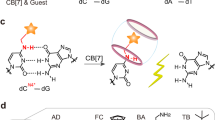
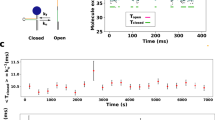
Abstract
The hybridization of short nucleic acid strands is a remarkable spontaneous process that is foundational to biotechnology and nanotechnology and plays a crucial role in gene expression, editing and DNA repair. Decades of research into the mechanism of hybridization have resulted in a deep understanding of its thermodynamics, but many questions remain regarding its kinetics and dynamics. Recent advances in experiments and molecular dynamics simulations of nucleic acids are enabling more direct insight into the structural dynamics of hybridization, which can test long-standing assumptions regarding its mechanism. In this Review, we summarize the current state of knowledge of hybridization kinetics, discuss the barriers to a molecular description of hybridization dynamics, and highlight the new approaches that have begun uncovering the dynamics of hybridization and the duplex ensemble. The kinetics and dynamics of hybridization are highly sensitive to the composition of nucleic acids, and we emphasize recent discoveries and open questions on the role of nucleobase sequence and chemical modifications.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Jiang, F. & Doudna, J. A. CRISPR–Cas9 structures and mechanisms. Annu. Rev. Biophys. 46, 505–529 (2017).
Klein, M., Eslami-Mossallam, B., Arroyo, D. G. & Depken, M. Hybridization kinetics explains CRISPR–Cas off-targeting rules. Cell Rep. 22, 1413–1423 (2018).
Seeman, N. C. & Sleiman, H. F. DNA nanotechnology. Nat. Rev. Mater. 3, 17068 (2017).
Schneider, F., Möritz, N. & Dietz, H. The sequence of events during folding of a DNA origami. Sci. Adv. 5, eaaw1412 (2019).
Song, J. et al. Reconfiguration of DNA molecular arrays driven by information relay. Science 357, eaan3377 (2017).
Cumberworth, A., Frenkel, D. & Reinhardt, A. Simulations of DNA-origami self-assembly reveal design-dependent nucleation barriers. Nano Lett. 22, 6916–6922 (2022).
Zhan, P. et al. Recent advances in DNA origami-engineered nanomaterials and applications. Chem. Rev. 123, 3976–4050 (2023).
Wei, X., Nangreave, J. & Liu, Y. Uncovering the self-assembly of DNA nanostructures by thermodynamics and kinetics. Acc. Chem. Res. 47, 1861–1870 (2014).
Jungmann, R. et al. Single-molecule kinetics and super-resolution microscopy by fluorescence imaging of transient binding on DNA origami. Nano Lett. 10, 4756–4761 (2010).
Strauss, S. & Jungmann, R. Up to 100-fold speed-up and multiplexing in optimized DNA-PAINT. Nat. Methods 17, 789–791 (2020).
Gray, D. M. & Tinoco, I. Jr A new approach to the study of sequence‐dependent properties of polynucleotides. Biopolymers 9, 223–244 (1970).
SantaLucia, J. Jr A unified view of polymer, dumbbell, and oligonucleotide DNA nearest-neighbor thermodynamics. Proc. Natl Acad. Sci. USA 95, 1460–1465 (1998).
SantaLucia, J. Jr & Hicks, D. The thermodynamics of DNA structural motifs. Annu. Rev. Biophys. Biomol. Struct. 33, 415–440 (2004).
Xia, T. et al. Thermodynamic parameters for an expanded nearest-neighbor model for formation of RNA duplexes with Watson–Crick base pairs. Biochemistry 37, 14719–14735 (1998).
Zuber, J., Schroeder, S. J., Sun, H., Turner, D. H. & Mathews, D. H. Nearest neighbor rules for RNA helix folding thermodynamics: improved end effects. Nucleic Acids Res. 50, 5251–5262 (2022).
Markham, N. R. & Zuker, M. in Bioinformatics: Structure, Function, and Applications (ed. Keith, J. M.) 3–31 (Humana, 2008).
Zadeh, J. N. et al. NUPACK: analysis and design of nucleic acid systems. J. Comput. Chem. 32, 170–173 (2011).
Rejali, N. A., Ye, F. D., Zuiter, A. M., Keller, C. C. & Wittwer, C. T. Nearest-neighbour transition-state analysis for nucleic acid kinetics. Nucleic Acids Res. 49, 4574–4585 (2021).
Zhang, J. X. et al. Predicting DNA hybridization kinetics from sequence. Nat. Chem. 10, 91–98 (2018).
Hertel, S. et al. The stability and number of nucleating interactions determine DNA hybridization rates in the absence of secondary structure. Nucleic Acids Res. 50, 7829–7841 (2022).
Todisco, M. & Szostak, J. W. Hybridization kinetics of out-of-equilibrium mixtures of short RNA oligonucleotides. Nucleic Acids Res. 50, 9647–9662 (2022).
Phan, T. T., Phan, T. M. & Schmit, J. D. Beneficial and detrimental effects of non-specific binding during DNA hybridization. Biophys. J. 122, 835–848 (2023).
Untergasser, A. et al. Primer3 — new capabilities and interfaces. Nucleic Acids Res. 40, e115–e115 (2012).
Wang, J. S. & Zhang, D. Y. Simulation-guided DNA probe design for consistently ultraspecific hybridization. Nat. Chem. 7, 545–553 (2015).
Haley, N. E. et al. Design of hidden thermodynamic driving for non-equilibrium systems via mismatch elimination during DNA strand displacement. Nat. Commun. 11, 2562 (2020).
Ouldridge, T. E., Šulc, P., Romano, F., Doye, J. P. & Louis, A. A. DNA hybridization kinetics: zippering, internal displacement and sequence dependence. Nucleic Acids Res. 41, 8886–8895 (2013).
Hinckley, D. M., Lequieu, J. P. & de Pablo, J. J. Coarse-grained modeling of DNA oligomer hybridization: length, sequence, and salt effects. J. Chem. Phys. 141, 07B613_611 (2014).
Jones, M. S., Ashwood, B., Tokmakoff, A. & Ferguson, A. L. Determining sequence-dependent DNA oligonucleotide hybridization and dehybridization mechanisms using coarse-grained molecular simulation, Markov state models, and infrared spectroscopy. J. Am. Chem. Soc. 143, 17395–17411 (2021).
Sanstead, P. J. & Tokmakoff, A. Direct observation of activated kinetics and downhill dynamics in DNA dehybridization. J. Phys. Chem. B 122, 3088–3100 (2018).
Chen, C., Wang, W., Wang, Z., Wei, F. & Zhao, X. S. Influence of secondary structure on kinetics and reaction mechanism of DNA hybridization. Nucleic Acids Res. 35, 2875–2884 (2007).
Wang, H., Li, B., Kim, Y.-J., Kwon, O.-H. & Granick, S. Intermediate states of molecular self-assembly from liquid-cell electron microscopy. Proc. Natl Acad. Sci. USA 117, 1283–1292 (2020).
Neupane, K., Wang, F. & Woodside, M. Direct measurement of sequence-dependent transition path times and conformational diffusion in DNA duplex formation. Proc. Natl Acad. Sci. USA 114, 1329–1334 (2017).
