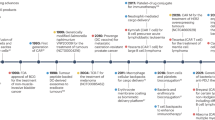
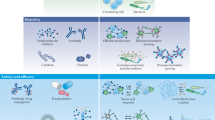
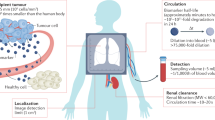
Abstract
Synthetic biology seeks to redesign biological systems to perform novel functions in a predictable manner. Recent advances in bacterial and mammalian cell engineering include the development of cells that function in biological samples or within the body as minimally invasive diagnostics or theranostics for the real-time regulation of complex diseased states. Ex vivo and in vivo cell-based biosensors and therapeutics have been developed to target a wide range of diseases including cancer, microbiome dysbiosis and autoimmune and metabolic diseases. While probiotic therapies have advanced to clinical trials, chimeric antigen receptor (CAR) T cell therapies have received regulatory approval, exemplifying the clinical potential of cellular therapies. This Review discusses preclinical and clinical applications of bacterial and mammalian sensing and drug delivery platforms as well as the underlying biological designs that could enable new classes of cell diagnostics and therapeutics. Additionally, we describe challenges that must be overcome for more rapid and safer clinical use of engineered systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hornick, J. L. Limited biopsies of soft tissue tumors: the contemporary role of immunohistochemistry and molecular diagnostics. Mod. Pathol. 32, 27–37 (2019).
Litwin, M. S. & Tan, H.-J. The diagnosis and treatment of prostate cancer: a review. JAMA 317, 2532–2542 (2017).
Smetherman, D. H. Screening, imaging, and image-guided biopsy techniques for breast cancer. Surg. Clin. North. Am. 93, 309–327 (2013).
Robbins, P. F. et al. A pilot trial using lymphocytes genetically engineered with an NY-ESO-1-reactive T-cell receptor: long-term follow-up and correlates with response. Clin. Cancer Res. 21, 1019–1027 (2015).
Seebacher, N. A., Stacy, A. E., Porter, G. M. & Merlot, A. M. Clinical development of targeted and immune based anti-cancer therapies. J. Exp. Clin. Cancer Res. 38, 156 (2019).
Cameron, D. E., Bashor, C. J. & Collins, J. J. A brief history of synthetic biology. Nat. Rev. Microbiol. 12, 381–390 (2014).
Way, J. C., Collins, J. J., Keasling, J. D. & Silver, P. A. Integrating biological redesign: where synthetic biology came from and where it needs to go. Cell 157, 151–161 (2014).
Kojima, R., Aubel, D. & Fussenegger, M. Toward a world of theranostic medication: programming biological sentinel systems for therapeutic intervention. Adv. Drug Deliv. Rev. 105, 66–76 (2016).
Braendstrup, P., Levine, B. L. & Ruella, M. The long road to the first FDA-approved gene therapy: chimeric antigen receptor T cells targeting CD19. Cytotherapy 22, 57–69 (2020).
FDA. FDA approves CAR-T cell therapy to treat adults with certain types of large B-cell lymphoma. US Food and Drug Administration https://www.fda.gov/news-events/press-announcements/fda-approves-car-t-cell-therapy-treat-adults-certain-types-large-b-cell-lymphoma (2017).
FDA. FDA approves brexucabtagene autoleucel for relapsed or refractory mantle cell lymphoma. US Food and Drug Administration https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-brexucabtagene-autoleucel-relapsed-or-refractory-mantle-cell-lymphoma (2020).
FDA. FDA approves lisocabtagene maraleucel for relapsed or refractory large B-cell lymphoma. US Food and Drug Administration https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-lisocabtagene-maraleucel-relapsed-or-refractory-large-b-cell-lymphoma (2021).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2020).
Lu, T. K., Mimee, M., Citorik, R. J. & Pepper, K. Engineering the Microbiome for Human Health Applications. The Chemistry of Microbiomes: Proceedings of a Seminar Series (National Academies Press (US), 2017).
Gibson, D. G. et al. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 6, 343–345 (2009).
Goodwin, S., McPherson, J. D. & McCombie, W. R. Coming of age: ten years of next-generation sequencing technologies. Nat. Rev. Genet. 17, 333–351 (2016).
Sarnaik, A., Liu, A., Nielsen, D. & Varman, A. M. High-throughput screening for efficient microbial biotechnology. Curr. Opin. Biotechnol. 64, 141–150 (2020).
Dijkstra, K. K. et al. Generation of tumor-reactive T cells by co-culture of peripheral blood lymphocytes and tumor organoids. Cell 174, 1586–1598.e12 (2018).
Harimoto, T. et al. Rapid screening of engineered microbial therapies in a 3D multicellular model. Proc. Natl Acad. Sci. USA 116, 9002–9007 (2019).
