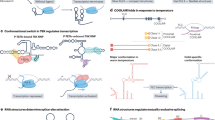
Abstract
The versatility of RNA in sensing and interacting with small molecules, proteins and other nucleic acids while encoding genetic instructions for protein translation makes it a powerful substrate for engineering biological systems. RNA devices integrate cellular information sensing, processing and actuation of specific signals into defined functions and have yielded programmable biological systems and novel therapeutics of increasing sophistication. However, challenges centred on expanding the range of analytes that can be sensed and adding new mechanisms of action have hindered the full realization of the field’s promise. Here, we describe recent advances that address these limitations and point to a significant maturation of synthetic RNA-based devices.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nshogozabahizi, J. C., Aubrey, K. L., Ross, J. A. & Thakor, N. Applications and limitations of regulatory RNA elements in synthetic biology and biotechnology. J. Appl. Microbiol. 127, 968–984 (2019).
Kim, J. & Franco, E. RNA nanotechnology in synthetic biology. Curr. Opin. Biotechnol. 63, 135–141 (2020).
Schmidt, C. M. & Smolke, C. D. RNA switches for synthetic biology. Cold Spring Harb. Perspect. Biol. 11, 135–141 (2019).
Park, S. V. et al. Catalytic RNA, ribozyme, and its applications in synthetic biology. Biotechnol. Adv. 37, 107452 (2019).
Win, M. N., Liang, J. C. & Smolke, C. D. Frameworks for programming biological function through RNA parts and devices. Chem. Biol. 16, 298–310 (2009).
Liang, J. C., Bloom, R. J. & Smolke, C. D. Engineering biological systems with synthetic RNA molecules. Mol. Cell 43, 915–926 (2011).
Nahvi, A. et al. Genetic control by a metabolite binding mRNA. Chem. Biol. 9, 1043–1049 (2002).
Winkler, W. C., Cohen-Chalamish, S. & Breaker, R. R. An mRNA structure that controls gene expression by binding FMN. Proc. Natl Acad. Sci. USA 99, 15908–15913 (2002).
Sudarsan, N., Barrick, J. E. & Breaker, R. R. Metabolite-binding RNA domains are present in the genes of eukaryotes. RNA 9, 644–647 (2003).
Isaacs, F. J. et al. Engineered riboregulators enable post-transcriptional control of gene expression. Nat. Biotechnol. 22, 841–847 (2004).
Suess, B. & Weigand, J. E. Engineered riboswitches: overview, problems and trends. RNA Biol. 5, 24–29 (2008).
McKeague, M., Wong, R. S. & Smolke, C. D. Opportunities in the design and application of RNA for gene expression control. Nucleic Acids Res. 44, 2987–2999 (2016).
Westbrook, A. M. & Lucks, J. B. Achieving large dynamic range control of gene expression with a compact RNA transcription–translation regulator. Nucleic Acids Res. 45, 5614–5624 (2017).
Kim, J. et al. De novo-designed translation-repressing riboregulators for multi-input cellular logic. Nat. Chem. Biol. 15, 1173–1182 (2019).
Rauch, S., Jones, K. A. & Dickinson, B. C. Small molecule-inducible RNA-targeting systems for temporal control of RNA regulation. ACS Cent. Sci. 6, 1987–1996 (2020).
Chappell, J., Westbrook, A., Verosloff, M. & Lucks, J. B. Computational design of small transcription activating RNAs for versatile and dynamic gene regulation. Nat. Commun. 8, 1051 (2017).
Anzalone, A. V., Lin, A. J., Zairis, S., Rabadan, R. & Cornish, V. W. Reprogramming eukaryotic translation with ligand-responsive synthetic RNA switches. Nat. Methods 13, 453–458 (2016).
Spöring, M., Finke, M. & Hartig, J. S. Aptamers in RNA-based switches of gene expression. Curr. Opin. Biotechnol. 63, 34–40 (2020).
Kawasaki, S., Ono, H., Hirosawa, M. & Saito, H. RNA and protein-based nanodevices for mammalian post-transcriptional circuits. Curr. Opin. Biotechnol. 63, 99–110 (2020).