Alvey, H. S., Gottardo, F. L., Nikolova, E. N. & Al-Hashimi, H. M. Widespread transient Hoogsteen base pairs in canonical duplex DNA with variable energetics. Nat. Commun. 5, 4786 (2014).
Dupuis, N. F., Holmstrom, E. D. & Nesbitt, D. J. Single-molecule kinetics reveal cation-promoted DNA duplex formation through ordering of single-stranded helices. Biophys. J. 105, 756–766 (2013).
Dubini, R. C. et al. 1H NMR chemical exchange techniques reveal local and global effects of oxidized cytosine derivatives. ACS Phys. Chem. Au 2, 237–246 (2022).
Ashwood, B., Sanstead, P. J., Dai, Q., He, C. & Tokmakoff, A. 5-Carboxylcytosine and cytosine protonation distinctly alter the stability and dehybridization dynamics of the DNA duplex. J. Phys. Chem. B 124, 627–640 (2019).
Ghosh, S. et al. Nearest-neighbor parameters for predicting DNA duplex stability in diverse molecular crowding conditions. Proc. Natl Acad. Sci. USA 117, 14194–14201 (2020).
Shi, H. et al. NMR chemical exchange measurements reveal that N6-methyladenosine slows RNA annealing. J. Am. Chem. Soc. 141, 19988–19993 (2019).
Rangadurai, A., Szymaski, E. S., Kimsey, I. J., Shi, H. & Al-Hashimi, H. M. Characterizing micro-to-millisecond chemical exchange in nucleic acids using off-resonance R1ρ relaxation dispersion. Prog. Nucl. Mag. Reson. Spectrosc. 112, 55–102 (2019).
Holmstrom, E. D. & Nesbitt, D. J. Biophysical insights from temperature-dependent single-molecule förster resonance energy transfer. Annu. Rev. Phys. Chem. 67, 441–465 (2016).
Chung, H. S. Transition path times measured by single-molecule spectroscopy. J. Mol. Biol. 430, 409–423 (2018).
Hoffer, N. Q. & Woodside, M. T. Probing microscopic conformational dynamics in folding reactions by measuring transition paths. Curr. Opin. Chem. Biol. 53, 68–74 (2019).
Dans, P. D., Walther, J., Gómez, H. & Orozco, M. Multiscale simulation of DNA. Curr. Opin. Struc. Biol. 37, 29–45 (2016).
Sponer, J. et al. RNA structural dynamics as captured by molecular simulations: a comprehensive overview. Chem. Rev. 118, 4177–4338 (2018).
Lipfert, J., Doniach, S., Das, R. & Herschlag, D. Understanding nucleic acid–ion interactions. Annu. Rev. Biochem. 83, 813–841 (2014).
Abou Assi, H., Garavís, M., González, C. & Damha, M. J. i-Motif DNA: structural features and significance to cell biology. Nucleic Acids Res. 46, 8038–8056 (2018).
Grün, J. T. & Schwalbe, H. Folding dynamics of polymorphic G‐quadruplex structures. Biopolymers 113, e23477 (2022).
Marin-Gonzalez, A., Vilhena, J., Perez, R. & Moreno-Herrero, F. A molecular view of DNA flexibility. Q. Rev. Biophys. 54, e8 (2021).
Craig, M. E., Crothers, D. M. & Doty, P. Relaxation kinetics of dimer formation by self complementary oligonucleotides. J. Mol. Biol. 62, 383–401 (1971).
Pörschke, D. & Eigen, M. Co-operative non-enzymatic base recognition III. Kinetics of the helix–coil transition of the oligoribouridylic· oligoriboadenylic acid system and of oligoriboadenylic acid alone at acidic pH. J. Mol. Biol. 62, 361–381 (1971).
Jin, R. & Maibaum, L. Mechanisms of DNA hybridization: transition path analysis of a simulation-informed Markov model. J. Chem. Phys. 150, 105103 (2019).
Ashwood, B., Jones, M. S., Ferguson, A. L. & Tokmakoff, A. Disruption of energetic and dynamic base pairing cooperativity in DNA duplexes by an abasic site. Proc. Natl Acad. Sci. USA 120, e2219124120 (2023).
Poland, D. & Scheraga, H. A. Theory of Helix–Coil Transitions in Biopolymers (Academic, 1970).
Manning, G. S. On the application of polyelectrolyte limiting laws to the helix–coil transition of DNA. V. Ionic effects on renaturation kinetics. Biopolymers 15, 1333–1343 (1976).
Hart, D. J., Jeong, J., Gumbart, J. C. & Kim, H. D. Weak tension accelerates hybridization and dehybridization of short oligonucleotides. Nucleic Acids Res. 51, 3030–3040 (2023).
Whitley, K. D., Comstock, M. J. & Chemla, Y. R. Elasticity of the transition state for oligonucleotide hybridization. Nucleic Acids Res. 45, 547–555 (2017).
Yakovchuk, P., Protozanova, E. & Frank-Kamenetskii, M. D. Base-stacking and base-pairing contributions into thermal stability of the DNA double helix. Nucleic Acids Res. 34, 564–574 (2006).
Zacharias, M. Base-pairing and base-stacking contributions to double-stranded DNA formation. J. Phys. Chem. B 124, 10345–10352 (2020).
Altun, A., Garcia-Ratés, M., Neese, F. & Bistoni, G. Unveiling the complex pattern of intermolecular interactions responsible for the stability of the DNA duplex. Chem. Sci. 12, 12785–12793 (2021).
Son, I., Shek, Y. L., Dubins, D. N. & Chalikian, T. V. Hydration changes accompanying helix-to-coil DNA transitions. J. Am. Chem. Soc. 136, 4040–4047 (2014).
Huguet, J. M., Ribezzi-Crivellari, M., Bizarro, C. V. & Ritort, F. Derivation of nearest-neighbor DNA parameters in magnesium from single molecule experiments. Nucleic Acids Res. 45, 12921–12931 (2017).
Owczarzy, R., Moreira, B. G., You, Y., Behlke, M. A. & Walder, J. A. Predicting stability of DNA duplexes in solutions containing magnesium and monovalent cations. Biochem 47, 5336–5353 (2008).
Rouzina, I. & Bloomfield, V. A. Heat capacity effects on the melting of DNA. 2. Analysis of nearest-neighbor base pair effects. Biophys. J. 77, 3252–3255 (1999).
Mikulecky, P. J. & Feig, A. L. Heat capacity changes associated with nucleic acid folding. Biopolymers 82, 38–58 (2006).
Rissone, P., Rico-Pasto, M., Smith, S. & Ritort, F. DNA calorimetric force spectroscopy at single base pair resolution. Prperint at https://arxiv.org/abs/2404.18785 (2024).
Bergonzo, C., Grishaev, A. & Bottaro, S. Conformational heterogeneity of UCAAUC RNA oligonucleotide from molecular dynamics simulations, SAXS, and NMR experiments. RNA 28, 937–946 (2022).
Plumridge, A., Meisburger, S. P., Andresen, K. & Pollack, L. The impact of base stacking on the conformations and electrostatics of single-stranded DNA. Nucleic Acids Res. 45, 3932–3943 (2017).
