Abstract
The targeted pollination strategy has shown positive results in directing honey bees to crop flowers offering nectar along with pollen as reward. Kiwifruit is a functionally dioecious species, which relies on bees to transport pollen from staminate to pistillate nectarless flowers. Following the targeted pollination procedures recently validated, we first developed a mimic odor (KM) based on kiwifruit floral volatiles for which bees showed the highest level of generalization to the natural floral scent, although the response towards pistillate flowers was higher than towards staminate flowers. Then, in the field, feeding colonies KM-scented sucrose solution resulted in higher amounts of kiwifruit pollen collected by honey bees compared to control colonies fed unscented sucrose solution. Our results support the hypothesis that olfactory conditioning bees biases their foraging preferences in a nectarless crop, given the higher visitation to target flowers despite having provided the mimic odor paired with a sugar reward.
Similar content being viewed by others
Introduction
In the last decades, different strategies have been proposed to cope with an increasing demand for pollination services in agricultural settings worldwide1, such as preserving areas of semi-natural habitats for enhancing wild pollinator assemblages2, managing alternative bee species3, or recurring to hand pollination practices4. Nevertheless, global food production mostly relies on a single pollinator species, the western honey bee Apis mellifera5,6. Given the honey bee dominance as managed pollinator in agroecosystems, many efforts have been made to guide them to the target crop7. Recently, the targeted pollination strategy has proven to be successful in diverse pollinator-dependent crops, such as sunflower8, pear, apple9 and almond10. Briefly, this strategy implies conditioning bees to simple odorant mixtures mimicking the floral scent of the target crop to bias their foraging preferences with the potentiality to increase yields. So far, this procedure has shown positive results in directing honey bee foragers to flowers offering nectar along with pollen as reward. However, it remains to be assessed the extent to which this strategy can be implemented with nectarless crop flowers.
Kiwifruit (Actinidia chinensis var. deliciosa (A. Chev.) A. Chev.) is a functionally dioecious species, which requires the transport of pollen from staminate flowers of polleniser vines to pistillate flowers of fruiting vines for successful pollination11. Kiwifruit pollination depends highly on insects, and to a lesser extent on wind12,13,14. Indeed, to obtain fruits of marketable size and quality, pistillate flowers must receive enough viable pollen and produce hundreds of seeds12,15. Although pistillate flowers appear to be hermaphroditic, their stamens produce sterile pollen (pseudopollen) and are possibly involved in pollinator attraction as in other Actinidia species11,16. Nevertheless, kiwifruit flowers are claimed to be unattractive to honey bees due to the lack of nectar, particularly in the presence of competing flora17,18,19. Anyhow, producers rely on the honey bee as primary managed pollinator in kiwifruit orchards12,15 but frequently employ artificial pollination techniques to ensure pollination in different geographic regions including Argentina20,21, Italy22, Portugal23, and the United States24.
Previous studies report that honey bees have a preference for pistillate flowers compared to staminate flowers17,25, even though the pseudopollen appears to be of low nutritional value26. Such preference would indicate that honey bees are able to discriminate between both types of flowers based for instance on visual and olfactory cues27. Indeed, the floral volatiles of different female and male kiwifruit cultivars showed great qualitative similarity but some quantitative differences, particularly in the compounds perceived by the honey bee28. These authors suggested that the close match between floral volatiles of female and male cultivars would facilitate honey bee movements between pistillate and staminate flowers, while dissimilarities across the odor profiles of cultivars would hinder cross-pollination.
Our research aimed to determine the effectiveness of the targeted pollination strategy in nectarless dioecious crops offering pollen as reward. Our goals were to (1) develop a synthetic odorant mixture based on kiwifruit floral volatiles that honey bees would generalize towards the natural scent of pistillate and staminate flowers, and (2) evaluate the effect of conditioning honey bee colonies in a kiwifruit orchard with the developed mimic odor on directing pollinators to the target crop. We formulated a synthetic odorant mixture (KM) which honey bees poorly discriminated from the natural scent of kiwifruit flowers under laboratory conditions. In the field, the amount of kiwifruit pollen collected in colonies fed KM-scented sucrose solution resulted higher than the control colonies fed unscented sucrose solution, suggesting a biased preference towards the target crop.
Results
Identification of kiwifruit floral volatiles
To account for the presence of the volatile compounds previously reported for kiwifruit flowers28,29 in the cultivars most frequently planted in Argentina, volatiles were collected in situ by dynamic headspace sampling in two orchards. We found no qualitative differences between samples from flowers of male and female kiwifruit cultivars. The six volatile compounds previously reported to provoke honey bee antennal responses were identified in all samples, accounting for 46–62% of the sampled volatile blends (Supplementary Table S1). Specifically, three of these antennally active compounds were among the most abundant compounds (α-farnesene, (6Z,9Z)-heptadecadiene, and (8Z)-heptadecene). Other major compounds corresponded to the saturated hydrocarbons n-tridecane, n-pentadecane, and n-heptadecane, present in both types of flowers (data not shown).
Having confirmed the presence of the antenally active floral volatiles of pistillate and staminate kiwifruit flowers, different odorant mixtures were formulated taking into account the commercially available compounds.
Similarity of mimic odors to the natural kiwifruit floral scent
Six synthetic mixtures (KM, KM I–V) comprising three compounds each were proposed as candidate mimic odors of kiwifruit flowers and were used to test honey bee perception in comparison with the natural kiwifruit floral scent in an olfactory proboscis extension (PER) paradigm30,31.
First, we evaluated the ability of honey bees to generalize each mixture of odorants to the natural scent of kiwifruit pistillate and staminate flowers by means of absolute conditioning assays. After five training trials with each candidate mixture, bees showed high levels of response (≥ 80%) towards the learned stimulus (Fig. 1). However, when presented with the natural floral scent during the test phase, the conditioned bees were less responsive. Significant differences were found between the response levels of bees trained with each mixture towards the natural floral scent, regardless of the type of flower presented (pistillate or staminate) (GLMM: effect of CS, χ25 = 18.84, p < 0.01; effect of flower, χ21 = 0.08, p = 0.7789; Supplementary Table S2). Among the six mixtures evaluated, the KM showed the highest level of generalization to the natural floral scent, significantly higher than the mixture KM V (t-ratio = − 3.16 p = 0.02), although no significant differences were found between mixtures KM and KM II, III or IV (KM vs KM II, t-ratio = − 1.4 p = 0.7081; KM vs KM III, t-ratio = 0.784 p = 0.9702; KM vs KM IV, t-ratio = − 2.48 p = 0.1327), and responses were marginally different between KM and KM I (KM vs KM I, t-ratio = − 2.69 p = 0.08) (Supplementary Table S3).
