fig. 2a1, a2).
a1, a2 SEM surface imaging of M2 membrane (interfacial layer) with a3 water droplet and contact angle on the surface. b1, b2 SEM images of M3 membrane (interfacial layer) with b3 water droplet and contact angle on the surface and c1, c2 SEM images of support/bottom layer in M4, M5 and M6 membranes with c3 water droplet and contact angle on the surface.
The granular particles also demonstrated uneven height distribution between gaps, a surface characteristic closely mimicking the tiny bristle structure of Gecko feet. The presence of microscale gaps and rough surface facilitates air trapping and mechanical locking between the membrane surfaces. The superhydrophobic nature of this surface is shown in Fig. 2a3, confirmed through contact angle of water droplet. On the other hand, the intermediate layer involving hydrophilic additive (PAN+CuO) in M3 membrane demonstrated particle distribution in fibrous morphology providing roughness and hydrophilic surface characteristic (Fig. 2b). Building on these results, the support layer was deposited by electrospinning polyetherimide (PEI) fibers onto the M1, M2 and M3 membrane surfaces. This sequential process produced a Janus multilayer membrane, designated as M6, M4 and M5 respectively. The support layer demonstrated a highly interconnected fibrous structure with uniform size distribution, as shown in Fig. 2c1. (Size distribution of particles and fibers are shown in Fig. S1). Furthermore, the PEI fibers demonstrated hydrophilic affinity from the water contact measured on the fiber surface (Fig. 2c3). It’s worth stating that the M4, M5, and M6 membranes have the same architecture, only that M4 and M5 contain an interfacial/intermediate layer while M6 doesn’t.
The WCA recorded from membrane surfaces depicted in Figs. 1, 2 depends on several variables, such as the membrane’s surface chemistry, topography, roughness, and surface energy47. A typical PVDF membrane fabricated through the NIPS method in water as the coagulation bath exhibits a WCA value of 77o 48. A typical glass substrate used to cast PVDF membrane using NIPS conditions in this work showed a WCA of less than 90 degrees (Fig. S2). The developed membrane (M1) precipitated using a mixture of non-solvents on a sandpaper substrate displayed a significantly greater WCA of about 131.5o. The increase in hydrophobicity is reasonable considering published research that demonstrated the substantial impact of coagulation media and substrate on membrane surface morphology and, consequently, membrane hydrophobicity11,47. The mimicked rough surface of M2 displayed a superhydrophobic nature with WCA of 151.8o °, which gives this surface the ability to “self-clean”, a similar characteristic of a Gecko’s feet. The interaction between the water droplet and the membrane surface in this case shows the Cassie-Baxter state. However, various measurements of WCA on the electrosprayed interfacial layer were taken as optimization parameters and displayed in Fig. S3. The WCA was found to decrease with an increase in spraying time. The water contact angle (WCA) of membranes is influenced by surface roughness and molecular interactions. Increased roughness from longer spraying time of the composite solution of PVDF-HFP and CuO, enhances air pockets at the membrane-water interface. However, the hydrophilic nature of the CuO dominates, reducing WCA despite the observed increased roughness. Because of the PAN and CuO particles synergistic influence, the M3 surface displayed a hydrophilic character with a WCA value of 27.2o °, whereas the WCA angle of the bottom PEI fiber layer in M4, M5, and M6 showed a value of 41.4o depicting a Wenzel state due to the roughness induced by the fibers.
To confirm the surface roughness on critical surfaces of the membranes responsible for hydrophobicity and interfacial adhesion, the average surface roughness (Ra) was obtained from the AFM imaging over a given sampling length. This value was related to the microporous structure and uniformity on the surface of the membranes1. Notably, from Fig. 3a, the textured surface of M1 membrane demonstrated a roughness (Ra) value of 148 nm, indicating the influence of sandpaper on the surface roughness due to observed textures in SEM images of the membrane in Fig. 1b. Although the values of Ra don’t fully explain the dependency of surface roughness on WCA values, the higher roughness in PVDF membrane (M1) is a key point in explaining the higher WCA49. However, it’s worth mentioning that the texture and surface porosity on M1 membrane introduced water-enclosed air pockets, producing an air-water droplet interface, essential for increasing the WCA of the surface44,50.
The rough morphology of M2 membrane surface was quantitatively supported by the AFM data displayed in Fig. 3b, which shows an average surface roughness (Ra) of approximately 665 nm. The potential interfacial area for fiber interlocking is maximized by the textured landscape depicted in the 3d topographic image. As can be observed, critical surface characteristics are represented by the peaks and valleys in the AFM picture, where fibers can mechanically engage and embed themselves into the granule-formed crevices. It is noteworthy that surfaces with longer sprayed duration produced higher surface roughness (Fig. S4). However, the main reason for selecting 15-min spraying period was the uniform distribution of particles and the hydrophilic effect of CuO at higher spraying durations.
The FT-IR spectra obtained from various layers of the proposed membrane samples are shown in Fig. 4. The sandpaper cast PVDF membrane represented by M1 and made the top layer and similar at the top for all other membranes. PEI fibers made up the bottom layers in membranes M4, M5 and M6, and the intermediate layers comprising the M2 and M3 membranes with composites of PVDF-HFP+CuO and PAN+CuO layers, respectively. In M1 membrane, the peaks formed at around 1072 cm−1 and 1278 cm−1 were attributed to the α- and β- phases, which are mostly found in the PVDF membrane surface fabricated through the NIPS method. The highly intense peaks occurring at around 1171 cm−1 correspond to the C–F bonds, which are highly distributed within the polymer chain and the peaks just below 880 cm−1 indicate the presence of C–C bonds51,52. In M2 membrane surface, the peak attributed to –CF2 stretching modes appeared at 1175 cm−1 53. For PVDF-HFP, this peak was observed at 1168 cm−1 (Fig. S5). A susceptible reason for this is attributed to the interaction between the polymeric matrix and the dispersed CuO37. This is like the findings explained elsewhere in the literature where additives in a polymeric PVDF-HFP matrix interact with the bonds and correspondingly results in peaks shift54. The peak present at around 2243 cm−1 in M3 corresponds to –C ≡ N of PAN polymer while the vibrations noticed at 2984 cm−1 and 1666 cm−1 are attributed to sp3 C–H and amide group bonds respectively55,56. However, considering the surfaces of M4, M5 and M6 which are spectra of PEI fibers, stretching and bending motions of –C–N bonds are shown by the peaks at 1359 cm−1 and 746 cm−1. Finally, the different etheric functional groups in polyetherimide are confirmed by the peaks formed at 1273 cm−1 and 1235 cm−1 57. It is worth nothing that certain similarities were observed in the spectra of M1, M2 and M3 membranes. M1 membrane, which was PVDF membrane, predominately contains C–F as the only free fluorinated carbon bond. In M2 membrane, various fluorinated carbon bonds are present such as the CF2 and CF3. This is reflected in the intensity of the bond stretching observed above 1170 cm−1 in both membranes. Furthermore, the spectra of M1 and M3 exhibit notable similarities. Given the short electrospraying duration, full surface coverage of PAN + CuO is unlikely (in M3). Consequently, ATR-mode FT-IR analysis of M3 subsequently reflect the spectra of M1, with additional peaks corresponding to the presence of PAN on the surface.
