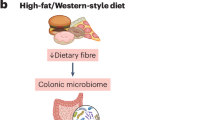
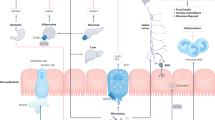
Abstract
Dietary fibres constitute a heterogeneous class of nutrients that are key in the prevention of various chronic diseases. Most dietary fibres are fermented by the gut microbiome and may, thereby, modulate the gut microbial ecology and metabolism, impacting human health. Dietary fibres may influence the occurrence of specific bacterial taxa, with this effect varying between individuals. The effect of dietary fibres on microbial diversity is a matter of debate. Most intervention studies with dietary fibres in the context of obesity and related metabolic disorders reveal the need for an accurate assessment of the microbiome to better understand the variable response to dietary fibres. Epidemiological studies confirm that a high dietary fibre intake is strongly associated with a reduced occurrence of many types of cancer. However, there is a need to determine the impact of intervention with specific dietary fibres on cancer risk, therapy efficacy and toxicity, as well as in cancer cachexia. In this Review, we summarize the mechanisms by which the gut microbiome can mediate the physiological benefits of dietary fibres in the contexts of obesity, cardiometabolic diseases and cancer, their incidence being clearly linked to low dietary fibre intake.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mathers, J. C. Dietary fibre and health: the story so far. Proc. Nutr. Soc. 82, 120–129 (2023).
Stephen, A. M. et al. Dietary fibre in Europe: current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 30, 149–190 (2017). This review article defines dietary fibre and describes the characteristics of all known dietary fibres.
Puhlmann, M. L. & de Vos, W. M. Intrinsic dietary fibers and the gut microbiome: rediscovering the benefits of the plant cell matrix for human health. Front. Immunol. 13, 954845 (2022).
Müller, M., Canfora, E. E. & Blaak, E. E. Gastrointestinal transit time, glucose homeostasis and metabolic health: modulation by dietary fibers. Nutrients 10, 275 (2018).
Carmody, R. N. et al. Cooking shapes the structure and function of the gut microbiome. Nat. Microbiol. 4, 2052–2063 (2019).
Cao, S. & Li, C. Influence of resistant starch in whole rice on human gut microbiota — from correlation implications to possible causal mechanisms. J. Agric. Food Chem. 70, 12760–12771 (2022).
Delzenne, N. M. et al. Nutritional interest of dietary fiber and prebiotics in obesity: lessons from the MyNewGut consortium. Clin. Nutr. 39, 414–424 (2020).
McKeown, N. M., Fahey, G. C. Jr., Slavin, J. & van der Kamp, J. W. Fibre intake for optimal health: how can healthcare professionals support people to reach dietary recommendations? BMJ 378, e054370 (2022).
Armet, A. M. et al. Rethinking healthy eating in light of the gut microbiome. Cell Host Microbe 30, 764–785 (2022).
Deehan, E. C. et al. Modulation of the gastrointestinal microbiome with nondigestible fermentable carbohydrates to improve human health. Microbiol. Spectr. https://doi.org/10.1128/microbiolspec.BAD-0019-2017 (2017).
GBD2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1736–1788 (2018).
Kok, C. R., Rose, D. & Hutkins, R. Predicting personalized responses to dietary fiber interventions: opportunities for modulation of the gut microbiome to improve health. Annu. Rev. Food Sci. Technol. 14, 157–182 (2023).
Wardman, J. F., Bains, R. K., Rahfeld, P. & Withers, S. G. Carbohydrate-active enzymes (CAZymes) in the gut microbiome. Nat. Rev. Microbiol. 20, 542–556 (2022).
Krautkramer, K. A., Fan, J. & Bäckhed, F. Gut microbial metabolites as multi-kingdom intermediates. Nat. Rev. Microbiol. 19, 77–94 (2021). This article provides an overview of the metabolic pathways involved in gut microbial metabolite production and their implication for host health.
Canfora, E. E., Meex, R. C. R., Venema, K. & Blaak, E. E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 15, 261–273 (2019).
So, D. et al. Dietary fiber intervention on gut microbiota composition in healthy adults: a systematic review and meta-analysis. Am. J. Clin. Nutr. 107, 965–983 (2018).
Tap, J. et al. Gut microbiota richness promotes its stability upon increased dietary fibre intake in healthy adults. Environ. Microbiol. 17, 4954–4964 (2015).
