Abstract
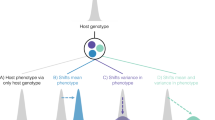
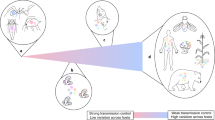
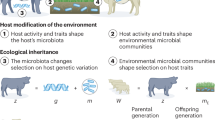
Microbiomes contribute to variation in many plant and animal traits, suggesting that microbiome-mediated traits could evolve through selection on the host. However, for such evolution to occur, microbiomes must exhibit sufficient heritability to contribute to host adaptation. Previous work has attempted to estimate the heritability of a variety of microbiome attributes. Here we show that most published estimates are limited to vertebrate and plant hosts, but significant heritability of microbiome attributes has been frequently reported. This indicates that microbiomes could evolve in response to host-level selection, but studies across a wider range of hosts are necessary before general conclusions can be made. We suggest future studies focus on standardizing heritability measurements for the purpose of meta-analyses and investigate the role of the environment in contributing to heritable microbiome variation. This could have important implications for the use of microbiomes in conservation, agriculture and medicine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McFall-Ngai, M. et al. Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl Acad. Sci. USA 110, 3229–3236 (2013).
Koskella, B. & Bergelson, J. The study of host-microbiome (co)evolution across levels of selection. Phil. Trans. R. Soc. B 375, 20190604 (2020).
Bisschop, K. et al. Microbiome heritability and its role in adaptation of hosts to novel resources. Front. Microbiol. 13, 703183 (2022).
Henry, L. P., Bruijning, M., Forsberg, S. K. G. & Ayroles, J. F. The microbiome extends host evolutionary potential. Nat. Commun. 12, 5141 (2021).
Levins, R. & Lewontin, R. The Dialectical Biologist (Harvard Univ. Press, 1985).
van Opstal, E. J. & Bordenstein, S. R. Rethinking heritability of the microbiome. Science 349, 1172–1173 (2015).
Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 (2018).
He, Y. et al. Exploring methods to summarize gut microbiota composition for microbiability estimation and phenotypic prediction in swine. J. Anim. Sci. 100, skac231 (2022).
Aliakbari, A. et al. Microbiability and microbiome-wide association analyses of feed efficiency and performance traits in pigs. Genet. Sel. Evol. 54, 29 (2022).
Zilber-Rosenberg, I. & Rosenberg, E. Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol. Rev. 32, 723–735 (2008).
Rosenberg, E. & Zilber-Rosenberg, I. The hologenome concept of evolution after 10 years. Microbiome 6, 78 (2018).
Moran, N. A. & Sloan, D. B. The hologenome concept: helpful or hollow? PLoS Biol. 13, e1002311 (2015).
Douglas, A. E. & Werren, J. H. Holes in the hologenome: why host-microbe symbioses are not holobionts. mBio 7, e02099-15 (2016).
Oliver, K. M., Degnan, P. H., Burke, G. R. & Moran, N. A. Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu. Rev. Entomol. 55, 247–266 (2010).
McFall-Ngai, M. J. The importance of microbes in animal development: lessons from the squid–Vibrio symbiosis. Annu. Rev. Microbiol. 68, 177–194 (2014).
Bolnick, D. I. et al. Major histocompatibility complex class IIb polymorphism influences gut microbiota composition and diversity. Mol. Ecol. 23, 4831–4845 (2014).
Feldman, M. W. & Ramachandran, S. Missing compared to what? Revisiting heritability, genes and culture. Phil. Trans. R. Soc. B 373, 20170064 (2018).
Raulo, A. et al. Social networks strongly predict the gut microbiota of wild mice. ISME J. 15, 2601–2613 (2021).
Meadow, J. F. et al. Humans differ in their personal microbial cloud. PeerJ 3, e1258 (2015).
Campos-Cerda, F. & Bohannan, B. J. M. The nidobiome: a framework for understanding microbiome assembly in neonates. Trends Ecol. Evol. 35, 573–582 (2020).
Tavalire, H. F. et al. Shared environment and genetics shape the gut microbiome after infant adoption. mBio 12, e00548-21 (2021).
Chapron, L., Lartaud, F., Le Bris, N., Peru, E. & Galand, P. E. Local variability in microbiome composition and growth suggests habitat preferences for two reef-building cold-water coral species. Front. Microbiol. 11, 275 (2020).