Endy, D. Foundations for engineering biology. Nature 438, 449–453 (2005).
Barrick, J. E. & Breaker, R. R. The distributions, mechanisms, and structures of metabolite-binding riboswitches. Genome Biol. 8, R239 (2007).
Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990).
Lauridsen, L. H., Doessing, H. B., Long, K. S. & Nielsen, A. T. in Synthetic Metabolic Pathways: Methods and Protocols (eds. Jensen, M. K. & Keasling, J. D.) 291–306 (Springer, 2018).
Baird, G. S. Where are all the aptamers? Am. J. Clin. Pathol. 134, 529–531 (2010).
Dunn, M. R., Jimenez, R. M. & Chaput, J. C. Analysis of aptamer discovery and technology. Nat. Rev. Chem. 1, 0076 (2017).
McKeague, M. & DeRosa, M. C. Challenges and opportunities for small molecule aptamer development. J. Nucleic Acids 2012, 1–20 (2012).
McKeague, M. et al. Analysis of in vitro aptamer selection parameters. J. Mol. Evol. 81, 150–161 (2015).
Valencia-Sanchez, M. A., Liu, J., Hannon, G. J. & Parker, R. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev. 20, 515–524 (2006).
Bloom, R. J., Winkler, S. M. & Smolke, C. D. Synthetic feedback control using an RNAi-based gene-regulatory device. J. Biol. Eng. 9, 5 (2015).
Lucks, J. B., Qi, L., Mutalik, V. K., Wang, D. & Arkin, A. P. Versatile RNA-sensing transcriptional regulators for engineering genetic networks. Proc. Natl Acad. Sci. USA 108, 8617–8622 (2011).
Ausländer, S. & Fussenegger, M. Synthetic RNA-based switches for mammalian gene expression control. Curr. Opin. Biotechnol. 48, 54–60 (2017).
Chappell, J. et al. The centrality of RNA for engineering gene expression. Biotechnol. J. 8, 1379–1395 (2013).
Till, P., Toepel, J., Bühler, B., Mach, R. L. & Mach-Aigner, A. R. Regulatory systems for gene expression control in cyanobacteria. Appl. Microbiol. Biotechnol. 104, 1977–1991 (2020).
Bayer, T. S. & Smolke, C. D. Programmable ligand-controlled riboregulators of eukaryotic gene expression. Nat. Biotechnol. 23, 337–343 (2005).
Kawasaki, S., Fujita, Y., Nagaike, T., Tomita, K. & Saito, H. Synthetic mRNA devices that detect endogenous proteins and distinguish mammalian cells. Nucleic Acids Res. 45, e117–e117 (2017).
Paige, J. S., Nguyen-Duc, T., Song, W. & Jaffrey, S. R. Fluorescence imaging of cellular metabolites with RNA. Science 335, 1194–1194 (2012).
Jepsen, M. D. E. et al. Development of a genetically encodable FRET system using fluorescent RNA aptamers. Nat. Commun. 9, 18 (2018).
Wurmthaler, L. A., Sack, M., Gense, K., Hartig, J. S. & Gamerdinger, M. A tetracycline-dependent ribozyme switch allows conditional induction of gene expression in Caenorhabditis elegans. Nat. Commun. 10, 491 (2019).
Gu, H., Furukawa, K. & Breaker, R. R. Engineered allosteric ribozymes that sense the bacterial second messenger cyclic diguanosyl 5′-monophosphate. Anal. Chem. 84, 4935–4941 (2012).
Niu, T. et al. Engineering a glucosamine-6-phosphate responsive glmS ribozyme switch enables dynamic control of metabolic flux in Bacillus subtilis for overproduction of N-acetylglucosamine. ACS Synth. Biol. 7, 2423–2435 (2018).
Sterner, R. C. & Sterner, R. M. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 11, 1–11 (2021).
Di Stasi, A. et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N. Engl. J. Med. 365, 1673–1683 (2011).
Chen, Y. Y., Jensen, M. C. & Smolke, C. D. Genetic control of mammalian T-cell proliferation with synthetic RNA regulatory systems. Proc. Natl Acad. Sci. USA 107, 8531–8536 (2010).
