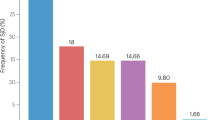
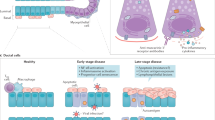
Abstract
Sjögren syndrome or Sjögren disease is a chronic form of autoimmune epithelitis characterized by lymphocytic infiltration of the exocrine glands, particularly the salivary and lacrimal glands, leading to progressive glandular dysfunction and subsequent xerostomia and xerophthalmia. Other common manifestations include pain and fatigue, various systemic manifestations and non-Hodgkin’s lymphoma. Sjögren syndrome is therefore a complex and disabling disease associated with a reduced quality of life and with considerable long-term damage. Most of the available treatments are merely symptomatic with limited efficacy in both preventing glandular damage and suppressing systemic disease activity. In the past 10 years, great progress has been made in understanding the pathophysiology of Sjögren syndrome, opening new avenues towards a more targeted and individualized therapeutic approach to the disease. Indeed, several randomized controlled trials have just been completed or are poised to commence evaluating the effectiveness of novel drugs targeting both innate and adaptive immune pathways, including pro-inflammatory cytokines, the type I interferon system, B cell activation, B cell and T cell co-stimulation pathway, and ectopic germinal centre formation. Novel clinical trials are also ongoing exploring various targeted approaches (that is, IgG recycling inhibition, nuclease therapy and CAR-T cell therapy) for Sjögren syndrome.
Key points
-
New insights into Sjögren syndrome pathophysiology are opening new avenues in the treatment of Sjögren syndrome, with novel molecules in the pipeline.
-
Different therapeutic strategies are under investigation that interfere with activation of interferon pathways including agents that degrade circulating RNA, agents that interfere with Toll-like receptor activation and inhibitors of downstream signalling.
-
Novel strategies that target B cell hyperactivity have shown promising results, including anti-BAFF antibodies, agents that target B cell receptor signalling and antibody-depleting drugs.
-
Targeting B cell and/or T cell co-stimulation pathways, including targeting the CD40–CD40 ligand axis and the ICOS–ICOS ligand axis, have also shown promise.
-
A miscellanea of additional novel agents, including drugs that target T cells and pro-inflammatory cytokines, are in the pipeline and could offer novel therapeutic possibilities in the near future.
-
Novel insights into clinical, molecular and histopathological stratification, based on multiomics techniques, emerging imaging biomarkers and new composite outcome measures, are paving new avenues for personalized medicine in Sjögren syndrome.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mariette, X. & Criswell, L. A. Primary Sjögren’s Syndrome. N. Engl. J. Med. 379, 97 (2018).
Brito-Zerón, P. et al. Sjögren syndrome. Nat. Rev. Dis. Prim. 2, 16047 (2016).
Segal, B. M., Pogatchnik, B., Henn, L., Rudser, K. & Sivils, K. M. Pain severity and neuropathic pain symptoms in primary Sjögren’s syndrome: a comparison study of seropositive and seronegative Sjögren’s syndrome patients. Arthritis Care Res. 65, 1291–1298 (2013).
Mæland, E., Miyamoto, S. T., Hammenfors, D., Valim, V. & Jonsson, M. V. Understanding fatigue in Sjögren’s syndrome: outcome measures, biomarkers and possible interventions. Front. Immunol. 12, 703079 (2021).
Unger, J. et al. The experiences of functioning and health of patients with primary Sjögren’s syndrome: a multicenter qualitative European study. Front. Med. 8, 770422 (2021).
Beydon, M. et al. Epidemiology of Sjogren syndrome. Nat. Rev. Rheumatol. 20, 158–169 (2024).
McCoy, S. S. et al. National Sjögren’s foundation survey: burden of oral and systemic involvement on quality of life. J. Rheumatol. 48, 1029–1036 (2021).
Tarn, J. et al. Primary Sjögren’s syndrome: longitudinal real-world, observational data on health-related quality of life. J. Intern. Med. 291, 849–855 (2022).
Baer, A. N. & Hammitt, K. M. Sjogren’s disease, not syndrome. Arthritis Rheumatol. 73, 1347–1348 (2021).
Baldini, C., Bartoloni, E. & Bombardieri, M. Highlights of the 15th International Symposium on Sjogren’s Syndrome. Clin. Exp. Rheumatol. 40, 2201–2210 (2022).
Brito-Zerón, P. et al. Efficacy and safety of topical and systemic medications: a systematic literature review informing the EULAR recommendations for the management of Sjögren’s syndrome. RMD Open. 5, e001064 (2019).
Vivino, F. B. et al. Sjogren’s syndrome: an update on disease pathogenesis, clinical manifestations and treatment. Clin. Immunol. 203, 81–121 (2019).
