Abstract
Competition between motor and declarative memory systems, both involved simultaneously in motor learning, has been shown to reduce motor consolidation. Here, we investigated this conflict during the learning of a sequential finger-tapping task (SFTT) scheduled for either the morning or the afternoon. Sixty participants, divided into four groups, trained on SFTT at either 10 a.m. or 3 p.m., and retested five hours later. To disrupt the conflict between the two memories, two groups underwent declarative learning immediately after SFTT training, involving word list training (G10DL and G3DL), while the two other groups (G10CTR and G3CTR) experienced no additional learning. The results revealed that after morning training without additional learning (C10CTR), skill consolidation deteriorated, while the addition of declarative learning (G10DL) significantly attenuated this decay, stabilizing consolidation. Afternoon training showed skill stabilization for both groups (G3CTR and G3DL). These results suggest that weaker consolidation after morning training may be due to an important competition between motor and declarative memories within the same motor task.
Similar content being viewed by others
Introduction
The acquisition and mastery of motor skills requires practice1. The formation of a varied motor repertoire is developed and maintained over several timescales, ranging from a single session to extended periods, spanning days, weeks, or even months2. Depending on the motor task, a single practice session can lead to rapid enhancements in performance (e.g. accuracy and/or speed)3,4,5 until the emergence of an asymptotic level6. This learning phase, known as motor acquisition, represents the initial stage in the formation of new memories of motor skills (motor memory or procedural memory). After acquisition, the consolidation of motor skill memory is unstable and susceptible to interference7. Retroactive interference, that is the disruption in retention of skill performance caused by the immediate learning of a second task, demonstrates well that a skill memory is initially vulnerable8. Thus, a consolidation period is necessary to transform this initially unstable memory into a robust motor memory9,10. The simple passage of time during the day and/or a period of sleep stabilizes motor memory11,12 or even further improves it, commonly referred to as off-line learning7,13.
Interference in the motor learning process is a well-study phenomenon. Typically, retroactive interference produces partial forgetting14 or limits off-line learning10,15. For instance, the consolidation of a motor sequence, such as a sequential finger-tapping task (SFTT), where participants learn to tap a specific sequence as accurately and fast as possible, is interfered with by the acquisition of a second motor sequence introduced immediately after the practice of the first one15. Retroactive interference can arise not only within the same memory system (i.e., competition between motor skills) but also between different memory systems (i.e., competition between motor and declarative memories, the latter is the conscious memory of events and facts)16. Brown and Robertson (2007) demonstrated that SFTT learning is disrupted by subsequent declarative learning, such as word list training, and vice versa17. Note that in the previous study17, the SFTT task was purely motor, i.e., without any declarative component (implicit learning).
Interestingly, motor and declarative memories can compete within the same motor task. Learning a new musical piece on the piano is a representative example. The pianist must learn the partition and at the same time perform well on the piano keyboard (speed, accuracy, and rhythm). In the laboratory, the SFTT can be designed to involve improvement in both motor execution (i.e., implicit/motor learning) and learning of the sequence order (i.e., explicit/declarative learning). In this case, a competition between the declarative and motor memories affects motor consolidation18,19. Intriguingly, introducing declarative learning (e.g., word list learning) immediately after the SFTT learning can improve skill consolidation20. This is because the declarative learning acts on the declarative component of the SFTT during the consolidation phase, preventing so the conflict between the two memories and allowing thus the development of offline motor learning during the consolidation phase.
Recently, using a SFTT engaging both motor and declarative components (i.e., explicit task), we found that the consolidation, not the acquisition phase, was influenced by the time-of-day in which the practice took place21,22. Precisely, we observed a stabilization and an improvement of motor skill 24 h after afternoon and evening training, respectively, while the motor skill deteriorated after morning training. We propose that this deterioration may occur over time between the morning training and the subsequent night of sleep. Precisely, our investigation aimed to ascertain whether the conflict between motor and declarative memory systems is more present in the morning than in the afternoon. To explore this hypothesis, we employed declarative learning, by using word list training, aimed to disrupt the conflict between the two memory systems20. This learning was administered immediately after SFTT learning, engaging both motor and declarative components, and scheduled either in the morning or the afternoon.