Wong, R. S., Chen, Y. Y. & Smolke, C. D. Regulation of T cell proliferation with drug-responsive microRNA switches. Nucleic Acids Res. 46, 1541–1552 (2018).
Liu, Y. et al. Directing cellular information flow via CRISPR signal conductors. Nat. Methods 13, 938–944 (2016).
Liu, Y. et al. Engineering cell signaling using tunable CRISPR–Cpf1-based transcription factors. Nat. Commun. 8, 2095 (2017).
Tang, W., Hu, J. H. & Liu, D. R. Aptazyme-embedded guide RNAs enable ligand-responsive genome editing and transcriptional activation. Nat. Commun. 8, 15939 (2017).
Hoffmann, M. D. et al. Cell-specific CRISPR–Cas9 activation by microRNA-dependent expression of anti-CRISPR proteins. Nucleic Acids Res. 47, e75 (2019).
Hirosawa, M. et al. Cell-type-specific genome editing with a microRNA-responsive CRISPR–Cas9 switch. Nucleic Acids Res. 45, e118 (2017).
Hirosawa, M., Fujita, Y. & Saito, H. Cell-type-specific CRISPR activation with microRNA-responsive AcrllA4 switch. ACS Synth. Biol. 8, 1575–1582 (2019).
Lee, J. et al. Tissue-restricted genome editing in vivo specified by microRNA-repressible anti-CRISPR proteins. RNA 25, 1421–1431 (2019).
Davis, K. M., Pattanayak, V., Thompson, D. B., Zuris, J. A. & Liu, D. R. Small molecule-triggered Cas9 protein with improved genome-editing specificity. Nat. Chem. Biol. 11, 316–318 (2015).
Robbins, P. D., Tahara, H. & Ghivizzani, S. C. Viral vectors for gene therapy. Trends Biotechnol. 16, 35–40 (1998).
Strobel, B. et al. Riboswitch-mediated attenuation of transgene cytotoxicity increases adeno-associated virus vector yields in HEK-293 cells. Mol. Ther. 23, 1582–1591 (2015).
Takahashi, K. & Yokobayashi, Y. Reversible gene regulation in mammalian cells using riboswitch-engineered vesicular stomatitis virus vector. ACS Synth. Biol. 8, 1976–1982 (2019).
Reid, C. A., Nettesheim, E. R., Connor, T. B. & Lipinski, D. M. Development of an inducible anti-VEGF rAAV gene therapy strategy for the treatment of wet AMD. Sci. Rep. 8, 11763 (2018).
Strobel, B. et al. A small-molecule-responsive riboswitch enables conditional induction of viral vector-mediated gene expression in mice. ACS Synth. Biol. 9, 1292–1305 (2020).
Han, S. R. et al. Targeted suicide gene therapy for liver cancer based on ribozyme-mediated RNA replacement through post-transcriptional regulation. Mol. Ther. Nucleic Acids 23, 154–168 (2021).
Wang, H. et al. Characterization of a bifunctional synthetic RNA aptamer and a truncated form for ability to inhibit growth of non-small cell lung cancer. Sci. Rep. 9, 18836 (2019).
Worgall, S., Wolff, G., Falck-Pedersen, E. & Crystal, R. G. Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration. Hum. Gene Ther. 8, 37–44 (1997).
Uehata, T. & Takeuchi, O. RNA recognition and immunity-innate immune sensing and its posttranscriptional regulation mechanisms. Cells 9, E1701 (2020).
Ireton, R. C., Wilkins, C. & Gale, M. RNA PAMPs as molecular tools for evaluating RIG-I function in innate immunity. Methods Mol. Biol. 1656, 119–129 (2017).
Kell, A. M. & Gale, M. RIG-I in RNA virus recognition. Virology 479–480, 110–121 (2015).
Wu, M. Z., Asahara, H., Tzertzinis, G. & Roy, B. Synthesis of low immunogenicity RNA with high-temperature in vitro transcription. RNA 26, 345–360 (2020).
Andries, O. et al. N1-Methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. J. Control. Rel. 217, 337–344 (2015).