Fasano, S. et al. Pathogenesis of primary Sjögren’s syndrome beyond B lymphocytes. Clin. Exp. Rheumatol. 38, 315–323 (2020).
Fox, R. I., Fox, C. M., Gottenberg, J. E. & Dörner, T. Treatment of Sjögren’s syndrome: current therapy and future directions. Rheumatology 60, 2066–2074 (2021).
Retamozo, S., Sisó-Almirall, A., Flores-Chávez, A., Ramos-Casals, M. & Brito-Zerón, P. An update of targeted therapeutic options for primary Sjögren syndrome: current status and future development. Expert. Opin. Pharmacother. 22, 2359–2371 (2021).
Ridgewell, D., Thalayasingam, N. & Ng, W. F. Sjögren’s syndrome: shedding light on emerging and key drug targets. Expert. Opin. Ther. Targets 26, 869–882 (2022).
Zhan, Q. et al. Pathogenesis and treatment of Sjogren’s syndrome: review and update. Front. Immunol. 14, 1127417 (2023).
Fox, R. I., Fox, C. M. & McCoy, S. S. Emerging treatment for Sjögren’s disease: a review of recent phase II and III trials. Expert. Opin. Emerg. Drugs 28, 107–120 (2023).
Bettacchioli, E. et al. Association of combined anti-Ro52/TRIM21 and anti-Ro60/SSA antibodies with increased Sjögren disease severity through interferon pathway activation. Arthritis Rheumatol. 76, 751–762 (2023).
James, J. A. et al. Unique Sjögren’s syndrome patient subsets defined by molecular features. Rheumatology 59, 860–868 (2020).
Brito-Zerón, P., Retamozo, S. & Ramos-Casals, M. Phenotyping Sjögren’s syndrome: towards a personalised management of the disease. Clin. Exp. Rheumatol. 36, 198–209 (2018).
Tarn, J. R. et al. Symptom-based stratification of patients with primary Sjögren’s syndrome: multi-dimensional characterisation of international observational cohorts and reanalyses of randomised clinical trials. Lancet Rheumatol. 1, e85–e94 (2019).
McCoy, S. S. et al. Symptom-based cluster analysis categorizes Sjögren’s disease subtypes: an international cohort study highlighting disease severity and treatment discordance. Arthritis Rheumatol. 74, 1569–1579 (2022).
Noaiseh, G. & Baer, A. N. Toward better outcomes in Sjögren’s syndrome: the promise of a stratified medicine approach. Best. Pract. Res. Clin. Rheumatol. 34, 101475 (2020).
Devauchelle-Pensec, V. et al. French national diagnostic and care protocol for Sjogren’s disease. Rev. Med. Interne 44, 423–457 (2023).
de Wolff, L., Arends, S., van Nimwegen, J. F. & Bootsma, H. Ten years of the ESSDAI: is it fit for purpose? Clin. Exp. Rheumatol. 38, 283–290 (2020).
Seror, R. et al. Development and preliminary validation of the Sjögren’s Tool for Assessing Response (STAR): a consensual composite score for assessing treatment effect in primary Sjögren’s syndrome. Ann. Rheum. Dis. 81, 979–989 (2022).
Brito-Zerón, P. et al. Influence of geolocation and ethnicity on the phenotypic expression of primary Sjögren’s syndrome at diagnosis in 8310 patients: a cross-sectional study from the Big Data Sjögren Project Consortium. Ann. Rheum. Dis. 76, 1042–1050 (2017).
Nocturne, G. & Mariette, X. Advances in understanding the pathogenesis of primary Sjögren’s syndrome. Nat. Rev. Rheumatol. 9, 544–556 (2013).
Thorlacius, G. E., Björk, A. & Wahren-Herlenius, M. Genetics and epigenetics of primary Sjögren syndrome: implications for future therapies. Nat. Rev. Rheumatol. 19, 288–306 (2023).
Verstappen, G. M., Pringle, S., Bootsma, H. & Kroese, F. G. M. Epithelial-immune cell interplay in primary Sjögren syndrome salivary gland pathogenesis. Nat. Rev. Rheumatol. 17, 333–348 (2021).
Chatzis, L., Vlachoyiannopoulos, P. G., Tzioufas, A. G. & Goules, A. V. New frontiers in precision medicine for Sjogren’s syndrome. Expert. Rev. Clin. Immunol. 17, 127–141 (2021).
Björk, A., Mofors, J. & Wahren-Herlenius, M. Environmental factors in the pathogenesis of primary Sjögren’s syndrome. J. Intern. Med. 287, 475–492 (2020).
Lessard, C. J. et al. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjögren’s syndrome. Nat. Genet. 45, 1284–1292 (2013).
Yin, H. et al. Association of bone morphogenetic protein 6 with exocrine gland dysfunction in patients with Sjögren’s syndrome and in mice. Arthritis Rheum. 65, 3228–3238 (2013).