Mondal, B., Chakraborty, D., Hori, N., Nguyen, H. & Thirumalai, D. Competition between stacking and divalent cation mediated electrostatic interactions determines the conformations of short DNA sequences. J. Chem. Theory Comput. 20, 2934–2946 (2024).
Nüesch, M. F. et al. Nanosecond chain dynamics of single-stranded nucleic acids. Nat. Commun. 15, 6010 (2024).
Camunas-Soler, J., Ribezzi-Crivellari, M. & Ritort, F. Elastic properties of nucleic acids by single-molecule force spectroscopy. Annu. Rev. Biophys. 45, 65–84 (2016).
Dans, P. D. et al. The static and dynamic structural heterogeneities of B-DNA: extending Calladine–Dickerson rules. Nucleic Acids Res. 47, 11090–11102 (2019).
Nikolova, E. N. et al. Transient Hoogsteen base pairs in canonical duplex DNA. Nature 470, 498–502 (2011).
Vafabakhsh, R. & Ha, T. Extreme bendability of DNA less than 100 base pairs long revealed by single-molecule cyclization. Science 337, 1097–1101 (2012).
Da Rosa, G. et al. Sequence-dependent structural properties of B-DNA: what have we learned in 40 years? Biophys. Rev. 13, 995–1005 (2021).
Sun, T. et al. Bottom-up coarse-grained modeling of DNA. Front. Mol. Biosci. 8, 645527 (2021).
Printz, M. P. & von Hippel, P. H. Hydrogen exchange studies of DNA structure. Proc. Natl Acad. Sci. USA 53, 363–370 (1965).
von Hippel, P. H., Johnson, N. P. & Marcus, A. H. Fifty years of DNA ‘breathing’: reflections on old and new approaches. Biopolymers 99, 923–954 (2013).
Frank-Kamenetskii, M. D. & Prakash, S. Fluctuations in the DNA double helix: a critical review. Phys. Life Rev. 11, 153–170 (2014).
Manghi, M. & Destainville, N. Physics of base-pairing dynamics in DNA. Phys. Rep. 631, 1–41 (2016).
McGhee, J. D. & Von Hippel, P. H. Formaldehyde as a probe of DNA structure. 4. Mechanism of the initial reaction of formaldehyde with DNA. Biochemistry 16, 3276–3293 (1977).
Lukashin, A. V., Vologodskii, A. V., Frank-Kamenetskii, M. D. & Lyubchenko, Y. L. Fluctuational opening of the double helix as revealed by theoretical and experimental study of DNA interaction with formaldehyde. J. Mol. Biol. 108, 665–682 (1976).
Guéron, M. & Leroy, J.-L. Studies of base pair kinetics by NMR measurement of proton exchange. Methods Enzymol. 261, 383–413 (1995).
Leroy, J. L., Kochoyan, M., Huynh-Dinh, T. & Guéron, M. Characterization of base-pair opening in deoxynucleotide duplexes using catalyzed exchange of the imino proton. J. Mol. Biol. 200, 223–238 (1988).
Várnai, P., Canalia, M. & Leroy, J.-L. Opening mechanism of G·T/U pairs in DNA and RNA duplexes: a combined study of imino proton exchange and molecular dynamics simulation. J. Am. Chem. Soc. 126, 14659–14667 (2004).
Giudice, E., Várnai, P. & Lavery, R. Base pair opening within B‐DNA: free energy pathways for GC and AT pairs from umbrella sampling simulations. Nucleic Acids Res. 31, 1434–1443 (2003).
Priyakumar, U. D. & MacKerell, A. D. Computational approaches for investigating base flipping in oligonucleotides. Chem. Rev. 106, 489–505 (2006).
Coman, D. & Russu, I. M. A nuclear magnetic resonance investigation of the energetics of basepair opening pathways in DNA. Biophys. J. 89, 3285–3292 (2005).
Hoogsteen, K. The structure of crystals containing a hydrogen-bonded complex of 1-methylthymine and 9-methyladenine. Acta Crystallogr. 12, 822–823 (1959).
Palmer, A. G. & Massi, F. Characterization of the dynamics of biomacromolecules using rotating-frame spin relaxation NMR spectroscopy. Chem. Rev. 106, 1700–1719 (2006).
Ray, D. & Andricioaei, I. Free energy landscape and conformational kinetics of hoogsteen base pairing in DNA vs. RNA. Biophys. J. 119, 1568–1579 (2020).
Vreede, J., Pérez de Alba Ortíz, A., Bolhuis, P. G. & Swenson, D. W. Atomistic insight into the kinetic pathways for Watson–Crick to Hoogsteen transitions in DNA. Nucleic Acids Res. 47, 11069–11076 (2019).
Krueger, A., Protozanova, E. & Frank-Kamenetskii, M. D. Sequence-dependent basepair opening in DNA double helix. Biophys. J. 90, 3091–3099 (2006).
Rangadurai, A. et al. Why are Hoogsteen base pairs energetically disfavored in A-RNA compared to B-DNA? Nucleic Acids Res. 46, 11099–11114 (2018).
Nonin, S., Leroy, J.-L. & Guéron, M. Terminal base pairs of oligodeoxynucleotides: imino proton exchange and fraying. Biochemistry 34, 10652–10659 (1995).
Sanstead, P. J., Stevenson, P. & Tokmakoff, A. Sequence-dependent mechanism of DNA oligonucleotide dehybridization resolved through infrared spectroscopy. J. Am. Chem. Soc. 138, 11792–11801 (2016).
Pörschke, D. A direct measurement of the unzippering rate of a nucleic acid double helix. Biophys. Chem. 2, 97–101 (1974).
Chen, X., Zhou, Y., Qu, P. & Zhao, X. S. Base-by-base dynamics in DNA hybridization probed by fluorescence correlation spectroscopy. J. Am. Chem. Soc. 130, 16947–16952 (2008).
Araque, J. & Robert, M. Lattice model of oligonucleotide hybridization in solution. II. Specificity and cooperativity. J. Chem. Phys. 144, 125101 (2016).
Sanstead, P. J. & Tokmakoff, A. A lattice model for the interpretation of oligonucleotide hybridization experiments. J. Chem. Phys. 150, 185104 (2019).
Zgarbová, M., Otyepka, M., Sponer, J., Lankas, F. & Jurecka, P. Base pair fraying in molecular dynamics simulations of DNA and RNA. J. Chem. Theory Comput. 10, 3177–3189 (2014).
Pinamonti, G., Paul, F., Noé, F., Rodriguez, A. & Bussi, G. The mechanism of RNA base fraying: molecular dynamics simulations analyzed with core-set Markov state models. J. Chem. Phys. 150, 154123 (2019).
Dabin, A. & Stirnemann, G. Atomistic simulations of RNA duplex thermal denaturation: sequence-and forcefield-dependence. Biophys. Chem. 307, 107167 (2024).
Patel, D. J. et al. Premelting and melting transitions in the d(CGCGAATTCGCG) self-complementary duplex in solution. Biochemistry 21, 428–436 (1982).
Ashwood, B. et al. Molecular insight into how the position of an abasic site modifies DNA duplex stability and dynamics. Biophys. J. 123, 118–133 (2024).