Karikó, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Wesselhoeft, R. A. et al. RNA circularization diminishes immunogenicity and can extend translation duration in vivo. Mol. Cell 74, 508–520.e4 (2019).
Pardi, N., Hogan, M. J. & Weissman, D. Recent advances in mRNA vaccine technology. Curr. Opin. Immunol. 65, 14–20 (2020).
Wadhwa, A., Aljabbari, A., Lokras, A., Foged, C. & Thakur, A. Opportunities and challenges in the delivery of mRNA-based vaccines. Pharmaceutics 12, E102 (2020).
Dua, P., Kim, S. & Lee, D. Nucleic acid aptamers targeting cell-surface proteins. Methods 54, 215–225 (2011).
Narberhaus, F., Waldminghaus, T. & Chowdhury, S. RNA thermometers. FEMS Microbiol. Rev. 30, 3–16 (2006).
Neupert, J., Karcher, D. & Bock, R. Design of simple synthetic RNA thermometers for temperature-controlled gene expression in Escherichia coli. Nucleic Acids Res. 36, e124–e124 (2008).
Lotz, T. S. et al. A light-responsive RNA aptamer for an azobenzene derivative. Nucleic Acids Res. 47, 2029–2040 (2019).
Gold, L. et al. Aptamer-based multiplexed proteomic technology for biomarker discovery. PLoS ONE 5, e15004 (2010).
Darmostuk, M., Rimpelova, S., Gbelcova, H. & Ruml, T. Current approaches in SELEX: an update to aptamer selection technology. Biotechnol. Adv. 33, 1141–1161 (2015).
Wayment-Steele, H., Wu, M., Gotrik, M. & Das, R. in Methods in Enzymology Vol. 623 Ch. 18 (ed. Hargrove, A. E.) 417–450 (Academic, 2019).
Davis, J. H. & Szostak, J. W. Isolation of high-affinity GTP aptamers from partially structured RNA libraries. Proc. Natl Acad. Sci. USA 99, 11616–11621 (2002).
Kohlberger, M. & Gadermaier, G. SELEX: critical factors and optimization strategies for successful aptamer selection. Biotechnol. Appl. Biochem. https://doi.org/10.1002/bab.2244 (2021).
Komarova, N. & Kuznetsov, A. Inside the black box: what makes SELEX better? Molecules 24, E3598 (2019).
Ricci, F., Vallée-Bélisle, A., Simon, A. J., Porchetta, A. & Plaxco, K. W. Using nature’s “tricks” to rationally tune the binding properties of biomolecular receptors. Acc. Chem. Res. 49, 1884–1892 (2016).
Townshend, B., Xiang, J. S., Manzanarez, G., Hayden, E. J. & Smolke, C. D. A multiplexed, automated evolution pipeline enables scalable discovery and characterization of biosensors. Nat. Commun. 12, 1437 (2021).
Hasegawa, H., Savory, N., Abe, K. & Ikebukuro, K. Methods for improving aptamer binding affinity. Molecules 21, 421 (2016).
Kalra, P., Dhiman, A., Cho, W. C., Bruno, J. G. & Sharma, T. K. Simple methods and rational design for enhancing aptamer sensitivity and specificity. Front. Mol. Biosci. 5, 41 (2018).
Stoltenburg, R., Nikolaus, N. & Strehlitz, B. Capture-SELEX: selection of DNA aptamers for aminoglycoside antibiotics. J. Anal. Methods Chem. 2012, 1–14 (2012).
Boussebayle, A., Groher, F. & Suess, B. RNA-based Capture-SELEX for the selection of small molecule-binding aptamers. Methods 161, 10–15 (2019).
Koizumi, M., Soukup, G. A., Kerr, J. N. & Breaker, R. R. Allosteric selection of ribozymes that respond to the second messengers cGMP and cAMP. Nat. Struct. Biol. 6, 1062–1071 (1999).
Xiang, J. S. et al. Massively parallel RNA device engineering in mammalian cells with RNA-seq. Nat. Commun. 10, 4327 (2019).
Strobel, B. et al. High-throughput identification of synthetic riboswitches by barcode-free amplicon-sequencing in human cells. Nat. Commun. 11, 1–12 (2020).