Nakamura, H. et al. Lysosome-associated membrane protein 3 misexpression in salivary glands induces a Sjögren’s syndrome-like phenotype in mice. Ann. Rheum. Dis. 80, 1031–1039 (2021).
Mo, Y. Q. et al. Lysosomal exocytosis of HSP70 stimulates monocytic BMP6 expression in Sjögren’s syndrome. J. Clin. Invest. 132, e152780 (2022).
Tang, Y. et al. The role of epithelial cells in the immunopathogenesis of Sjögren’s syndrome. J. Leukoc. Biol. 115, 57–67 (2023).
Yura, Y. & Hamada, M. Outline of salivary gland pathogenesis of Sjögren’s syndrome and current therapeutic approaches. Int. J. Mol. Sci. 24, 11179 (2023).
Wang, Y. et al. TLR7 signaling drives the development of Sjögren’s syndrome. Front. Immunol. 12, 676010 (2021).
Nezos, A. et al. Type I and II interferon signatures in Sjogren’s syndrome pathogenesis: contributions in distinct clinical phenotypes and Sjogren’s related lymphomagenesis. J. Autoimmun. 63, 47–58 (2015).
Hu, X., Li, J., Fu, M., Zhao, X. & Wang, W. The JAK/STAT signaling pathway: from bench to clinic. Signal. Transduct. Target. Ther. 6, 402 (2021).
Marketos, N., Cinoku, I., Rapti, A. & Mavragani, C. P. Type I interferon signature in Sjögren’s syndrome: pathophysiological and clinical implications. Clin. Exp. Rheumatol. 37, 185–191 (2019).
Del Papa, N. et al. The role of interferons in the pathogenesis of Sjögren’s syndrome and future therapeutic perspectives. Biomolecules 11, 251 (2021).
Mavragani, C. P. Mechanisms and new strategies for primary Sjögren’s syndrome. Annu. Rev. Med. 68, 331–343 (2017).
de Paiva, C. S. & Pflugfelder, S. C. Mechanisms of disease in Sjögren syndrome — new developments and directions. Int. J. Mol. Sci. 21, 650 (2020).
Sequi-Sabater, J. M. & Beretta, L. Defining the role of monocytes in Sjögren’s syndrome. Int. J. Mol. Sci. 23, 12765 (2022).
Bodewes, I. L. A., Björk, A., Versnel, M. A. & Wahren-Herlenius, M. Innate immunity and interferons in the pathogenesis of Sjögren’s syndrome. Rheumatology 60, 2561–2573 (2019).
Qi, J. et al. Interleukin-12 exacerbates Sjögren’s syndrome through induction of myeloid-derived suppressor cells. Mol. Med. Rep. 20, 1131–1138 (2019).
Roescher, N., Tak, P. P. & Illei, G. G. Cytokines in Sjögren’s syndrome: potential therapeutic targets. Ann. Rheum. Dis. 69, 945–948 (2010).
Zhou, Y. et al. Novel small molecule tyrosine kinase 2 pseudokinase ligands block cytokine-induced TYK2-mediated signaling pathways. Front. Immunol. 13, 884399 (2022).
Ullrich, K. A. et al. Immunology of IL-12: an update on functional activities and implications for disease. EXCLI J. 19, 1563–1589 (2020).
Nocturne, G. & Mariette, X. B cells in the pathogenesis of primary Sjögren syndrome. Nat. Rev. Rheumatol. 14, 133–145 (2018).
Baldini, C., Ferro, F., Elefante, E. & Bombardieri, S. Biomarkers for Sjögren’s syndrome. Biomark. Med. 12, 275–286 (2018).
Baldini C., Berardicurti O., Giacomelli R., Bombardieri M. Is minor salivary gland biopsy still mandatory in Sjogren’s syndrome? Does seronegative Sjogren’s syndrome exist? Autoimmun. Rev. 23, 103425 (2023).
Bombardieri, M. et al. One year in review 2020: pathogenesis of primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 38, 3–9 (2020).
Kroese, F. G. M., Haacke, E. A. & Bombardieri, M. The role of salivary gland histopathology in primary Sjögren’s syndrome: promises and pitfalls. Clin. Exp. Rheumatol. 36, 222–233 (2018).
Goules, A. V. & Tzioufas, A. G. Lymphomagenesis in Sjögren’s syndrome: predictive biomarkers towards precision medicine. Autoimmun. Rev. 18, 137–143 (2019).
Szodoray, P. & Jonsson, R. The BAFF/APRIL system in systemic autoimmune diseases with a special emphasis on Sjögren’s syndrome. Scand. J. Immunol. 62, 421–428 (2005).
Pontarini, E., Lucchesi, D. & Bombardieri, M. Current views on the pathogenesis of Sjögren’s syndrome. Curr. Opin. Rheumatol. 30, 215–221 (2018).