Roughgarden, J. Holobiont evolution: population theory for the hologenome. Am. Nat. 201, 763–778 (2023).
van Vliet, S. & Doebeli, M. The role of multilevel selection in host microbiome evolution. Proc. Natl Acad. Sci. USA 116, 20591–20597 (2019).
Week, B., Morris, A. H. & Bohannan, B. J. M. The evolution of microbiome-mediated traits. Preprint at bioRxiv https://doi.org/10.1101/2024.03.29.587374 (2024).
Sesardic, N. Heritability and indirect causation. Philos. Sci. 70, 1002–1014 (2003).
Lynch, K. E. & Bourrat, P. Interpreting heritability causally. Philos. Sci. 84, 14–34 (2017).
Falconer, D. S. & MacKay, T. F. C. Introduction to Quantitative Genetics (Prentice Hall, 1996).
Polderman, T. J. C. et al. Meta-analysis of the heritability of human traits based on fifty years of twin studies. Nat. Genet. 47, 702–709 (2015).
Grieneisen, L. et al. Gut microbiome heritability is nearly universal but environmentally contingent. Science 373, 181–186 (2021).
Siddaway, A. P., Wood, A. M. & Hedges, L. V. How to do a systematic review: a best practice guide for conducting and reporting narrative reviews, meta-analyses and meta-syntheses. Annu. Rev. Psychol. 70, 747–770 (2019).
Kurilshikov, A. et al. Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat. Genet. 53, 156–165 (2021).
Goodrich, J. K., Davenport, E. R., Clark, A. G. & Ley, R. E. The relationship between the human genome and microbiome comes into view. Annu. Rev. Genet. 51, 413–433 (2017).
Mousseau, T. A. & Roff, D. A. Natural selection and the heritability of fitness components. Heredity 59, 181–197 (1987).
Dochtermann, N. A., Schwab, T., Anderson Berdal, M., Dalos, J. & Royauté, R. The heritability of behavior: a meta-analysis. J. Hered. 110, 403–410 (2019).
Goodrich, J. K. et al. Human genetics shape the gut microbiome. Cell 159, 789–799 (2014).
Goodrich, J. K. et al. Genetic determinants of the gut microbiome in UK twins. Cell Host Microbe 19, 731–743 (2016).
Beaumont, M. et al. Heritable components of the human fecal microbiome are associated with visceral fat. Genome Biol. 17, 189 (2016).
Mueller, U. G. & Linksvayer, T. A. Microbiome breeding: conceptual and practical issues. Trends Microbiol. 30, 997–1011 (2022).
Ryu, E. P. & Davenport, E. R. Host genetic determinants of the microbiome across animals: from Caenorhabditis elegans to cattle. Annu. Rev. Anim. Biosci. 10, 203–226 (2022).
Wen, C. et al. The gut microbiota is largely independent of host genetics in regulating fat deposition in chickens. ISME J. 13, 1422–1436 (2019).
Difford, G. F. et al. Host genetics and the rumen microbiome jointly associate with methane emissions in dairy cows. PLoS Genet. 14, e1007580 (2018).
Li, F. et al. Host genetics influence the rumen microbiota and heritable rumen microbial features associate with feed efficiency in cattle. Microbiome 7, 92 (2019).
Wallace, R. J. et al. A heritable subset of the core rumen microbiome dictates dairy cow productivity and emissions. Sci. Adv. 5, eaav8391 (2019).
Sasson, G. et al. Heritable bovine rumen bacteria are phylogenetically related and correlated with the cow’s capacity to harvest energy from its feed. mBio 8, e00703-17 (2017).
Bergamaschi, M. et al. Heritability and genome-wide association of swine gut microbiome features with growth and fatness parameters. Sci. Rep. 10, 10134 (2020).
Camarinha-Silva, A. et al. Host genome influence on gut microbial composition and microbial prediction of complex traits in pigs. Genetics 206, 1637–1644 (2017).
Chen, C. et al. Contribution of host genetics to the variation of microbial composition of cecum lumen and feces in pigs. Front. Microbiol. 9, 2626 (2018).
Ramayo-Caldas, Y. et al. Gut eukaryotic communities in pigs: diversity, composition and host genetics contribution. Anim. Microbiome 2, 18 (2020).