Jalili-Firoozinezhad, S. et al. A complex human gut microbiome cultured in an anaerobic intestine-on-a-chip. Nat. Biomed. Eng. 3, 520–531 (2019).
Fischbach, M. A., Bluestone, J. A. & Lim, W. A. Cell-based therapeutics: the next pillar of medicine. Sci. Transl Med. 5, 179ps7 (2013).
Lim, W. A. & June, C. H. The principles of engineering immune cell to treat cancer. Cell 168, 724–740 (2017).
Riglar, D. T. & Silver, P. A. Engineering bacteria for diagnostic and therapeutic applications. Nat. Rev. Microbiol. 16, 214–225 (2018).
Landry, B. P. & Tabor, J. J. Engineering diagnostic and therapeutic gut bacteria. Microbiol. Spectr. 5, 5 (2017).
Gardner, T. S., Cantor, C. R. & Collins, J. J. Construction of a genetic toggle switch in Escherichia coli. Nature 403, 339–342 (2000).
Elowitz, M. B. & Leibler, S. A synthetic oscillatory network of transcriptional regulators. Nature 403, 335–338 (2000).
Khalil, A. S. & Collins, J. J. Synthetic biology: applications come of age. Nat. Rev. Genet. 11, 367–379 (2010).
Jannetto, P. J. & Fitzgerald, R. L. Effective use of mass spectrometry in the clinical laboratory. Clin. Chem. 62, 92–98 (2016).
Anderson, N. L. The clinical plasma proteome: a survey of clinical assays for proteins in plasma and serum. Clin. Chem. 56, 177–185 (2010).
McNerney, M. P., Michel, C. L., Kishore, K., Standeven, J. & Styczynski, M. P. Dynamic and tunable metabolite control for robust minimal-equipment assessment of serum zinc. Nat. Commun. 10, 5514 (2019). This article demonstrates ways that bacterial diagnostics can be tuned to respond to physiologically relevant concentrations and to function in biological samples.
McNerney, M. P., Piorino, F., Michel, C. L. & Styczynski, M. P. Active analyte import improves the dynamic range and sensitivity of a vitamin B12 biosensor. ACS Synth. Biol. 9, 402–411 (2020).
Courbet, A., Endy, D., Renard, E., Molina, F. & Bonnet, J. Detection of pathological biomarkers in human clinical samples via amplifying genetic switches and logic gates. Sci. Transl Med. 7, 289ra83 (2015).
Watstein, D. M. & Styczynski, M. P. Development of a pigment-based whole-cell zinc biosensor for human serum. ACS Synth. Biol. 7, 267–275 (2018).
Mukherjee, S. & Bassler, B. L. Bacterial quorum sensing in complex and dynamically changing environments. Nat. Rev. Microbiol. 17, 371–382 (2019).
Holowko, M. B., Wang, H., Jayaraman, P. & Poh, C. L. Biosensing Vibrio cholerae with genetically engineered Escherichia coli. ACS Synth. Biol. 5, 1275–1283 (2016).
Ostrov, N. et al. A modular yeast biosensor for low-cost point-of-care pathogen detection. Sci. Adv. 3, e1603221 (2017).
Carter, S. R., Rodemeyer, M., Garfinkel, M. S. & Friedman, R. M. Synthetic biology and the U.S. biotechnology regulatory system: challenges and options. US Department of Energy Office of Scientific and Technical Information https://www.osti.gov/biblio/1169537 (2014)
Silverman, A. D., Karim, A. S. & Jewett, M. C. Cell-free gene expression: an expanded repertoire of applications. Nat. Rev. Genet. 21, 151–170 (2020).
Pardee, K. et al. Paper-based synthetic gene networks. Cell 159, 940–954 (2014).
Pardee, K. et al. Rapid, low-cost detection of zika virus using programmable biomolecular components. Cell 165, 1255–1266 (2016).
Joung, J. et al. Point-of-care testing for COVID-19 using SHERLOCK diagnostics. Preprint at medRxiv https://doi.org/10.1101/2020.05.04.20091231 (2020).
McNerney, M. P. et al. Point-of-care biomarker quantification enabled by sample-specific calibration. Sci. Adv. 5, eaax4473 (2019).
Wen, K. Y. et al. A cell-free biosensor for detecting quorum sensing molecules in P. aeruginosa-infected respiratory samples. ACS Synth. Biol. 6, 2293–2301 (2017).
Danino, T. et al. Programmable probiotics for detection of cancer in urine. Sci. Transl Med. 7, 289ra84 (2015).
Daeffler, K. N.-M. et al. Engineering bacterial thiosulfate and tetrathionate sensors for detecting gut inflammation. Mol. Syst. Biol. 13, 923 (2017).
Mimee, M. et al. An ingestible bacterial-electronic system to monitor gastrointestinal health. Science 360, 915–918 (2018). This article demonstrates that bacterial sensors interface with an electronic capsule to provide real-time reporting of the gut composition.