Nicolucci, A. C. et al. Prebiotics reduce body fat and alter intestinal microbiota in children who are overweight or with obesity. Gastroenterology 153, 711–722 (2017).
Reimer, R. A. et al. Effect of chicory inulin-type fructan-containing snack bars on the human gut microbiota in low dietary fiber consumers in a randomized crossover trial. Am. J. Clin. Nutr. 111, 1286–1296 (2020).
Hiel, S. et al. Effects of a diet based on inulin-rich vegetables on gut health and nutritional behavior in healthy humans. Am. J. Clin. Nutr. 109, 1683–1695 (2019).
Hiel, S. et al. Link between gut microbiota and health outcomes in inulin-treated obese patients: lessons from the Food4Gut multicenter randomized placebo-controlled trial. Clin. Nutr. 39, 3618–3628 (2020).
Scorletti, E. et al. Synbiotics alter fecal microbiomes, but not liver fat or fibrosis, in a randomized trial of patients with nonalcoholic fatty liver disease. Gastroenterology 158, 1597–1610.e7 (2020).
Deehan, E. C. et al. Elucidating the role of the gut microbiota in the physiological effects of dietary fiber. Microbiome 10, 77 (2022).
Martínez, I., Kim, J., Duffy, P. R., Schlegel, V. L. & Walter, J. Resistant starches types 2 and 4 have differential effects on the composition of the fecal microbiota in human subjects. PLoS ONE 5, e15046 (2010).
Deehan, E. C. et al. Precision microbiome modulation with discrete dietary fiber structures directs short-chain fatty acid production. Cell Host Microbe 27, 389–404.e6 (2020). This study demonstrates that small differences in dietary fibre structure distinctly affect the gut microbiome.
Dewulf, E. et al. Insight into the prebiotic concept: lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 62, 1112–1121 (2012).
Reimer, R. A. et al. Inulin-type fructans and whey protein both modulate appetite but only fructans alter gut microbiota in adults with overweight/obesity: a randomized controlled trial. Mol. Nutr. Food Res. 61, 1700484 (2017).
Salazar, N. et al. Inulin-type fructans modulate intestinal Bifidobacterium species populations and decrease fecal short-chain fatty acids in obese women. Clin. Nutr. 34, 501–507 (2015).
Ruben, L., Julien, T. & Muriel, D. Exploring Bifidobacterium species community and functional variations with human gut microbiome structure and health beyond infancy. Microbiome Res. Rep. 2, 9 (2023).
Davis, L. M., Martínez, I., Walter, J., Goin, C. & Hutkins, R. W. Barcoded pyrosequencing reveals that consumption of galactooligosaccharides results in a highly specific bifidogenic response in humans. PLoS ONE 6, e25200 (2011).
Button, J. E. et al. Dosing a synbiotic of human milk oligosaccharides and B. infantis leads to reversible engraftment in healthy adult microbiomes without antibiotics. Cell Host Microbe 30, 712–725.e7 (2022).
Bajorek, S. et al. B. infantis EVC001 is well-tolerated and improves human milk oligosaccharide utilization in preterm infants in the neonatal intensive care unit. Front. Pediatr. 9, 795970 (2021).
Frese, S. A. et al. Persistence of supplemented Bifidobacterium longum subsp. infantis EVC001 in breastfed infants. mSphere 2, e00501-17 (2017).
Benítez-Páez, A. et al. A multi-omics approach to unraveling the microbiome-mediated effects of arabinoxylan oligosaccharides in overweight humans. mSystems 4, e00209-19 (2019).
Nguyen, N. K. et al. Gut microbiota modulation with long-chain corn bran arabinoxylan in adults with overweight and obesity is linked to an individualized temporal increase in fecal propionate. Microbiome 8, 118 (2020).
Rodriguez, J., Olivares, M. & Delzenne, N. M. Implication of the gut microbiota in metabolic inflammation associated with nutritional disorders and obesity. Mol. Nutr. Food Res. 65, e1900481 (2021).
Ostrowski, M. P. et al. Mechanistic insights into consumption of the food additive xanthan gum by the human gut microbiota. Nat. Microbiol. 7, 556–569 (2022).
Van den Abbeele, P. et al. Arabinoxylans and inulin differentially modulate the mucosal and luminal gut microbiota and mucin-degradation in humanized rats. Environ. Microbiol. 13, 2667–2680 (2011).