Org, E. et al. Genetic and environmental control of host-gut microbiota interactions. Genome Res. 25, 1558–1569 (2015).
Doms, S. et al. Key features of the genetic architecture and evolution of host-microbe interactions revealed by high-resolution genetic mapping of the mucosa-associated gut microbiome in hybrid mice. eLife 11, e75419 (2022).
Wu, J. et al. Honey bee genetics shape the strain-level structure of gut microbiota in social transmission. Microbiome 9, 225 (2021).
Early, A. M., Shanmugarajah, N., Buchon, N. & Clark, A. G. Drosophila genotype influences commensal bacterial levels. PLoS ONE 12, e0170332 (2017).
Quigley, K. M., Willis, B. L. & Bay, L. K. Heritability of the Symbiodinium community in vertically- and horizontally-transmitting broadcast spawning corals. Sci. Rep. 7, 8219 (2017).
Jia, S. et al. The pass-on effect of tetracycline-induced honey bee (Apis mellifera) gut community dysbiosis. Front. Microbiol. 12, 781746 (2022).
Kowallik, V., Das, A. & Mikheyev, A. S. Experimental inheritance of antibiotic acquired dysbiosis affects host phenotypes across generations. Front. Microbiol. 13, 1030771 (2022).
Wagner, M. R. Prioritizing host phenotype to understand microbiome heritability in plants. New Phytol. 232, 502–509 (2021).
Deng, S. et al. Genome wide association study reveals plant loci controlling heritability of the rhizosphere microbiome. ISME J. 15, 3181–3194 (2021).
Wallace, J. G., Kremling, K. A., Kovar, L. L. & Buckler, E. S. Quantitative genetics of the maize leaf microbiome. Phytobiomes J. 2, 208–224 (2018).
Gopal, M. & Gupta, A. Microbiome selection could spur next-generation plant breeding strategies. Front. Microbiol. 7, 1971 (2016).
Wagner, M. R. et al. Host genotype and age shape the leaf and root microbiomes of a wild perennial plant. Nat. Commun. 7, 12151 (2016).
Schweitzer, J. A. et al. Plant–soil–microorganism interactions: heritable relationship between plant genotype and associated soil microorganisms. Ecology 89, 773–781 (2008).
Walters, W. A. et al. Large-scale replicated field study of maize rhizosphere identifies heritable microbes. Proc. Natl Acad. Sci. USA 115, 7368–7373 (2018).
Martínez-Álvaro, M. et al. Bovine host genome acts on rumen microbiome function linked to methane emissions. Commun. Biol. 5, 350 (2022).
Meng, H. et al. Body weight selection affects quantitative genetic correlated responses in gut microbiota. PLoS ONE 9, e89862 (2014).
Mignon-Grasteau, S. et al. Impact of selection for digestive efficiency on microbiota composition in the chicken. PLoS ONE 10, e0135488 (2015).
Moraitou, M. et al. Ecology, not host phylogeny, shapes the oral microbiome in closely related species. Mol. Biol. Evol. 39, msac263 (2022).
Lamit, L. J., Holeski, L. M., Flores-Rentería, L., Whitham, T. G. & Gehring, C. A. Tree genotype influences ectomycorrhizal fungal community structure: ecological and evolutionary implications. Fungal Ecol. 24, 124–134 (2016).
Callahan, B. J., McMurdie, P. J. & Holmes, S. P. Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME J. 11, 2639–2643 (2017).
Huang, C. et al. Meta-analysis reveals the vaginal microbiome is a better predictor of earlier than later preterm birth. BMC Biol. 21, 199 (2023).
Pasaniuc, B. & Price, A. L. Dissecting the genetics of complex traits using summary association statistics. Nat. Rev. Genet. 18, 117–127 (2017).
Sollis, E. et al. The NHGRI-EBI GWAS Catalog: knowledgebase and deposition resource. Nucleic Acids Res. 51, D977–D985 (2023).
Petersen, C. et al. Host and microbiome jointly contribute to environmental adaptation. ISME J. 17, 1953–1965 (2023).
Mueller, E. A., Wisnoski, N. I., Peralta, A. L. & Lennon, J. T. Microbial rescue effects: how microbiomes can save hosts from extinction. Funct. Ecol. 34, 2055–2064 (2020).