We hypothesized that if the weaker consolidation after morning training is due to a strong conflict between motor and declarative memories within the SFTT, the declarative learning (i.e., word list training) would improve motor consolidation compared to SFTT learning without additional declarative learning. In this case, we also expected no differences in consolidation between morning and afternoon training.
Results
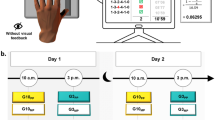
Sixty healthy adults performed a sequential finger-tapping task (SFTT) on a computer keyboard (Fig. 1a). Participants were divided into four groups: two groups underwent a ‘Declarative learning’ session after a ‘Training’ session of SFTT, either at 10 a.m. (G10DL) or at 3 p.m. (G3DL), and two groups without any additional ‘Declarative learning’ after a ‘Training’ session of SFTT, either at 10 a.m. (G10CTR) or at 3 p.m. (G3CTR). Figure 1 illustrates and details the experimental design. Hereafter, we present the main results for the skill, while the separate analysis for speed and accuracy is detailed in the Supplementary information.
Illustration of experimental device and procedure. (a) The sequential finger-tapping task. Each key was affected to a specific finger of the participants’ left hand: 0 (thumb), 1 (index), 2 (middle), 3 (ring), and 4 (little). Participants were instructed to tap the given sequence (1-3-2-4-1-0) as accurately and as fast as possible. Six consecutive sequences composed one trial. Accuracy was quantified by the number of incorrect sequences (Errors) within each single trial. Movement duration (MD) was delineated as the time from the beginning (initial pressure on the key ‘0’) and the end (final pressure on the key '0' after the 6th sequence) of the trial. Motor skill was defined as a composite ratio of duration and accuracy. (b) Experimental procedure. All participants performed a ‘Training’ session at 10 a.m. (G10DL and G10CTR) or at 3 p.m. (G3DL and G3CTR). 'Training' session consisted of 48 trials, organized into 12 blocks of 4 trials each, with a 5-s rest between trials and a 30-s rest between blocks. The pre-test (T1) and post-test (T2) encompassed the first two trials (1 and 2) and the last two trials (47 and 48), respectively. Immediately after the ‘Training’ session, only G10DL and G3DL received a ‘Declarative learning’ session involving the acquisition of a list of 16 words. Each word was presented for 2 s, and participants were subsequently asked to recall as many words as possible. This list and recall procedure was repeated 5 times, with the first recall designated as the pre-test for the declarative learning (T1DL). A free recall was also conducted 10 min later as the post-test for the declarative learning (T2DL). No additional session was presented for G10CTR and G3CTR. Subsequently, 5 h later, all participants performed the ‘Retest’ session comprising two additional trials of finger-tapping task (T3).
Figure 2a shows the average values (+ SD) of motor skill for the four groups (G10DL, G3DL, G10CTR, and G3CTR) and the three sessions (T1, T2, and T3). A repeated measures (rm) ANOVA revealed a significant main effect of session (F2,112 = 306.86, p < 0.001, η2 = 0.85) and an interaction effect group x session (F6,112 = 3.93, p = 0.001, η2 = 0.17) but no significant effect of group (F3,56 = 0.82, p = 0.49, η2 = 0.04). Post-hoc analysis did not show a significant difference between groups in T1 (p > 0.7 in all cases), indicating that the initial level of skill was comparable across groups. Moreover, all Bayesian equivalence tests between groups in T1 demonstrated that the overlapping hypothesis Bayes Factor (BFOH01) was superior to 2.55 and the non-overlapping hypothesis Bayes Factor (BFNOH01) was superior to 2.58. This suggests that the data are 2.6 times more likely to support the null hypothesis than the alternative one and 2.6 times more likely to lie in the equivalence than in the non-equivalence region.
Motor skill performance for the G10DL, G3DL, G10CTR and G3CTR groups. (a) Average values and standard deviations (+ SD) of skill in T1, T2, and T3 for each group. (b) Average values and SD of the percentage of acquisition gain in skill (T1_T2). (c) Average values and SD of the percentage of consolidation gain in skill (T2_T3). Dots represent individual data. Stars indicate significant differences between sessions or groups (✧ p < 0.001; ✦ p < 0.05). Triangles indicate significant differences from the value zero (Δ p < 0.001).