Chung, W. S. et al. Modulation of the human gut microbiota by dietary fibres occurs at the species level. BMC Biol. 14, 3 (2016).
Dalile, B., Van Oudenhove, L., Vervliet, B. & Verbeke, K. The role of short-chain fatty acids in microbiota-gut-brain communication. Nat. Rev. Gastroenterol. Hepatol. 16, 461–478 (2019).
Koh, A., De Vadder, F., Kovatcheva-Datchary, P. & Bäckhed, F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165, 1332–1345 (2016).
Roberfroid, M. B. Caloric value of inulin and oligofructose. J. Nutr. 129, 1436s–1437s (1999).
Canfora, E. E., Jocken, J. W. & Blaak, E. E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 11, 577–591 (2015).
Bindels, L. B. et al. Gut microbiota-derived propionate reduces cancer cell proliferation in the liver. Br. J. Cancer 107, 1337–1344 (2012).
Korpela, K. Diet, microbiota, and metabolic health: trade-off between saccharolytic and proteolytic fermentation. Annu. Rev. Food Sci. Technol. 9, 65–84 (2018).
Sampson, T. R. et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell 167, 1469–1480.e12 (2016).
Brinkworth, G. D., Noakes, M., Clifton, P. M. & Bird, A. R. Comparative effects of very low-carbohydrate, high-fat and high-carbohydrate, low-fat weight-loss diets on bowel habit and faecal short-chain fatty acids and bacterial populations. Br. J. Nutr. 101, 1493–1502 (2009).
Hoyles, L. et al. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat. Med. 24, 1070–1080 (2018).
Delzenne, N. M., Cani, P. D., Everard, A., Neyrinck, A. M. & Bindels, L. B. Gut microorganisms as promising targets for the management of type 2 diabetes. Diabetologia 58, 2206–2217 (2015).
Bansal, T., Alaniz, R. C., Wood, T. K. & Jayaraman, A. The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation. Proc. Natl Acad. Sci. USA 107, 228–233 (2010).
Beaumont, M. et al. The gut microbiota metabolite indole alleviates liver inflammation in mice. FASEB J. 32, fj201800544 (2018).
Diether, N. E. & Willing, B. P. Microbial fermentation of dietary protein: an important factor in diet-microbe-host interaction. Microorganisms 7, 19 (2019).
Nishimoto, Y. et al. Resistant maltodextrin intake reduces virulent metabolites in the gut environment: a randomized control study in a Japanese cohort. Front. Microbiol. 13, 644146 (2022).
Gutiérrez-Díaz, I. et al. Adherence to a Mediterranean diet influences the fecal metabolic profile of microbial-derived phenolics in a Spanish cohort of middle-age and older people. J. Agric. Food Chem. 65, 586–595 (2017).
Verbeke, K. A. et al. Towards microbial fermentation metabolites as markers for health benefits of prebiotics. Nutr. Res. Rev. 28, 42–66 (2015).
Oliphant, K. & Allen-Vercoe, E. Macronutrient metabolism by the human gut microbiome: major fermentation by-products and their impact on host health. Microbiome 7, 91 (2019).
Yehia, N. A. et al. Association of branched chain fatty acids with cardiometabolic disorders in humans: a systematic review. Nutr. Rev. 81, 180–190 (2023).
Aslamy, A. et al. Increased plasma branched short-chain fatty acids and improved glucose homeostasis: the Microbiome and Insulin Longitudinal Evaluation Study (MILES). Diabetes 73, 385–390 (2024).
Rios-Covian, D. et al. An overview on fecal branched short-chain fatty acids along human life and as related with body mass index: associated dietary and anthropometric factors. Front. Microbiol. 11, 973 (2020).
Wang, X., Gibson, G. R., Sailer, M., Theis, S. & Rastall, R. A. Prebiotics inhibit proteolysis by gut bacteria in a host diet-dependent manner: a three-stage continuous in vitro gut model experiment. Appl. Environ. Microbiol. 86, e02730-19 (2020).
Clarke, S. T. et al. β2-1 Fructan supplementation alters host immune responses in a manner consistent with increased exposure to microbial components: results from a double-blinded, randomised, cross-over study in healthy adults. Br. J. Nutr. 115, 1748–1759 (2016).