Plumridge, A., Andresen, K. & Pollack, L. Visualizing disordered single-stranded RNA: connecting sequence, structure, and electrostatics. J. Am. Chem. Soc. 142, 109–119 (2019).
Grotz, K. K. et al. Dispersion correction alleviates dye stacking of single-stranded DNA and RNA in simulations of single-molecule fluorescence experiments. J. Phys. Chem. B 122, 11626–11639 (2018).
Gao, Y., Wolf, L. K. & Georgiadis, R. M. Secondary structure effects on DNA hybridization kinetics: a solution versus surface comparison. Nucleic Acids Res. 34, 3370–3377 (2006).
Hata, H., Kitajima, T. & Suyama, A. Influence of thermodynamically unfavorable secondary structures on DNA hybridization kinetics. Nucleic Acids Res. 46, 782–791 (2018).
Schreck, J. S. et al. DNA hairpins destabilize duplexes primarily by promoting melting rather than by inhibiting hybridization. Nucleic Acids Res. 43, 6181–6190 (2015).
Nakano, M. et al. Local thermodynamics of the water molecules around single-and double-stranded DNA studied by grid inhomogeneous solvation theory. Chem. Phys. Lett. 660, 250–255 (2016).
Chen, H. et al. Ionic strength-dependent persistence lengths of single-stranded RNA and DNA. Proc. Natl Acad. Sci. USA 109, 799–804 (2012).
Meisburger, S. P. et al. Polyelectrolyte properties of single stranded DNA measured using SAXS and single‐molecule FRET: beyond the wormlike chain model. Biopolymers 99, 1032–1045 (2013).
Sim, A. Y., Lipfert, J., Herschlag, D. & Doniach, S. Salt dependence of the radius of gyration and flexibility of single-stranded DNA in solution probed by small-angle x-ray scattering. Phys. Rev. E 86, 021901 (2012).
Viader-Godoy, X., Manosas, M. & Ritort, F. Stacking correlation length in single-stranded DNA. Nucleic Acids Res. 52, 13243–13254 (2024).
Jacobson, D. R., McIntosh, D. B., Stevens, M. J., Rubinstein, M. & Saleh, O. A. Single-stranded nucleic acid elasticity arises from internal electrostatic tension. Proc. Natl Acad. Sci. USA 114, 5095–5100 (2017).
McIntosh, D. B., Duggan, G., Gouil, Q. & Saleh, O. A. Sequence-dependent elasticity and electrostatics of single-stranded DNA: signatures of base-stacking. Biophys. J. 106, 659–666 (2014).
Banerjee, A., Anand, M., Kalita, S. & Ganji, M. Single-molecule analysis of DNA base-stacking energetics using patterned DNA nanostructures. Nat. Nanotechnol. 18, 1474–1482 (2023).
Oweida, T. J., Kim, H. S., Donald, J. M., Singh, A. & Yingling, Y. G. Assessment of AMBER force fields for simulations of ssDNA. J. Chem. Theory Comput. 17, 1208–1217 (2021).
Bottaro, S., Bussi, G., Kennedy, S. D., Turner, D. H. & Lindorff-Larsen, K. Conformational ensembles of RNA oligonucleotides from integrating NMR and molecular simulations. Sci. Adv. 4, eaar8521 (2018).
Wetmur, J. G. & Davidson, N. Kinetics of renaturation of DNA. J. Mol. Biol. 31, 349–370 (1968).
Schickinger, M., Zacharias, M. & Dietz, H. Tethered multifluorophore motion reveals equilibrium transition kinetics of single DNA double helices. Proc. Natl Acad. Sci. USA 115, E7512–E7521 (2018).
Chen, Y.-I. et al. Measuring DNA hybridization kinetics in live cells using a time-resolved 3D single-molecule tracking method. J. Am. Chem. Soc. 141, 15747–15750 (2019).
Wong, K. L. & Liu, J. Factors and methods to modulate DNA hybridization kinetics. Biotechnol. J. 16, 2000338 (2021).
Pörschke, D., Uhlenbeck, O. C. & Martin, F. H. Thermodynamics and kinetics of the helix‐coil transition of oligomers containing GC base pairs. Biopolymers 12, 1313–1335 (1973).
Eigen, M. Methods for investigation of ionic reactions in aqueous solutions with half-times as short as 10–9 sec. Application to neutralization and hydrolysis reactions. Discuss. Faraday Soc. 17, 194–205 (1954).
Rauzan, B. et al. Kinetics and thermodynamics of DNA, RNA, and hybrid duplex formation. Biochemistry 52, 765–772 (2013).
Williams, A. P., Longfellow, C. E., Freier, S. M., Kierzek, R. & Turner, D. H. Laser temperature-jump, spectroscopic, and thermodynamic study of salt effects on duplex formation by dGCATGC. Biochemistry 28, 4283–4291 (1989).
Carrillo-Nava, E., Mejía-Radillo, Y. & Hinz, H.-J. Dodecamer DNA duplex formation is characterized by second-order kinetics, positive activation energies, and a dependence on sequence and Mg2+ ion concentration. Biochemistry 47, 13153–13157 (2008).
Saunders, M. & Ross, P. D. A simple model of the reaction between polyadenylic acid and polyuridylic acid. Biochem. Biophys. Res. Commun. 3, 314–318 (1960).
Thompson, M. C. et al. Temperature-jump solution X-ray scattering reveals distinct motions in a dynamic enzyme. Nat. Chem. 11, 1058–1066 (2019).
Narayanan, R. et al. Exploring the energy landscape of nucleic acid hairpins using laser temperature-jump and microfluidic mixing. J. Am. Chem. Soc. 134, 18952–18963 (2012).
Ding, F. et al. Displacement and dissociation of oligonucleotides during DNA hairpin closure under strain. Nucleic Acids Res. 50, 12082–12093 (2022).
Hoose, A., Vellacott, R., Storch, M., Freemont, P. S. & Ryadnov, M. G. DNA synthesis technologies to close the gene writing gap. Nat. Rev. Chem. 7, 144–161 (2023).
Schoen, I., Krammer, H. & Braun, D. Hybridization kinetics is different inside cells. Proc. Natl Acad. Sci. USA 106, 21649–21654 (2009).
Menssen, R. J. & Tokmakoff, A. Length-dependent melting kinetics of short DNA oligonucleotides using temperature-jump IR spectroscopy. J. Phys. Chem. B 123, 756–767 (2019).
Lukacs, G. L. et al. Size-dependent DNA mobility in cytoplasm and nucleus. J. Biol. Chem. 275, 1625–1629 (2000).
Ashwood, B. et al. Thermodynamics and kinetics of DNA and RNA dinucleotide hybridization to gaps and overhangs. Biophys. J. 122, 3323–3339 (2023).
Xiao, S., Sharpe, D. J., Chakraborty, D. & Wales, D. J. Energy landscapes and hybridization pathways for DNA hexamer duplexes. J. Phys. Chem. Lett. 10, 6771–6779 (2019).
Murugan, R. Lattice model on the rate of DNA hybridization. Phys. Rev. E 105, 064410 (2022).