Skill significantly improved after training for all groups (post-hoc comparisons between T1 and T2: p < 0.001). Figure 2b further illustrates this result by depicting the average (+ SD) acquisition gains (T1_T2) in motor skill. The comparison of the T1_T2 gain with the reference value zero (0) confirmed significant enhancement for all groups (G10DL: t = 11.60, p < 0.001, d = 3.00; G3DL: t = 8.05, p < 0.001, d = 2.08; G10CTR: t = 9.77, p < 0.001, d = 2.52; G3CTR: t = 7.48, p < 0.001, d = 1.93). The T1_T2 gain did not differ significantly between groups (one-way ANOVA: F3,56 = 0.27, p = 0.85, η2 = 0.01; Bayesian equivalence tests: BFOH01 > 2.17 and BFNOH01 > 2.19 in all cases). This similar skill improvement during the ‘Training’ session suggests that the acquisition process is independent of the time-of-day, as also indicated in a previous study21.
Notable differences emerged between groups in motor skill consolidation. Specifically, there was a significant deterioration in skill performance between T2 and T3 for the G10CTR (post-hoc analysis: p < 0.001), while there was no significant difference between T2 and T3 for the G10DL, the G3DL, and the G3CTR (for all, post-hoc analysis: p = 0.26). This result indicates a partial forgetting for the G10CTR with the passage of time and consolidation for the other groups. To further refine this finding, Fig. 2c illustrates the average (+ SD) consolidation gains (T2_T3). The comparison of the T2_T3 gain with the reference value zero (0) revealed a significant deterioration for the G10CTR (t = − 5.19, p < 0.001, d = − 1.34; 14/15 participants decreased their performance) and a marginal deterioration for the G10DL (t =− 1.98, p = 0.07, d = − 0.51; 10/15 participants decreased their performance). On the other hand, we observed a stabilization for the G3DL (t = 0.99, p = 0.34, d = 0.26; 8/15 participants increased their performance) and the G3CTR (t = 1.32, p = 0.21, d = 0.34; 6/15 participants increased their performance). These results indicate that declarative learning after SFTT was beneficial for the G10DL group, as this group stabilized skill motor memory in comparison to the G10CTR group. Indeed, one-way ANOVA on T2_T3 gain confirmed these findings by indicating a group effect on the consolidation (F3,56 = 9.75, p < 0.001, η2 = 0.34). The post-hoc analysis showed more deterioration for G10CTR than G10DL (p = 0.02), while no difference was found between G3DL and G3CTR (p = 0.68; Bayesian equivalence tests: BFOH01 = 2.72 and BFNOH01 = 2.77), suggesting that the declarative learning modified the consolidation only after the morning training. It is worth noting that the G10DL exhibited marginally difference with weaker consolidation from G3DL (p = 0.054; Bayesian equivalence tests: BFOH01 = 0.56 and BFNOH01 = 0.56) and G3CTR (p = 0.053; Bayesian equivalence tests: BFOH01 = 0.43 and BFNOH01 = 0.43). Finally, G10CTR showed significant weaker consolidation than G3CTR (p < 0.001), supporting a different effect of simple passage of time on consolidation based on the time-of-day. Taken together, these results suggest that declarative learning limited skill deterioration during the consolidation phase for the morning group, without any effect for the afternoon group.
It is important to emphasize that the different effects of declarative learning on SFTT according to time-of-day are not attributable to differences in the acquisition of the word list. Indeed, Fig. 3 shows the average values (+ SD) of the number of words quoted in T1DL and T2DL. Permutation tests showed a significant improvement in the number of words quoted at the end of learning for both groups (T1DL vs T2DL; T < − 4.80, p < 0.001 in both cases). Furthermore, no significant differences were observed between the groups for either the initial level (T1DL; T = − 0.63, p = 0.59) or the level at the end of learning (T2DL; T = 1.37, p = 0.41).