Perino, A., Demagny, H., Velazquez-Villegas, L. & Schoonjans, K. Molecular physiology of bile acid signaling in health, disease, and aging. Physiol. Rev. 101, 683–731 (2021).
Pushpass, R. G., Alzoufairi, S., Jackson, K. G. & Lovegrove, J. A. Circulating bile acids as a link between the gut microbiota and cardiovascular health: impact of prebiotics, probiotics and polyphenol-rich foods. Nutr. Res. Rev. 35, 161–180 (2022).
Olivares, M. et al. The Janus face of cereals: wheat-derived prebiotics counteract the detrimental effect of gluten on metabolic homeostasis in mice fed a high-fat/high-sucrose diet. Mol. Nutr. Food Res. 63, e1900632 (2019).
Hiel, S. et al. Inulin improves postprandial hypertriglyceridemia by modulating gene expression in the small intestine. Nutrients 10, 532 (2018).
Neyrinck, A. M. et al. Intestinal sucrase as a novel target contributing to the regulation of glycemia by prebiotics. PLoS ONE 11, e0160488 (2016).
Pinart, M. et al. Gut microbiome composition in obese and non-obese persons: a systematic review and meta-analysis. Nutrients 14, 12 (2021).
Thomas, A. M. et al. Gut OncoMicrobiome Signatures (GOMS) as next-generation biomarkers for cancer immunotherapy. Nat. Rev. Clin. Oncol. 20, 583–603 (2023).
Sze, M. A. & Schloss, P. D. Looking for a signal in the noise: revisiting obesity and the microbiome. mBio 7, e01018-16 (2016).
Lin, Y. et al. Combing fecal microbial community data to identify consistent obesity-specific microbial signatures and shared metabolic pathways. iScience 26, 106476 (2023).
Chanda, D. & De, D. Meta-analysis reveals obesity associated gut microbial alteration patterns and reproducible contributors of functional shift. Gut Microbes 16, 2304900 (2024).
Thomas, A. M. et al. Author correction: Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat. Med. 25, 1948 (2019).
Yonekura, S. et al. Cancer induces a stress ileopathy depending on β-adrenergic receptors and promoting dysbiosis that contributes to carcinogenesis. Cancer Discov. 12, 1128–1151 (2022).
Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 (2018). This study shows that host genetics cannot explain all microbiome characteristics, which are influenced by diet as a key environmental factor.
Zhang, C. et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J. 4, 232–241 (2010).
Klingbeil, E. & de La Serre, C. B. Microbiota modulation by eating patterns and diet composition: impact on food intake. Am. J. Physiol. Regul. Integr. Comp. Physiol. 315, R1254–R1260 (2018).
Park, E. M. et al. Targeting the gut and tumor microbiota in cancer. Nat. Med. 28, 690–703 (2022).
Reynolds, A. et al. Carbohydrate quality and human health: a series of systematic reviews and meta-analyses. Lancet 393, 434–445 (2019).
Neuhouser, M. L. The importance of healthy dietary patterns in chronic disease prevention. Nutr. Res. 70, 3–6 (2019).
Lotfi, K., Saneei, P., Hajhashemy, Z. & Esmaillzadeh, A. Adherence to the Mediterranean diet, five-year weight change, and risk of overweight and obesity: a systematic review and dose-response meta-analysis of prospective cohort studies. Adv. Nutr. 13, 152–166 (2022).
Huwiler, V. V. et al. Prolonged isolated soluble dietary fibre supplementation in overweight and obese patients: a systematic review with meta-analysis of randomised controlled trials. Nutrients 14, 2627 (2022).
Cerdó, T., García-Santos, J. A., Bermúdez, M, G. & Campoy, C. The role of probiotics and prebiotics in the prevention and treatment of obesity. Nutrients 11, 635 (2019).
Suhr, J., Vuholm, S., Iversen, K. N., Landberg, R. & Kristensen, M. Wholegrain rye, but not wholegrain wheat, lowers body weight and fat mass compared with refined wheat: a 6-week randomized study. Eur. J. Clin. Nutr. 71, 959–967 (2017).
Wanders, A. J. et al. The effects of bulking, viscous and gel-forming dietary fibres on satiation. Br. J. Nutr. 109, 1330–1337 (2013).
Burley, V. J., Paul, A. W. & Blundell, J. E. Influence of a high-fibre food (myco-protein) on appetite: effects on satiation (within meals) and satiety (following meals). Eur. J. Clin. Nutr. 47, 409–418 (1993).