Pontarini, E. et al. Blocking T cell co-stimulation in primary Sjögren’s syndrome: rationale, clinical efficacy and modulation of peripheral and salivary gland biomarkers. Clin. Exp. Rheumatol. 38, 222–227 (2020).
Pontarini, E. et al. Unique expansion of IL-21+ Tfh and Tph cells under control of ICOS identifies Sjögren’s syndrome with ectopic germinal centres and MALT lymphoma. Ann. Rheum. Dis. 79, 1588–1599 (2020).
Raeber, M. E., Zurbuchen, Y., Impellizzieri, D. & Boyman, O. The role of cytokines in T-cell memory in health and disease. Immunol. Rev. 283, 176–193 (2018).
Rivière, E. et al. Interleukin-7/interferon axis drives T cell and salivary gland epithelial cell interactions in Sjögren’s syndrome. Arthritis Rheumatol. 73, 631–640 (2021).
Hillen, M. R. et al. High soluble IL-7 receptor expression in Sjögren’s syndrome identifies patients with increased immunopathology and dryness. Ann. Rheum. Dis. 75, 1735–1736 (2016).
Alunno, A. et al. T regulatory and T helper 17 cells in primary Sjögren’s syndrome: facts and perspectives. Mediators Inflamm. 2015, 243723 (2015).
Zhang, X. et al. CD8+ cells regulate the T helper-17 response in an experimental murine model of Sjögren syndrome. Mucosal Immunol. 7, 417–427 (2014).
Barone, F. et al. IL-22 regulates lymphoid chemokine production and assembly of tertiary lymphoid organs. Proc. Natl Acad. Sci. USA 112, 11024–11029 (2015).
Noack, M. & Miossec, P. Th17 and regulatory T cell balance in autoimmune and inflammatory diseases. Autoimmun. Rev. 13, 668–677 (2014).
Bodewes, I. L. A., Gottenberg, J. E., van Helden-Meeuwsen, C. G., Mariette, X. & Versnel, M. A. Hydroxychloroquine treatment downregulates systemic interferon activation in primary Sjögren’s syndrome in the JOQUER randomized trial. Rheumatology 59, 107–11 (2019).
Gottenberg, J. E. et al. Effects of hydroxychloroquine on symptomatic improvement in primary Sjögren syndrome: the JOQUER randomized clinical trial. JAMA 312, 249–258 (2014).
Gandolfo, S. & Ciccia, F. JAK/STAT pathway targeting in primary Sjögren syndrome. Rheumatol. Immunol. Res. 3, 95–102 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02049437 (2021).
Burge, D. J. et al. Safety, pharmacokinetics, and pharmacodynamics of RSLV-132, an RNase-Fc fusion protein in systemic lupus erythematosus: a randomized, double-blind, placebo-controlled study. Lupus 26, 825–834 (2017).
Posada, J. et al. Improvement of severe fatigue following nuclease therapy in patients with primary Sjögren’s syndrome: a randomized clinical trial. Arthritis Rheumatol. 73, 143–150 (2021).
Hamkour, S. et al. Leflunomide/hydroxychloroquine combination therapy targets type I IFN-associated proteins in patients with Sjögren’s syndrome that show potential to predict and monitor clinical response. RMD Open. 9, e002979 (2023).
van der Heijden, E. H. M. et al. Leflunomide–hydroxychloroquine combination therapy in patients with primary Sjögren’s syndrome (RepurpSS-I): a placebo-controlled, double-blinded, randomised clinical trial. Lancet Rheumatol. 2, e260–e269 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02780674 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03817424 (2020).
Bodewes, I. L. A. et al. TBK1: a key regulator and potential treatment target for interferon positive Sjögren’s syndrome, systemic lupus erythematosus and systemic sclerosis. J. Autoimmun. 91, 97–102 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05383677 (2022).
Lee, J. et al. JAK-1 inhibition suppresses interferon-induced BAFF production in human salivary gland: potential therapeutic strategy for primary Sjögren’s syndrome. Arthritis Rheumatol. 70, 2057–2066 (2018).
Price, E. et al. Safety and efficacy of filgotinib, lanraplenib and tirabrutinib in Sjogren’s syndrome: a randomized, phase 2, double-blind, placebo-controlled study. Rheumatology 61, 4797–4808 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05087589 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04496960 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04916756 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05016297 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05946941 (2024).
Kingston, P., Blauvelt, A., Strober, B. & Armstrong, A. W. Deucravacitinib: a novel TYK2 inhibitor for the treatment of moderate-to-severe psoriasis. J. Psoriasis Psoriatic Arthritis 8, 156–165 (2023).