Discussion
In this study, we investigated to what extent the conflict between motor and declarative memory impacts consolidation during a sequential finger-tapping task (SFTT) according to a morning (10 a.m.) or an afternoon (3 p.m.) training. We showed a deterioration of motor skill (i.e., partial forgetting) 5 h after morning training (G10CTR) compared to its stabilization for afternoon training (G3CTR). Interestingly, declarative learning involving word list training, applied immediately after the acquisition of the SFTT in the morning (G10DL), significantly attenuated the deterioration (i.e., no forgetting) in skill performance normally observed after morning training (see G10CTR). No modulation was induced by the declarative learning on consolidation for the afternoon training (G3DL). It is worth mentioning that these differences in consolidation cannot be attributed to different levels of acquisition since performance either for the SFTT or the word list (declarative learning) was similar according to the time-of-day of practice and groups.
Considering the SFTT learning without additional learning (G10CTR and G3CTR), our results reaffirmed our prior findings, in which we highlighted that the consolidation process varied according to the time-of-day of practice21,22. Specifically, our prior work revealed better skill consolidation, tested after a night of sleep, when training was scheduled in the afternoon (stabilization of performance) and evening (enhancement of performance; i.e., off-line learning), compared to a partial forgetting in memory retention (decline in motor performance) following morning training. Interestingly, in the present study, this pattern remained consistent even when the retention interval was limited exclusively to the wakefulness period. Indeed, when morning and afternoon groups are compared with the same retention interval involving wakefulness without sleep, as in this study, only the morning group exhibited a noticeable forgetting.
Consistently with the existing literature, our study supports that skill consolidation improvement can occur when declarative learning is introduced following a SFTT engaging both motor and declarative components20. However, it is worth mentioning that in our study, the control groups did not perform any additional task, such as a vowel-counting task used in other studies20,23. Thus, we cannot exclude that engaging in any task (other than word-list learning) might be enough to improve this consolidation. Brown et al.20 used a word list training and a vowel counting task after training on a finger-tapping task engaging both motor and declarative memories. They showed enhanced motor consolidation on the finger-tapping task exclusively after the word list training, suggesting that declarative learning (i.e., word list training) disrupts the competition between motor and declarative memories during consolidation.
As described previously, we observed, specifically for morning training, a better consolidation, that is no partial forgetting, in the presence of this declarative learning compared to no additional learning. Following the literature, this phenomenon is explained by the fact that declarative learning (here, a word list) acts on the declarative component of the SFTT, by blocking its interference on the motor memory, releasing thus the development of offline motor learning during the consolidation phase18.
The neurophysiological support that could explain these results may arise from the activation of the dorsolateral prefrontal cortex (DLPFC), which acts as an intermediate structure linking motor and declarative memory systems. The DLPFC plays a crucial role in facilitating the interaction between striatal and hippocampal systems during the acquisition of a SFTT, thereby ensuring the consolidation of learning24,25,26. According to Tunovic et al., the disruption of DLPFC or the activation primary motor cortex (M1), by means of repetitive transcranial magnetic stimulation (rTMS), resulted in improved consolidation during wakefulness, as the declarative learning, indicating that neural network involving these two structures regulates subsequent memory consolidation19. We propose that the activation of this network may exhibit variation throughout the day influencing the conflict between motor and declarative memories, as already demonstrated in other cerebral structures during movement27 or in other networks with a variation of functional connectivity in rest-state28.
It is of interest that the conflict between motor and declarative memory systems arises in the morning but not in the afternoon. Indeed, motor consolidation was stable after afternoon training and declarative learning had no impact on subsequent consolidation (G3CTR and G3int). This result suggests a potentially better resistance to memory conflict in the afternoon compared to the morning. A plausible hypothesis to explain this finding results in the accumulation of physical activity across the day, which can impact processes crucial for motor learning29. Indeed, the longer the duration between waking up and training, the greater the accumulation of daily motor activity (more time when the training took place at 3 p.m. than 10 a.m.). Moreover, several studies highlighted the beneficial effect of physical activity on consolidation, which could create a conducive environment for consolidation by influencing the cellular mechanisms underlying plasticity, as shown with moderate and intense aerobic activities performed prior to learning30. Recent findings by Chen et al. demonstrated that physical activity carried out after learning can reduce the conflict between motor and declarative memories, optimizing the utilization of neural resources23. Overall, these studies prompt us to consider the effects of accumulated daily activities, even at lower intensities than aerobic activities, on further consolidation.