Janus membrane interface analysis (Interlocking action)
This study investigated the interaction between the active hydrophobic surface and the support hydrophilic layer of a multi-layer Janus membrane. The weak interaction between the NIPS-cast membrane and the electro-spun PEI layer often results in delamination, compromising the membrane’s performance in hypersaline treatment. To address this, a gecko-inspired adhesion mechanism was developed to engineer a robust interface between these layers. To achieve the desired interface properties, a highly porous, thin active PVDF surface was developed using substrate-induced NIPS. The interface was further engineered using PVDF-HFP and CuO, leveraging their highly hydrophobic and polarizable nature. Measurements from SEM images (Fig. S1) using ImageJ software revealed that the average spacing on the electro-sprayed particles’ surface was approximately 3.47 μm, while the average diameter of the electrospun PEI fibers was 1.30 μm. This size ratio allowed the M2 surface to effectively accommodate, penetrate, and anchor the nanofibrous layers. The penetration and anchoring of fibers were further facilitated by the strength of the electrospinning potential, which ensures sufficient ejection and deposition force. To validate the importance of surface engineering, the hydrophilic, polarizable membrane (M3) composed of PAN and CuO particles demonstrates a possible weak interaction due to the formation of a beaded thin fabric structure with high packing irregularities, typically due to low concentration and higher operational voltage58,59. The resulting low wettability of this layer also contradicted the gecko-type adhesion behavior, further highlighting the significance of surface modifications introduced in M2.
The adhesion behavior of the M4 multilayer Janus membrane was evaluated using a peeling test. Physical force was applied to peel the PEI layer deposited on the rough M2 surface, and the peeled-up surface was analyzed via SEM to understand the proposed adhesion mechanism (Fig. 5). The results revealed that the fibrous PEI network was intermingled with the rough, granular surface of the M2 membrane. Several factors contributed to the observed adhesion mechanism, including mechanical action, surface roughness enhancement, and frictional forces (Fig. 5b). To begin with, multiple contact points could be observed from PEI nanofibers anchored and held fixed within the textured sprayed surface, which can improve the mechanical strength of this physical interaction. Also, the granular structure of the electro-sprayed particles fixed on the M1 surface provided a high surface area that can enhance the penetration of the deposited fibers, leading to stronger physical adhesion. Another reinforcement worthy of explanation is the frictional force that enhances the resistance of fibers shearing, sliding or detaching from the interlocked points, a feature like the gecko-type adhesion. The FTIR analysis of the peeled interface of the Janus membrane indicated plethora of peaks belonging to both PVDF-HFP in the electrosprayed layer and PEI fibers from the support (Fig. S6). Furthermore, the interaction of fibers and particles in the interfacial zone was also confirmed from the WCA shown in Fig. 5a3 which drops to 112.2o from 151.8o due to the anchored hydrophilic PEI fibers.
Comparatively, the peeled surfaces of M5 and M6 membranes under SEM are shown in Fig. S7. The image revealed significant differences in fiber distribution, anchorage density, and interfacial architecture, which can profoundly influence membrane stability and performance in membrane distillation applications. M5 displays a relatively sparse fibrous distribution with fewer and poorer anchoring points across the surface. This morphology suggests weak adhesion between the layers, potentially predisposing the membrane to interfacial separation during prolonged use. M6 membrane on the other hand demonstrated fibers bridging at various points on the surface. However, this entanglement is very weak and unsupported and can easily be disturbed especially in the WGMD system, where the presence of water in the gap will pose a threat to the wetting and detachment of layers. Collectively, these observations highlight the critical role of fiber architecture and interfacial morphology in determining the structural integrity of Janus Membranes. The interlocking fiber network in M4 likely promotes superior adhesion and mechanical robustness, positioning it as a promising candidate for high-performance membrane distillation systems.
The Janus multilayer membrane (M4) cross-sectional image is shown in Fig. 6, which shows distinct morphologies between the layers and interlocking structures. EDS elemental composition and mapping of the interface between hydrophobic and hydrophilic layers shows the distribution of the main components of the PVDF (C, F) and PEI (C, N) polymer and the presence of CuO particles at the interface. Furthermore, the EDS mapped images show a sharp change in the elemental distribution (especially F), which verifies the interfacial region in the Janus membrane.
a Janus membrane interface cross sectional SEM image. b EDS spectra obtained from a region around the Janus membrane interface highlighting the elemental composition. c EDS mapping highlighting the distribution of elements at the interface of Janus membranes. d Janus membrane cross-section SEM image.
Janus membrane interfacial layer polarization
The zeta potential data for the Janus membrane surfaces near the interfacial layer at different pH values are displayed in Fig. 7a. It was found that under both basic and acidic conditions, the M1 membrane skin surface maintains a negative zeta potential value. At neutral conditions, the zeta potential value was nearly −17 mV, indicating a stronger negative potential. Over a broad pH range, the electrosprayed PVDF-HFP+CuO membrane (M2) retains positive zeta potential, though it does change only at higher pH levels to neutral or slightly negative values60. The interaction of CuO nanoparticles with the PVDF-HFP matrix is likely the cause of this phenomenon, which points to a surface rich in polarizable functional groups. On the other hand, CuO was recognized as a polar substance that can contribute to surface polarization due to its localized surface charges61. In contrast, dipole formations were shown to be supported by PVDF-HFP, which forms the rough matrix, when it encounters charged species. Therefore, this polarizability can help make rough surfaces more dynamically sensitive to interactions.
In line with these results, it is anticipated that polyetherimide, which was described as having a significantly negative potential, will interact more favorably with M2 surface than with M162. Strong adhesion is promoted by an electrostatic interaction between the negatively charged PEI fibers and the positive zeta potential of M2 at lower pH values. Van der Waals forces and polar contacts can still promote advantageous interfacial interactions at higher pH levels, when M2 is closer to a neutral zeta potential. M1, on the other hand, would mostly encounter repulsive electrostatic forces due to its continuously negative zeta potential over the pH range, which would limit its ability to interact with PEI.
Moreover, the polarizability of the interfacial layer could be explained relative to the PVDF phases present at the interface. Out of many phases known for PVDF-HFP, the β-phase appeared to be highly electro-active which is attributed to the alignment of dipoles in its configuration63. This configuration which is referred to as TTTT state was reported in the literature to have the maximum dipole moment and hence could increase the interactions between dipoles64. As can be seen in Fig. 7b, the FT-IR spectra of the M2 membrane surface show distinctive peaks at 1400 cm−1, 1276 cm−1 and 840 cm−1 which corresponds to the β-phase and other peaks at 762 cm−1 and 975 cm−1 correspond to the α-phase. Noticeably, the peaks of the β-phase were found to be highly intense while the α-peaks are hardly seen. To confirm the availability of the β-phase and the disappearance of α-phase in M2 membrane surface, XRD analysis was conducted, and the result was displayed in Fig. 7c. As can be observed, the peak at 20.02o was broad and clear and corresponds to the β-phase crystal planes while that just above 18o corresponds to the α-phase crystal planes63. These findings suggest that the proposed interface in this work is highly polarized and electroactive which opens to various physical dipolar interactions. Hence, the CuO and PVDF-HFP layer deposited through the electro-spraying process provided a polarizable and superhydrophobic surface. The ionic character of CuO and PVDF-HFP’s dipolar nature are the causes of the polarizability, respectively65,66.