Morand, E. et al. Deucravacitinib, a tyrosine kinase 2 inhibitor, in systemic lupus erythematosus: a phase II, randomized, double-blind, placebo-controlled trial. Arthritis Rheumatol. 75, 242–252 (2023).
Kahlenberg, J. M. et al. POS0112 deucravacitinib reduces interferons, b-cell pathways, and serological biomarkers of systemic lupus disease activity: pharmacodynamic analysis from the phase 2 paisley study. Ann. Rheum. Dis. 82, 271–271 (2023).
Grigoriadou, S. et al. B cell depletion with rituximab in the treatment of primary Sjögren’s syndrome: what have we learnt? Clin. Exp. Rheumatol. 37, 217–224 (2019).
Pijpe, J. et al. Rituximab treatment in patients with primary Sjögren’s syndrome: an open-label phase II study. Arthritis Rheum. 52, 2740–2750 (2005).
Devauchelle-Pensec, V. et al. Improvement of Sjögren’s syndrome after two infusions of rituximab (anti-CD20). Arthritis Rheum. 57, 310–317 (2007).
Dass, S. et al. Reduction of fatigue in Sjögren syndrome with rituximab: results of a randomised, double-blind, placebo-controlled pilot study. Ann. Rheum. Dis. 67, 1541–1544 (2008).
Meijer, J. M., Pijpe, J., Vissink, A., Kallenberg, C. G. & Bootsma, H. Treatment of primary Sjögren syndrome with rituximab: extended follow-up, safety and efficacy of retreatment. Ann. Rheum. Dis. 68, 284–285 (2009).
Meijer, J. M. et al. Effectiveness of rituximab treatment in primary Sjögren’s syndrome: a randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 62, 960–968 (2010).
Meiners, P. M. et al. Responsiveness of disease activity indices ESSPRI and ESSDAI in patients with primary Sjögren’s syndrome treated with rituximab. Ann. Rheum. Dis. 71, 1297–1302 (2012).
Gottenberg, J. E. et al. Efficacy of rituximab in systemic manifestations of primary Sjögren’s syndrome: results in 78 patients of the AutoImmune and Rituximab registry. Ann. Rheum. Dis. 72, 1026–1031 (2013).
St Clair, E. W. et al. Rituximab therapy for primary Sjögren’s syndrome: an open-label clinical trial and mechanistic analysis. Arthritis Rheum. 65, 1097–1106 (2013).
Carubbi, F. et al. Efficacy and safety of rituximab treatment in early primary Sjögren’s syndrome: a prospective, multi-center, follow-up study. Arthritis Res. Ther. 15, R172 (2013).
Devauchelle-Pensec, V. et al. Treatment of primary Sjögren syndrome with rituximab: a randomized trial. Ann. Intern. Med. 160, 233–242 (2014).
Bowman, S. J. et al. Randomized controlled trial of rituximab and cost-effectiveness analysis in treating fatigue and oral dryness in primary Sjögren’s syndrome. Arthritis Rheumatol. 69, 1440–1450 (2017).
Brown, S. et al. The TRACTISS protocol: a randomised double blind placebo controlled clinical trial of anti-B-cell therapy in patients with primary Sjögren’s Syndrome. BMC Musculoskelet. Disord. 15, 21 (2014).
Hamza, N. et al. Persistence of immunoglobulin-producing cells in parotid salivary glands of patients with primary Sjögren’s syndrome after B cell depletion therapy. Ann. Rheum. Dis. 71, 1881–1887 (2012).
Cambridge, G. et al. Circulating levels of B lymphocyte stimulator in patients with rheumatoid arthritis following rituximab treatment: relationships with B cell depletion, circulating antibodies, and clinical relapse. Arthritis Rheum. 54, 723–732 (2006).
Lavie, F. et al. Increase of B cell-activating factor of the TNF family (BAFF) after rituximab treatment: insights into a new regulating system of BAFF production. Ann. Rheum. Dis. 66, 700–703 (2007).
Dorner, T., Kinnman, N. & Tak, P. P. Targeting B cells in immune-mediated inflammatory disease: a comprehensive review of mechanisms of action and identification of biomarkers. Pharmacol. Ther. 125, 464–475 (2010).
Broeren, M. G. A. et al. Proteogenomic analysis of the autoreactive B cell repertoire in blood and tissues of patients with Sjögren’s syndrome. Ann. Rheum. Dis. 81, 644–652 (2022).
Mariette, X. et al. A randomized, phase II study of sequential belimumab and rituximab in primary Sjögren’s syndrome. JCI Insight 7, e163030 (2022).
Pezot, M. et al. Obinutuzumab in patients with Sjogren’s disease immunised against rituximab. Ann. Rheum. Dis. 83, 407–408 (2024).
Wong, D. P. et al. A BAFF ligand-based CAR-T cell targeting three receptors and multiple B cell cancers. Nat. Commun. 13, 217 (2022).