Odunsi, S. T. et al. Effect of alginate on satiation, appetite, gastric function, and selected gut satiety hormones in overweight and obesity. Obesity 18, 1579–1584 (2010).
Rodriguez, J. et al. Discovery of the gut microbial signature driving the efficacy of prebiotic intervention in obese patients. Gut 69, 1975–1987 (2020). This study reports the microbial features that drive the metabolic response to inulin in obesity.
Rodriguez, J. et al. Physical activity enhances the improvement of body mass index and metabolism by inulin: a multicenter randomized placebo-controlled trial performed in obese individuals. BMC Med. 20, 110 (2022).
Morel, F. B. et al. α-Galacto-oligosaccharides dose-dependently reduce appetite and decrease inflammation in overweight adults. J. Nutr. 145, 2052–2059 (2015).
EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on the substantiation of health claims related to arabinoxylan produced from wheat endosperm and reduction of post-prandial glycaemic responses (ID 830) pursuant to Article 13(1) of Regulation (EC) no 1924/2006. EFSA J. https://doi.org/10.2903/j.efsa.2011.2205 (2011).
Müller, M. et al. Circulating but not faecal short-chain fatty acids are related to insulin sensitivity, lipolysis and GLP-1 concentrations in humans. Sci. Rep. 9, 12515 (2019).
van Deuren, T., Blaak, E. E. & Canfora, E. E. Butyrate to combat obesity and obesity-associated metabolic disorders: current status and future implications for therapeutic use. Obes. Rev. 23, e13498 (2022).
van Deuren, T. et al. Butyrate and hexanoate-enriched triglycerides increase postprandrial systemic butyrate and hexanoate in men with overweight/obesity: a double-blind placebo-controlled randomized crossover trial. Front. Nutr. 9, 1066950 (2022).
Canfora, E. E. et al. Fiber mixture-specific effect on distal colonic fermentation and metabolic health in lean but not in prediabetic men. Gut Microbes 14, 2009297 (2022).
O’Keefe, S. J. The association between dietary fibre deficiency and high-income lifestyle-associated diseases: Burkitt’s hypothesis revisited. Lancet Gastroenterol. Hepatol. 4, 984–996 (2019). This viewpoint article discusses the evidence that fibre deficiency increases the risk of colon, liver and breast cancer and the underlying mechanisms.
Papadimitriou, N. et al. A prospective diet-wide association study for risk of colorectal cancer in EPIC. Clin. Gastroenterol. Hepatol. 20, 864–873.e13 (2022).
Li, N. et al. Dietary carbohydrate intake is associated with a lower risk of breast cancer: a meta-analysis of cohort studies. Nutr. Res. 100, 70–92 (2022).
Nucci, D. et al. Dietary fiber intake and risk of pancreatic cancer: systematic review and meta-analysis of observational studies. Int. J. Environ. Res. Public Health 18, 11556 (2021).
Yu, E. Y. W. et al. Grain and dietary fiber intake and bladder cancer risk: a pooled analysis of prospective cohort studies. Am. J. Clin. Nutr. 112, 1252–1266 (2020).
Khodavandi, A., Alizadeh, F. & Razis, A. F. A. Association between dietary intake and risk of ovarian cancer: a systematic review and meta-analysis. Eur. J. Nutr. 60, 1707–1736 (2021).
Li, H., Mao, H., Yu, Y. & Nan, Y. Association between dietary fiber and endometrial cancer: a meta-analysis. Nutr. Cancer 72, 959–967 (2020).
Kuan, A. S. et al. Diet and risk of glioma: combined analysis of 3 large prospective studies in the UK and USA. Neuro Oncol. 21, 944–952 (2019).
US Food & Drug Administration. 21 CFR 101.76 Health claims: fiber-containing grain products, fruits, and vegetables and cancer, 58 FR 2578 (FDA, 1993).
Taper, H. S. & Roberfroid, M. B. Inulin/oligofructose and anticancer therapy. Br. J. Nutr. 87, S283–S286 (2002).
Han, K. et al. Generation of systemic antitumour immunity via the in situ modulation of the gut microbiome by an orally administered inulin gel. Nat. Biomed. Eng. 5, 1377–1388 (2021).
Li, Y. et al. Prebiotic-induced anti-tumor immunity attenuates tumor growth. Cell Rep. 30, 1753–1766.e6 (2020).