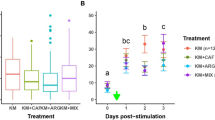
Odor generalization of memories from six candidate odorant mixtures to natural kiwifruit floral scent. After training bees to different synthetic mixtures used as conditioned stimulus (CS) during an absolute classical conditioning of the proboscis extension reflex (PER; left panel), their ability to generalize the CS toward the natural scent of pistillate and staminate kiwifruit flowers was tested (right panel). Significant differences were found between the response levels of bees trained with each mixture towards the natural floral scent (GLMM: effect of CS, χ25 = 18.84, p < 0.01; effect of flower, χ21 = 0.08, p = 0.7789; Supplementary Table S2). Bees trained with KM presented higher levels of response towards both types of flowers, although not significantly different from KM II and KM III (Supplementary Table S3). The number of bees tested is shown in parentheses.
To better assess bee’s ability to discriminate between a synthetic mixture and the natural kiwifruit floral scent, differential conditionings were performed. For this purpose, we selected the synthetic mixtures with the highest and lowest levels of generalization (KM and KM V, respectively), as well as a commercial extract (JM) as control. In a first series of assays, we presented the two candidate mixtures as rewarded conditioned stimulus (CS+) and the natural floral scent of either pistillate or staminate flowers or JM as unrewarded conditioned stimulus (CS−).
Bee’s discrimination ability was affected by the pair of conditioned stimuli presented (Fig. 2; GLM: effect of conditioned stimuli, χ24 = 21.33 p < 0.001; Supplementary Table S2). Bees trained with KM as CS+ and the natural scent of either pistillate or staminate flowers as CS− exhibited a diminished ability to discriminate between both stimuli compared to those trained with KM V as CS+ (KM+/pistillate flowers− vs KM V+/pistillate flowers−, t-ratio = 2.44 p = 0.11; KM+/staminate flowers− vs KM V+/staminate flowers−, t-ratio = 3.08 p < 0.05; Supplementary Table S4). On the other hand, the former was able to better discriminate the rewarded synthetic mixture from the unrewarded JM (KM+/pistillate flowers− vs KM+/JM−, t-ratio = − 2.83 p < 0.05; KM+/staminate flowers− vs KM+/JM−, t-ratio = − 2.46 p = 0.11; Supplementary Table S4).
Odor discrimination between synthetic mixtures and the natural scent of kiwifruit flowers. Discrimination was evaluated by means of a discrimination index (DI) calculated as the difference between the bee response towards the rewarded conditioned stimulus (CS+) minus the response to the unrewarded conditioned stimulus (CS−) in the last pair of training trials (mean ± s.e) (see more details in Statistics). Each conditioning series consisted of four training trials in which a synthetic mixture was presented as CS+ (KM or KM V) and either floral extracts of kiwifruit flowers (pistillate or staminate) or a commercial extract (JM) as CS− (see Supplementary Fig. S1). The DIs of the conditioning series with KM as CS+ and the floral extracts as CS− were lower. P values indicate differences between Dis in trial four between series (post hoc comparisons, *, p < 0.05). The number of bees tested is shown in parentheses.
Finally, in the series in which the CS+ was the natural scent of either pistillate or staminate flowers and the CS− were the synthetic mixtures or JM, bees showed similar discrimination abilities between both stimuli (Supplementary Fig. S2) (GLM: effect of conditioned sitmuli, χ24 = 3.33 p = 0.5046; Supplementary Table S2).
Based on the obtained results in both type of conditionings, KM was selected as the synthetic mixture which better mimics the kiwifruit floral scent, since it showed the highest level of generalization among candidate formulations, while it presented the lowest discrimination index among them.
Honeybee colony foraging activity
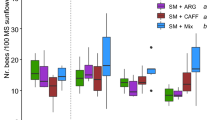
The offering of KM-scented food to colonies in the field showed varying impact on the number of incoming foragers at the hive entrance in the three years evaluated (Fig. 3). In 2019, when activity was assessed on days 2 and 8 after treatment, the total number of incoming bees in SS + KM-treated colonies resulted higher than in control colonies (SS), although such differences were not significant (Fig. 3A; GLMM: effect of treatment, χ21 = 0.158 p = 0.6912; Supplementary Table S2). A similar pattern was observed in the number of bees carrying pollen on their hind legs (Fig. 3D; GLMM: effect of treatment, χ21 = 0.03 p = 0.8624; Supplementary Table S2). In 2020, statistically higher activity levels were observed in SS + KM-treated colonies (Fig. 3B; GLMM: effect of treatment, χ21 = 4.54 p < 0.05; Supplementary Table S2). However, no differences were found in the number of pollen foragers between treatments (Fig. 3E; GLMM: effect of treatment, χ21 = 0.006 p = 0.9384; Supplementary Table S2). Finally, in 2021, when colonies were monitored on days 1, 2 and 3 after treatment, similar levels of activity were found in both groups of colonies in terms of total incoming foragers (Fig. 3C; GLMM: effect of treatment, χ21 = 0.245 p = 0.6210; Supplementary Table S2), as well as incoming pollen-foragers (Fig. 3F; GLMM: effect of treatment, χ21 = 0.156 p = 0.6927; Supplementary Table S2).
Effect of kiwifruit mimic odor on the activity of colonies. The total number of incoming bees per minute and the number of bees carrying pollen loads were monitored after feeding colonies unscented sucrose solution (SS) or kiwifruit mimic scented food (SS + KM) in years 2019 (A, D), 2020 (B, E) and 2021 (C, F). Asterisk indicates significant difference between treatments (*p < 0.05). Circles indicate the mean values and bars represent two standard errors. The number of colonies evaluated is shown in parentheses.