Additional factors could also explain our results, such as circadian cortisol variations. Sale et al. showed plasticity variation in the primary motor cortex (M1) with modulation of corticospinal excitability (CSE) after induction of long-term potentiation (LTP-like)31. Their findings indicated a higher increase of CSE in the afternoon compared to the morning, aligning with the natural diminution of cortisol across the day. Notably, this modulation of CSE post-training can predict further consolidation18,19. Additionally, Dolfen et al. increased the cortisol level by stress induction during acquisition and showed an increase in the recruitment of motor areas and a progressive disengagement of hippocampocortical networks during acquisition, proving unfavorable to consolidation32. We can assume that the naturally high level of cortisol in the morning31 would then have a detrimental impact on the consolidation process. The authors underline, however, that cortisol is not the only factor in brain modulation, since participants who did not respond to stress with increased cortisol also showed these brain modulations.
Overall, we highlighted that the conflict between the motor and declarative memory systems during SFTT learning played a role in the distinct patterns of consolidation observed throughout the day. Although we proposed a few explanatory leads, a comprehensive investigation is necessary, focusing particularly on the modulation of networks associated with motor consolidation. Specifically, delving into the roles of the DLPFC and M1, as well as the interplay between the striatum and hippocampus, according to the time-of-day would provide a deeper understanding of the varying degrees of conflict among different memory systems according to the time-of-day and the specific forgetting observed following morning training. Moreover, it would be interesting in future investigations to compare the specific impact of declarative learning tasks with tasks that do not engage declarative memory. This approach would clarify the specific impact of declarative learning on motor memory consolidation. Additionally, since the majority of participants in the current study had an intermediate chronotype, it could be interesting to expand this research and include individuals with more extreme chronotypes, such as morning or evening. For instance, the elderly population, presenting a more prominent morning chronotype33,34, could be an interesting model to provide a more comprehensive understanding of the real link between chronotype and memory consolidation, and thus, broaden the applicability of our findings.
Methods
Participants
Sixty (n = 60) adults participated in our study after giving their written informed consent. All were right-handed (mean score 0.8 ± 0.2), as determined by their scores on the Edinburgh handedness questionnaire35, and free from any neurological or physical disorder. Musicians and professional typists were intentionally excluded from the experiments due to the specific motor task involving hand dexterity. We included four groups in our experimental protocol: (i) the G10DL group (n = 15, 6 females and 9 males, mean age: 23 ± 7 years old), the participants of this group were trained at 10 a.m. on the sequential finger-tapping task (SFTT) and just after they received a word list training (declarative learning); (ii) the G3DL group (n = 15, 9 females and 6 males, mean age: 23 ± 3 years old), the participants of this group were trained at 3 p.m. on the SFTT and just after they received the same word list training; (iii) G10CTR group (n = 15, 6 females and 9 males, mean age: 24 ± 3 years old), the participants of this group were trained at 10 a.m. on the SFTT only, without any additional training; (iv) the G3CTR group (n = 15, 5 females and 10 males, mean age: 26 ± 6 years old), the participants of this group were trained at 3 p.m. on the SFTT only, without any additional training. The experimental protocol received approval from an ethics committee (Comité de Protection des Personnes—Région EST) and adhered to the standards set by the Declaration of Helsinki.
All participants were explicitly instructed to abstain from consuming drugs or alcohol throughout the entire duration of the experiment and the night preceding it. They were also requested not to alter their regular sleep patterns at home, to engage in their typical daily activities (e.g., cooking, computer use, handicrafts), and to refrain from participating in intensive physical activities during the 24 h leading up to the experiments. All participants adhered to a standard diurnal activity schedule, alternating between 8 a.m. ± 1 h and 12 a.m. ± 1 h, synchronized with the night. In addition, we examined the chronotype and the sleep quality of each participant using the Morningness-Eveningness Questionnaire36 and the Pittsburgh Sleep Quality Index37, respectively. There were no significant differences observed between groups concerning chronotype (one-way ANOVA: F3,56 = 0.55, p = 0.65, η2 = 0.03) and sleep quality (one-way ANOVA: F3,56 = 1.50, p = 0.22, η2 = 0.07). Detailed results from all groups can be found in the Supplementary information in Table S1.