Singh, V. et al. Dysregulated microbial fermentation of soluble fiber induces cholestatic liver cancer. Cell 175, 679–694.e22 (2018).
Oliero, M. et al. Inulin impacts tumorigenesis promotion by colibactin-producing Escherichia coli in Apc(Min/+) mice. Front. Microbiol. 14, 1067505 (2023).
Debras, C. et al. Fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) and cancer risk in the prospective NutriNet-Santé cohort. J. Nutr. 152, 1059–1069 (2022).
Schatzkin, A. et al. Lack of effect of a low-fat, high-fiber diet on the recurrence of colorectal adenomas. Polyp Prevention Trial Study Group. N. Engl. J. Med. 342, 1149–1155 (2000).
Sansbury, L. B. et al. The effect of strict adherence to a high-fiber, high-fruit and -vegetable, and low-fat eating pattern on adenoma recurrence. Am. J. Epidemiol. 170, 576–584 (2009). This re-analysis of an interventional trial highlights that super compliers to a high-fibre, high-fruit and high-vegetable, and low-fat eating pattern displayed reduced odds of adenoma recurrence.
Le Leu, R. K. et al. Butyrylated starch intake can prevent red meat-induced O6-methyl-2-deoxyguanosine adducts in human rectal tissue: a randomised clinical trial. Br. J. Nutr. 114, 220–230 (2015).
Wu, W. T., Cheng, H. C. & Chen, H. L. Ameliorative effects of konjac glucomannan on human faecal β-glucuronidase activity, secondary bile acid levels and faecal water toxicity towards Caco-2 cells. Br. J. Nutr. 105, 593–600 (2011).
Windey, K. et al. Wheat bran extract alters colonic fermentation and microbial composition, but does not affect faecal water toxicity: a randomised controlled trial in healthy subjects. Br. J. Nutr. 113, 225–238 (2015).
Worthley, D. L. et al. A human, double-blind, placebo-controlled, crossover trial of prebiotic, probiotic, and synbiotic supplementation: effects on luminal, inflammatory, epigenetic, and epithelial biomarkers of colorectal cancer. Am. J. Clin. Nutr. 90, 578–586 (2009).
Donohoe, D. R. et al. A gnotobiotic mouse model demonstrates that dietary fiber protects against colorectal tumorigenesis in a microbiota- and butyrate-dependent manner. Cancer Discov. 4, 1387–1397 (2014).
Wortmann, E. et al. Secondary bile acid production by gut bacteria promotes Western diet-associated colorectal cancer. Preprint at bioRxiv https://doi.org/10.1101/2023.03.17.533140 (2023).
Thibaut, M. M. & Bindels, L. B. Crosstalk between bile acid-activated receptors and microbiome in entero-hepatic inflammation. Trends Mol. Med. 28, 223–236 (2022).
Bindels, L. B. et al. Resistant starch can improve insulin sensitivity independently of the gut microbiota. Microbiome 5, 12 (2017).
Jenkins, A. L., Vuksan, V. & Jenkins, D. A. in CRC Handbook of Dietary Fiber in Human Nutrition (ed. Spiller, G. A.) 423–430 (CRC Press, 2001).
Byrd, D. A. et al. Circulating bile acids and adenoma recurrence in the context of adherence to a high-fiber, high-fruit and vegetable, and low-fat dietary intervention. Clin. Transl. Gastroenterol. 13, e00533 (2022).
Lampe, J. W. et al. Colonic mucosal and exfoliome transcriptomic profiling and fecal microbiome response to a flaxseed lignan extract intervention in humans. Am. J. Clin. Nutr. 110, 377–390 (2019).
Wallace, B. D. et al. Alleviating cancer drug toxicity by inhibiting a bacterial enzyme. Science 330, 831–835 (2010).
Wiseman, M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Proc. Nutr. Soc. 67, 253–256 (2008).
Chrysostomou, D., Roberts, L. A., Marchesi, J. R. & Kinross, J. M. Gut microbiota modulation of efficacy and toxicity of cancer chemotherapy and immunotherapy. Gastroenterology 164, 198–213 (2023).
Kang, X., Lau, H. C. & Yu, J. Modulating gut microbiome in cancer immunotherapy: harnessing microbes to enhance treatment efficacy. Cell Rep. Med. 5, 101478 (2024).