Cho, I. & Blaser, M. J. The human microbiome: at the interface of health and disease. Nat. Rev. Genet. 13, 260–270 (2012).
Sands, B. E. Biomarkers of inflammation in inflammatory bowel disease. Gastroenterology 149, 1275–1285.e2 (2015).
Vermeire, S., Van Assche, G. & Rutgeerts, P. C-reactive protein as a marker for inflammatory bowel disease. Inflamm. Bowel Dis. 10, 661–665 (2004).
Lehmann, F. S., Burri, E. & Beglinger, C. The role and utility of faecal markers in inflammatory bowel disease. Ther. Adv. Gastroenterol. 8, 23–36 (2015).
Tian, T., Wang, Z. & Zhang, J. Pathomechanisms of oxidative stress in inflammatory bowel disease and potential antioxidant therapies. Oxid. Med. Cell. Longev. 2017, 4535194 (2017).
Winter, S. E. et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 467, 426–429 (2010).
Riglar, D. T. et al. Engineered bacteria can function in the mammalian gut long-term as live diagnostics of inflammation. Nat. Biotechnol. 35, 653–658 (2017). This article demonstrates that bacterial cells can respond to inflammation and ‘remember’ exposure to inflammation for long periods of time, enabling diagnosis of gut inflammation from plating of stool samples from mice fed the engineered bacteria.
Potvin-Trottier, L., Lord, N. D., Vinnicombe, G. & Paulsson, J. Synchronous long-term oscillations in a synthetic gene circuit. Nature 538, 514–517 (2016).
Riglar, D. T. et al. Bacterial variability in the mammalian gut captured by a single-cell synthetic oscillator. Nat. Commun. 10, 4665 (2019).
McCarthy, E. F. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop. J. 26, 154–158 (2006).
Forbes, N. S. Engineering the perfect (bacterial) cancer therapy. Nat. Rev. Cancer 10, 785–794 (2010).
Gniadek, T. J. et al. A phase I, dose escalation, single dose trial of oral attenuated salmonella typhimurium containing human IL-2 in patients with metastatic gastrointestinal cancers. J. Immunother. 43, 217–221 (2020). This report provides a current example of a genetically modified Salmonella strain expressing human IL-2 that is being tested for treating metastic cancer.
Leventhal, D. S. et al. Immunotherapy with engineered bacteria by targeting the STING pathway for anti-tumor immunity. Nat. Commun. 11, 2739 (2020).
Toso, J. F. et al. Phase I study of the intravenous administration of attenuated Salmonella typhimurium to patients with metastatic melanoma. J. Clin. Oncol. 20, 142–152 (2002).
Chien, T., Doshi, A. & Danino, T. Advances in bacterial cancer therapies using synthetic biology. Curr. Opin. Syst. Biol. 5, 1–8 (2017).
Piñero-Lambea, C. et al. Programming controlled adhesion of E. coli to target surfaces, cells, and tumors with synthetic adhesins. ACS Synth. Biol. 4, 463–473 (2015).
Ryan, R. M. et al. Bacterial delivery of a novel cytolysin to hypoxic areas of solid tumors. Gene Ther. 16, 329–339 (2009).
Anderson, J. C., Clarke, E. J., Arkin, A. P. & Voigt, C. A. Environmentally controlled invasion of cancer cells by engineered bacteria. J. Mol. Biol. 355, 619–627 (2006).
Panteli, J. T. & Forbes, N. S. Engineered bacteria detect spatial profiles in glucose concentration within solid tumor cell masses. Biotechnol. Bioeng. 113, 2474–2484 (2016).
Zheng, J. H. et al. Two-step enhanced cancer immunotherapy with engineered Salmonella typhimurium secreting heterologous flagellin. Sci. Transl Med. 9, eaak9537 (2017).
Duong, M. T.-Q., Qin, Y., You, S.-H. & Min, J.-J. Bacteria-cancer interactions: bacteria-based cancer therapy. Exp. Mol. Med. 51, 1–15 (2019).
Din, M. O. et al. Synchronized cycles of bacterial lysis for in vivo delivery. Nature 536, 81–85 (2016). This study uses an elegant synthetic circuit to affect population-level behaviour such that an anticancer payload is delivered to tumours upon bacterial accumulation in the tumour microenvironment.
Chowdhury, S. et al. Programmable bacteria induce durable tumor regression and systemic antitumor immunity. Nat. Med. 25, 1057–1063 (2019).
Nejman, D. et al. The human tumor microbiome is composed of tumor type-specific intracellular bacteria. Science 368, 973–980 (2020).
Riquelme, E. et al. Tumor microbiome diversity and composition influence pancreatic cancer outcomes. Cell 178, 795–806.e12 (2019).