Howard, J. F. Jr., Vu, T. & Mozaffar, T. CAR T-cell therapy in autoimmune disease. N. Engl. J. Med. 390, 1629–1631 (2024).
Wilhelm, A. et al. Selective CAR-T cell mediated B cell depletion suppresses interferon signature in SLE. JCI Insight https://doi.org/10.1172/jci.insight.179433 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05085431 (2021).
Steinfeld, S. D. et al. Epratuzumab (humanised anti-CD22 antibody) in primary Sjögren’s syndrome: an open-label phase I/II study. Arthritis Res. Ther. 8, R129 (2006).
Gottenberg, J. E. et al. Efficacy of epratuzumab, an anti-CD22 monoclonal IgG antibody, in systemic lupus erythematosus patients with associated Sjögren’s syndrome: post hoc analyses from the EMBODY trials. Arthritis Rheumatol. 70, 763–773 (2018).
Clowse, M. E. et al. Efficacy and safety of epratuzumab in moderately to severely active systemic lupus erythematosus: results from two phase III randomized, double-blind, placebo-controlled trials. Arthritis Rheumatol. 69, 362–375 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02962895 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05350072 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05349214 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02614716 (2018).
Xu, D. et al. Efficacy and safety of telitacicept in primary Sjögren’s syndrome: a randomized, double-blind, placebo-controlled, phase 2 trial. Rheumatology 63, 698–705 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05673993 (2023).
Tkachenko, A., Kupcova, K. & Havranek, O. B-cell receptor signaling and beyond: the role of Igα (CD79a)/Igβ (CD79b) in normal and malignant B cells. Int. J. Mol. Sci. 25, 10 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04186871 (2024).
Dorner, T. et al. Efficacy and safety of remibrutinib, a selective potent oral BTK inhibitor, in Sjögren’s syndrome: results from a randomised, double-blind, placebo-controlled phase 2 trial. Ann. Rheum. Dis. 83, 360–371 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04700280 (2023).
Juarez, M. et al. A phase 2 randomized, double-blind, placebo-controlled, proof-of-concept study of oral seletalisib in primary Sjögren’s syndrome. Rheumatology 60, 1364–1375 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02775916 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03627065 (2021).
Scuron, M. D., Fay, B. L., Connell, A. J., Oliver, J. & Smith, P. A. The PI3Kδ inhibitor parsaclisib ameliorates pathology and reduces autoantibody formation in preclinical models of systemic lupus erythematosus and Sjögren’s syndrome. Int. Immunopharmacol. 98, 107904 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04968912 (2024).
Bhandari, V. & Bril, V. FcRN receptor antagonists in the management of myasthenia gravis. Front. Neurol. 14, 1229112 (2023).
Hubbard, J. et al. POS1130 designing of a phase 2, multicenter, randomized, placebo-controlled, double-blind study to assess the efficacy and safety of nipocalimab, an FCRN inhibitor, in adults with primary Sjögren’s syndrome. Ann. Rheum. Dis. 82, 892–893 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05817669 (2024).
Baer, A. N. et al. Efficacy and safety of abatacept in active primary Sjögren’s syndrome: results of a phase III, randomised, placebo-controlled trial. Ann. Rheum. Dis. 80, 339–348 (2021).
van Nimwegen, J. F. et al. Abatacept treatment for patients with early active primary Sjögren’s syndrome: a single-centre, randomised, double-blind, placebo-controlled, phase 3 trial (ASAP-III study). Lancet Rheumatol. 2, e153–e163 (2020).
de Wolff, L. et al. Long-term abatacept treatment for 48 weeks in patients with primary Sjögren’s syndrome: the open-label extension phase of the ASAP-III trial. Semin. Arthritis Rheum. 53, 151955 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02843659 (2018).
Li, P., Jin, Y., Zhao, R., Xue, Z. & Ji, J. Expression of ICOS in the salivary glands of patients with primary Sjogren’s syndrome and its molecular mechanism. Mol. Med Rep. 26, 348 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00344448 (2015).
U.S. Food and Drug Administration. Efalizumab (marketed as raptiva) information. FDA https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/efalizumab-marketed-raptiva-information (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05217472 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02291029 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03905525 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04541589 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04129164 (2024).
Fisher, B. et al. FRI0174 subcutaneous dosing of the novel antI-CD40 antibody iscalimab achieves target drug exposure and clinical efficacy in primary Sjögren’s syndrome; results of a phasE IIA randomised open label two arm parallel group triaL. Ann. Rheum. Dis. 78, 760–761 (2019).
Robles-Carrillo, L. et al. Anti-CD40L immune complexes potently activate platelets in vitro and cause thrombosis in FCGR2A transgenic mice. J. Immunol. 185, 1577–1583 (2010).