Taper, H. S. & Roberfroid, M. B. Nontoxic potentiation of cancer chemotherapy by dietary oligofructose or inulin. Nutr. Cancer 38, 1–5 (2000).
Encarnação, J. C. et al. Butyrate, a dietary fiber derivative that improves irinotecan effect in colon cancer cells. J. Nutr. Biochem. 56, 183–192 (2018).
Then, C. K., Paillas, S., Wang, X., Hampson, A. & Kiltie, A. E. Association of Bacteroides acidifaciens relative abundance with high-fibre diet-associated radiosensitisation. BMC Biol. 18, 102 (2020).
Suran, M. Melanoma immunotherapy responses to dietary fiber and probiotics. JAMA 327, 1117 (2022).
Spencer, C. N. et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374, 1632–1640 (2021).
Nomura, M. et al. Association of short-chain fatty acids in the gut microbiome with clinical response to treatment with nivolumab or pembrolizumab in patients with solid cancer tumors. JAMA Netw. Open 3, e202895 (2020).
Zhang, S. L. et al. Pectin supplement significantly enhanced the anti-PD-1 efficacy in tumor-bearing mice humanized with gut microbiota from patients with colorectal cancer. Theranostics 11, 4155–4170 (2021).
Coutzac, C. et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat. Commun. 11, 2168 (2020).
Smith, C. L. et al. Lactobacillus fermentum BR11 and fructo-oligosaccharide partially reduce jejunal inflammation in a model of intestinal mucositis in rats. Nutr. Cancer 60, 757–767 (2008).
Galdino, F. M. P. et al. Pretreatment and treatment with fructo-oligosaccharides attenuate intestinal mucositis induced by 5-FU in mice. J. Funct. Foods 49, 485–492 (2018).
Gallotti, B. et al. Effects of dietary fibre intake in chemotherapy-induced mucositis in murine model. Br. J. Nutr. 126, 853–864 (2021).
Sahasrabudhe, N. M. et al. Dietary fiber pectin directly blocks Toll-like receptor 2-1 and prevents doxorubicin-induced ileitis. Front. Immunol. 9, 383 (2018).
Argilés, J. M., López-Soriano, F. J., Stemmler, B. & Busquets, S. Cancer-associated cachexia — understanding the tumour macroenvironment and microenvironment to improve management. Nat. Rev. Clin. Oncol. 20, 250–264 (2023).
Fearon, K. et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 12, 489–495 (2011).
Pryce, B. R., Wang, D. J., Zimmers, T. A., Ostrowski, M. C. & Guttridge, D. C. Cancer cachexia: involvement of an expanding macroenvironment. Cancer Cell 41, 581–584 (2023).
Ubachs, J. et al. Gut microbiota and short-chain fatty acid alterations in cachectic cancer patients. J. Cachexia Sarcopenia Muscle 12, 2007–2021 (2021).
Pötgens, S. A. et al. Multi-compartment metabolomics and metagenomics reveal major hepatic and intestinal disturbances in cancer cachectic mice. J. Cachexia Sarcopenia Muscle 12, 456–475 (2021).
Bindels, L. B. et al. Synbiotic approach restores intestinal homeostasis and prolongs survival in leukaemic mice with cachexia. ISME J. 10, 1456–1470 (2016).
Lefevre, C. & Bindels, L. B. Role of the gut microbiome in skeletal muscle physiology and pathophysiology. Curr. Osteoporos. Rep. 20, 422–432 (2022).
Jiang, Y. et al. The altered tight junctions: an important gateway of bacterial translocation in cachexia patients with advanced gastric cancer. J. Interferon Cytokine Res. 34, 518–525 (2014).
Bindels, L. B. et al. Increased gut permeability in cancer cachexia: mechanisms and clinical relevance. Oncotarget 9, 18224–18238 (2018).
Pötgens, S. A. et al. Gut microbiome alterations at acute myeloid leukemia diagnosis are associated with muscle weakness and anorexia. Haematologica 109, 3194–3208 (2024).
Bindels, L. B. et al. Non digestible oligosaccharides modulate the gut microbiota to control the development of leukemia and associated cachexia in mice. PLoS ONE 10, e0131009 (2015). Together with Pötgens et al. (2021), this study highlights that cancerous mice with cachexia exhibit alterations in gut microbiota composition and functions associated with intestinal disturbance.