Membrane bulk properties
One of the crucial factors in determining membrane performance in MD is liquid entry pressure, which also assesses the membrane’s pore wetting resistance. From Table 1, the single-layer membrane (M1) recorded a LEP of 1.6 bar. This value agrees with Franken’s equation related to the maximum pore size and the water contact angle of the active surface, as well as with the desired reported LEP values in the literature for a long-term operation of membranes67,68. The concentration of the host active polymer and the higher WCA produced, as explained in the preceding sections, resulted in the higher LEP. As a result, the deposition of the interfacial layer in M2 and M3 membranes resulted in a slight increase in LEP to 1.7 bar. This can be attributed to the electro-sprayed particles, which could obstruct a portion of the membrane’s surface porosity. Furthermore, it was observed that the LEP values further increased with the co-deposition of the electro-spun PEI fibers in multi-layer membranes due to the decrease in pore size and increase in membrane thickness consequently increasing the density of the membranes and LEP (Table 1). The modest rise suggests that membrane-linked pores are not significantly affected by the hydrophilic PEI layer.
The pore size distribution (PSD) data for all developed membranes is illustrated in Fig. 8a, which depicts narrow PSD in all membranes and relatively large pores in the initial membrane layer M1 attributed to the effects of casting substrate, NIPS process and membrane thickness13,44,69. However, as can be seen in Table 1, the net membrane thickness affects the mean pore size of the membranes due to the addition of subsequent layers. M4 and M5 Janus membranes with multiple layers exhibit higher thickness and smaller pore sizes, which results in higher LEP. The thickness of the support hydrophilic layer falls within the range of 60–70 µm and was evaluated by comparing the thickness of the active layer before and after co-deposition.
The stress-strain relationship of fabricated membranes with different layers (M1, M4, and M6) was examined to develop a better understanding of the interfacial influence on the mechanical stability of the developed Janus membrane. Table 2 provides details of Young’s modulus, maximum stress, and elongation at break of the examined multiple membranes. The single-layer membrane (M1) achieved a maximum stress of approx. 3.4 MPa and an elongation strain of approximately 28.4% as shown in Fig. 8b. Typically, PVDF flat sheet membranes through the NIPS process lead to a consistent (dense) composition and structure with effective stress distribution, responsible for the observed uniform deformation and smoothness in the curve44,70. The mechanical behavior of M4, a multiple-layer membrane with an interlocking interface, was distinct. Over a strain range of around 15–25%, the stress-strain curve showed a plateau region at approximately 3.20 MPa. After this point, M4 showed a progressive rise in strain, peaking at about 45.1% before failing due to the fibrous PEI layer. The interlocking interface appears to be essential for improving adhesion and promoting stress transmission between layers, allowing the membrane to withstand greater elongation. Nevertheless, the stress plateau indicates the existence of shear and interfacial friction forces, which would have counterbalanced the advantages of improved adhesion by adding resistance to deformation. Because of these characteristics, the triple-layer M4 membrane has demonstrated superior mechanical stability. Conversely, the M6 dual-layer membrane without an interfacial layer showed the least effective mechanical performance, revealing a sudden stress reduction, indicating premature failure in the layers, after reaching a maximum stress of about 2.4 MPa at a strain of about 25%. Under tensile stress, delamination resulted from ineffective load transfer between layers caused by a lack of interfacial connection deteriorates the mechanical response. These findings emphasize the importance of interfacial adhesion between a flat sheet and fiber support to preserve the structural integrity and enhance the mechanical characteristics of multilayer membranes.
Comparison of the interface interaction with Gecko mode adhesion
The naturally occurring microscopic adhesion system of geckos and the adhesion mechanism displayed by M2 surface and M4 multilayer membrane’s interfacial surface are quite comparable. Setae, which branch into even finer structures (spatulae) at their tips, are micro- and nanoscale structures used by geckos. By maximizing contact area, these hierarchical structures allow van der Waals forces to produce strong adhesion without the need for adhesives. Likewise, the roughened surface through electrospraying found in M2 membrane imitates this hierarchical structure. The granules function as a microscale counterpart of gecko setae, and the interconnecting fibers mimic the spatulae that embed themselves in the rough surface. For both systems to generate robust adhesion, a mixture of secondary interactions, contact area maximization, and surface roughness is required. The high value of water contact angle of M2 membrane surface emphasizes even more how similar the superhydrophobic qualities of geckos are to their ability to self-clean and retain adhesion in a variety of environments. Additionally, gecko adhesion is flexible and adaptable, enabling robust attachment on a variety of surfaces. Similar versatility is provided by the variable zeta potential in M2 surface, which can encourage contact in a variety of pH ranges and replicate the Gecko-type adaptable adhesive behavior41,42.
Membrane distillation evaluation
Using a bi-functional interfacial layer and successive layer-by-layer co-deposition, a delamination-free Janus membrane was developed in this research. To increase permeate flux during WGMD treatment of high-salinity water (up to 70,000 ppm), the main objectives were to improve water uptake and retain adhesion between the hydrophobic and hydrophilic layers during the MD test. The membrane’s performance was assessed using MD experiments, such as treating high-salinity feed for 24 h, long-term test operating for 60 h, and evaluating antifouling and anti-wetting properties. Following MD tests, other characterization methods were used to examine surface characteristics, including WCA measurements, SEM imaging, and EDS analysis. Firstly, the WGMD results with flux and salt rejection for all developed membranes are displayed in Fig. 9. After 24 h of testing, the single-layer M1 membrane showed a permeate flux of 21.70 kgm−2 h−1 and salt rejection of 99.97%. Because of the concomitant increase in the interfacial surface area of the membrane facing the feed side, surface roughness has been reported to improve membrane performance in MD71. According to earlier research, it is easier to induce roughness using a textured surface like sandpaper in the NIPS process than it is to use surface chemical modification, which raises the net cost of membrane fabrication and results in a less sustainable and environmentally friendly product13. On the other hand, dual-layer membranes involving CuO particles with hydrophobic (M2) and hydrophilic (M3) polymer additive demonstrated a flux of 26.61 kg m−2 h−1 with 99.99% and 27.3 kg m−2 h−1 with 99.92% salt rejection, respectively. Due to its higher thermal conductivity, the presence of CuO on the permeate side of the membrane was responsible for the abatement of temperature polarization and consequently increased in flux relative to the single-layer membrane (M1). CuO nanoparticles also improved selectivity through hierarchical architectures, improved adhesion and membrane stability under ideal spraying circumstances and composite ratios used in this work. According to previous research, nanoparticles enhance the fabrication of multilayer Janus membranes for MD applications by encouraging faster water absorption and reduced mass transfer resistance72. Zhang et al. demonstrated the use of ferrous oxide nanoparticles as adhesion materials, which enhanced performance through improved interfacial bonding24. Sarwar et al. explained the excellent interactions of CuO particles with the nanofibers matrix, which resulted in excellent mechanical property73.