Townshend, B., Kennedy, A. B., Xiang, J. S. & Smolke, C. D. High-throughput cellular RNA device engineering. Nat. Methods 12, 989–994 (2015).
Nomura, Y., Chien, H.-C. & Yokobayashi, Y. Direct screening for ribozyme activity in mammalian cells. Chem. Commun. 53, 12540–12543 (2017).
Gotrik, M. et al. Direct selection of fluorescence-enhancing RNA aptamers. J. Am. Chem. Soc. 140, 3583–3591 (2018).
Zhao, B. S., Roundtree, I. A. & He, C. Post-transcriptional gene regulation by mRNA modifications. Nat. Rev. Mol. Cell Biol. 18, 31–42 (2017).
Holley, R. W., Everett, G. A., Madison, J. T. & Zamir, A. Nucleotide sequences in the yeast alanine transfer ribonucleic acid. J. Biol. Chem. 240, 2122–2128 (1965).
Pereira, M. et al. Impact of tRNA modifications and tRNA-modifying enzymes on proteostasis and human disease. Int. J. Mol. Sci. 19, E3738 (2018).
Dominissini, D. et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature 485, 201–206 (2012).
Meyer, K. D. et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell 149, 1635–1646 (2012).
Jaffrey, S. R. & Kharas, M. G. Emerging links between m6A and misregulated mRNA methylation in cancer. Genome Med. 9, 2 (2017).
Liu, J. et al. A METTL3–METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat. Chem. Biol. 10, 93–95 (2014).
Wilson, C., Chen, P. J., Miao, Z. & Liu, D. R. Programmable m6A modification of cellular RNAs with a Cas13-directed methyltransferase. Nat. Biotechnol. 38, 1431–1440 (2020).
Xiao, W. et al. Nuclear m6A reader YTHDC1 regulates mRNA splicing. Mol. Cell 61, 507–519 (2016).
Rees, H. A. & Liu, D. R. Base editing: precision chemistry on the genome and transcriptome of living cells. Nat. Rev. Genet. 19, 770–788 (2018).
Marina, R. J., Brannan, K. W., Dong, K. D., Yee, B. A. & Yeo, G. W. Evaluation of engineered CRISPR-cas-mediated systems for site-specific RNA editing. Cell Rep. 33, 108350 (2020).
Qu, L. et al. Programmable RNA editing by recruiting endogenous ADAR using engineered RNAs. Nat. Biotechnol. 37, 1059–1069 (2019).
Cox, D. B. T. et al. RNA editing with CRISPR–Cas13. Science 358, 1019–1027 (2017).
Rauch, S. et al. Programmable RNA-guided RNA effector proteins built from human parts. Cell 178, 122–134.e12 (2019).
Abudayyeh, O. O. et al. A cytosine deaminase for programmable single-base RNA editing. Science 365, 382–386 (2019).
Salzman, J. Circular RNA expression: its potential regulation and function. Trends Genet. 32, 309–316 (2016).
Lasda, E. & Parker, R. Circular RNAs: diversity of form and function. RNA 20, 1829–1842 (2014).
Akhter, R. Circular RNA and Alzheimer’s disease. Adv. Exp. Med. Biol. 1087, 239–243 (2018).
Altesha, M.-A., Ni, T., Khan, A., Liu, K. & Zheng, X. Circular RNA in cardiovascular disease. J. Cell Physiol. 234, 5588–5600 (2019).
Han, B., Chao, J. & Yao, H. Circular RNA and its mechanisms in disease: from the bench to the clinic. Pharmacol. Ther. 187, 31–44 (2018).
Prats, A.-C. et al. Circular RNA, the key for translation. Int. J. Mol. Sci. 21, 8591 (2020).
Legnini, I. et al. Circ-ZNF609 is a circular RNA that can be translated and functions in myogenesis. Mol. Cell 66, 22–37.e9 (2017).
Pamudurti, N. R. et al. Translation of circRNAs. Mol. Cell 66, 9–21.e7 (2017).
Litke, J. L. & Jaffrey, S. R. Highly efficient expression of circular RNA aptamers in cells using autocatalytic transcripts. Nat. Biotechnol. 37, 667–675 (2019).