Grabenhorst, L., Sturzenegger, F., Hasler, M., Schuler, B. & Tinnefeld, P. Single-molecule FRET at 10 MHz count rates. J. Am. Chem. Soc. 146, 3539–3544 (2024).
Bastos, M., Castro, V., Mrevlishvili, G. & Teixeira, J. Hydration of ds-DNA and ss-DNA by neutron quasielastic scattering. Biophys. J. 86, 3822–3827 (2004).
Kuchuk, K. & Sivan, U. Hydration structure of a single DNA molecule revealed by frequency-modulation atomic force microscopy. Nano Lett. 18, 2733–2737 (2018).
Tripathi, P., Firouzbakht, A., Gruebele, M. & Wanunu, M. Direct observation of single-protein transition state passage by nanopore ionic current jumps. J. Phys. Chem. Lett. 13, 5918–5924 (2022).
Sturzenegger, F. et al. Transition path times of coupled folding and binding reveal the formation of an encounter complex. Nat. Commun. 9, 4708 (2018).
Kim, J.-Y. & Chung, H. S. Disordered proteins follow diverse transition paths as they fold and bind to a partner. Science 368, 1253–1257 (2020).
Wilson, H. & Wang, Q. ABEL-FRET: tether-free single-molecule FRET with hydrodynamic profiling. Nat. Methods 18, 816–820 (2021).
Jang, S. S. et al. Characterizing the conformational free-energy landscape of RNA stem-loops using single-molecule field-effect transistors. J. Am. Chem. Soc. 145, 402–412 (2022).
Needham, L.-M. et al. Label-free detection and profiling of individual solution-phase molecules. Nature 629, 1062–1068 (2024).
Zerze, Gl. H., Stillinger, F. H. & Debenedetti, P. G. Thermodynamics of DNA hybridization from atomistic simulations. J. Phys. Chem. B 125, 771–779 (2021).
Love, O. et al. Assessing the current state of amber force field modifications for DNA — 2023 edition. J. Chem. Theory Comput. 19, 4299–4307 (2023).
Liebl, K. & Zacharias, M. Tumuc1: a new accurate DNA force field consistent with high-level quantum chemistry. J. Chem. Theory Comput. 17, 7096–7105 (2021).
Liebl, K. & Zacharias, M. Toward force fields with improved base stacking descriptions. J. Chem. Theory Comput. 19, 1529–1536 (2023).
Liebl, K. & Zacharias, M. The development of nucleic acids force fields: from an unchallenged past to a competitive future. Biophys. J. 122, 2841–2851 (2022).
Yoo, J., Winogradoff, D. & Aksimentiev, A. Molecular dynamics simulations of DNA–DNA and DNA–protein interactions. Curr. Opin. Struct. Biol. 64, 88–96 (2020).
Kuhrova, P. et al. Improving the performance of the amber RNA force field by tuning the hydrogen-bonding interactions. J. Chem. Theory Comput. 15, 3288–3305 (2019).
Pan, A. C. et al. Atomic-level characterization of protein–protein association. Proc. Natl Acad. Sci. USA 116, 4244–4249 (2019).
Maciejczyk, M., Spasic, A., Liwo, A. & Scheraga, H. A. DNA duplex formation with a coarse-grained model. J. Chem. Theory Comput. 10, 5020–5035 (2014).
Markegard, C. B., Fu, I. W., Reddy, K. A. & Nguyen, H. D. Coarse-grained simulation study of sequence effects on DNA hybridization in a concentrated environment. J. Phys. Chem. B 119, 1823–1834 (2015).
Fritz, D., Koschke, K., Harmandaris, V. A., van der Vegt, N. F. & Kremer, K. Multiscale modeling of soft matter: scaling of dynamics. Phys. Chem. Chem. Phys. 13, 10412–10420 (2011).
Ingólfsson, H. I. et al. The power of coarse graining in biomolecular simulations. Wiley Interdiscip. Rev. Comput. Mol. Sci. 4, 225–248 (2014).
Hinckley, D. M., Freeman, G. S., Whitmer, J. K. & de Pablo, J. J. An experimentally-informed coarse-grained 3-site-per-nucleotide model of DNA: structure, thermodynamics, and dynamics of hybridization. J. Chem. Phys. 139, 10B604_601 (2013).
Ouldridge, T. E., Louis, A. A. & Doye, J. P. Structural, mechanical, and thermodynamic properties of a coarse-grained DNA model. J. Chem. Phys. 134, 02B627 (2011).
Snodin, B. E. et al. Introducing improved structural properties and salt dependence into a coarse-grained model of DNA. J. Chem. Phys. 142, 234901 (2015).
Chakraborty, D., Hori, N. & Thirumalai, D. Sequence-dependent three interaction site model for single-and double-stranded DNA. J. Chem. Theory Comput. 14, 3763–3779 (2018).
Bernetti, M. & Bussi, G. Integrating experimental data with molecular simulations to investigate RNA structural dynamics. Curr. Opin. Struct. Biol. 78, 102503 (2023).
Chodera, J. D. & Noé, F. Markov state models of biomolecular conformational dynamics. Curr. Opin. Struct. Biol. 25, 135–144 (2014).
Husic, B. E. & Pande, V. S. Markov state models: from an art to a science. J. Am. Chem. Soc. 140, 2386–2396 (2018).
Noé, F. et al. Dynamical fingerprints for probing individual relaxation processes in biomolecular dynamics with simulations and kinetic experiments. Proc. Natl Acad. Sci. USA 108, 4822–4827 (2011).
Remington, J. M., McCullagh, M. & Kohler, B. Molecular dynamics simulations of 2-aminopurine-labeled dinucleoside monophosphates reveal multiscale stacking kinetics. J. Phys. Chem. B 123, 2291–2304 (2019).
Pinamonti, G. et al. Predicting the kinetics of RNA oligonucleotides using Markov state models. J. Chem. Theory Comput. 13, 926–934 (2017).
Bozovic, O. et al. Real-time observation of ligand-induced allosteric transitions in a PDZ domain. Proc. Natl Acad. Sci. USA 117, 26031–26039 (2020).
Wang, H., Xu, Z., Mao, S. & Granick, S. Experimental guidelines to image transient single-molecule events using graphene liquid cell electron microscopy. ACS Nano 16, 18526–18537 (2022).
Varghese, N. et al. Binding of DNA nucleobases and nucleosides with graphene. ChemPhysChem 10, 206–210 (2009).
Husale, B. S. et al. ssDNA binding reveals the atomic structure of graphene. Langmuir 26, 18078–18082 (2010).
Truex, K., Chung, H. S., Louis, J. M. & Eaton, W. A. Testing landscape theory for biomolecular processes with single molecule fluorescence spectroscopy. Phys. Rev. Lett. 115, 018101 (2015).
Gladrow, J., Ribezzi-Crivellari, M., Ritort, F. & Keyser, U. F. Experimental evidence of symmetry breaking of transition-path times. Nat. Commun. 10, 55 (2019).
Neupane, K., Hoffer, N. Q. & Woodside, M. Measuring the local velocity along transition paths during the folding of single biological molecules. Phys. Rev. Lett. 121, 018102 (2018).