Given that kiwifruit flowers do not offer nectar, we installed pollen traps at the entrance of hives to better assess the collection of kiwifruit pollen. Figure 4 depicts the effect of the offering of KM-scented food in the number and weight of kiwifruit pollen loads. In 2019, marginal differences were observed in the number of kiwifruit pollen loads trapped in KM-treated colonies compared to control colonies (SS) (Fig. 4A; GLMM: effect of treatment, χ21 = 2.57 p = 0.11; Supplementary Table S2). However, the analysis of the dry weight of pollen loads revealed a significant interaction between treatment and time (Fig. 4C; GLMM: effect of treatment*days, χ21 = 3.98 p < 0.05; Supplementary Table S2). Although there was no difference between treatments 2 days after treating the colonies (SS vs SS + KM, t-ratio = 0.173 p = 0.8646), kiwifruit pollen loads resulted significantly heavier in KM-treated colonies on day 8 (SS vs SS + KM, t-ratio = − 2.754 p < 0.05) (Supplementary Table S5). In 2021, the number of kiwifruit pollen loads trapped in KM-treated colonies was significantly higher than in control colonies, though not across all days measured (Fig. 4B; GLMM: significant treatment*days interaction, χ22 = 9.89 p < 0.01; Supplementary Table S2). Larger amounts of pollen loads were collected on days 1 and 3, but not on day 2 (comparisons SS vs SS + KM: day 1, t-ratio = − 4.096 p < 0.001; day 2, t-ratio = − 1.532 p = 0.1385; day 3, t-ratio = − 4.109 p < 0.001; Supplementary Table S5). Instead, the weight of pollen loads collected in KM-treated colonies resulted consistently higher on the 3 days evaluated (Fig. 4D; GLMM: effect of treatment, χ22 = 4.33 p < 0.05; Supplementary Table S2).
Effect of kiwifruit mimic odor on the collection of kiwifruit pollen. The number and weight of trapped kiwifruit (white) pollen loads was monitored after feeding colonies unscented sucrose solution (SS) or kiwifruit mimic scented food (SS + KM) in years 2019 (A, C), and 2021 (B, D). Asterisks indicate significant difference between treatments (*p < 0.05; ***p < 0.001). Circles indicate the mean values and bars represent two standard errors. The number of colonies evaluated is shown in parentheses.
Finally, in 2020, the number of incoming bees carrying white pollen loads was recorded as an indirect measure of pollen foraging on kiwifruit flowers (see “Methods”). An increase in the number of incoming foragers with kiwifruit pollen on their hind legs was observed in colonies treated with KM-scented food on day 2 (see Supplementary Fig. S3, Table S2; GLM: effect of treatment, χ21 = 8.89 p < 0.01).
Discussion
Our study supports the potential use of the targeted pollination strategy in kiwifruit, a nectarless dioecious crop, with the aim of improving honey bee pollination services. Firstly, we developed a synthetic odorant mixture (KM) which honey bees poorly discriminated from the natural scent of kiwifruit flowers under laboratory conditions. Then, we tested the effect of conditioning honey bee colonies with KM in the field during three consecutive years. While conditioned colonies presented similar levels of foraging activity (i.e. incoming bees), they showed enhanced kiwifruit pollen collection, suggesting a biased preference towards the target crop.
The volatile profiles of the kiwifruit flowers sampled bore resemblance to those previously reported for female and male cultivars of this species28,29. Specifically, among the major compounds, we found three of the six compounds reported to have elicited antennal responses in honey bees29. These qualitative results suggest variability in the relative contribution of certain compounds in the samples collected, such as 2-phenylethanol in staminate flowers, in line with previous reports which documented either higher or lower amounts of this floral volatile in different male cultivars compared to the Hayward female cultivar28. Additional studies are needed for a quantitative analysis of different cultivars frequently used in the studied production region with an appropriate number of replicates to better evaluate the variability of volatile profiles among kiwifruit flowers of different cultivars and geographies, which was not the aim of the present work.
With the aim of developing a single synthetic blend mimicking both types of flowers, we selected as kiwifruit mimic odor the 3-compound mixture for which bees showed the highest level of generalization to the natural floral scent, although the level of responses towards pistillate flowers was higher than towards staminate flowers. Such difference could be due to the observed quantitive variation in the volatile profile of both type of flowers. In this sense, it is well established that honey bees rely on a few key compounds when processing complex odorant mixtures such as floral blends32. Recently, it has been demonstrated that honey bees conditioned to mimic odors comprising 3 or 4 volatiles present in target crop flowers were less capable of discriminating between each mimic odor and the respective natural floral scent in differential conditioning protocols when the former was paired to a sucrose reward and the latter was not rewarded8,9,10. Herein, the differential conditioning assays suggest that bees were not able to clearly discriminate between the rewarded mimic odor and the unrewarded scent of kiwifruit flowers, evinced by the low values of the discrimination index. On the other hand, the reduction of the response levels towards both types of flowes in the absolute conditionings might be explained by differences in the concentration of the stimuli presented, given that the synthetic mixture involved pure odorants, and thus, higher concentrations compared to the flowers. In this regard, previous works show that absolute odor concentrations might be a salient feature used by honey bees in odor recognition and discrimination33. It is worth mentioning that discrimination abilities were not symmetrical in the differential conditionings when the floral extracts were rewarded and the mimics were not, in line with results obtained for other mimic odors7. Such asymmetry denotes the complexity of the olfactory environment and how honey bees extract important cues for the exploitation of floral resources34. It is well known that olfactory information is processed in neural networks, which exhibit dynamic activity patterns according to experience, being the mushroom body a central structure of the honey bee brain where sensory information is converted to value-based information35. During associative learning events, most of the neurons in this centre show increased activity when presented a rewarded stimulus, while activity is mostly lost when the stimulus is not accompanied by a reward35. To better understand the underlying mechanism of the observed asymmetric responses to the mimic odor and the natural kiwifruit floral scent, optical imaging techniques could be used to compare neural activity patterns generated by rewarded or unrewarded simple or complex odorant mixtures.
In the field, feeding colonies KM-scented sucrose solution resulted in higher amounts of kiwifruit pollen collected by honey bees compared to control colonies. These findings are consistent with mimic odors previously developed for sunflower8, pear and apple9 and almond10, which positively affected honey bee foraging preferences towards each target crop. Yet, these crop flowers offer nectar as reward while kiwifruit pistillate and staminate flowers do not. It is worth mentioning, though, that pear flowers offer low-quality nectar in terms of sugar concentrarion but high-quality pollen, being this the reward mainly exploited by honey bees36. In pear orchards, treating colonies with scented sugar solution ultimately resulted in a higher number of fruits per tree9, which could be explained by a percentage of conditioned foragers being able to switch to pollen collection based on the actual profitability of the resources encountered in pear flowers37. In our study, KM-treated colonies consistently increased the collection of kiwifruit pollen despite having provided the mimic odor paired with a sugar reward. Such increases in the collection of kiwifruit pollen (although we were not able to distinguish between the fertile pollen of staminate flowers and the pseudopollen of pistillate ones) could translate into higher yields if both types of flowers received similar number of honey bee visits. However, differences in the response levels of conditioned honey bees towards staminate and pistillate flowers were herein observed under laboratory conditions. Such differentiation is in agreement with the results of Goodwin and Congdon38, supporting the hypothesis that they are able to distinguish between staminate and pistillate flowers based on the taste and/or odour of the two types of pollen. These authors observed that nectar foragers were more responsive to artificially collected pistillate pollen applied on their antennae in one out of two assays under the PER-paradigm. It is not trivial to mention that nectar and pollen foragers differ in their sensitivity to odors39, and, in particular, in the latter, the delivery of pollen on the tarsi performed slightly better than when applied on the antennae40. In view of the complexity of honey bee olfactory perception and the specific requirements of kiwifruit pollination, future field studies should test the effect of the mimic odor on the attractiveness of both types of flowers for pollen foragers, as well as their transition rates between flowers and vines to evaluate the impact on fruit production. Interestingly, in a pilot trial carried out in a third commercial orchard near Mar del Plata, province of Buenos Aires, Argentina, the fruit set of kiwifruit plants in the surroundings of treated colonies resulted 32% higher than in control plants (N = 30 branches near KM-treated colonies, N = 25 branches near control colonies, unpublished data).