Experimental procedure
All participants were comfortably seated in front of a keyboard. We employed a computerized version of the SFTT, previously used in our studies21,22. Precisely, the participants were instructed to tap as accurately and as fast as possible the following sequence with their non-dominant hand: 1-3-2-4-1-0. A specific finger is affected at each key: 0-thumb, 1-index, 2-middle, 3-ring, and 4-little. A single trial comprised the completion of 6 sequences in succession. At the onset of each trial, participants initiated a chronometer by pressing the key ‘0’ with their thumb. Subsequently, they performed the 6 sequences consecutively. Pressing the key ‘0’ at the end of the 6th sequence stopped the chronometer and marked the end of the trial (Fig. 1a).
All participants completed two sessions (Fig. 1b). During the ‘Training’ session, they carried out 48 trials organized into 12 blocks of 4 trials. There was a 5-s rest interval between individual trials and a 30-s rest interval between blocks. The scores from the first two trials (1 and 2) and the last two trials (47 and 48) constituted the pre-test (T1) and post-test (T2), respectively. The remaining intermediate trials (n = 44; 3–46) constituted the training trials. After a retention interval of 5 h, all participants completed the second 'Retest’ session consisting of two trials (T3).
The groups G10CTR and G3CTR exclusively carried out the ‘Training’ and ‘Retest’ sessions, to examine the effect of the simple passage of time on the acquisition (training) and consolidation (retest) process according to the time-of-day (i.e., acquisition at 10 a.m. or 3 p.m. and consolidation at 3 p.m. or 8 p.m., respectively) (Fig. 1b).
Regarding the two other groups (G10DL and G3DL), ten minutes after the ‘Training’ session, each participant learned (declarative learning) a list of 16 words (from the California Verbal Learning Test French version38,39. Words were presented on a computer screen for 2 s each, with participants recalling as many words as possible after the list completion. Word presentation and recall were repeated five times. The first recall was designated as the pre-test measure (T1DL). Subsequently, participants were prompted to recall the word list once more, 10 min following the last verbal recall, to recall the word list again 10 min after the final verbal recall, constituting the post-test measure (T2DL) to assess the acquisition of the word list. Precisely, we wanted to alter the declarative component of the previously acquired finger sequence learning with the word list training, hypothesizing that this will facilitate motor skill consolidation during the simple passage of time20.
Data recording
Movement accuracy and duration were computed for each trial. The accuracy (‘Error rate’) was defined as the number of false sequences throughout one trial (0 = no false trial; 6 = all trials false). Any trial in which participants made one or more mistakes in a sequence was considered false (Fig. 1a). The error rate was calculated as the percentage of errors relative to the total number of errors during a trial:
Movement duration was defined as the time elapsed (in seconds) from the start of the trial (when the participant pressed the key ‘0’) to the end of the trial (when the participant pressed the key ‘0’ at the end of the 6th sequence).
These two parameters (Movement duration and Error rate) are related by the speed-accuracy tradeoff function40. Consequently, motor skill improvement (i.e., the training-related change in the speed-accuracy trade-off function) is not possible when duration and accuracy change with the same magnitude in opposite directions. For that reason, we calculate a composite ratio (in arbitrary units, a.u) to characterize motor skill as follows21,22,41:
In this formula, skill increases if the duration decreases and/or if the number of errors decreases.
Gains between sessions were calculated following a simple proportional formula:
Statistical analysis
For all data, we verified the normality and sphericity using the Shapiro–Wilk and Mauchly’s tests, respectively, and then applied the appropriate statistical analysis. The significance level was fixed at 0.05 and the observed power was superior to 0.8. We first applied a repeated measures (rm) ANOVA on the skill with group as between-subjects factor (G10DL, G3DL, G10CTR, and G3CTR) and session as within-subjects factor (T1, T2, and T3). Gains in motor skill (T1_T2 and T2_T3) were compared with the reference value ‘zero’ (t-tests). Additionally, we used one-way ANOVA between groups. Post-hoc analyses were carried out using Newman-Keuls tests.