Wesselhoeft, R. A., Kowalski, P. S. & Anderson, D. G. Engineering circular RNA for potent and stable translation in eukaryotic cells. Nat. Commun. 9, 2629 (2018).
Meganck, R. M. et al. Engineering highly efficient backsplicing and translation of synthetic circRNAs. Mol. Ther. Nucleic Acids 23, 821–834 (2021).
Paige, J. S., Wu, K. Y. & Jaffrey, S. R. RNA mimics of green fluorescent protein. Science 333, 642–646 (2011).
Rossbach, O. Artificial circular RNA sponges targeting microRNAs as a novel tool in molecular biology. Mol. Ther. Nucleic Acids 17, 452–454 (2019).
Jost, I. et al. Functional sequestration of microRNA-122 from hepatitis C virus by circular RNA sponges. RNA Biol. 15, 1032–1039 (2018).
Blight, K. J., McKeating, J. A. & Rice, C. M. Highly permissive cell lines for subgenomic and genomic hepatitis C virus RNA replication. J. Virol. 76, 13001–13014 (2002).
Schreiner, S., Didio, A., Hung, L.-H. & Bindereif, A. Design and application of circular RNAs with protein-sponge function. Nucleic Acids Res. 48, 12326–12335 (2020).
Li, X., Yang, L. & Chen, L.-L. The biogenesis, functions, and challenges of circular RNAs. Mol. Cell 71, 428–442 (2018).
Delebecque, C. J., Lindner, A. B., Silver, P. A. & Aldaye, F. A. Organization of intracellular reactions with rationally designed RNA assemblies. Science 333, 470–474 (2011).
Sachdeva, G., Garg, A., Godding, D., Way, J. C. & Silver, P. A. In vivo co-localization of enzymes on RNA scaffolds increases metabolic production in a geometrically dependent manner. Nucleic Acids Res. 42, 9493–9503 (2014).
Shibata, T. et al. Protein-driven RNA nanostructured devices that function in vitro and control mammalian cell fate. Nat. Commun. 8, 540 (2017).
WHO. WHO coronavirus (COVID-19) dashboard with vaccination data. World Health Organization https://covid19.who.int/info (2020).
Polack, F. P. et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020).
Corbett, K. S. et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 586, 567–571 (2020).
Slaoui, M. & Hepburn, M. Developing safe and effective covid vaccines — Operation Warp Speed’s strategy and approach. N. Engl. J. Med. 383, 1701–1703 (2020).
Bell, J. Moderna founder’s next big play in RNA raises $440M. BioPharma Dive https://www.biopharmadive.com/news/laronde-endless-rna-series-b-flagship-moderna/605740/ (2021).
Bell, J. Venture capital pours more money into RNA medicines with the launch of Replicate. BioPharma Dive https://www.biopharmadive.com/news/replicate-launch-rna-ehlers-apple-tree/606210/ (2021).
Al Idrus, A. Shape builds out RNA editing tech with a major $112M funding boost. FierceBiotech https://www.fiercebiotech.com/biotech/shape-therapeutics-reels-112m-to-spur-rna-editing-tech (2021).
Baek, M. et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science 373, 871–876 (2021).
Senior, A. W. et al. Improved protein structure prediction using potentials from deep learning. Nature 577, 706–710 (2020).
Kuhlman, B. & Bradley, P. Advances in protein structure prediction and design. Nat. Rev. Mol. Cell Biol. 20, 681–697 (2019).
Langan, R. A. et al. De novo design of bioactive protein switches. Nature 572, 205–210 (2019).
Zhang, K. et al. Cryo-EM structure of a 40 kDa SAM-IV riboswitch RNA at 3.7 Å resolution. Nat. Commun. 10, 5511 (2019).
Mustoe, A. M., Lama, N. N., Irving, P. S., Olson, S. W. & Weeks, K. M. RNA base-pairing complexity in living cells visualized by correlated chemical probing. Proc. Natl Acad. Sci. USA 116, 24574–24582 (2019).
Yesselman, J. D. et al. Computational design of three-dimensional RNA structure and function. Nat. Nanotechnol. 14, 866–873 (2019).