Other recent attempts to improve honey bee pollination of kiwifruit have involved the spraying of female flowers or whole branches with non-specific attractants, such as plant natural extracts41 or pheromonal compounds42. Although these methods have shown success in increasing honey bee visitations and the numbers of seeds per fruit, they would require considerable labor and/or mechanical force, implying high costs. On the other hand the targeted pollination strategy has the advantage of being compatible with a common practice of beekeepers, which is feeding sugar syrup to colonies in kiwifruit orchards43 and of being crop-specific, in contrast with pheromone-based attractants which can trigger innate responses not related to the foraging behavior of honey bees7.
Our study presents novel insights into whether the use of mimic odors supplied with a sugar reward within the colonies has the potential to direct honey bees to a target crop offering pollen as reward. Our results support the hypothesis that olfactory conditioning bees biases their foraging preferences, resulting in higher visitation to target flowers even when the actual floral reward differs from the one previously experienced inside the hive. Additional studies are needed to understand the duration of the effect and its impact on cross pollination and yield, looking into the dynamics among nectar and pollen foragers activated within the colony, as well as the differences between the proportion of kiwifruit pollen collected from each flower type.
Methods
Study sites, chemicals and honeybee colonies
Kiwifruit (Actinidia chinensis var. deliciosa (A. Chev.) A. Chev.) floral volatiles were collected in a commercial orchard, ‘El Abrojito’, near Miramar, province of Buenos Aires, Argentina (38° 12′ 54″ S, 57° 53′ 41″ W), during the blooming season of 2018. Vines of female cultivar Hayward were interplanted with vines of three male cultivars (Matua, Tomuri and Chieftain) to ensure pollen availability throughout the blooming period. Additional volatile collections were carried out in a second commercial orchard, ‘Dos Abriles’, near Navarro, province of Buenos Aires, Argentina (33° 47′ 48″ S, 59° 44′ 13″ W), during the blooming season of 2018. Vines of female cultivar Hayward were interplanted with vines of male cultivars Chico malo, Matua, Tomuri and Chieftain.
The identification of volatile compounds was carried out at the Chemical Ecology Laboratory of Facultad de Química (Universidad de la República, Montevideo, Uruguay). Chemical compounds used to prepare the synthetic mixtures were obtained from Sigma-Aldrich, Steinheim, Germany, and from Tokyo Chemical Industry (TCI), Tokyo, Japan. The six synthetic mixtures (KM, KM I–V) were composed by farnesene (mixture of isomers, product #W383902), nonanal (product #N30803), 2-phenylethanol (product #77861), 1-heptadecene (product #H0372). For details of their proportions in the mixtures see Patent PCT/IB2022/05828744. Jasmine mimic (JM) was a commercial extract obtained from Laboratorios Helvética S.A., Buenos Aires, Argentina, which was chosen based on its presumed dissimilarity with the kiwifruit floral scent.
Conditioning experiments were performed with naïve bee foragers captured at the entrance of Langstroth-type hives located at in the Experimental Field of the School of Exact and Natural Sciences of the University of Buenos Aires (34° 32′ S, 58° 26′ W). Absolute olfactory conditionings were done during November 2017 and 2018, while differential olfactory conditionings were carried out from November 2020 to February 2021.
Field experiments were performed in the commercial orchard ‘Dos Abriles’ (5 ha) during the kiwifruit blooming seasons of 2019, 2020 and 2021. Commercial honeybee colonies (Langstroth-type hives) providing pollination services were placed along the borders of the plantation in groups. All colonies were inspected before conducting the study. Experimental colonies contained a mated queen, 4–6 brood frames, limited food reserves and 20,000 worker strength. During each year 2–4 groups of experimental colonies were randomly assigned to the treatments (see section Feeding protocol). A total of 40 experimental colonies were used in 2019, 22 in 2020, and 21 in 2021.
Collection and identification of kiwifruit floral volatiles
To account for the presence of the volatile compounds previously reported for kiwifruit flowers28,29 in the cultivars most frequently planted in Argentina, volatiles were collected in situ by dynamic headspace sampling. A branch of each cultivar (i.e., female and male vines) bearing 4–12 recently opened flowers was enclosed in polyester oven bags (30 × 45 cm) gently sealed around the stems with plastic seals for a period of 7.5 h (between 8:00 a.m. and 15:30 p.m., coinciding with bee foraging activity). Floral volatiles were trapped on 100 mg of HayeSep Q 80/100 mesh (product #10301-U, Sigma-Aldrich) with a flow rate of ca. 1 L/min and eluted using 2 mL of n-hexane (product # 650552, Sigma-Aldrich). Samples were stored at − 20 °C until analysis.
Analyses of volatiles, with special focus on the compounds which provoked active antennal responses in honeybees29, were performed in a GC–MS instrument (QP–2010, Shimadzu Corp.). One-microliter samples were injected in a non-polar column (Rtx–5MS, Restek, USA) (30 m × 0.25 mm, 0.25 µm). The injection port was operated in the splitless mode with helium as carrier gas with a constant column flow of 1.0 mL/min. The initial oven temperature was 40 °C (held for 1 min), ramped at 7 °C/min to 280 °C (held for 5 min). The injector temperature was 280 °C. The mass spectrometer was operated in the electron ionization mode at 70 eV, in the scan mode (mass range: m/z 30–350) with an ion source temperature of 200 °C. Compounds were identified based on their mass spectra and retention indices in comparison with those of the NIST17 and Adams’ database and in some cases, of synthetic standards45. Experimental ion fragmentation patterns of the floral volatile compounds are available upon request.