Kenkhuis, M. F. et al. Longitudinal associations of fiber, vegetable, and fruit intake with quality of life and fatigue in colorectal cancer survivors up to 24 months posttreatment. Am. J. Clin. Nutr. 115, 822–832 (2022).
Tollosa, D. N., Tavener, M., Hure, A. & James, E. L. Adherence to multiple health behaviours in cancer survivors: a systematic review and meta-analysis. J. Cancer Surviv. 13, 327–343 (2019).
Walter, J., Armet, A. M., Finlay, B. B. & Shanahan, F. Establishing or exaggerating causality for the gut microbiome: lessons from human microbiota-associated rodents. Cell 180, 221–232 (2020).
Babbar, N., Dejonghe, W., Gatti, M., Sforza, S. & Elst, K. Pectic oligosaccharides from agricultural by-products: production, characterization and health benefits. Crit. Rev. Biotechnol. 36, 594–606 (2016).
Arzami, A. N., Ho, T. M. & Mikkonen, K. S. Valorization of cereal by-product hemicelluloses: fractionation and purity considerations. Food Res. Int. 151, 110818 (2022).
Turck, D. et al. Scientific advice related to nutrient profiling for the development of harmonised mandatory front-of-pack nutrition labelling and the setting of nutrient profiles for restricting nutrition and health claims on foods. EFSA J. 20, e07259 (2022).
Pascale, N., Gu, F., Larsen, N., Jespersen, L. & Respondek, F. The potential of pectins to modulate the human gut microbiota evaluated by in vitro fermentation: a systematic review. Nutrients 14, 3629 (2022).
Hipsley, E. H. Dietary “fibre” and pregnancy toxaemia. Br. Med. J. 2, 420–422 (1953).
Burkitt, D. P., Walker, A. R. & Painter, N. S. Dietary fiber and disease. JAMA 229, 1068–1074 (1974).
Trowell, H. Ischemic heart disease and dietary fiber. Am. J. Clin. Nutr. 25, 926–932 (1972).
Asp, N. G., Johansson, C. G., Hallmer, H. & Siljeström, M. Rapid enzymatic assay of insoluble and soluble dietary fiber. J. Agric. Food Chem. 31, 476–482 (1983).
Prosky, L. et al. Determination of total dietary fiber in foods and food products: collaborative study. J. Assoc. Off. Anal. Chem. 68, 677–679 (1985).
Gibson, G. R. & Roberfroid, M. B. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J. Nutr. 125, 1401–1412 (1995).
Gibson, G. R. et al. Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 14, 491–502 (2017).
Bindels, L. B., Delzenne, N. M., Cani, P. D. & Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 12, 303–310 (2015).
Acknowledgements
L.B.B. is a Collen-Francqui Research Professor and is grateful for the support of the Francqui Fondation. N.M.D. is a recipient of grants from the Fonds de la Recherche Scientifique (FRS)‐FNRS (grant numbers: PINT‐MULTI R.8013.19 (NEURON, call 2019), PDR T.0068.19 and PDR T008524).
Author information
Authors and Affiliations
Contributions
N.M.D. led the work (content, writing and discussion). All authors researched data for the article, contributed to the discussion of the content, and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Microbiology thanks Rachel Carmody, Yolanda Sanz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
FDA: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-101#101.76
Institute for Health Metrics and Evaluation: https://www.healthdata.org/
WHO fact sheet for cancer: https://www.who.int/news-room/fact-sheets/detail/cancer
WHO fact sheet for obesity and overweight: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Delzenne, N.M., Bindels, L.B., Neyrinck, A.M. et al. The gut microbiome and dietary fibres: implications in obesity, cardiometabolic diseases and cancer. Nat Rev Microbiol 23, 225–238 (2025). https://doi.org/10.1038/s41579-024-01108-z
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41579-024-01108-z
This article is cited by
-
Association of a newly proposed dietary index for gut microbiota with phenotypic age acceleration: a cross-sectional study of NHANES 1999–2018
Journal of Health, Population and Nutrition (2025)
-
Early-life and concurrent predictors of the healthy adolescent microbiome in a cohort study
Genome Medicine (2025)
-
Gut microbiota L-ornithine promotes resistance to obesity through metabolites mediated immunosuppressive macrophages
Cellular and Molecular Life Sciences (2025)
-
Interest of inulin in obesity: comparison of the prebiotic effect of edible-food sources versus purified inulin from chicory root
European Journal of Nutrition (2025)