We also performed the same analyses on the movement duration (see Supplementary information). Given that the error rate did not conform to a normal distribution (Shapiro–Wilk test, p < 0.05), we used two-tailed permutation tests (5000 permutations; MATLAB function mult_comp_perm_t1). P values were corrected for multiple comparisons using the Benjamini–Hochberg False Discovery Rate (MATLAB function fdr_bh) (see Supplementary information). The same permutation test was employed for the word list acquisition (T1DL and T2DL) to ensure that both groups acquired the same level of declarative learning.
The ANOVA effect size is expressed in terms of partial eta squared (η2) and is categorized as small (≥ 0.01), moderate (≥ 0.07), or large (≥ 0.14). For t-tests, effect sizes were measured using Cohen's d and classified as small (≥ 0.20), moderate (≥ 0.50), or large (≥ 0.80). In cases where null effects were found, Bayesian equivalence analysis was conducted with a practical equivalence region (ROPE) of [− 0.1, 0.1] and a Cauchy scale before 0.70742. This type of analysis quantifies the evidence in favor of the null hypothesis and discriminates the ‘absence of evidence’ and the ‘evidence of absence’, thereby strengthening and enhancing the reliability of conclusions.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author upon reasonable request.
References
Krakauer, J. W., Hadjiosif, A. M., Xu, J., Wong, A. L. & Haith, A. M. Motor learning. In Comprehensive Physiology Vol. 9 613–663 (Wiley, USA, 2019).
Dayan, E. & Cohen, L. G. Neuroplasticity subserving motor skill learning. Neuron 72, 443–454 (2011).
Karni, A. et al. Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature 377, 155–158 (1995).
Korman, M., Raz, N., Flash, T. & Karni, A. Multiple shifts in the representation of a motor sequence during the acquisition of skilled performance. Proc. Natl. Acad. Sci. 100, 12492–12497 (2003).
Kvint, S. et al. Acquisition and retention of motor sequences: The effects of time of the day and sleep. Arch. Ital. Biol. 149, 303–312 (2011).
Doyon, J. & Benali, H. Reorganization and plasticity in the adult brain during learning of motor skills. Curr. Opin. Neurobiol. 15, 161–167 (2005).
Robertson, E. M., Pascual-Leone, A. & Miall, R. C. Current concepts in procedural consolidation. Nat. Rev. Neurosci. 5, 576–582 (2004).
Robertson, E. M. New insights in human memory interference and consolidation. Curr. Biol. 22, R66–R71 (2012).
Krakauer, J. W. & Shadmehr, R. Consolidation of motor memory. Trends Neurosci. 29, 58–64 (2006).
Korman, M. et al. Daytime sleep condenses the time course of motor memory consolidation. Nat. Neurosci. 10, 1206–1213 (2007).
Walker, M. P., Brakefield, T., Morgan, A., Hobson, J. A. & Stickgold, R. Practice with sleep makes perfect: Sleep-dependent motor skill learning. Neuron 35, 205–211 (2002).
Doyon, J. et al. Contribution of night and day sleep vs. simple passage of time to the consolidation of motor sequence and visuomotor adaptation learning. Exp. Brain Res. 195, 15–26 (2009).
Walker, M. P. A refined model of sleep and the time course of memory formation. Behav. Brain Sci. 28, 51–64 (2005).
Albouy, G. et al. Cerebral activity associated with transient sleep-facilitated reduction in motor memory vulnerability to interference. Sci. Rep. 6, 34948 (2016).
Walker, M. P., Brakefield, T., Allan Hobson, J. & Stickgold, R. Dissociable stages of human memory consolidation and reconsolidation. Nature 425, 616–620 (2003).
Robertson, E. M. From creation to consolidation: A novel framework for memory processing. PLoS Biol. 7, e1000019 (2009).
Brown, R. M. & Robertson, E. M. Off-line processing: Reciprocal interactions between declarative and procedural memories. J. Neurosci. 27, 10468–10475 (2007).
Breton, J. & Robertson, E. M. Flipping the switch: Mechanisms that regulate memory consolidation. Trends Cogn. Sci. 18, 629–634 (2014).
Tunovic, S., Press, D. Z. & Robertson, E. M. A physiological signal that prevents motor skill improvements during consolidation. J. Neurosci. 34, 5302–5310 (2014).
Brown, R. M. & Robertson, E. M. Inducing motor skill improvements with a declarative task. Nat. Neurosci. 10, 148–149 (2007).