St, E. W. et al. OP0143 efficacy and safety of DAZODALIBEP (VIB4920/HZN4920) in subjects with Sjögren’s syndrome: a phase 2, randomized, double-blind, placebo-controlled, proof of concept sTUDY. Ann. Rheum. Dis. 82, 95–95 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT06245408 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT06104124 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04684654 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04572841 (2024).
He, J. et al. Efficacy and safety of low-dose interleukin 2 for primary Sjögren syndrome: a randomized clinical trial. JAMA Netw. Open. 5, e2241451 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04093531 (2022).
Vendel, A. C. et al. B and T lymphocyte attenuator regulates B cell receptor signaling by targeting Syk and BLNK. J. Immunol. 182, 1509–1517 (2009).
Mavragani, C. P. & Moutsopoulos, H. M. Sjögren’s syndrome: old and new therapeutic targets. J. Autoimmun. 110, 102364 (2020).
Wang, B. et al. Targeted therapy for primary Sjögren’s syndrome: where are we now? BioDrugs 35, 593–610 (2021).
Tan, P. H., Ji, J., Hsing, C. H., Tan, R. & Ji, R. R. Emerging roles of type-I interferons in neuroinflammation, neurological diseases, and long-haul COVID. Int. J. Mol. Sci. 23, 14394 (2022).
de Oliveira, F. R. et al. Neurological and inflammatory manifestations in Sjögren’s syndrome: the role of the kynurenine metabolic pathway. Int. J. Mol. Sci. 19, 3953 (2018).
Maria, N. I. et al. Association of increased Treg cell levels with elevated indoleamine 2,3-dioxygenase activity and an imbalanced kynurenine pathway in interferon-positive primary Sjögren’s syndrome. Arthritis Rheumatol. 68, 1688–1699 (2016).
Tarn, J. R. et al. Symptom-based stratification of patients with primary Sjogren’s syndrome: multi-dimensional characterisation of international observational cohorts and reanalyses of randomised clinical trials. Lancet Rheumatol. 1, e85–e94 (2019).
Soret, P. et al. A new molecular classification to drive precision treatment strategies in primary Sjögren’s syndrome. Nat. Commun. 12, 3523 (2021).
Warner, B. et al. GZMK+CD8+ T cells target a specific acinar cell type in Sjogren’s disease. Preprint at Research Square https://doi.org/10.21203/rs.3.rs-3601404/v1 (2023).
Sarkar, I. et al. Aberrant signaling of immune cells in Sjögren’s syndrome patient subgroups upon interferon stimulation. Front. Immunol. 13, 854183 (2022).
van der Kroef, M. et al. Cytometry by time of flight identifies distinct signatures in patients with systemic sclerosis, systemic lupus erythematosus and Sjögrens syndrome. Eur. J. Immunol. 50, 119–129 (2020).
van Ginkel, M. S. et al. Increased diagnostic accuracy of the labial gland biopsy in primary Sjögren syndrome when multiple histopathological features are included. Arthritis Rheumatol. 76, 421–428 (2024).
Luo, D. et al. Unraveling the transcriptome-based network of Tfh cells in primary Sjogren syndrome: insights from a systems biology approach. Front. Immunol. 14, 1216379 (2023).
Nguyen, Y. et al. Identification of distinct subgroups of Sjögren’s disease by cluster analysis based on clinical and biological manifestations: data from the cross-sectional Paris-Saclay and the prospective ASSESS cohorts. Lancet Rheumatol. 6, e216–e225 (2024).
Lorenzon, M. et al. Salivary gland ultrasound in primary Sjögren’s syndrome: current and future perspectives. Open. Access. Rheumatol. 14, 147–160 (2022).
Fulvio, G. et al. UHFUS: a valuable tool in evaluating exocrine gland abnormalities in Sjögren’s disease. Diagnostics 13, 2771 (2023).
Felten, R., Scher, F., Sibilia, J., Gottenberg, J. E. & Arnaud, L. The pipeline of targeted therapies under clinical development for primary Sjögren’s syndrome: a systematic review of trials. Autoimmun. Rev. 18, 576–582 (2019).
Felten, R. et al. Identification of new candidate drugs for primary Sjögren’s syndrome using a drug repurposing transcriptomic approach. Rheumatology 26, 3715–3723 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03247686 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05113004 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04988087 (2023).
Bai, W. et al. A multi-center, open-label, randomized study to explore efficacy and safety of baricitinib in active primary Sjogren’s syndrome patients. Trials 24, 112 (2023).
Felten, R. et al. Interleukin 6 receptor inhibition in primary Sjögren syndrome: a multicentre double-blind randomised placebo-controlled trial. Ann. Rheum. Dis. 80, 329–338 (2020).
Norheim, K. B., Harboe, E., Gøransson, L. G. & Omdal, R. Interleukin-1 inhibition and fatigue in primary Sjögren’s syndrome — a double blind, randomised clinical trial. PLoS One 7, e30123 (2012).