Watkins, A. M., Rangan, R. & Das, R. FARFAR2: improved de novo Rosetta prediction of complex global RNA folds. Structure 28, 963–976.e6 (2020).
Townshend, R. J. L. et al. Geometric deep learning of RNA structure. Science 373, 1047–1051 (2021).
Rosa, S. S., Prazeres, D. M. F., Azevedo, A. M. & Marques, M. P. C. mRNA vaccines manufacturing: challenges and bottlenecks. Vaccine 39, 2190–2200 (2021).
Shi, H. et al. Bias in RNA-seq library preparation: current challenges and solutions. Biomed. Res. Int. 2021, 6647597 (2021).
Yu, A.-M., Choi, Y. H. & Tu, M.-J. RNA drugs and RNA targets for small molecules: principles, progress, and challenges. Pharmacol. Rev. 72, 862–898 (2020).
Schlick, T. & Pyle, A. M. Opportunities and challenges in RNA structural modeling and design. Biophysical J. 113, 225–234 (2017).
Potter, K., Cremona, N. & Wise, J. A. in Encyclopedia of Biological Chemistry 2nd edn (eds Lennarz, W. J. & Lane, M. D.) 59–64 (Academic, 2013).
Wilkinson, M. E., Charenton, C. & Nagai, K. RNA splicing by the spliceosome. Annu. Rev. Biochem. 89, 359–388 (2020).
Chen, L.-L. The biogenesis and emerging roles of circular RNAs. Nat. Rev. Mol. Cell Biol. 17, 205–211 (2016).
Zhang, X. et al. Circular RNA circNRIP1 acts as a microRNA-149-5p sponge to promote gastric cancer progression via the AKT1/mTOR pathway. Mol. Cancer 18, 20 (2019).
Cesana, M. et al. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell 147, 358–369 (2011).
Merkle, T. et al. Precise RNA editing by recruiting endogenous ADARs with antisense oligonucleotides. Nat. Biotechnol. 37, 133–138 (2019).
Acknowledgements
The authors thank J. Payne, P. Srinivasan and B. Townshend for valuable feedback in the preparation of this Review. This work was supported by the National Institutes of Health (NIH) (grant to C.D.S.), National Science Foundation (NSF) (graduate fellowships to P.B.D. and M.K.) and Howard Hughes Medical Institute (HHMI) (Gilliam graduate fellowship to M.K.). C.D.S. is a Chan Zuckerberg Biohub investigator.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Genetics thanks James M. Carothers, James J. Collins and Hirohide Saito for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Sensor
-
An element that can detect signals, including nucleic acid sequences, proteins, small molecules or non-biological stimuli such as temperature and light.
- Actuator
-
An element that can control a process or event.
- Gene-regulatory RNA elements
-
RNA elements that control expression of a gene.
- RNA devices
-
Engineered genetically encoded RNA elements that combine sensing and actuation activities.
- Riboswitches
-
Natural RNA elements that conditionally regulate gene expression in response to binding of a small molecule.
- Aptamers
-
Nucleic acid sequences that can bind a particular ligand, such as a small molecule or protein.
- Ribozyme switch
-
A type of riboswitch that uses a ribozyme, an RNA element that acts through cleaving RNA, to encode the actuation component.
Rights and permissions
About this article
Cite this article
Dykstra, P.B., Kaplan, M. & Smolke, C.D. Engineering synthetic RNA devices for cell control. Nat Rev Genet 23, 215–228 (2022). https://doi.org/10.1038/s41576-021-00436-7
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41576-021-00436-7
This article is cited by
-
Protein-inducible ribosomal frameshifting enables programmable translational control for genetic circuit design in Escherichia coli
Journal of Biological Engineering (2026)
-
Transcriptome-wide identification of glycoRNAs by Clier-seq pipeline
Science China Life Sciences (2026)
-
Determining structures of RNA conformers using AFM and deep neural networks
Nature (2025)
-
Programmable solid-state condensates for spatiotemporal control of mammalian gene expression
Nature Chemical Biology (2025)
-
Mammalian synthetic gene circuits for biopharmaceutical development & manufacture
npj Systems Biology and Applications (2025)