Ozdemir, T., Fedorec, A. J. H., Danino, T. & Barnes, C. P. Synthetic biology and engineered live biotherapeutics: toward increasing system complexity. Cell Syst. 7, 5–16 (2018).
Spisni, E. et al. Cyclooxygenase-2 silencing for the treatment of colitis: a combined in vivo strategy based on RNA interference and engineered Escherichia coli. Mol. Ther. 23, 278–289 (2015).
Citorik, R. J., Mimee, M. & Lu, T. K. Sequence-specific antimicrobials using efficiently delivered RNA-guided nucleases. Nat. Biotechnol. 32, 1141–1145 (2014).
Bikard, D. et al. Exploiting CRISPR–Cas nucleases to produce sequence-specific antimicrobials. Nat. Biotechnol. 32, 1146–1150 (2014).
Flint, H. J., Scott, K. P., Louis, P. & Duncan, S. H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 9, 577–589 (2012).
Charbonneau, M. R., Isabella, V. M., Li, N. & Kurtz, C. B. Developing a new class of engineered live bacterial therapeutics to treat human diseases. Nat. Commun. 11, 1738 (2020).
Kurtz, C. B. et al. An engineered E. coli Nissle improves hyperammonemia and survival in mice and shows dose-dependent exposure in healthy humans. Sci. Transl Med. 11, eaau7975 (2019).
Isabella, V. M. et al. Development of a synthetic live bacterial therapeutic for the human metabolic disease phenylketonuria. Nat. Biotechnol. 36, 857–864 (2018). This study develops a bacterial therapeutic to treat phenylketonuria, which is the basis of a treatment currently in phase II clinical trials.
Hwang, I. Y. et al. Engineered probiotic Escherichia coli can eliminate and prevent Pseudomonas aeruginosa gut infection in animal models. Nat. Commun. 8, 15028 (2017).
Hsu, B. B., Way, J. C. & Silver, P. A. Stable neutralization of a virulence factor in bacteria using temperate phage in the mammalian gut. mSystems 5, 1 (2020).
Center for Biologics Evaluation and Research. Early clinical trials with live biotherapeutic products: chemistry, manufacturing, and control information; guidance for industry. US Food and Drug Administration https://www.fda.gov/regulatory-information/search-fda-guidance-documents/early-clinical-trials-live-biotherapeutic-products-chemistry-manufacturing-and-control-information (2018).
The European Pharmacopoeia Commission. Live Biotherapeutic Products for Human Use Vol. 9 (Council of Europe, 2019).
Dang, L. H., Bettegowda, C., Huso, D. L., Kinzler, K. W. & Vogelstein, B. Combination bacteriolytic therapy for the treatment of experimental tumors. Proc. Natl Acad. Sci. USA 98, 15155–15160 (2001).
Miano, A., Liao, M. J. & Hasty, J. Inducible cell-to-cell signaling for tunable dynamics in microbial communities. Nat. Commun. 11, 1193 (2020).
Mandell, D. J. et al. Biocontainment of genetically modified organisms by synthetic protein design. Nature 518, 55–60 (2015).
Liao, M. J., Din, M. O., Tsimring, L. & Hasty, J. Rock-paper-scissors: engineered population dynamics increase genetic stability. Science 365, 1045–1049 (2019).
Soucy, S. M., Huang, J. & Gogarten, J. P. Horizontal gene transfer: building the web of life. Nat. Rev. Genet. 16, 472–482 (2015).
Ma, N. J. & Isaacs, F. J. Genomic recoding broadly obstructs the propagation of horizontally transferred genetic elements. Cell Syst. 3, 199–207 (2016).
Lienert, F., Lohmueller, J. J., Garg, A. & Silver, P. A. Synthetic biology in mammalian cells: next generation research tools and therapeutics. Nat. Rev. Mol. Cell Biol. 15, 95–107 (2014).
Kitada, T., DiAndreth, B., Teague, B. & Weiss, R. Programming gene and engineered-cell therapies with synthetic biology. Science 359, eaad1067 (2018).
Xie, M. & Fussenegger, M. Designing cell function: assembly of synthetic gene circuits for cell biology applications. Nat. Rev. Mol. Cell Biol. 19, 507–525 (2018).
Sedlmayer, F., Aubel, D. & Fussenegger, M. Synthetic gene circuits for the detection, elimination and prevention of disease. Nat. Biomed. Eng. 2, 399–415 (2018).
Hicks, M., Bachmann, T. T. & Wang, B. Synthetic biology enables programmable cell-based biosensors. ChemPhysChem 21, 132–144 (2020).
Ausländer, D. et al. A designer cell-based histamine-specific human allergy profiler. Nat. Commun. 5, 4408 (2014).
Tastanova, A. et al. Synthetic biology-based cellular biomedical tattoo for detection of hypercalcemia associated with cancer. Sci. Transl Med. 10, eaap8562 (2018).