Similarity of mimic odors to the natural kiwifruit floral scent
The main volatiles of kiwifruit flowers known to elicit bee antennal responses were taken into account to formulate different synthetic mixtures of three compounds each that would imitate the natural floral scent. Due to the difficulty of synthesizing hydrocarbon standards which were not commercially available, they were disregarded (6Z,9Z-heptadecadiene) or replaced by a commercially available isomer (1-heptadecene was used instead of 8Z-heptadecene). Six candidate mixtures were used to test honey bee perception in comparison with the natural kiwifruit floral scent in an olfactory proboscis extension paradigm30,31.
To assay the PER, harnessed bees were placed in front of a continuous airflow (50 mL/s). On each conditioning assay, a candidate odorant mixture (4 µL impregnated on a 30 × 3 mm filter paper inside a syringe) was delivered through a secondary airstream (6.25 mL/s) injected into the main airflow through a system of valves controlled by a computer. On the other hand, the natural scent of kiwifruit flowers was swept from a 100-g sample of either pistillate or staminate flowers contained in a Büchner flask by means of a continuous airflow. Only those bees that showed the unconditioned response (the reflexive extension of the proboscis after applying a 50% weight/weight (hence sucrose solution to the antennae) and did not respond to the mechanical air flow stimulus were used. Trials lasted 39 s which consisted in 15 s of clean air, followed by 6 s of odor presentation (CS) and lastly, 20 s of clean air. A drop of sucrose solution (50% weight/weight, henceforth w/w) was presented as reward (US) during the last 3 s of CS delivery. Bee response (PER) was evaluated during the first 3 s of the CS presentation. Bees that responded in the first trial were discarded, as well as those which did not elicit the reflexive response at the end of the experiment.
Returning honeybees foragers were randomly captured at the hive entrance and then anesthetized at − 4 °C for 1 min and individually harnessed in metal tubes, restraining their movement except for their antennae and mouthparts. After feeding the bees a droplet of 50% w/w sucrose solution, they were kept in darkness in an incubator (32 °C, 55% relative humidity) for about 1.5 h prior to conditioning experiments.
Absolute olfactory conditioning
First, we evaluated the ability of honey bees to generalize each mixture of odorants to the natural scent of kiwifruit pistillate and staminate flowers by means of absolute conditioning assays. Each assay consisted of five training trials during which one of the six candidate mixtures (KM, KM I–V) was presented as CS with an intertrial interval of 15 min. During the test phase, harnessed bees were presented with the natural kiwifruit scent of either pistillate or staminate flowers without reward as novel odors 15 min after the last training trial.
Kiwifruit flowers (from female and male cultivars) used as novel odors were collected in the commercial orchard ‘Dos Abriles’ during the blooming season and taken to the laboratory to be used in the absolute conditionings within the following 2 days.
Differential olfactory conditioning
As a second step, we evaluated the ability of honey bees to discriminate between a synthetic mixture (KM or KM V) and the natural kiwifruit floral scent. To this end, bees were subjected to a differential conditioning protocol31, consisting of four rewarded learning trials (CS+) and four unrewarded learning trials (CS−) in a pseudo-randomized order (−++−−+−+) with an interval trial interval of 20 min. During the test phase, bee response towards both odors without reward was evaluated 20 min after the last training trial.
Extracts of kiwifruit floral volatiles were used as conditioned stimuli (either as CS+ or CS−). An aliquot (5 µL) of an extract of either pistillate or staminate flowers was impregnated on filter paper inside a syringe (and injected in the airflow as described above) and renewed every 6 trials. The use of floral extracts as an alternative to the presentation of freshly collected flowers has the advantage of minimizing the release of volatiles emitted from the damaged tissues or wilting flowers46, while allowing for performing assays after the blooming season. The similarity of both methodologies was validated by means of absolute conditioning assays (data not shown).
Feeding protocol in field experiments
We used the offering of scented food within the hive as a standardized procedure to establish long-term olfactory memory in honey bees7. Scented food was obtained by diluting 50 µl of KM per liter of sucrose solution (50% w/w). Colonies were fed in situ in a single event, by pouring either 500 mL of KM-scented sucrose solution (SS + KM; N = 23 in 2019, N = 12 in 2020, N = 13 in 2021) or 500 ml of unscented 50% w/w sucrose solution (SS; N = 17 in 2019, N = 10 in 2020, N = 8 in 2021) as control, over the top of the central frames of the hives7. Depending on the year, treatments were done when kiwifruit female vines were 40–50% in bloom in 2019–2020, and 80% in 2021.
Honeybee colony foraging activity
The effect of KM-scented food on the general colony activity was assessed by recording the number of incoming foragers at the hive entrance. We registered the number of incoming bees for 1 min, once before treatment (day 0) and on the following mornings after treatment (between 10:30 a.m. and 12:30 p.m.; once per morning on days 2 and 8 in 2019, on day 2 in 2020, and on days 1, 2, 3 in 2021). Incoming bees were recorded as pollen-foragers if they carried pollen loads on their hind legs. For each year, we compared the activity level between SS + KM-treated colonies (N = 17 in 2019, N = 12 in 2020, N = 13 in 2021) and SS-treated colonies (N = 11 in 2019, N = 10 in 2020, N = 8 in 2021).
Additionally, we evaluated the collection of kiwifruit pollen by means of conventional pollen traps (frontal-entrance type). Pollen traps were installed for 4 h each day at the same time (before noon), once before treatment and on the following days after treatment (on the same days when colony activity was measured each year). Pollen samples of each colony were stored individually at − 2 0 °C until analysis. At the laboratory, pollen pellets were sorted by color, counted and weighed. White pellets were identified as coming from kiwifruit flowers based on their color47. Additionally, pollen harvested from anthers of kiwifruit pistillate and staminate flowers, as well as from foragers captured on each flower type served as voucher specimens for the preparation of reference slides for morphological identification of the trapped pollen48. For each year, we compared the number and dry weight of kiwifruit pollen loads between of SS + KM-treated colonies (N = 6 in 2019, N = 6 in 2021) and SS-treated colonies (N = 6 in 2019, N = 5 in 2021). During 2020, restricted access to the laboratory due to COVID-19 pandemic hindered the processing of pollen samples. Instead, the number of incoming bees carrying white pollen loads was recorded as an indirect measure of pollen foraging (see above). It is worth mentioning that the number of foragers with kiwifruit pollen loads can be underestimated due to changes in the color appearance under varying sunlight conditions.