Truong, C. et al. Time of day and sleep effects on motor acquisition and consolidation. NPJ Sci. Learn. 8, 1–9 (2023).
Truong, C. et al. Time-of-day effects on skill acquisition and consolidation after physical and mental practices. Sci. Rep. 12, 5933–5933 (2022).
Chen, J., Roig, M. & Wright, D. L. Exercise reduces competition between procedural and declarative memory systems. Eneuro https://doi.org/10.1523/ENEURO.0070-20.2020 (2020).
Albouy, G. et al. Neural correlates of performance variability during motor sequence acquisition. Neuroimage 60, 324–331 (2012).
Albouy, G. et al. Interaction between hippocampal and striatal systems predicts subsequent consolidation of motor sequence memory. PLoS One 8, e59490 (2013).
Freedberg, M., Toader, A. C., Wassermann, E. M. & Voss, J. L. Competitive and cooperative interactions between medial temporal and striatal learning systems. Neuropsychologia 136, 107257 (2020).
Bonzano, L., Roccatagliata, L., Ruggeri, P., Papaxanthis, C. & Bove, M. Frontoparietal cortex and cerebellum contribution to the update of actual and mental motor performance during the day. Sci. Rep. 6, 30126 (2016).
Iester, C. et al. Time-of-day influences resting-state functional cortical connectivity. Front. Neurosci. https://doi.org/10.3389/fnins.2023.1192674 (2023).
Gueugneau, N., Schweighofer, N. & Papaxanthis, C. Daily update of motor predictions by physical activity. Sci. Rep. 5, 17933 (2015).
Wanner, P., Cheng, F.-H. & Steib, S. Effects of acute cardiovascular exercise on motor memory encoding and consolidation: A systematic review with meta-analysis. Neurosci. Biobehav. Rev. 116, 365–381 (2020).
Sale, M. V., Ridding, M. C. & Nordstrom, M. A. Cortisol inhibits neuroplasticity induction in human motor cortex. J. Neurosci. 28, 8285–8293 (2008).
Dolfen, N. et al. Stress modulates the balance between hippocampal and motor networks during motor memory processing. Cereb. Cortex 31, 1365–1382 (2021).
Hicks, H., Meyer, K. & Watts, A. Differential effects of chronotype on physical activity and cognitive performance in older adults. Front. Epidemiol. 3, 1–16 (2023).
May, C. P., Hasher, L. & Stoltzfus, E. R. Optimal time of day and the magnitude of age differences in memory. Psychol. Sci. 4, 326–330 (1993).
Oldfield, R. C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 9, 97–113 (1971).
Horne, J. A. & Ostberg, O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int. J. Chronobiol. 4, 97–110 (1976).
Buysse, D. J., Reynolds, C. F., Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 28, 193–213 (1989).
Nolin, P. Analyses psychométriques de l ’ adaptation française du California Verbal Learning Test. Revue 20, 43–60 (1999).
Delis, D. C., Kramer, J. H., Kaplan, E. & Thompkins, B. A. O. CVLT: California Verbal Learning Test-Adult Version: Manual (Psychological Corporation, 1987).
Shmuelof, L., Krakauer, J. W. & Mazzoni, P. How is a motor skill learned? Change and invariance at the levels of task success and trajectory control. J. Neurophysiol. 108, 578–594 (2012).
Ruffino, C. et al. Acquisition and consolidation processes following motor imagery practice. Sci. Rep. 11, 2295 (2021).
Morey, R. D. & Rouder, J. N. Bayes factor approaches for testing interval null hypotheses. Psychol. Methods 16, 406–419 (2011).
Author information
Authors and Affiliations
Contributions
T.C., P.C. and R.C. designed the experiment; T.C. recorded the data, analysed the data, and developed figures; T.C., P.C., and R.C. wrote the manuscript; all co-authors read and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Truong, C., Papaxanthis, C. & Ruffino, C. Unraveling the time-of-day influences on motor consolidation through the motor-declarative memory conflict. Sci Rep 14, 22195 (2024). https://doi.org/10.1038/s41598-024-69336-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-69336-0
This article is cited by
-
The effects of declarative learning on early and late motor skill learning
npj Science of Learning (2025)