Furthermore, the permeate flux of triple layer membranes M4 and M5 increased to 37.2 kg m−2 h−1 and 28.62 kg m−2 h−1, respectively, compared to their predecessors (M2 and M3). This improvement is attributed to the hydrophilic layer’s (PEI fibers) ability to draw vapor closer to the permeate side for condensation. With controlled membrane layer thicknesses and interfacial surfaces, the multilayer Janus membrane provided low heat and mass transfer resistance due to the intrinsic properties of CuO and PEI fibers. However, the performance difference between M4 and M5 was linked to the interaction between the hydrophilic fibers and the composite adhesion layer. While both membranes had identical PEI thicknesses, M5 exhibited a gradual decline in flux after 10 h due to early delamination of the PEI fibers. This delamination likely arose from weak physical interactions and interfacial stress caused by water retention in the membrane. In contrast, M4 maintained steady flux and strong salt rejection, demonstrating superior MD performance. As can be in Fig. S8a, certain regions around M5 membrane show detachments or poor attachment between the layers of the Janus membrane. However, the results from MD suggested that the layers detached completely as the performance drops. To support this claim, image of the detached layers in the MD module was taken and shown in Fig. S8b, c. One more important reason worthy of mentioning here is the hydrophilicity of the interfacial layer. As PAN and CuO both possesses inherent wettability, the accumulation of water at this region can exert stress and pressure on the support layer, leading to an ultimate washing and detachment.
On the other hand, the M6 Janus membrane without interfacial layer exhibited significantly lower performance showing gradual decrease in flux, underscoring the importance of the interfacial composite layer in enhancing adhesion and vapor flux. Hence, material selection played a critical role, as layer adhesion and effective heat transfer across the membrane surface diffused thermal gradients, ensuring uniform temperature distribution and enhancement in membrane performance in MD. The performance of M4 was benchmarked against previously reported Janus membranes in Table 3, confirming its suitability for treating highly saline water with optimal water permeation and rejection.
The improved flux performance of Janus membranes developed in this work can be understood from the design of the membrane layer. The increase in interfacial surface area on the feed side of the membrane has been shown to enhance membrane performance due to surface roughness. Greater membrane roughness can induce turbulence at the feed-membrane interface, disrupting both the thermal and concentration polarization layers. This disruption raises the temperature at the membrane-feed interface, thereby reducing the temperature gradient between the feed bulk and this interface, ultimately improving net thermal efficiency (Fig. S9). Consequently, this effect increases the temperature gradient between the membrane’s feed-side surface and the permeate bulk. Considering various optimization parameters, including thermodynamic properties and mechanical stability, a Janus membrane was designed with a rough hydrophobic PVDF active layer, a thin interlayer of PVDF-HFP-CuO, and a hydrophilic PEI layer. The hydrophobic PVDF layer with thin interfacial surface enhances vapor transport due to the higher thermal conductivity of CuO, while the hydrophilic PEI, with lower thermal conductivity, helps maintain a strong heat gradient across the membrane, further improving thermal efficiency. More so, Janus membranes in WGMD are susceptible to improved flux due to the presence of a hydrophilic layer which favors and facilitate faster condensation of vapor and as such reducing the mass transfer distance resulting in a high flux performance.
Membrane wetting resistance evaluation (long-term MD test)
The fabricated Janus membranes with interfacial layers (M4 and M5) were assessed under prolonged WGMD test. After 48 h of operation, the M5 membrane showed a flux of 27.15 kg m−2 h−1 and 99.90% salt rejection (Fig. 10). A sharp decline in flux was noted in M5 membrane after 10 h indicating delamination of hydrophilic layer due to poor adhesion provided by the interfacial layer composite involving PAN and CuO. However, over 60 h of WGMD testing, the M4 Janus membrane maintained a flux of 36.23 kg m−2 h−1 and a salt rejection of 99.97%, indicating long-term stability. Since the interfacial/composite layer in Janus membrane (M4) is superhydrophobic with hierarchical granular structures as explained earlier, the long-term stability of the membrane is due to the robust hydrophobic layer of Janus membrane induced with self-cleaning effect from the synergistic impact of CuO particles as well as the strength of the electric field in the electrospraying process binding the particles in the matrix of the PVDF membrane. Consequently, the higher LEP and nominal pore size of the membrane (M4) provided stable rejection performance in MD. The observed drop in flux for the M4 membrane during prolonged operation was minimal and could be attributed to the combined effects of membrane surface wetting and the aggressive chloride ions (Cl−) present in the high-salinity feed. These factors likely induced minor surface modifications that marginally impacted flux over time. Notably, the M4 membrane exhibited no delaminated layers after long-term testing, with the layer attachment and thickness remaining nearly identical to their initial states before testing (Fig. S10). This result highlights the superior adhesion and structural integrity provided by the interfacial layer of PVDF-HFP and CuO in M4 membrane, which enabled it to outperform M5 in maintaining stable flux and salt rejection over an extended operational period. A schematic representation of M4 and M5 Janus membranes’ performance before and after MD is depicted in Fig. S11.
a, b Long-term WGMD performance of fabricated Janus membranes in terms of flux and rejection, respectively. SEM surface morphology and WCA (inset) of c active layer of Janus membranes and d PEI support after 60 h MD testing and e AFM imaging of the active layer of Janus membrane after long term testing. Where a Cartesian direction is shown with different colours indicating x-, y- and x-axis. The legend shows the coordinates in both directions.
It is worth mentioning that the hydrophobic active layer in M4 Janus membrane was stable after 60 h with minimal change in the surface porosity (from the SEM image) and WCA (105.2o) as seen in Fig. 10c. The membrane stability is also established from the surface roughness of the active hydrophobic layer after long term test, observed from the AFM image (Fig. 10e), where the Ra value of the active surface was measured as 135 nm. In a similar scenario, surface analysis was performed on the hydrophilic PEI layer; the interconnected fibers remained stable, and the morphology remained like that observed prior to the WGMD (Fig. 10d). A literature comparison is given in Table 3, highlighting the stability of the Janus membrane M4 in hypersaline treatment. In a similar vein, M4 membrane performance in desalination was compared with commercial PVDF membrane. Even though M4 membrane has comparable pore size to the commercial membrane used for comparison (~0.44 µm), the outstanding performance displayed by M4 was confirmed in terms of both flux and salt rejection (Fig. 11), highlighting the importance of developed Janus membrane in improving efficiency of MD process for desalination of highly saline water.