Statistics
All statistical analyses were performed with R v4.2.149, using the glmmTMB package50. Models in general were simplified as follows: significance of the different terms was tested starting from the higher-order terms model using likelihood ratio tests to compare between models51. Non-significant terms (p > 0.05) were removed (see Supplementary Table S2). We considered the use of generalized linear and generalized linear mixed-effect models (GLM and GLMM) because they allow analyzing response variables whose errors are not normally distributed, avoiding the transformation of the response variable or the adoption of non-parametric methods52. All models were inspected for over-/underdispersion, zero inflation and distribution of the residuals. Scaled residuals were simulated from the fitted model using the DHARMa package53. Post hoc comparisons using contrast matrixes were performed with the emmeans function54 (Supplementary Tables S3–S5).
To analyze differences in generalization of each synthetic mixture to kiwifruit flowers, we compared the responses of each bee (if it extended the proboscis or not) to both types of flowers (pistillate or staminate) during the testing trials by means of a GLMM following a binomial (Bernoulli) error distribution. We considered the mixtures presented as CS (a six-level factor) and the type of flower (two-level factor) as fixed effects, while the bee was included as random effect to account for repeated testing of the same bee. Only those bees that extended the proboscis in the last training trial were considered, to ensure they had effectively established the odor-reward association.
The ability of bees to discriminate between two odors (CS+ and CS−) was analyzed by means of a discrimination index (DI), calculated as the difference between the bee response to the CS+ minus the response to the CS− in the last pair of training trials. The DI could take values of 0 or 1. A bee was able to discriminate between both stimuli only when it exhibited PER towards CS+ but not towards CS− (DI = 1). Differences in the DI obtained in the different series of conditionings (given by the CS+ and CS− presented) were assessed by means of a GLM following a binomial (Bernoulli) error distribution, in which we considered the conditioned stimuli (a five-level factor) as fixed effect. First, we analyzed the series of conditionings in which a synthetic candidate mixture (either FKV or FKII) was presented as CS+ and the natural floral extracts (either of female or male vines) or JM were presented as CS−. Secondly, we analyzed the series of assays in which of the natural floral extracts or JM were presented as CS+ and the two synthetic candidate mixtures were presented as CS−.
The rates of incoming bees of each year were analyzed separately by means of GLMMs (GLM in year 2020) following a negative binomial error distribution to account for the overdispersion of the data. We considered treatment (a two-level factor) and days (a two-level factor) as fixed effects, the rate of incoming bees before the offering of food (to control for pre-existing colony differences) as covariate (a quantitative fixed effect variable), and colony as a random factor to account for repeated measurements in years 2019 and 2021. Similarly, the rates of incoming bees with pollen (and kiwifruit pollen in year 2020) were analyzed with GLMMs or a GLM following a Poisson or negative binomial error distributions, considering the same parameters mentioned. Interaction terms (treatment*days) were initially included in all models but removed if nonsignificant.
The number of kiwifruit pollen loads collected in pollen traps in years 2019 and 2021 was analyzed separately by means of a GLMM following a negative binomial error distribution to account for the overdispersion of the data. We considered treatment (a two-level factor) and days (a two-level factor) as fixed effects, the number of pollen loads before the offering of food as covariate, and colony as a random factor to account for repeated measurements. Interaction terms (treatment*days) were initially included in the models but removed if nonsignificant.
The dry weight of kiwifruit pollen loads collected in pollen traps in years 2019 and 2021 was analyzed separately by means of a GLMM following a Gaussian error distribution. We considered treatment (a two-level factor) and days (a three-level factor) as fixed effects, the weight of pollen loads before the offering of food as covariate, and colony as a random factor to account for repeated measurements. Interaction terms (treatment*days) were initially included in the models but removed if nonsignificant.
Statement on the experimental research and field studies
The producers granted permission for plant measurements, which were done in situ. All methods and assays performed in this field study comply with national legislation and guidelines of the University of Buenos Aires and CONICET-Argentina.
Data availability
The datasets generated for this study are available on request to the corresponding author.
References
Aizen, M. A. et al. Global agricultural productivity is threatened by increasing pollinator dependence without a parallel increase in crop diversification. Glob. Change Biol. 25(10), 3516–3527. https://doi.org/10.1111/gcb.14736 (2019).
Garibaldi, L. A. et al. From research to action: Enhancing crop yield through wild pollinators. Front. Ecol. Environ. 12(8), 439–447. https://doi.org/10.1890/130330 (2014).
Osterman, J. et al. Global trends in the number and diversity of managed pollinator species. Agric. Ecosyst. Environ. 322, 107653. https://doi.org/10.1016/j.agee.2021.107653 (2021).
Wurz, A., Grass, I. & Tscharntke, T. Hand pollination of global crops—A systematic review. Basic Appl. Ecol. 56, 299–321. https://doi.org/10.1016/j.baae.2021.08.008 (2021).
Klein, A. M. et al. Importance of pollinators in changing landscapes for world crops. Proc. Royal Society B Biol. Sci. 274(1608), 303–313. https://doi.org/10.1098/rspb.2006.3721 (2007).
IPBES (2016). Summary for policymakers of the assessment report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on pollinators, pollination and food production. S.G. Potts, V.L. Imperatriz-Fonseca, H. T. Ngo, J.C. Biesmeijer, T.D. Breeze, L.V. Dicks, L.A. Garibaldi, R. Hill, J. Settele, A.J. Vanbergen, M.A. Aizen, S.A. Cunningham, C. Eardley, B.8 M. Freitas, N. Gallai, P. G. Kevan, A. Kovács-Hostyánszki, P.K. Kwapong, J. Li, X. Li, D.J. Martins, G. Nates-Parra, J.S. Pettis, R. Rader y B.F. Viana (Eds.). Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services, Bonn, Germany. 36 pages
Farina, W. M., Arenas, A., Estravis-Barcala, M. C. & Palottini, F. Targeted crop pollination by training honey bees: Advances and perspectives. Front. Bee Sci. 1, 1253157. https://doi.org/10.3389/frbee.2023.1253157 (2023).
Farina, W. M., Arenas, A., Díaz, P. C., Martin, C. S. & Estravis Barcala, M. C. Learning of a mimic odor within beehives improves pollination service efficiency in a commercial crop. Curr. Biol. 30(21), 4284–4290. https://doi.org/10.1016/j.cub.2020.08.018 (2020).