Trevelline, B. K., Fontaine, S. S., Hartup, B. K. & Kohl, K. D. Conservation biology needs a microbial renaissance: a call for the consideration of host-associated microbiota in wildlife management practices. Proc. R. Soc. B 286, 20182448 (2019).
Davenport, E. R. et al. Genome-wide association studies of the human gut microbiota. PLoS ONE 10, e0140301 (2015).
Si, J., Lee, S., Park, J. M., Sung, J. & Ko, G. Genetic associations and shared environmental effects on the skin microbiome of Korean twins. BMC Genomics 16, 992 (2015).
Turpin, W. et al. Association of host genome with intestinal microbial composition in a large healthy cohort. Nat. Genet. 48, 1413–1417 (2016).
Xie, H. et al. Shotgun metagenomics of 250 adult twins reveals genetic and environmental impacts on the gut microbiome. Cell Syst. 3, 572–584.e3 (2016).
Lim, M. Y. et al. The effect of heritability and host genetics on the gut microbiota and metabolic syndrome. Gut 66, 1031–1038 (2017).
Demmitt, B. A. et al. Genetic influences on the human oral microbiome. BMC Genomics 18, 659 (2017).
Gomez, A. et al. Host genetic control of the oral microbiome in health and disease. Cell Host Microbe 22, 269–278.e3 (2017).
Zierer, J. et al. The fecal metabolome as a functional readout of the gut microbiome. Nat. Genet. 50, 790–795 (2018).
Ishida, S. et al. Genome-wide association studies and heritability analysis reveal the involvement of host genetics in the Japanese gut microbiota. Commun. Biol. 3, 686 (2020).
Xu, F. et al. The interplay between host genetics and the gut microbiome reveals common and distinct microbiome features for complex human diseases. Microbiome 8, 145 (2020).
Adebayo, A. S. et al. The urinary tract microbiome in older women exhibits host genetic and environmental influences. Cell Host Microbe 28, 298–305.e3 (2020).
Wright, M. L. et al. Vaginal microbiome Lactobacillus crispatus is heritable among European American women. Commun. Biol. 4, 872 (2021).
Gacesa, R. et al. Environmental factors shaping the gut microbiome in a Dutch population. Nature 604, 732–739 (2022).
Corby, P. M. A. et al. Mutans streptococci in preschool twins. Arch. Oral Biol. 50, 347–351 (2005).
Corby, P. M. et al. Heritability of oral microbial species in caries-active and caries-free twins. Twin Res. Hum. Genet. 10, 821–828 (2007).
Lim, M. Y. et al. Analysis of the association between host genetics, smoking and sputum microbiota in healthy humans. Sci. Rep. 6, 23745 (2016).
Esberg, A., Haworth, S., Kuja-Halkola, R., Magnusson, P. K. E. & Johansson, I. Heritability of oral microbiota and immune responses to oral bacteria. Microorganisms 8, 1126 (2020).
Boulund, U. et al. Gut microbiome associations with host genotype vary across ethnicities and potentially influence cardiometabolic traits. Cell Host Microbe 30, 1464–1480.e6 (2022).
Saborío-Montero, A. et al. Structural equation models to disentangle the biological relationship between microbiota and complex traits: methane production in dairy cattle as a case of study. J. Anim. Breed. Genet. 137, 36–48 (2019).
Abbas, W. et al. Influence of host genetics in shaping the rumen bacterial community in beef cattle. Sci. Rep. 10, 15101 (2020).
Saborío-Montero, A. et al. A dimensional reduction approach to modulate the core ruminal microbiome associated with methane emissions via selective breeding. J. Dairy Sci. 104, 8135–8151 (2021).
Cardinale, S. & Kadarmideen, H. N. Host genome-metagenome analyses using combinatorial network methods reveal key metagenomic and host genetic features for methane emission and feed efficiency in cattle. Front. Genet. 13, 795717 (2022).
Lu, D. et al. Host contributes to longitudinal diversity of fecal microbiota in swine selected for lean growth. Microbiome 6, 4 (2018).
Aliakbari, A. et al. Genetic relationships between feed efficiency and gut microbiome in pig lines selected for residual feed intake. J. Anim. Breed. Genet. 138, 491–507 (2021).
O’Connor, A., Quizon, P. M., Albright, J. E., Lin, F. T. & Bennett, B. J. Responsiveness of cardiometabolic-related microbiota to diet is influenced by host genetics. Mamm. Genome 25, 583–599 (2014).