Membrane fouling and scaling evaluation
Two obstacles that have also hampered the integration of MD applications are membrane fouling and scaling, both of which have an impact on the permeate flux or rejection during MD evaluation. The anti-wetting characteristics of the membrane are frequently compromised, and its porous structure is destroyed when foulants are present in the feed stream. To assess the fouling and scaling properties of the membrane, MD operation was set up for the Janus membrane (M4) in a 70,000-ppm saline feed that was combined with 20 mM solution each of SDS, MgCl2, CaCl2 and Na2CO3. Figure 12a illustrates how, for various foulant combinations, the membrane’s flow stays nearly constant throughout the 24-h testing period, with saline rejection of >99%.
After the MD test, the membrane was removed from the module, dried, and imaged under SEM (Fig. 12b). The measurements of the water contact angle, and EDS were made and reported inclusively. The initial WCA of the active layer was reduced to 115°, demonstrating a slight decrease in hydrophobicity due to the foulants’ synergistic action. Although the porous nature of the membrane was not greatly affected, the decrease in the WCA of the active skin can be attributed to the interfacial energy increase, which is primarily contributed by the interaction of the ionic species with aggressive Cl− driven by the thermal effect from the hot feed. As shown on the EDS spectrum in Fig. 12c, almost no traces of elements on the membrane surface originates from the foulants, except few traces of S from SDS and Mg, which might be attributed to the small ionic size and high ionic charge of Mg ions. The Janus membrane reported in this work was compared with a brief description of prior fouling results from the literature in Table 4. The superior interaction between the layers, as well as the wettability of the active membrane surface in the created Janus membrane, provided stable flux and consistent rejection under a combination of foulants in the MD test.
Discussion
The proposed bio-inspired multilayer Janus membrane demonstrates a breakthrough in desalination performance by addressing critical challenges in Janus membrane fabrication such as delamination. A robust interfacial layer engineered with a gecko-inspired adhesion mechanism was achieved by electrospraying a composite consisting of PVDF-HFP+CuO between a PVDF flat sheet membrane and PEI electrospun fiber support. The synergistic influence of CuO particles in the interface and affinity of active and support layers provided enhanced and stable performance in WGMD, displaying a flux of 36.23 kg m−2 h−1 and 99.97% saline rejection over 60 h of continuous operation. This highlights the significance of strategic material selection and interface engineering in advancing desalination technologies, positioning this membrane design as a sustainable and efficient solution for treating hypersaline water.
Methods
Materials
Polyvinylidene fluoride (PVDF) (Mw ~534,000 by GPC, powder), polyetherimide (PEI), lithium chloride (LiCl) (≥99.98 trace metal basis), copper (ii) oxide (CuO) (nanopowder, <50 nm particle size), poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP) (Mw ~400,000), poly(acrylonitrile) (PAN) (Mw ~ 150,000), Dimethylformamide (DMF, 99.9%), N-methyl-pyrrolidone (NMP), and 2-propanol (Isopropanol, 99.9%) were all purchased from Sigma-Aldrich and used without any modifications. De-ionized water from a laboratory-sized Millipore system was used in membrane fabrication and testing.
Methodology
A predetermined amount of PVDF powder was weighed and oven-treated at 60 °C for 1 h to eliminate any residual moisture. From this treated powder, 3.4 g was measured and added to a 2 wt.% LiCl solution prepared in DMF. The mixture was continuously agitated at 200 rpm for 24 hours to produce a homogeneous dope solution with a final PVDF concentration of 17 wt.%. The resulting dope solution was allowed to degas at room temperature before being cast on a 400-grit sandpaper casting substrate using a doctor blade. To ensure efficient phase separation, the solution was cast at a thickness of 120 µm and immediately immersed in a coagulation bath containing a warm water-isopropanol mixture (80:20 v/v) maintained at a temperature of 35 ± 0.3 °C. After successful phase inversion, the membrane was cleaned in deionized (DI) water for 24 hours while still attached to the substrate to remove residual solvents (Fig. 13a). After drying at room temperature, the resulting membrane, referred to as M1, was subsequently detached from the sandpaper substrate for further processing. This method of using sandpaper casting substrate provided a uniform flat sheet, unsupported porous membrane as reported in other studies13,44.
The fabricated single-layer flat-sheet membrane, designated as M1, was placed on a flat plate with the skin side facing up. A composite solution, prepared by mixing CuO and PVDF-HFP in a 5:5 wt.% ratio in a DMF solvent, was loaded into a 10 mL syringe for electro-spraying. The solution was sprayed for 15 minutes at a high voltage of 22.0 kV and a flow rate of 1.5 mL/h. The 15-minute spraying duration was determined as optimal based on the surface morphology and wettability investigation conducted at intervals of 15, 30, and 60 minutes. After spraying, the resulting membrane, labeled M2, was dried in an oven at 60°C. Similarly, another membrane (M3) was fabricated for comparison using the same spraying conditions but with a composite solution of PAN and CuO in the same 5:5 wt.% ratio.
For the support layer deposition, membranes M1, M2, and M3 were subsequently mounted onto a rotating drum covered with aluminum foil in an electrospinning apparatus to create Janus membranes. The membranes obtained after electrospinning were labeled as M4, M5, and M6, respectively. A 20 wt.% PEI solution, prepared by dissolving 2.0 g of PEI polymer in 8.0 g of NMP, was used for fiber spinning. Electrospinning was conducted for 3 hours with the following parameters: a flow rate of 1.0 mL/h, a voltage of 13.5 kV, and a drum rotation speed of 100 rpm (Fig. 13b). To evaluate the role of the interfacial layer, the M6 membrane was fabricated with PVDF active layer and PEI support layer without any electrosprayed layer in between. A summary of the fabricated membranes and their compositions is provided in Table 5.
Membrane characterization
The surface morphology of all membrane layers was analyzed using scanning electron microscopy (SEM, JEOL JSM-6610LV). Prior to imaging, the membrane surface was sputter-coated with gold using a DII-29030SCTR smart coater to ensure conductivity. SEM imaging was performed at varying magnifications to capture detailed morphological features. To further investigate the elemental composition of the membrane layers, energy-dispersive spectroscopy (EDS) was conducted in conjunction with SEM. Additionally, atomic force microscopy (AFM) (Nanosurf) was also used to evaluate the membrane surface topography and surface roughness.
The chemical composition of the membrane surfaces was characterized using Fourier Transform Infrared (FT-IR) spectroscopy (Bruker INVENIO series). FT-IR spectra were recorded within the wavenumber range of 600–4000 cm⁻¹ to identify characteristic peaks associated with membrane surfaces. Furthermore, the interfacial polarizable characteristics were studied using the X-Ray Diffraction (XRD) (Rigaku Ultima IV) and Zeta potential analyzer.
The wettability of the membrane surfaces was evaluated using water contact angle (WCA) measurements. The WCA was determined using the sessile drop method, where approximately 5 µL of water droplet was dispensed and measurement was performed automatically using Famas software. The average water contact angle was calculated based on measurements taken at several points on the membrane surface.