Hoffer, N. Q., Neupane, K. & Woodside, M. T. Measuring the average shape of transition paths during the folding of a single biological molecule. Proc. Natl Acad. Sci. USA 116, 8125–8130 (2019).
Hoffer, N. Q., Neupane, K. & Woodside, M. T. Observing the base-by-base search for native structure along transition paths during the folding of single nucleic acid hairpins. Proc. Natl Acad. Sci. USA 118, e2101006118 (2021).
Lyons, A., Devi, A., Hoffer, N. Q. & Woodside, M. T. Quantifying the properties of nonproductive attempts at thermally activated energy-barrier crossing through direct observation. Phys. Rev. X 14, 011017 (2024).
Neupane, K. et al. Transition path times for nucleic acid folding determined from energy-landscape analysis of single-molecule trajectories. Phys. Rev. Lett. 109, 068102 (2012).
Pyo, A. G., Hoffer, N. Q., Neupane, K. & Woodside, M. T. Transition-path properties for folding reactions in the limit of small barriers. J. Chem. Phys. 149, 115101 (2018).
Ansari, A. & Kuznetsov, S. V. Is hairpin formation in single-stranded polynucleotide diffusion-controlled? J. Phys. Chem. B 109, 12982–12989 (2005).
Hori, N., Denesyuk, N. A. & Thirumalai, D. Frictional effects on RNA folding: speed limit and Kramers turnover. J. Phys. Chem. B 122, 11279–11288 (2018).
Ngo, T. T. et al. Effects of cytosine modifications on DNA flexibility and nucleosome mechanical stability. Nat. Commun. 7, 10813 (2016).
Rossetti, G. et al. The structural impact of DNA mismatches. Nucleic Acids Res. 43, 4309–4321 (2015).
Sanstead, P. J., Ashwood, B., Dai, Q., He, C. & Tokmakoff, A. Oxidized derivatives of 5-methylcytosine alter the stability and dehybridization dynamics of duplex DNA. J. Phys. Chem. B 124, 1160–1174 (2020).
Teng, X. & Hwang, W. Effect of methylation on local mechanics and hydration structure of DNA. Biophys. J. 114, 1791–1803 (2018).
Sabahi, A., Guidry, J., Inamati, G. B., Manoharan, M. & Wittung-Stafshede, P. Hybridization of 2′-ribose modified mixed-sequence oligonucleotides: thermodynamic and kinetic studies. Nucleic Acids Res. 29, 2163–2170 (2001).
Christensen, U., Jacobsen, N., Rajwanshi, V. K., Wengel, J. & Koch, T. Stopped-flow kinetics of locked nucleic acid (LNA)–oligonucleotide duplex formation: studies of LNA–DNA and DNA–DNA interactions. Biochem. J. 354, 481–484 (2001).
Rajasekaran, T. et al. Backbone hydrocarbon-constrained nucleic acids modulate hybridization kinetics for RNA. J. Am. Chem. Soc. 144, 1941–1950 (2022).
Gold, B., Stone, M. P. & Marky, L. A. Looking for Waldo: a potential thermodynamic signature to DNA damage. Acc. Chem. Res. 47, 1446–1454 (2014).
Gelfand, C. A., Plum, G. E., Grollman, A. P., Johnson, F. & Breslauer, K. J. Thermodynamic consequences of an abasic lesion in duplex DNA are strongly dependent on base sequence. Biochemistry 37, 7321–7327 (1998).
Testa, S. M., Disney, M. D., Turner, D. H. & Kierzek, R. Thermodynamics of RNA–RNA duplexes with 2- or 4-thiouridines: implications for antisense design and targeting a group I intron. Biochemistry 38, 16655–16662 (1999).
Wang, D., Kreutzer, D. A. & Essigmann, J. M. Mutagenicity and repair of oxidative DNA damage: insights from studies using defined lesions. Mutat. Res. 400, 99–115 (1998).
Wang, W., Xu, J., Chong, J. & Wang, D. Structural basis of DNA lesion recognition for eukaryotic transcription-coupled nucleotide excision repair. DNA Repair. 71, 43–55 (2018).
Hardwick, J. S., Lane, A. N. & Brown, T. Epigenetic modifications of cytosine: biophysical properties, regulation, and function in mammalian DNA. BioEssays 40, 1700199 (2018).
Jones, J. D., Monroe, J. & Koutmou, K. S. A molecular‐level perspective on the frequency, distribution, and consequences of messenger RNA modifications. Wiley Interdiscip. Rev. RNA 11, e1586 (2020).
Shen, X. & Corey, D. R. Chemistry, mechanism and clinical status of antisense oligonucleotides and duplex RNAs. Nucleic Acids Res. 46, 1584–1600 (2018).
Bauer, J., Reichl, A. & Tinnefeld, P. Kinetic referencing allows identification of epigenetic cytosine modifications by single-molecule hybridization kinetics and superresolution DNA-PAINT microscopy. ACS Nano 18, 1496–1503 (2023).
Todisco, M., Ding, D. & Szostak, J. W. Transient states during oligonucleotides hybridization: insights from annealing kinetics of mismatches and bulges. Nucleic Acids Res. 52, 2174–2187 (2023).
Zhao, X.-C. et al. 5-Methyl-cytosine stabilizes DNA but hinders DNA hybridization revealed by magnetic tweezers and simulations. Nucleic Acids Res. 50, 12344–12354 (2022).
Liu, B. et al. A quantitative model predicts how m6A reshapes the kinetic landscape of nucleic acid hybridization and conformational transitions. Nat. Commun. 12, 5201 (2021).
Tawa, K. & Knoll, W. Mismatching base‐pair dependence of the kinetics of DNA–DNA hybridization studied by surface plasmon fluorescence spectroscopy. Nucleic Acids Res. 32, 2372–2377 (2004).
Rajasekaran, T. et al. Systematic investigation of tether length and phosphorus configuration in backbone constrained macrocyclic nucleic acids to modulate binding kinetics for RNA. J. Org. Chem. 88, 3599–3614 (2023).
Naiser, T., Kayser, J., Mai, T., Michel, W. & Ott, A. Position dependent mismatch discrimination on DNA microarrays — experiments and model. BMC Bioinformatics 9, 509 (2008).
Chen, J., Dupradeau, F.-Y., Case, D. A., Turner, C. J. & Stubbe, J. DNA oligonucleotides with A, T, G or C opposite an abasic site: structure and dynamics. Nucleic Acids Res. 36, 253–262 (2008).
Kierzek, R., Burkard, M. E. & Turner, D. H. Thermodynamics of single mismatches in RNA duplexes. Biochemistry 38, 14214–14223 (1999).
Woodside, M. T. et al. Direct measurement of the full, sequence-dependent folding landscape of a nucleic acid. Science 314, 1001–1004 (2006).
Manuel, A. P., Lambert, J. & Woodside, M. T. Reconstructing folding energy landscapes from splitting probability analysis of single-molecule trajectories. Proc. Natl Acad. Sci. USA 112, 7183–7188 (2015).
McCauley, M. J. et al. Quantifying the stability of oxidatively damaged DNA by single-molecule DNA stretching. Nucleic Acids Res. 46, 4033–4043 (2018).