Goldner, W. Cancer-related hypercalcemia. J. Oncol. Pract. 12, 426–432 (2016).
Clay, T. M. et al. Efficient transfer of a tumor antigen-reactive TCR to human peripheral blood lymphocytes confers anti-tumor reactivity. J. Immunol. 163, 507 (1999).
Rapoport, A. P. et al. NY-ESO-1–specific TCR-engineered T cells mediate sustained antigen-specific antitumor effects in myeloma. Nat. Med. 21, 914–921 (2015).
Morgan, R. A. et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 314, 126 (2006).
Ping, Y., Liu, C. & Zhang, Y. T-cell receptor-engineered T cells for cancer treatment: current status and future directions. Protein Cell 9, 254–266 (2018).
Eshhar, Z., Waks, T., Gross, G. & Schindler, D. G. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the γ or ζ subunits of the immunoglobulin and T-cell receptors. Proc. Natl Acad. Sci. USA 90, 720–724 (1993).
Gross, G., Gorochov, G., Waks, T. & Eshhar, Z. Generation of effector T cells expressing chimeric T cell receptor with antibody type-specificity. Transplant. Proc. 21, 127–130 (1989).
Sadelain, M., Rivière, I. & Riddell, S. Therapeutic T cell engineering. Nature 545, 423–431 (2017).
Hong, M., Clubb, J. D. & Chen, Y. Y. Engineering CAR-T cells for next-generation cancer therapy. Cancer Cell 38, 473–488 (2020).
Levine, B. L., Miskin, J., Wonnacott, K. & Keir, C. Global manufacturing of CAR T cell therapy. Mol. Ther. Methods Clin. Dev. 4, 92–101 (2017).
Majzner, R. G. & Mackall, C. L. Clinical lessons learned from the first leg of the CAR T cell journey. Nat. Med. 25, 1341–1355 (2019).
Shah, N. N. & Fry, T. J. Mechanisms of resistance to CAR T cell therapy. Nat. Rev. Clin. Oncol. 16, 372–385 (2019).
Perales, M.-A., Kebriaei, P., Kean, L. S. & Sadelain, M. Building a safer and faster CAR: seatbelts, airbags, and CRISPR. Biol. Blood Marrow Transpl. 24, 27–31 (2018).
Caliendo, F., Dukhinova, M. & Siciliano, V. Engineered cell-based therapeutics: synthetic biology meets immunology. Front. Bioeng. Biotechnol. 7, 43 (2019).
Esensten, J. H., Bluestone, J. A. & Lim, W. A. Engineering therapeutic T cells: from synthetic biology to clinical trials. Annu. Rev. Pathol. Mech. Dis. 12, 305–330 (2017).
Diaconu, I. et al. Inducible caspase-9 selectively modulates the toxicities of CD19-specific chimeric antigen receptor-modified T cells. Mol. Ther. 25, 580–592 (2017).
Casucci, M. et al. Extracellular NGFR spacers allow efficient tracking and enrichment of fully functional CAR-T cells co-expressing a suicide gene. Front. Immunol. 9, 507 (2018).
Fedorov, V. D., Themeli, M. & Sadelain, M. PD-1- and CTLA-4-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci. Transl Med. 5, 215ra172 (2013).
Chakravarti, D., Caraballo, L. D., Weinberg, B. H. & Wong, W. W. Inducible gene switches with memory in human T cells for cellular immunotherapy. ACS Synth. Biol. 8, 1744–1754 (2019).
Wu, C.-Y., Roybal, K. T., Puchner, E. M., Onuffer, J. & Lim, W. A. Remote control of therapeutic T cells through a small molecule–gated chimeric receptor. Science 350, aab4077 (2015).
Richman, S. A. et al. Ligand-induced degradation of a CAR permits reversible remote control of CAR T cell activity in vitro and in vivo. Mol. Ther. J. Am. Soc. Gene Ther. 28, 1600–1613 (2020). This article demonstrates an approach to improving safety of CAR T cells, through engineering a CAR that contains a ligand-induced degradation domain so that it can be reversibly downregulated.
Guedan, S., Calderon, H., Posey, A. D. & Maus, M. V. Engineering and design of chimeric antigen receptors. Mol. Ther. Methods Clin. Dev. 12, 145–156 (2019).
Roybal, K. T. et al. Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell 164, 770–779 (2016).
Lee, J. et al. Rational design of a bifunctional AND-gate ligand to modulate cell–cell interactions. ACS Synth. Biol. 9, 191–197 (2020).
Stoiber, S. et al. Limitations in the design of chimeric antigen receptors for cancer therapy. Cells 8, 472 (2019).