The hydrophobicity of fabricated membranes was also assessed through liquid entry pressure (LEP) using a custom laboratory setup designed to measure the penetration pressure of water. The LEP was determined by gradually applying pressure to an enclosed water chamber in contact with the membrane surface, following the methodology described by Smolder and Franklin [45]. The LEP corresponds to the minimum pressure at which water penetrates the membrane pores and forms visible droplets on the opposite side. The membrane thickness was measured using a LITEMATIC VL-50A precision measurement device (Mitutoyo, Japan) at multiple points to ensure accuracy and consistency. The pore size distribution (PSD) of the membranes was analyzed using a porometer (3Gzh, Quantachrome Instruments, USA). The measurement involved forcing ethanol out of the membrane pores with controlled gas pressure, allowing the PSD to be determined. A discovery hybrid rheometer HR30 from TA Instruments with a tensile deformation module was used to assess the mechanical stability of the developed membranes. Samples were clamped meticulously and tested at a constant rate of 50 µm/s.
WGMD performance test
The performance of the fabricated membranes was evaluated using a laboratory-scale water gap membrane distillation system with a 5 mm gap distance, as shown in Fig. S12. The feed solution, containing 70,000 ppm NaCl, was heated to approximately 70 °C using a Julabo 601F heater tank, which automatically regulated the feed temperature and was connected to the membrane distillation (MD) module. The opposing side of the module, containing deionized water, was connected to a chiller maintained at 20 °C to cool a conduction plate. Water vapor permeating through the membrane pores is condensed in the water gap touching the plate. Support with perforations was utilized to rest the membrane and provide the needed gap in the module. Flow meters were installed on both the feed and permeate sides to regulate and monitor flow rates into and out of the system. Detailed test conditions of WGMD is provided in Table S1. The net distillation permeate flux was calculated using Eq. 1, which incorporates the weight of the collected water, the effective membrane area, and the duration of the test. The weight of permeated water was measured using a high-precision Ohaus PR2200 weighing scale connected to a computer for real-time data acquisition. The salt rejection performance was evaluated using a Hanna HI5321 conductivity/TDS device. The saline rejection percentage was calculated using Eq. 2, which relates the feed and permeate conductivity measurements recorded per minute. The permeate flux ‘J’ in (kg/m2h) is calculated as
where M is the mass of the permeate collected through the membrane in kg, A is the effective membrane area in m2, and t is the time taken for the testing in hours:
where S.R is the salt rejection (%), \({C}_{f}\) and \({C}_{p}\) are the concentration of feed and permeate in ppm, respectively.
Data availability
The authors declare that all data supporting the findings of this study are available within the article and attached Supplementary Information file.
References
Hosseini, Z. & Kargari, A. Fabrication and characterization of hydrophobic/hydrophilic dual-layer polyphenyl sulfone Janus membrane for application in direct contact membrane distillation. Desalination 571, 117100 (2024).
Li, M., Lu, K. J., Wang, L., Zhang, X. & Chung, T.-S. Janus membranes with asymmetric wettability via a layer-by-layer coating strategy for robust membrane distillation. J. Membr. Sci. 603, 118031 (2020).
Niknejad, A. S., Bazgir, S. & Kargari, A. Desalination by direct contact membrane distillation using a superhydrophobic nanofibrous poly (methyl methacrylate) membrane. Desalination 511, 115108 (2021).
Zare, S. & Kargari, A. in Emerging Technologies for Sustainable Desalination Handbook 107–156 (Elsevier, 2018). https://doi.org/10.1016/B978-0-12-815818-0.00004-7.
Li, C. et al. Antiwetting and antifouling janus membrane for desalination of saline oily wastewater by membrane distillation. ACS Appl Mater. Interfaces 11, 18456–18465 (2019).
Shirazi, M. M. A., Quist-Jensen, C. A. & Ali, A. in Experimental Methods for Membrane Applications in Desalination and Water Treatment 97–138 (IWA Publishing, 2024). https://doi.org/10.2166/9781789062977_0097.
Huang, Y.-X., Wang, Z., Hou, D. & Lin, S. Coaxially electrospun super-amphiphobic silica-based membrane for anti-surfactant-wetting membrane distillation. J. Membr. Sci. 531, 122–128 (2017).
Hussain, A., Janson, A., Matar, J. M. & Adham, S. Membrane distillation: recent technological developments and advancements in membrane materials. Emergent Mater. 5, 347–367 (2022).
Shirazi, M. M. A., Kargari, A., Tabatabaei, M., Ismail, A. F. & Matsuura, T. Concentration of glycerol from dilute glycerol wastewater using sweeping gas membrane distillation. Chem. Eng. Process.: Process Intensif. 78, 58–66 (2014).
Zare, S. & Kargari, A. CFD simulation and optimization of an energy-efficient direct contact membrane distillation (DCMD) desalination system. Chem. Eng. Res. Des. 188, 655–667 (2022).
Namdari, M., Zokaee Ashtiani, F. & Bonyadi, E. Development of a high flux Janus PVDF membrane for oily saline water desalination by membrane distillation via PDA-TEOS-APTES surface modification. Desalination 572, 117139 (2024).
Li, H., Feng, H., Li, M. & Zhang, X. Engineering a covalently constructed superomniphobic membrane for robust membrane distillation. J. Membr. Sci. 644, 120124 (2022).
Baroud, T. N. Tuning PVDF membrane porosity and wettability resistance via varying substrate morphology for the desalination of highly saline water. Membranes (Basel) 13, 395 (2023).
Zheng, R. et al. Preparation of omniphobic PVDF membrane with hierarchical structure for treating saline oily wastewater using direct contact membrane distillation. J. Membr. Sci. 555, 197–205 (2018).
Hu, J. et al. Anisotropic gypsum scaling of corrugated polyvinylidene fluoride hydrophobic membrane in direct contact membrane distillation. Water Res. 244, 120513 (2023).
Zhang, W., Hu, B., Wang, Z. & Li, B. Fabrication of omniphobic PVDF composite membrane with dual-scale hierarchical structure via chemical bonding for robust membrane distillation. J. Membr. Sci. 622, 119038 (2021).
Li, C. et al. Superhydrophobic PVDF membrane modified by dopamine self-polymerized nanoparticles for vacuum membrane distillation. Sep. Purif. Technol. 304, 122182 (2023).
Meng, L. et al. Janus membranes at the water-energy nexus: a critical review. Adv. Colloid Interface Sci. 318, 102937 (2023).
Gryta, M. Effectiveness of water desalination by membrane distillation process. Membranes (Basel) 2, 415–429 (2012).
El-Bourawi, M. S., Ding, Z., Ma, R. & Khayet, M. A framework for better understanding membrane distillation separation process. J. Membr. Sci. 285, 4–29 (2006).
Zhang, R., Sun, Y., Guo, Z. & Liu, W. Janus membranes with asymmetric wettability applied in oil/water emulsion separations. Adv. Sustain. Syst. 5, (2021).
Zhou, H. & Guo, Z. Superwetting Janus membranes: focusing on unidirectional transport behaviors and multiple applications. J. Mater. Chem. A Mater. 7, 12921–12950 (2019).
Li, H., Yang, J. & Xu, Z. Asymmetric surface engineering for Janus membranes. Adv. Mater. Interfaces 7 (2020).
Zhang, W. et al. Preparation Janus membrane via polytetrafluoroethylene membrane modification for enhanced performance of vacuum membrane distillation desalination. Sep. Purif. Technol. 313, 123465 (2023).