Nguyen, H. T., Hori, N. & Thirumalai, D. Condensates in RNA repeat sequences are heterogeneously organized and exhibit reptation dynamics. Nat. Chem. 14, 775–785 (2022).
Bellini, T. et al. Liquid crystal self-assembly of random-sequence DNA oligomers. Proc. Natl Acad. Sci. USA 109, 1110–1115 (2012).
Snodin, B. E. et al. Direct simulation of the self-assembly of a small DNA origami. ACS Nano 10, 1724–1737 (2016).
Ouldridge, T. E. DNA nanotechnology: understanding and optimisation through simulation. Mol. Phys. 113, 1–15 (2015).
Fonseca, P. et al. Multi-scale coarse-graining for the study of assembly pathways in DNA-brick self-assembly. J. Chem. Phys. 148, 134910 (2018).
Wang, J. et al. Probing heterogeneous folding pathways of DNA origami self-assembly at the molecular level with atomic force microscopy. Nano Lett. 22, 7173–7179 (2022).
Simmel, F. C., Yurke, B. & Singh, H. R. Principles and applications of nucleic acid strand displacement reactions. Chem. Rev. 119, 6326–6369 (2019).
Srinivas, N. et al. On the biophysics and kinetics of toehold-mediated DNA strand displacement. Nucleic Acids Res. 41, 10641–10658 (2013).
James, P. L., Brown, T. & Fox, K. R. Thermodynamic and kinetic stability of intermolecular triple helices containing different proportions of C+*GC and T*AT triplets. Nucleic Acids Res. 31, 5598–5606 (2003).
Hu, Y., Cecconello, A., Idili, A., Ricci, F. & Willner, I. Triplex DNA nanostructures: from basic properties to applications. Angew. Chem. Int. Ed. Engl. 56, 15210–15233 (2017).
Ouldridge, T. E., Johnston, I. G., Louis, A. A. & Doye, J. P. The self-assembly of DNA holliday junctions studied with a minimal model. J. Chem. Phys. 130, 065101 (2009).
Wang, W. et al. Holliday junction thermodynamics and structure: coarse-grained simulations and experiments. Sci. Rep. 6, 22863 (2016).
Zhou, L. et al. Non-enzymatic primer extension with strand displacement. eLife 8, e51888 (2019).
Hong, F. & Šulc, P. An emergent understanding of strand displacement in RNA biology. J. Struct. Biol. 207, 241–249 (2019).
Machinek, R. R., Ouldridge, T. E., Haley, N. E., Bath, J. & Turberfield, A. J. Programmable energy landscapes for kinetic control of DNA strand displacement. Nat. Commun. 5, 5324 (2014).
Irmisch, P., Ouldridge, T. E. & Seidel, R. Modeling DNA-strand displacement reactions in the presence of base-pair mismatches. J. Am. Chem. Soc. 142, 11451–11463 (2020).
Broadwater, D. B., Cook, A. W. & Kim, H. D. First passage time study of DNA strand displacement. Biophys. J. 120, 2400–2412 (2021).
Broadwater, D. B. & Kim, H. D. The effect of basepair mismatch on DNA strand displacement. Biophys. J. 110, 1476–1484 (2016).
Šulc, P., Ouldridge, T. E., Romano, F., Doye, J. P. & Louis, A. A. Modelling toehold-mediated RNA strand displacement. Biophys. J. 108, 1238–1247 (2015).
Smith, F. G., Goertz, J. P., Jurinović, K., Stevens, M. M. & Ouldridge, T. E. Strong sequence–dependence in RNA/DNA hybrid strand displacement kinetics. Nanoscale 16, 17624–17637 (2024).
Ratajczyk, E. J. et al. Controlling DNA-RNA strand displacement kinetics with base distribution. Preprint at bioRxiv https://doi.org/10.1101/2024.08.06.606789v1 (2024).
Zhang, D. Y. & Winfree, E. Control of DNA strand displacement kinetics using toehold exchange. J. Am. Chem. Soc. 131, 17303–17314 (2009).
Yurke, B. & Mills, A. P. Using DNA to power nanostructures. Genet. Program. Evol. Mach. 4, 111–122 (2003).
Reynaldo, L. P., Vologodskii, A. V., Neri, B. P. & Lyamichev, V. I. The kinetics of oligonucleotide replacements. J. Mol. Biol. 297, 511–520 (2000).
Di Michele, L. et al. Effect of inert tails on the thermodynamics of DNA hybridization. J. Am. Chem. Soc. 136, 6538–6541 (2014).
Todisco, M., Radakovic, A. & Szostak, J. W. RNA complexes with nicks and gaps: thermodynamic and kinetic effects of coaxial stacking and dangling ends. J. Am. Chem. Soc. 146, 18083–18094 (2024).
Abraham Punnoose, J. et al. High-throughput single-molecule quantification of individual base stacking energies in nucleic acids. Nat. Commun. 14, 631 (2023).
Walbrun, A. et al. Single-molecule force spectroscopy of toehold-mediated strand displacement. Nat. Commun. 15, 7564 (2024).
Ivani, I. et al. Parmbsc1: a refined force field for DNA simulations. Nat. Methods 13, 55–58 (2016).
Chaudhury, S. & Makarov, D. E. A harmonic transition state approximation for the duration of reactive events in complex molecular rearrangements. J. Chem. Phys. 133, 034118 (2010).
Marky, L. A. & Breslauer, K. J. Calculating thermodynamic data for transitions of any molecularity from equilibrium melting curves. Biopolymers 26, 1601–1620 (1987).
Mergny, J.-L. & Lacroix, L. Analysis of thermal melting curves. Oligonucleotides 13, 515–537 (2003).
Majikes, J. M., Zwolak, M. & Liddle, J. A. Best practice for improved accuracy: a critical reassessment of van’t Hoff analysis of melt curves. Biophys. J. 121, 1986–2001 (2022).
Bernasconi, C. Relaxation Kinetics (Academic, 1976).
Hopfinger, M. C., Kirkpatrick, C. C. & Znosko, B. M. Predictions and analyses of RNA nearest neighbor parameters for modified nucleotides. Nucleic Acids Res. 48, 8901–8913 (2020).
Denisov, V. P., Carlström, G., Venu, K. & Halle, B. Kinetics of DNA hydration. J. Mol. Biol. 268, 118–136 (1997).
Duboué-Dijon, E., Fogarty, A. C., Hynes, J. T. & Laage, D. Dynamical disorder in the DNA hydration shell. J. Am. Chem. Soc. 138, 7610–7620 (2016).
Dewey, T. & Turner, D. H. Laser temperature-jump study of stacking in adenylic acid polymers. Biochemistry 18, 5757–5762 (1979).
Lieblein, A. L., Buck, J., Schlepckow, K., Fürtig, B. & Schwalbe, H. Time‐resolved NMR spectroscopic studies of DNA i‐motif folding reveal kinetic partitioning. Angew. Chem. Int. Ed. Engl. 51, 250–253 (2012).
Stellwagen, E., Lu, Y. & Stellwagen, N. C. Unified description of electrophoresis and diffusion for DNA and other polyions. Biochemistry 42, 11745–11750 (2003).