Salzer, B. et al. Engineering AvidCARs for combinatorial antigen recognition and reversible control of CAR function. Nat. Commun. 11, 4166 (2020). This study shows a way to enhance CAR T cell specificity, through development of a low-affinity CAR that requires bivalent antigen binding and thus prevents off-tumour CAR activation.
Cho, J. H. et al. Engineering advanced logic and distributed computing in human CAR immune cells. Nat. Commun. 12, 792 (2021).
Cartellieri, M. et al. Switching CAR T cells on and off: a novel modular platform for retargeting of T cells to AML blasts. Blood Cancer J. 6, e458 (2016).
Cho, J. H., Collins, J. J. & Wong, W. W. Universal chimeric antigen receptors for multiplexed and logical control of T cell responses. Cell 173, 1426–1438.e11 (2018). This article presents a modular CAR system, composed of a universal receptor and adapter scFvs, that enables targeting of different antigens without having to entirely re-engineer the CAR.
Urbanska, K. et al. A universal strategy for adoptive immunotherapy of cancer through use of a novel T-cell antigen receptor. Cancer Res. 72, 1844–1852 (2012).
Loff, S. et al. Rapidly switchable universal CAR-T cells for treatment of CD123-positive leukemia. Mol. Ther. Oncolytics 17, 408–420 (2020).
Rosewell Shaw, A. & Suzuki, M. Oncolytic viruses partner with T-cell therapy for solid tumor treatment. Front. Immunol. 9, 2103 (2018).
Ajina, A. & Maher, J. Prospects for combined use of oncolytic viruses and CAR T-cells. J. Immunother. Cancer 5, 90 (2017).
Amor, C. et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583, 127–132 (2020).
Kirkland, J. L. & Tchkonia, T. Cellular senescence: a translational perspective. EBioMedicine 21, 21–28 (2017).
Baker, D. J. et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236 (2011).
Franceschi, C., Garagnani, P., Vitale, G., Capri, M. & Salvioli, S. Inflammaging and ‘Garb-aging’. Trends Endocrinol. Metab. 28, 199–212 (2017).
Higashikuni, Y., Chen, W. C. & Lu, T. K. Advancing therapeutic applications of synthetic gene circuits. Curr. Opin. Biotechnol. 47, 133–141 (2017).
Ho, P. & Chen, Y. Y. Mammalian synthetic biology in the age of genome editing and personalized medicine. Curr. Opin. Chem. Biol. 40, 57–64 (2017).
Schukur, L., Geering, B., Hamri, G. C.-E. & Fussenegger, M. Implantable synthetic cytokine converter cells with AND-gate logic treat experimental psoriasis. Sci. Transl Med. 7, 318ra201 (2015). This article develops theranostic cells to sense pro-inflammatory cytokines associated with psoriasis and to respond by expressing therapeutic anti-inflammatory cytokines.
Ye, H. et al. Self-adjusting synthetic gene circuit for correcting insulin resistance. Nat. Biomed. Eng. 1, 005 (2017).
Liu, Y. et al. Immunomimetic designer cells protect mice from MRSA infection. Cell 174, 259–270.e11 (2018).
Chassin, H. et al. Sensing and responding to allergic response cytokines through a genetically encoded circuit. Nat. Commun. 8, 1101 (2017).
Qudrat, A., Mosabbir, A. A. & Truong, K. Engineered proteins program mammalian cells to target inflammatory disease sites. Cell Chem. Biol. 24, 703–711.e2 (2017).
Smole, A., Lainšcˇek, D., Bezeljak, U., Horvat, S. & Jerala, R. A synthetic mammalian therapeutic gene circuit for sensing and suppressing inflammation. Mol. Ther. 25, 102–119 (2017).
Nowakowski, A., Walczak, P., Janowski, M. & Lukomska, B. Genetic engineering of mesenchymal stem cells for regenerative medicine. Stem Cell Dev. 24, 2219–2242 (2015).
Moon, H.-H. et al. MSC-based VEGF gene therapy in rat myocardial infarction model using facial amphipathic bile acid-conjugated polyethyleneimine. Biomaterials 35, 1744–1754 (2014).
Lai, T. et al. Over-expression of VEGF in marrow stromal cells promotes angiogenesis in rats with cerebral infarction via the synergistic effects of VEGF and Ang-2. J. Huazhong Univ. Sci. Technol. Med. Sci. 32, 724–731 (2012).
Serra, J. et al. Engineering of human mesenchymal stem/stromal cells with vascular endothelial growth factor-encoding minicircles for angiogenic ex vivo gene therapy. Hum. Gene Ther. 30, 316–329 (2019).
Rebar, E. J. et al. Induction of angiogenesis in a mouse model using engineered transcription factors. Nat. Med. 8, 1427–1432 (2002).
Fan, L. et al. Transplantation with survivin-engineered mesenchymal stem cells results in better prognosis in a rat model of myocardial infarction. Eur. J. Heart Fail. 11, 1023–1030 (2009).