Yang, H.-C., Zhong, W., Hou, J., Chen, V. & Xu, Z.-K. Janus hollow fiber membrane with a mussel-inspired coating on the lumen surface for direct contact membrane distillation. J. Membr. Sci. 523, 1–7 (2017).
Mohammadi Ghaleni, M. et al. Fabrication of Janus membranes for desalination of oil-contaminated saline water. ACS Appl Mater. Interfaces 10, 44871–44879 (2018).
Afsari, M., Shon, H. K. & Tijing, L. D. Janus membranes for membrane distillation: recent advances and challenges. Adv. Colloid Interface Sci. 289, 102362 (2021).
Lou, M. et al. Robust dual-layer Janus membranes with the incorporation of polyphenol/Fe3+ complex for enhanced anti-oil fouling performance in membrane distillation. Desalination 515, 115184 (2021).
Xia, Q.-C. et al. A hydrophilicity gradient control mechanism for fabricating delamination-free dual-layer membranes. J. Membr. Sci. 539, 392–402 (2017).
Meng, J. et al. Compatibilizing hydrophilic and hydrophobic polymers via spray coating for desalination. J. Mater. Chem. A: Mater. 8, 8462–8468 (2020).
Yang, H. et al. Janus membranes: creating asymmetry for energy efficiency. Adv. Mater. 30 (2018).
Su, R. et al. Biomimetic Janus membrane with unidirectional water transport ability for rapid oil/water separation. Sep Purif. Technol. 277, 119423 (2021).
Yang, H.-C. et al. Mussel-inspired modification of a polymer membrane for ultra-high water permeability and oil-in-water emulsion separation. J. Mater. Chem. A 2, 10225–10230 (2014).
Zuo, J., Chung, T.-S., O’Brien, G. S. & Kosar, W. Hydrophobic/hydrophilic PVDF/Ultem® dual-layer hollow fiber membranes with enhanced mechanical properties for vacuum membrane distillation. J. Membr. Sci. 523, 103–110 (2017).
Zahid, M., Rashid, A., Akram, S., Rehan, Z. A. & Razzaq, W. A Comprehensive review on polymeric nano-composite membranes for water treatment. J. Membr. Sci. Technol. 08, (2018).
Baghbanzadeh, M., Rana, D., Matsuura, T. & Lan, C. Q. Effects of hydrophilic CuO nanoparticles on properties and performance of PVDF VMD membranes. Desalination 369, 75–84 (2015).
Aniagor, C. O. et al. CuO nanoparticles as modifiers for membranes: a review of performance for water treatment. Mater. Today Commun. 32, 103896 (2022).
Said, B., M’rabet, S., Hsissou, R. & El Harfi, A. Synthesis of new low-cost organic ultrafiltration membrane made from Polysulfone/Polyetherimide blends and its application for soluble azoic dyes removal. J. Mater. Res. Technol. 9, 4763–4772 (2020).
Kaleekkal, N. J., Thanigaivelan, A., Rana, D. & Mohan, D. Studies on carboxylated graphene oxide incorporated polyetherimide mixed matrix ultrafiltration membranes. Mater. Chem. Phys. 186, 146–158 (2017).
Song, H.-M. et al. Asymmetric Janus membranes based on in situ mussel-inspired chemistry for efficient oil/water separation. J. Membr. Sci. 573, 126–134 (2019).
Autumn, K. et al. Adhesive force of a single gecko foot-hair. Nature 405, 681–685 (2000).
Autumn, K. et al. Evidence for van der Waals adhesion in gecko setae. Proc. Natl Acad. Sci. 99, 12252–12256 (2002).
Guillen, G. R., Pan, Y., Li, M. & Hoek, E. M. V. Preparation and characterization of membranes formed by nonsolvent induced phase separation: a review. Ind. Eng. Chem. Res. 50, 3798–3817 (2011).
Azeem, M. A., Lawal, D. U., Al Abdulgader, H. & Baroud, T. N. Enhanced performance of superhydrophobic polyvinylidene fluoride membrane with sandpaper texture for highly saline water desalination in air-gap membrane distillation. Desalination 528, 115603 (2022).
Chen, M. et al. Preparation of PVDF membrane via synergistically vapor and non-solvent-induced phase separation. Appl. Water Sci. 12, 161 (2022).
Zheng, L., Wu, Z., Zhang, Y., Wei, Y. & Wang, J. Effect of non-solvent additives on the morphology, pore structure, and direct contact membrane distillation performance of PVDF-CTFE hydrophobic membranes. J. Environ. Sci. 45, 28–39 (2016).
Wu, Z., Zheng, K., Zhang, G., Huang, L. & Zhou, S. Preparation of polysulfone-based nanofiber Janus membrane for membrane distillation containing organic pollutants. NPJ Clean. Water 7, 51 (2024).
Muchtar, S., Wahab, M. Y., Mulyati, S., Arahman, N. & Riza, M. Superior fouling resistant PVDF membrane with enhanced filtration performance fabricated by combined blending and the self-polymerization approach of dopamine. J. Water Process Eng. 28, 293–299 (2019).
Ghorabi, S. et al. Development of a novel dual-bioinspired method for synthesis of a hydrophobic/hydrophilic polyethersulfone coated membrane for membrane distillation. Desalination 517, 115242 (2021).
Ravi, J. et al. Polymeric membranes for desalination using membrane distillation: a review. Desalination 490, 114530 (2020).
Lin, Y. et al. Development of Janus membrane with controllable asymmetric wettability for highly-efficient oil/water emulsions separation. J. Membr. Sci. 606, 118141 (2020).
Yang, H. et al. Janus polyvinylidene fluoride membranes with controllable asymmetric configurations and opposing surface wettability fabricated via nanocasting for emulsion separation. Colloids Surf. A: Physicochem Eng. Asp. 616, 126120 (2021).
Pyo, M., Lee, K. H. & Lee, E.-J. Enhancing hydrophobicity through polydimethylsiloxane-blended poly(vinylidene fluoride-co-hexafluoropropylene) electrospun membranes: a well-designed approach. Polymer (Guildf.) 307, 127261 (2024).
Chang, J. & Kang, S. W. CO 2 separation through poly(vinylidene fluoride-co-hexafluoropropylene) membrane by selective ion channel formed by tetrafluoroboric acid. Chem. Eng. J. 306, 1189–1192 (2016).
Korkut, I. & Aydin, E. S. Electrospun PAN-PS membranes with improved hydrophobic properties for high-performance oil/water separation. Sep Purif. Technol. 331, 125590 (2024).
Zhang, B. et al. Facile synthesis of TiO2-PAN photocatalytic membrane with excellent photocatalytic performance for uranium extraction from seawater. Sep. Purif. Technol. 328, 125026 (2024).
Wang, J., Nie, L., Zhang, C. & Wang, B. Free radical graft polymerization of hydroxyethyl methacrylate and acrylic acid on polyetherimide membrane surface via redox system to improve anti-fouling and oil resistance performance. Mater. Today Commun. 38, 108197 (2024).