Farina, W. M., Arenas, A., Díaz, P. C., Susic Martin, C. & Corriale, M. J. In-hive learning of specific mimic odours as a tool to enhance honey bee foraging and pollination activities in pear and apple crops. Sci. Rep. 12(1), 20510. https://doi.org/10.1038/s41598-022-22985-5 (2022).
Farina, W. M. et al. Conditioning honeybees to a specific mimic odor increases foraging activity on a self-compatible almond variety. Apidologie 54(4), 40. https://doi.org/10.1007/s13592-023-01019-7 (2023).
Ferguson, A. R. (2016). Botanical description. In: R. Testolin et al. (eds.), The Kiwifruit Genome, Compendium of Plant Genomes, 1–13. https://doi.org/10.1007/978-3-319-32274-2_1
Costa, G., Testolin, R. & Vizzotto, G. Kiwifruit pollination: An unbiased estimate of wind and bee contribution. N. Z. J. Crop. Hortic. Sci. 21(2), 189–195. https://doi.org/10.1080/01140671.1993.9513767 (1993).
Vaissière, B. E., Rodet, G., Cousin, M., Botella, L. & Torré Grossa, J. P. Pollination effectiveness of honey bees (Hymenoptera: Apidae) in a kiwifruit orchard. J. Econ. Entomol. 89(2), 453–461. https://doi.org/10.1093/jee/89.2.453 (1996).
Goodwin, R. M., McBrydie, H. M. & Taylor, M. A. Wind and honey bee pollination of kiwifruit (Actinidia chinensis ‘HORT16A’). NZ J. Bot. 51(3), 229–240. https://doi.org/10.1080/0028825X.2013.806934 (2013).
Testolin, R., Vizzotio, G. & Costa, G. Kiwifruit pollination by wind and insects in Italy. N. Z. J. Crop. Hortic. Sci. 19(4), 381–384. https://doi.org/10.1080/01140671.1991.10422880 (1991).
Kawagoe, T. & Suzuki, N. Cryptic dioecy in Actinidia polygama: A test of the pollinator attraction hypothesis. Can. J. Bot. 82(2), 214–218. https://doi.org/10.1139/b03-150 (2004).
Palmer-Jones, T. & Clinch, P. G. Observations on the pollination of Chinese gooseberries variety ‘Hayward’. N. Z. J. Exp. Agric. 2(4), 455–458. https://doi.org/10.1080/03015521.1974.10427714 (1974).
Clinch, P.G. (1984). Kiwifruit pollination by honey bees 1. Tauranga observations, 1978–1981. New Zealand J. Exp. Agric. 12(1), 29–38. https://doi.org/10.1080/03015521.1984.10427785
Jay, D. & Jay, C. Observations of honeybees on Chinese gooseberries (‘Kiwifruit’) in New Zealand. Bee World 65(4), 155–166. https://doi.org/10.1080/0005772X.1984.11098804 (1984).
Sáez, A., Negri, P., Viel, M. & Aizen, M. A. Pollination efficiency of artificial and bee pollination practices in kiwifruit. Sci. Hortic. 246, 1017–1021. https://doi.org/10.1016/j.scienta.2018.11.072 (2019).
David, M.A., Yommi, A., Sánchez, E., Martinez, A., Murillo, N., Marcellán, O., Atela, O., & Palacio, M.A. (2022). Strategic use of honey bees (Apis mellifera L.) to increase the number and size of fruits in kiwifruit (Actinidia chinensis var. deliciosa). Eur. J. Agron. 133, 126420. https://doi.org/10.1016/j.eja.2021.126420
Tacconi, G., Michelotti, V., 2018. Artificial Pollination in Kiwifruit and Olive Trees. In: Segneanu, A.E., Orbeci, C., Lazau, C., Sfirloaga, P., Vlazan, P., Bandas, C., Grozescu, I. (Eds.), Pollination in Plants. InTech, p. 29. https://doi.org/10.5772/intechopen.74831
Castro, H. et al. Pollination as a key management tool in crop production: Kiwifruit orchards as a study case. Sci. Hortic. 290, 110533. https://doi.org/10.1016/j.scienta.2021.110533 (2021).
Abbate, A. P., Campbell, J. W., Vinson, E. L., & Williams, G. R. (2021). The pollination and fruit quality of two kiwifruit cultivars (Actinidia chinensis var. chinensis ‘AU Golden Sunshine’and ‘AU Gulf Coast Gold’)(Ericales: Actinidiaceae) grown in the southeastern United States. J. Econ. Entomol. 114(3), 1234–1241. https://doi.org/10.1093/jee/toab075
Goodwin, R.M., & Steven, D. (1993) Behaviour of honey bees visiting kiwifruit flowers, New Zealand J. Crop Hortic. Sci. 21:1, 17–24. https://doi.org/10.1080/01140671.1993.9513741
Jay, S. C. & Jay, D. H. The effect of kiwifruit (Actinidia deliciosa A Chev) and yellow flowered broom (Cytisus scoparius Link) pollen on the ovary development of worker honey bees (Apis mellifera L.). Apidologie 24(6), 557–563. https://doi.org/10.1051/apido:19930603 (1993).
von Frisch, K. The Dance Language and Orientation of Bees (Harvard University, 1967)
Twidle, A. M. et al. Identification of in situ flower volatiles from kiwifruit (Actinidia chinensis var. deliciosa) cultivars and their male pollenisers in a New Zealand orchard. Phytochemistry 141, 61–69. https://doi.org/10.1016/j.phytochem.2017.05.011 (2017).
Twidle, A. M. et al. Kiwifruit flower odor perception and recognition by honey bees, Apis mellifera. J. Agric. Food Chem. 63(23), 5597–5602. https://doi.org/10.1021/acs.jafc.5b01165 (2015).
Takeda, K. (1961). Classical conditioned response in the honey bee. J. Insect Physiol. 6 (3), 168–179. https://doi.org/10.1016/0022-1910(61)90060-9
Bitterman, M. E., Menzel, R., Fietz, A. & Schäfer, S. Classical conditioning of proboscis extension in honeybees (Apis mellifera ). J. Comp. Psychol. 97(2), 107–119. https://doi.org/10.1037/0735-7036.97.2.107 (1983).
Reinhard, J., Sinclair, M., Srinivasan, M. V. & Claudianos, C. Honeybees learn odour mixtures via a selection of key odorants. PloS One 5(2), e9110. https://doi.org/10.1371/journal.pone.0009110 (2010).
Wright, G. A., Thomson, M. G. & Smith, B. H. Odour concentration affects odour identity in honeybees. Proc. Royal Society B Biol. Sci. 272(1579), 2417–2422. https://doi.org/10.1098/rspb.2005.3252 (2005).