Mariette, X. et al. Inefficacy of infliximab in primary Sjögren’s syndrome: results of the randomized, controlled Trial of Remicade in Primary Sjögren’s Syndrome (TRIPSS). Arthritis Rheum. 50, 1270–1276 (2004).
Sankar, V. et al. Etanercept in Sjögren’s syndrome: a twelve-week randomized, double-blind, placebo-controlled pilot clinical trial. Arthritis Rheum. 50, 2240–2245 (2004).
Seror, R. et al. Low numbers of blood and salivary natural killer cells are associated with a better response to belimumab in primary Sjögren’s syndrome: results of the BELISS study. Arthritis Res. Ther. 17, 241 (2015).
Pontarini, E. et al. Treatment with belimumab restores B cell subsets and their expression of B cell activating factor receptor in patients with primary Sjogren’s syndrome. Rheumatology 54, 1429–1434 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04563195 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05124925 (2024).
Diekhoff, T. et al. Ianalumab (VAY736) in primary Sjögren’s syndrome: assessing disease activity using multi-modal ultrasound. Clin. Exp. Rheumatol. 38, 228–236 (2020).
Bowman, S. J. et al. Safety and efficacy of subcutaneous ianalumab (VAY736) in patients with primary Sjögren’s syndrome: a randomised, double-blind, placebo-controlled, phase 2b dose-finding trial. Lancet 399, 161–171 (2022).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02915159 (2020).
de Wolff, L., Arends, S., Verstappen, G. M. & Bootsma, H. Minimal added value of separate dryness assessments compared with overall dryness in ESSPRI in patients with Sjögren’s disease. Rheumatology 63, e29–e31 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02334306 (2019).
Bentley, D., Fisher, B. A., Barone, F., Kolb, F. A. & Attley, G. A randomized, double-blind, placebo-controlled, parallel group study on the effects of a cathepsin S inhibitor in primary Sjögren’s syndrome. Rheumatology 62, 3644–3653 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03239600 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04605978 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05781451 (2024).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01552681 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04981145 (2022).
Seror, R. et al. Defining disease activity states and clinically meaningful improvement in primary Sjögren’s syndrome with EULAR primary Sjögren’s syndrome disease activity (ESSDAI) and patient-reported indexes (ESSPRI). Ann. Rheum. Dis. 75, 382–389 (2016).
Cornec, D. et al. Development of the Sjögren’s syndrome responder index, a data-driven composite endpoint for assessing treatment efficacy. Rheumatology 54, 1699–1708 (2015).
Arends, S. et al. Composite of relevant endpoints for Sjogren’s syndrome (CRESS): development and validation of a novel outcome measure. Lancet Rheumatol. 3, e553–e562 (2021).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
C.B. and F.F. have participated in randomized clinical trials of deucravacitinib (for BMS), belimumab plus rituximab (for GSK), anti-CD40L therapy (for Horizon Therapeutics), anti-CD40 therapy (for Novartis) and anti-CD20 therapy (for Novartis). G.F. and G.L.R. have participated in randomized clinical trials of deucravacitinib (for BMS), anti-CD40L therapy (for Horizon Therapeutics), anti-CD40 therapy (for Novartis) and anti-CD20 therapy (for Novartis). C.B. has served as adviser or consultant for Aurina, GSK, Horizon Therapeutics/AMGEN, Johnson and Johnson and Sanofi.
Peer review
Peer review information
Nature Reviews Rheumatology thanks Robert I. Fox, Valerie Devauchelle-Pensec and Raphaèle Seror, who co-reviewed with Gregoire Martin de Fremont, for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
The NECESSITY project: https://www.necessity-h2020.eu/
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baldini, C., Fulvio, G., La Rocca, G. et al. Update on the pathophysiology and treatment of primary Sjögren syndrome. Nat Rev Rheumatol 20, 473–491 (2024). https://doi.org/10.1038/s41584-024-01135-3
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41584-024-01135-3
This article is cited by
-
Nipocalimab reduces Sjögren disease activity in phase II trial
Nature Reviews Rheumatology (2026)
-
Usefulness of salivary gland ultrasonography in Sjögren’s syndrome: The Tunisian Experience
Clinical Rheumatology (2026)
-
Ferroptosis in salivary gland disorders: mechanisms, biomarkers, and therapeutic perspectives
Apoptosis (2026)
-
Effects of cognitive-motor dual-task walking on gait and pain in primary Sjögren’s syndrome: a comparative cross-sectional study
Clinical Rheumatology (2026)
-
miRNA let-7f-5p-encapsulated labial gland MSC-derived EVs ameliorate experimental Sjögren’s syndrome by suppressing Th17 cells via targeting RORC/IL-17A signaling axis
Journal of Nanobiotechnology (2025)