Taye, Z. M. et al. Core and differentially abundant bacterial taxa in the rhizosphere of field grown Brassica napus genotypes: implications for canola breeding. Front. Microbiol. 10, 3007 (2020).
Peiffer, J. A. et al. Diversity and heritability of the maize rhizosphere microbiome under field conditions. Proc. Natl Acad. Sci. USA 110, 6548–6553 (2013).
Wagner, M. R., Roberts, J. H., Balint-Kurti, P. & Holland, J. B. Heterosis of leaf and rhizosphere microbiomes in field-grown maize. New Phytol. 228, 1055–1069 (2020).
Singer, E., Bonnette, J., Kenaley, S. C., Woyke, T. & Juenger, T. E. Plant compartment and genetic variation drive microbiome composition in switchgrass roots. Environ. Microbiol. Rep. 11, 185–195 (2019).
Sutherland, J., Bell, T., Trexler, R. V., Carlson, J. E. & Lasky, J. R. Host genomic influence on bacterial composition in the switchgrass rhizosphere. Mol. Ecol. 31, 3934–3950 (2022).
Spor, A. et al. Domestication-driven changes in plant traits associated with changes in the assembly of the rhizosphere microbiota in tetraploid wheat. Sci. Rep. 10, 12234 (2020).
Dalmastri, C., Chiarini, L., Cantale, C., Bevivino, A. & Tabacchioni, S. Soil type and maize cultivar affect the genetic diversity of maize root-associated Burkholderia cepacia populations. Microb. Ecol. 38, 273–284 (1999).
Horton, M. W. et al. Genome-wide association study of Arabidopsis thaliana leaf microbial community. Nat. Commun. 5, 5320 (2014).
Burge, M. N. Fungi in Biological Control Systems (Manchester Univ. Press, 1988).
Barry, C.-J. S. et al. How to estimate heritability: a guide for genetic epidemiologists. Int. J. Epidemiol. 52, 624–632 (2022).
Kruuk, L. E. B. Estimating genetic parameters in natural populations using the ‘animal model’. Phil. Trans. R. Soc. Lond. B 359, 873–890 (2004).
Bruijning, M. et al. Relative abundance data can misrepresent heritability of the microbiome. Microbiome 11, 222 (2023).
Acknowledgements
This work was supported by a grant from the Gordon and Betty Moore Foundation (award no. 10001). B.J.M.B. was also supported by awards from the Alexander von Humboldt Foundation (Alexander-von-Humboldt Stiftung) and the SETS Synthesis Center of the Swiss Federal Institute of Aquatic Science and Technology (EAWAG). We are grateful for input from B. Week, H. Schulenburg and K. Räsänen, which greatly improved this Perspective.
Author information
Authors and Affiliations
Contributions
B.J.M.B. conceived of the study, supervised the project and critically revised the paper. A.H.M. performed the research, wrote the initial draft of the paper and critically revised the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Reviewer recognition
Nature Microbiology thanks Taichi Suzuki and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Table 1
Supplementary Table 1. Microbiome heritability estimates. Note that we chose to present ‘number of individuals’, which represents the number of unique hosts sampled in the study. This may or may not align with the sample size for statistical purposes so we have indicated that in a footnote where necessary. Attributes must have both a significant test statistic and/or a heritability estimate above a chosen heritability threshold (such as >0.15 or >0.20) to be considered heritable. Percent heritable, % is the percentage of tested microbiome attributes that were considered heritable. Heritability range is the range between the minimum and maximum heritability estimates for this study.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morris, A.H., Bohannan, B.J.M. Estimates of microbiome heritability across hosts. Nat Microbiol 9, 3110–3119 (2024). https://doi.org/10.1038/s41564-024-01865-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41564-024-01865-w
This article is cited by
-
Breeding of microbiomes conferring salt tolerance to plants
Microbiome (2025)
-
Mendelian randomization analysis revealed a gut microbiota-eye axis in acute anterior uveitis
Eye (2025)
-
Applying evolutionary theory to understand host–microbiome evolution
Nature Ecology & Evolution (2025)
-
Genetics-mediated regulation of intestinal gene expression on microbiome contributes to human disease heritability
Molecular Systems Biology (2025)