Sandoz, J., Pham-Delègue, M., Renou, M. & Wadhams, L. Asymmetrical generalisation between pheromonal and floral odours in appetitive olfactory conditioning of the honey bee (Apis mellifera L.). J. Comp. Physiol. A 187, 559–568. https://doi.org/10.1007/s003590100228 (2001).
Menzel, R. The honeybee as a model for understanding the basis of cognition. Nat. Rev. Neurosci. 13(11), 758–768. https://doi.org/10.1038/nrn3357 (2012).
Quinet, M. et al. Do floral resources influence pollination rates and subsequent fruit set in pear (Pyrus communis L.) and apple (Malus x domestica Borkh) cultivars?. Eur. J. Agro 77, 59–69. https://doi.org/10.1016/j.eja.2016.04.001 (2016).
Arenas, A. & Kohlmaier, M. G. Nectar source profitability influences individual foraging preferences for pollen and pollen-foraging activity of honeybee colonies. Behav. Ecol. Sociobiol. 73, 1–10. https://doi.org/10.1007/s00265-019-2644-5 (2019).
Goodwin, R.M., & Congdon, N.M. (2018). Recognition and attractiveness of staminate and pistillate kiwifruit flowers (Actinidia deliciosa var. deliciosa) by honey bees (Apis mellifera L.). New Zealand J. Crop Hortic. Sci. 46(1), 72–80. https://doi.org/10.1080/01140671.2017.1362012
Moreno, E., Corriale, M. J. & Arenas, A. Differences in olfactory sensitivity and odor detection correlate with foraging task specialization in honeybees Apis mellifera. J. Insect Physiol. 141, 104416. https://doi.org/10.1016/j.jinsphys.2022.104416 (2022).
Nery, D., Moreno, E., & Arenas, A. (2020). Pollen reinforces learning in honey bee pollen foragers but not in nectar foragers. J. Exp. Biol. 223(22), jeb230250. https://doi.org/10.1242/jeb.230250
Meroi Arcerito, F. R. et al. Fragrance addition improves visitation by honeybees and fruit quality in kiwifruit (Actinidia deliciosa). J. Sci. Food Agric. 101(12), 5082–5088. https://doi.org/10.1002/jsfa.11153 (2021).
Jailyang, L., Sharma, N.C., Chandel, J.S., Rana, V.S., Rana, K., & Chauhan, P. (2022). Influence of Bee Scent and other indigenous bee attractants on bee activity and fruiting behaviour of kiwifruit (Actinidia deliciosa A. Chev.). Sci. Hortic. 295, 110869. https://doi.org/10.1016/j.scienta.2021.110869
Goodwin, R. M. & Houten, A. T. Feeding sugar syrup to honey bee (Apis mellifera) colonies to increase kiwifruit (Actinidia deliciosa) pollen collection: effects of frequency, quantity and time of day. J. Apicult. Res. 30(1), 41–49. https://doi.org/10.1080/00218839.1991.11101233 (1991).
Farina, W. M., Palottini, F., Estravis-Barcala, M. C. (2022b). Formulation and composition which promote targeted pollination by bees towards kiwifruit crops and related methods. PCT/IB2022/058287. Patents pending. https://patentscope.wipo.int/search/es/detail.jsf?docId=WO2023031882.
Adams, R. P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry (Allured Publishing Corporation, 2007).
Dobson, H.E.M. (1991). Analysis of flower and pollen volatiles. In Essential oils and waxes (pp. 231–251). Berlin, Heidelberg: Springer Berlin Heidelberg.
Goodwin, R. M. Kiwifruit flowers: Anther dehiscence and daily collection of pollen by honey bees. New Zealand J. Exp. Agric. 14(4), 449–452. https://doi.org/10.1080/03015521.1986.10423064 (1986).
Delaplane, K. S. et al. Standard methods for pollination research with Apis mellifera. J. Apicult. Res. 52(4), 1–28. https://doi.org/10.3896/IBRA.1.52.4.12 (2013).
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.Rproject.org/
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9(2), 378–400 (2017).
Chambers, J.M. (1992) Linear models In: Chambers JM, Hastie TJ (eds) Statistical Models in S. Wadsworth & Brooks/Cole, Belmont
Crawley, M. J. The R Book (Wiley, 2013).
Hartig, F. (2022). _DHARMa: Residual Diagnostics for Hierarchical (Multi-Level / Mixed) Regression Models. R package version 0.4.6. https://CRAN.R-project.org/package=DHARMa.
Lenth, R. (2022). emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.8.1-1. https://CRAN.R-project.org/package=emmeans.
Acknowledgements
Authors are grateful to the commercial orchards ‘El Abrojito’ and ‘Dos Abriles’ for their selfless support at the agricultural setting. Authors also thank the CONICET and the University of Buenos Aires and ANPCYT for financial support. Authors are also grateful to R Okada and two anonymous reviewers for their suggestions during the reviewing process of the manuscript.
Funding
This study was funded by Agencia Nacional de Promoción Científica y Tecnológica (Grant no. PICT 2019 2438), Secretaría de Ciencia y Técnica, Universidad de Buenos Aires (Grant no. UBACYT 2018 20020170100078BA), Consejo Nacional de Investigaciones Científicas y Técnicas (Grant no. PIP 112-201501-00633).
Author information
Authors and Affiliations
Contributions
W.M.F. conceived the research study and was responsible for the project administration and funding acquisition. W.M.F., M.C.E.B. and F.P. designed the experiments. All authors collected the data; M.C.E.B., F.P. and W.M.F. supervised data collection. M.C.E.B., F.P. and A.G. analysed the data. M.C.E.B., F.P. and W.M.F. wrote the manuscript and all authors edited it, and approved the submission.
Corresponding author
Ethics declarations
Competing interests
National Scientific and Technical Research Council of Argentina (CONICET) and the University of Buenos Aires have filed the patent application (PCT/IB2022/058287) on the commercial use of the kiwifruit formulation to improve honey bee pollination efficiency, in which W.M.F., M.C.E.B. and F.P. are coinventors. W.M.F. is coinventor and shareholder of Beeflow Corporation, the licensee of this technology. F.V. and A.G. declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Estravis-Barcala, M.C., Palottini, F., Verellen, F. et al. Sugar-conditioned honey bees can be biased towards a nectarless dioecious crop. Sci Rep 14, 18263 (2024). https://doi.org/10.1038/s41598-024-67917-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-67917-7