Sakharkar, M. K., Chow, V. T. K. & Kangueane, P. Distributions of exons and introns in the human genome. Silico Biol. 4, 387–393 (2004).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Kiani, A. A. et al. Over expression of HIF-1α in human mesenchymal stem cells increases their supportive functions for hematopoietic stem cells in an experimental co-culture model. Hematol. Amst. Neth. 19, 85–98 (2014).
Ku, T.-H. et al. Nucleic acid aptamers: an emerging tool for biotechnology and biomedical sensing. Sensors 15, 16281–16313 (2015).
McMahon, C. et al. Yeast surface display platform for rapid discovery of conformationally selective nanobodies. Nat. Struct. Mol. Biol. 25, 289–296 (2018).
McNerney, M. P., Watstein, D. M. & Styczynski, M. P. Precision metabolic engineering: the design of responsive, selective, and controllable metabolic systems. Metab. Eng. 31, 123–131 (2015).
Nielsen, A. A. K. et al. Genetic circuit design automation. Science 352, aac7341 (2016).
Lin, J. K. et al. Cost effectiveness of chimeric antigen receptor T-cell therapy in relapsed or refractory pediatric B-cell acute lymphoblastic leukemia. J. Clin. Oncol. 36, 3192–3202 (2018).
Qasim, W. et al. Molecular remission of infant B-ALL after infusion of universal TALEN gene-edited CAR T cells. Sci. Transl Med. 9, eaaj2013 (2017).
Benjamin, R. et al. Genome-edited, donor-derived allogeneic anti-CD19 chimeric antigen receptor T cells in paediatric and adult B-cell acute lymphoblastic leukaemia: results of two phase 1 studies. Lancet 396, 1885–1894 (2020).
FDA. FDA approves novel gene therapy to treat patients with a rare form of inherited vision loss. US Food and Drug Administration https://www.fda.gov/news-events/press-announcements/fda-approves-novel-gene-therapy-treat-patients-rare-form-inherited-vision-loss (2020).
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Sadelain, M., Brentjens, R. & Rivière, I. The promise and potential pitfalls of chimeric antigen receptors. Curr. Opin. Immunol. 21, 215–223 (2009).
Chmielewski, M., Kopecky, C., Hombach, A. A. & Abken, H. IL-12 release by engineered T cells expressing chimeric antigen receptors can effectively muster an antigen-independent macrophage response on tumor cells that have shut down tumor antigen expression. Cancer Res. 71, 5697–5706 (2011).
Vivier, E., Tomasello, E., Baratin, M., Walzer, T. & Ugolini, S. Functions of natural killer cells. Nat. Immunol. 9, 503–510 (2008).
Liu, E. L. et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N. Engl. J. Med. 382, 545–553 (2020).
Xie, G. et al. CAR-NK cells: a promising cellular immunotherapy for cancer. EBioMedicine 59, 102975 (2020).
Yang, Y. et al. Phase I study of random healthy donor-derived allogeneic natural killer cell therapy in patients with malignant lymphoma or advanced solid tumors. Cancer Immunol. Res. 4, 215–224 (2016).
Lupo, K. B. & Matosevic, S. Natural killer cells as allogeneic effectors in adoptive cancer immunotherapy. Cancers 11, 769 (2019).
Li, Y., Hermanson, D. L., Moriarity, B. S. & Kaufman, D. S. Human iPSC-derived natural killer cells engineered with chimeric antigen receptors enhance anti-tumor activity. Cell Stem Cell 23, 181–192.e5 (2018).
Lim, R. M., Rong, L., Zhen, A. J. & Xie, J. M. A universal CAR-NK cell targeting various epitopes of HIV-1 gp160. ACS Chem. Biol. 15, 2299–2310 (2020).
Klichinsky, M. et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat. Biotechnol. 38, 947–953 (2020). This work describes the therapeutic effect of CAR macrophages, and demonstrates the ability of CAR-engineered cells to target solid tumours.
Murphy, K. & Weaver, C. in Janeway’s Immunobiology 9th edn 355–365 (Garland Science, 2017).
Rana, J. & Biswas, M. Regulatory T cell therapy: current and future design perspectives. Cell. Immunol. 356, 104193 (2020).
Arce-Sillas, A. et al. Regulatory T cells: molecular actions on effector cells in immune regulation. J. Immunol. Res. 2016, 1720827 (2016).
Grossman, W. J. et al. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity 21, 589–601 (2004).
Brusko, T. M. et al. Human antigen-specific regulatory T cells generated by T cell receptor gene transfer. PLoS ONE 5, e11726 (2010).
Mohseni, Y. R. et al. The future of regulatory T cell therapy: promises and challenges of implementing CAR technology. Front. Immunol. 11, 13 (2020).