Jia, W., Kharraz, J. A., Guo, J. & An, A. K. Superhydrophobic (polyvinylidene fluoride-co-hexafluoropropylene)/ (polystyrene) composite membrane via a novel hybrid electrospin-electrospray process. J. Membr. Sci. 611, 118360 (2020).
Jia, W., Kharraz, J. A., Sun, J. & An, A. K. Hierarchical Janus membrane via a sequential electrospray coating method with wetting and fouling resistance for membrane distillation. Desalination 520, 115313 (2021).
Metwally, S. & Stachewicz, U. Surface potential and charges impact on cell responses on biomaterials interfaces for medical applications. Mater. Sci. Eng. C 104, 109883 (2019).
Mohamed, E. A. Green synthesis of copper & copper oxide nanoparticles using the extract of seedless dates. Heliyon 6, e03123 (2020).
Gholami, F., Asadi, A., Dolatshah, M. & Nazari, S. Modeling and optimization of surface modification process of ultrafiltration membranes by guanidine-based deep eutectic solvent. Heliyon 11, e41432 (2025).
Liu, Y. et al. Phase separation of a PVDF–HFP film on an ice substrate to achieve self-polarisation alignment. Nano Energy 106, 108082 (2023).
Shi, S. et al. Three-dimensional polypyrrole induced high-performance flexible piezoelectric nanogenerators for mechanical energy harvesting. Compos. Sci. Technol. 219, 109260 (2022).
Gradišar Centa, U., Mihelčič, M., Bobnar, V., Remškar, M. & Slemenik Perše, L. The effect of PVP on thermal, mechanical, and dielectric properties in PVDF-HFP/PVP thin film. Coatings 12, 1241 (2022).
Alotaibi, B. M., Altuijri, R., Atta, A., Abdeltwab, E. & Abdelhamied, M. M. Fabrication, structure and optical characteristics of CuO/polymer nanocomposites materials for optical devices. Int. J. Polym. Anal. Charact. 29, 562–572 (2024).
Guillen-Burrieza, E., Servi, A., Lalia, B. S. & Arafat, H. A. Membrane structure and surface morphology impact on the wetting of MD membranes. J. Membr. Sci. 483, 94–103 (2015).
Yazgan-Birgi, P., Hassan Ali, M. I. & Arafat, H. A. Estimation of liquid entry pressure in hydrophobic membranes using CFD tools. J. Membr. Sci. 552, 68–76 (2018).
Shekari, R., Dehban, A. & Kargari, A. Fabrication of PVDF membranes via VIPS-NIPS technique for water desalination: effect of preparation condition. Desalination Water Treat. 319, 100447 (2024).
Ji, H. et al. Janus membrane prepared via one step depositing coatings onto PVDF/PDMS membrane for simultaneous antiwetting and antifouling in DCMD. Desalination 539, 115964 (2022).
Kuang, Z., Long, R., Liu, Z. & Liu, W. Analysis of temperature and concentration polarizations for performance improvement in direct contact membrane distillation. Int J. Heat. Mass Transf. 145, 118724 (2019).
Farid, M. U. et al. Advancements in nanoenabled membrane distillation for a sustainable water‐energy‐environment nexus. Adv. Mater. 36, e2307950 (2024).
Sarwar, M. N. et al. Electrospun PVA/CuONPs/bitter gourd nanofibers with improved cytocompatibility and antibacterial properties: application as antibacterial wound dressing. Polymer (Basel) 14, 1361 (2022).
Khayet, M., García-Payo, M. C., García-Fernández, L. & Contreras-Martínez, J. Dual-layered electrospun nanofibrous membranes for membrane distillation. Desalination 426, 174–184 (2018).
Liao, Y., Loh, C.-H., Wang, R. & Fane, A. G. Electrospun superhydrophobic membranes with unique structures for membrane distillation. ACS Appl. Mater. Interfaces 6, 16035–16048 (2014).
Lalia, B. S., Guillen-Burrieza, E., Arafat, H. A. & Hashaikeh, R. Fabrication and characterization of polyvinylidenefluoride-co-hexafluoropropylene (PVDF-HFP) electrospun membranes for direct contact membrane distillation. J. Membr. Sci. 428, 104–115 (2013).
Woo, Y. C. et al. Electrospun dual-layer nonwoven membrane for desalination by air gap membrane distillation. Desalination 403, 187–198 (2017).
Zhu, Z. et al. Monolithic and self-roughened Janus fibrous membrane with superhydrophilic/omniphobic surface for robust antifouling and antiwetting membrane distillation. J. Membr. Sci. 615, 118499 (2020).
Prince, J. A. et al. Nanofiber based triple layer hydro-philic/-phobic membrane—a solution for pore wetting in membrane distillation. Sci. Rep. 4, 6949 (2014).
Meng, L., Chen, X., Cai, T., Tong, X. & Wang, Z. Surface energy-induced anti-wetting and anti-fouling enhancement of Janus membrane for membrane distillation. Water Res. 263, 122176 (2024).
Li, W. et al. Depositing of PVAM and GA onto PVDF/POTS-SiO2 to fabricate a Janus membrane for direct contact membrane distillation (DCMD). Desalination 582, 117661 (2024).
Zheng, L. et al. A novel Cu-BTC@PVA/PVDF Janus membrane with underwater-oleophobic/hydrophobic asymmetric wettability for anti-fouling membrane distillation. Sep. Purif. Technol. 299, 121807 (2022).
Feng, D., Li, X. & Wang, Z. Comparison of omniphobic membranes and Janus membranes with a dense hydrophilic surface layer for robust membrane distillation. J. Membr. Sci. 660, 120858 (2022).
Shi, D., Gong, T., Wang, R., Qing, W. & Shao, S. Control the hydrophilic layer thickness of Janus membranes by manipulating membrane wetting in membrane distillation. Water Res. 237, 119984 (2023).
Ozbey-Unal, B., Balcik, C. & Van der Bruggen, B. Development of a novel hydrophilic SiO2/PVDF Janus membrane via different modification methods for robust antiwetting and antifouling membrane distillation. J. Water Process Eng. 54, 104021 (2023).
Acknowledgements
The authors would like to thank all anonymous reviewers and editors for their insightful comments and suggestions on revising and improving the quality of this research work. The authors would also like to acknowledge the support provided by King Fahd University of Petroleum and Minerals (KFUPM) and Interdisciplinary Research Center for Membranes and Water Security through project INMW2107.
Author information
Authors and Affiliations
Contributions
Conceptualization, writing, editing and investigation was done by A.D.T.N.B acquired funding, provided resources, conducted reviews and project administrationM.A.A conceptualized the work, conducted reviews, editing and validation of the work.H.A and D.U.L contributed in investigation and reviewing.All authors have read and agreed to publish this version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Doguwa, A., Azeem, M.A., Ahmad, H. et al. Bio-inspired surface engineered multilayer Janus membrane for efficient desalination of highly saline water in membrane distillation. npj Clean Water 8, 60 (2025). https://doi.org/10.1038/s41545-025-00482-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41545-025-00482-2