Wirth, A. J., Liu, Y., Prigozhin, M. B., Schulten, K. & Gruebele, M. Comparing fast pressure jump and temperature jump protein folding experiments and simulations. J. Am. Chem. Soc. 137, 7152–7159 (2015).
Su, H. et al. Massively parallelized molecular force manipulation with on-demand thermal and optical control. J. Am. Chem. Soc. 143, 19466–19473 (2021).
Yin, Y. et al. Dynamics of spontaneous flipping of a mismatched base in DNA duplex. Proc. Natl Acad. Sci. USA 111, 8043–8048 (2014).
Applequist, J. & Damle, V. Thermodynamics of the helix-coil equilibrium in oligoadenylic acid from hypochromicity studies. J. Am. Chem. Soc. 87, 1450–1458 (1965).
Menssen, R. J., Kimmel, G. J. & Tokmakoff, A. Investigation into the mechanism and dynamics of DNA association and dissociation utilizing kinetic Monte Carlo simulations. J. Chem. Phys. 154, 045101 (2021).
Hudson, G. A., Bloomingdale, R. J. & Znosko, B. M. Thermodynamic contribution and nearest-neighbor parameters of pseudouridine–adenosine base pairs in oligoribonucleotides. RNA 19, 1474–1482 (2013).
Nardo, L. et al. Effects of non-CpG site methylation on DNA thermal stability: a fluorescence study. Nucleic Acids Res. 43, 10722–10733 (2015).
Kierzek, E. et al. Secondary structure prediction for RNA sequences including N6-methyladenosine. Nat. Commun. 13, 1271 (2022).
Zhou, H. et al. m1A and m1G disrupt A-RNA structure through the intrinsic instability of Hoogsteen base pairs. Nat. Struct. Mol. Biol. 23, 803–810 (2016).
Gruber, D. R. et al. Oxidative damage to epigenetically methylated sites affects DNA stability, dynamics and enzymatic demethylation. Nucleic Acids Res. 46, 10827–10839 (2018).
Ovcherenko, S. S. et al. Dynamics of 8-oxoguanine in DNA: decisive effects of base pairing and nucleotide context. J. Am. Chem. Soc. 145, 5613–5617 (2023).
Rangadurai, A. et al. Measuring thermodynamic preferences to form non-native conformations in nucleic acids using ultraviolet melting. Proc. Natl Acad. Sci. USA 119, e2112496119 (2022).
Dai, Q. et al. Weakened N3 hydrogen bonding by 5-formylcytosine and 5-carboxylcytosine reduces their base-pairing stability. ACS Chem. Biol. 11, 470–477 (2016).
Kou, Y., Koag, M.-C. & Lee, S. N7 methylation alters hydrogen-bonding patterns of guanine in duplex DNA. J. Am. Chem. Soc. 137, 14067–14070 (2015).
Afek, A. et al. DNA mismatches reveal conformational penalties in protein–DNA recognition. Nature 587, 291–296 (2020).
Rohs, R. et al. Origins of specificity in protein–DNA recognition. Annu. Rev. Biochem. 79, 233–269 (2010).
Acknowledgements
We acknowledge financial support from the National Institute of General Medical Sciences of the National Institutes of Health (award no. R01-GM118774) and the National Science Foundation under grant no. CHE-2155027. B.A. acknowledges support from the NSF Graduate Research Fellowship Program. We greatly appreciate discussions and feedback on the manuscript from P. Sanstead, M. Jones, M. Todisco, Y. Lee, N. Lewis, J. Schauss, L. Whaley-Mayda and K. Liebl. We thank S. Granick, T. Ouldridge, M. Todisco, H. Wang and M. Woodside for providing original files used in Figs. 3, 4 and 6.
Author information
Authors and Affiliations
Contributions
B.A. researched data for the article. Both authors contributed to the discussion of content, wrote the article then reviewed and/or edited the manuscript prior to submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Chemistry thanks Thomas Ouldridge, Khalid Salaita and Bryan Wei for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- All-atom molecular dynamics (MD) simulation
-
Numerical integration of Newton’s equations of motions for molecules described in terms of forces calculated at the atomic level. A force field is a specific parameterization of an interaction potential between atoms that are used to calculate the forces.
- Base-pairing cooperativity
-
The preference for forming a base pair at the end of an existing, intact segment of the base pairs relative to forming the first base pair in a region of unpaired bases.
- Coarse-grained molecular dynamics (MD) simulation
-
Molecular dynamics simulations where the number of interacting particles are reduced from all atoms to coarse sites with interactions governed by a particular force field.
- Diffusive models of reactions
-
Framework where reactions are modelled as the diffusion of a particle along a free-energy landscape with a characteristic conformational diffusion coefficient that is set by microscopic friction induced by interactions with solvent and intramolecular degrees of freedom.
- Ensemble spectroscopy
-
Spectroscopic methods whose observables arise from a macroscopic sample of many molecules and thereby report on the ensemble average properties of a chemical species. Time-dependent properties of species and reactions are probed by synchronizing the movement of the ensemble of molecules from the reactant states to product states.
- Hybridization dynamics
-
The pathways of time-dependent molecular configurations that oligonucleotides traverse as they transition between single-strand and duplex forms.
- Hybridization kinetics
-
The rates of exchange between duplex, single-strand, and intermediate states and the characterization of energetic barriers separating them.
- Markov-state model
-
A class of kinetic model that uses MD trajectories to identify long-lived molecular states and describes the equilibrium occupancy and probability fluxes between those states.
- oxDNA model
-
Coarse-grained model of DNA that models each nucleotide with three different interaction sites for hydrogen bonding, base stacking and backbone, and employs non-bonded potentials for non-bonded excluded volume, base stacking, cross stacking and electrostatics, as well as a bonded potential between backbone sites. It is parameterized against experimental mechanical properties, experimental hybridization thermodynamics and hybridization thermodynamics predicted by SantaLucia’s empirical nearest-neighbour model.
- Single-molecule spectroscopy
-
Experimental methods that isolate an observable from one molecule and resolve the time-trajectories of individual molecules within an ensemble.
- Three-site-per-nucleotide (3SPN.2) model
-
Coarse-grained model of DNA that uses three interaction sites to represent the phosphate, deoxyribose and nucleobase of each nucleotide, and employs non-bonded potentials for excluded volume, base stacking, cross stacking, base pairing and electrostatics, as well as bonded potential with bond, angle and dihedral contributions. It is parameterized against experimental structural data, duplex melting thermodynamics and free energies of base stacking.
- Trajectory
-
A time-series of periodic observations of a dynamical system. This could be an extended series of experimental observables or a series of molecular structures from a MD dynamics simulation.
- Transition paths
-
Trajectories from a reactant state that successfully cross a free-energy barrier to a reach a product state.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ashwood, B., Tokmakoff, A. Kinetics and dynamics of oligonucleotide hybridization. Nat Rev Chem 9, 305–327 (2025). https://doi.org/10.1038/s41570-025-00704-8
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41570-025-00704-8
This article is cited by
-
Advances and challenges in non-canonical nucleic acids data storage
Nature Communications (2026)