MacDonald, K. G. et al. Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor. J. Clin. Invest. 126, 1413–1424 (2016).
Acknowledgements
The authors acknowledge members of the Silver laboratory for helpful conversations and acknowledge support from Harvard Medical School’s Dean’s Initiative Award and DARPA (140D0420C0057).
Author information
Authors and Affiliations
Contributions
M.P.M., K.E.D., T.L.N. and T.Z.C. researched the literature and wrote the article. All authors contributed to discussions of the content and reviewed and/or edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Genetics thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Off-target effects
-
Unintended therapeutic consequences that occur when a drug binds to molecules in the body that are not the intended target of the drug.
- Synthetic biology
-
A multidisciplinary field of biological research that seeks to design and engineer biology akin to other engineering disciplines. Goals of the field are the development of molecules, cells and even organisms with novel functions.
- Chimeric antigen receptor
-
(CAR). A synthetic receptor that combines the specific, extracellular antigen-binding ability of an antibody and the T cell-activating abilities of the T cell receptor (intracellular stimulatory domains) to redirect immune cell action towards a disease antigen of interest.
- Cell-free systems
-
Mixtures of proteins, nucleic acids, salts and metabolites that resemble the cytoplasm of cells and can be used to enact genetic circuits.
- Probiotics
-
Beneficial microorganisms that are used to promote health. Probiotics can be naturally occurring microorganisms or microorganisms that have been engineered to sense and respond to a specific condition.
- Genetic circuits
-
DNA systems in which the presence of different combinations of signals leads to differential gene expression. Circuits usually consist of an input (that is, surface receptor binding to a ligand) and an output (that is, expression of therapeutics) that is enacted only after the input condition is satisfied.
- Transcription factors
-
Proteins that control the expression of a gene, generally either by activating or repressing its expression.
- Quorum-sensing systems
-
Cell to cell communication systems, prominent in prokaryotes, that use extracellular small molecules as signalling cues.
- Dysbiosis
-
An imbalance of the gut microbiome community. Disruption to gut bacterial homeostasis has been associated with human diseases such as inflammatory bowel diseases and irritable bowel syndrome.
- Chassis
-
The organism or cell line that houses a genetic circuit and is engineered to perform specific tasks.
- Biofilm
-
A collection of microorganisms associated with a primarily polysaccharide-based matrix. Biofilms are recalcitrant to antibiotic treatments.
- Alginate beads
-
Beads used to encapsulate cells to allow their implantation into the body while keeping them isolated from the host immune system. In addition to preventing an immune response, alginate encapsulation forms a semipermeable barrier to allow the diffusion of nutrients and gases.
- T cell receptor
-
(TCR). The antigen-binding complex natively expressed on the surface of T cells. Binding of antigen to the TCR is necessary for T cell activation.
- Antigen peptides
-
Short amino acid sequences that can be recognized by T cell receptors (TCRs) or antibodies expressed by cells of the adaptive immune system. Binding of an antigen to cell receptors or antibodies can trigger an immune response.
- Single-chain variable fragment
-
(scFV). A fusion protein consisting of the variable domains of the heavy and light chains of an antibody connected by a small linker. scFvs confer antigen-binding specificity to the chimeric antigen receptor (CAR).
- Autologous
-
A term to denote cells or tissue derived from a patient for the treatment of that same patient.
- Cytokine release syndrome
-
A potentially deadly adverse effect of chimeric antigen receptor (CAR) T cell therapy, resulting from the mass release of pro-inflammatory cytokine expression from activated T cells.
- Cytokines
-
Secreted proteins that bind to receptors on other cells that induce a change in that target cell, generally categorized as pro-inflammatory or anti-inflammatory.
- Boolean logic gates
-
Systems of binary, on/off signals used to generate complex behaviours in a system (for example, an AND-gated circuit necessitates the presence of two inputs before expression of the output).
- Allogeneic
-
A term to describe cells or tissue derived from one person used to treat a genetically different person (allotransplantation).
Rights and permissions
About this article
Cite this article
McNerney, M.P., Doiron, K.E., Ng, T.L. et al. Theranostic cells: emerging clinical applications of synthetic biology. Nat Rev Genet 22, 730–746 (2021). https://doi.org/10.1038/s41576-021-00383-3
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41576-021-00383-3
This article is cited by
-
Hyperspectral reporters for long-distance and wide-area detection of gene expression in living bacteria
Nature Biotechnology (2026)
-
Delivering living medicines with biomaterials
Nature Reviews Materials (2025)
-
On-demand treatment of metabolic diseases by a synthetic drug-inducible exocytosis system
Nature Communications (2025)
-
Ingestible optoelectronic capsules enable bidirectional communication with engineered microbes for controllable therapeutic interventions
Nature Microbiology (2025)
-
Smart closed-loop drug delivery systems
Nature Reviews Bioengineering (2025)