Abstract
This study was conducted to determine the heterogeneity of the quantitative and qualitative properties of fodder growth in cultivated hydroponic wheat fodder (HWF) in the growth tray area and to evaluate the impact on the environment. HWF was grown using nutrient film technique. Yield productivity (YP) of HWF in the growth tray area was divided into four characteristic zones (A, B, C, and D). The most fertile zone A accounted for only 22.3 ± 4.2% of the entire growth tray area, while zone B accounted for 44.7 ± 4.0%. Zones C and D, which accounted for 28.0 ± 1.3% and 5.0 ± 0.3% area, respectively, pose various problems for forage production, i.e., they negatively impact the quantity and quality of HWF, as well as the environment. If all areas in the growth tray support the highest fodder YP (zones A and B), then one kg of dry wheat grains will yield about 6–7 kg of HWF (consisting of 10.7–12.4% dry matter, 17.3–17.5% crude protein, 1.8–2.3% starch, 13.1–14.4% crude fiber, and 4.5–4.6% ether extract). Results of life cycle assessment show that HWF with YPs of 3–5 kg from one kg of dry grains (zones C and D) has the most adverse impact on the environment (150 and 220 kg CO2eq t−1). Under optimum conditions (zone A), CO2eq varied from 94 to 115 kg CO2eq t−1 of feed. Environmentally, HWF production had the most impact on marine aquatic ecotoxicity, abiotic depletion, global warming potential, and freshwater aquatic ecotoxicity.
Similar content being viewed by others
Introduction
The use of traditional agricultural practices for meeting the livestock needs of society is nearing its production limits1 due to the rapidly increasing human population2. Food supply depends not only on the productivity and efficiency of the supply chain, but its environmental sustainability is also particularly important3. Intensive livestock operations on an industrial scale, mostly in industrialized countries but also increasingly in developing countries, are a major source of environmental problems4. Livestock production contributes to greenhouse gas (GHG) emissions consisting of methane, nitrous oxide, and CO25,6,7. Livestock farming emits approximately 7.1 × 109 tonnes of CO2eq GHG per year, which accounts for 18% of the total anthropogenic GHG emissions8. The largest contribution (45%) consists of GHG generated during the animal feed production process9. Therefore, to increase efficiency and reduce the environmental impact of livestock production, new and innovative methods and the application of new technologies are necessary10,11.
Green fodder is an essential component of animal rations to increase the productivity and reproductivity of animals. Therefore, high quality green fodder should be included in the animal feeding rations more often. However, the production of green fodder using traditional agricultural practices is limited by the decreasing land areas for fodder cultivation12, lack of water13, high fertilization costs14. These limitations may be addressed by hydroponic wheat fodder (HWF) cultivation, which can help facilitate the feeding of animals, supplement their diet, and increase the efficiency of the livestock farming. Using the nutrient film technique (NFT)—the most common irrigation fluid supply system, fodder can be grown within 7 days15. In NFT, a stream of water containing dissolved nutrients for plant growth is continuously circulated past the plant roots through a watertight channel, also known as a growth tray16. Different cereal grains such as barley17, maize18,19, and alfalfa20 have already been grown and studied using this system. Recent studies suggest that wheat fodder15,21,22 can be grown this way as well.
Hydroponics, which is performed in controlled environments, refers to a versatile, soilless plant growth system that increases resource use efficiency and autonomy of food production and ensures the reuse of nutrients21,23. This cultivation system can reduce both waste and resources used, as well as the adverse environmental impact of crop production24. Hydroponic systems achieve considerable climate control in modern greenhouses, and they can improve water and nutrient supply by controlling irrigation type and frequency, nutrient concentration, electrical conductivity, pH, and nutrient composition25. The reuse of nutrients allows producers to control resource use, avoid soil and groundwater contamination, and reduce environmental pollution26. Recent research has found that HWF can be grown without the use of any fertilizers or other additives. Growth processes during the first 7 days depend only on the activities of hydrolytic enzymes (mainly α-amylase) that release stored nutrients from the seed during germination15, thus determining the quantity and quality of the hydroponic fodder27. The quantity of hydroponic fodder can be evaluated by several properties: fodder yield per unit area, dry matter content, and the productivity of germinated seeds. There are only two quantitative indicators of hydroponic fodder evaluation: seed productivity and dry matter content. Fazaeli et al.17 found that 6.65 kg of fodder can be obtained from 1 kg of seeds by growing barley hydroponically for 7 days, while maize and wheat grown hydroponically produce 7.47 kg kg−128 and 6.04 kg kg−121 of fodder, respectively.
Researchers agree that the areas of arable land in the world are not homogeneous and the yield and its qualitative properties are often not the same throughout the entire cultivated area29. Although precision agriculture technologies are increasingly being used on farms30, assessing the heterogeneity of qualitative and quantitative properties in the same field is still quite expensive and difficult. The situation is somewhat different for hydroponically grown fodder. There is no evidence that the yield of hydroponically grown fodder is the same throughout the cultivated area, and therefore, its heterogeneity is not evaluated. However, the quality of the fodder growing system can only be determined if the properties of the yield of the cultivated fodder are determined along with their heterogeneity in the cultivated area. The main evaluation criteria for concentrated feed are crude protein, crude fiber, ether extract, and starch content. Similar to the procedure for evaluating quantitative properties, the evaluation of qualitative properties also requires accounting for their heterogeneity in the cultivated area. It is therefore reasonable to hypothesize that the YP of HWF and its qualitative and quantitative properties are not evenly distributed throughout the area of the growth tray.
It should be noted that there is no established relationship between quantitative properties (e.g., productivity of germinated seeds) and environmental impact. There is currently little data to support the use of qualitative and quantitative evaluation criteria to evaluate the efficiency and sustainability of hydroponically grown fodder. Life cycle assessment (LCA) is widely used to assess the environmental impact of any product; it is a global tool that is used to evaluate the environmental impact of a product, process, or activity throughout its life cycle31,32. LCA has been widely used to evaluate agricultural products obtained using conventional methods33,34,35,36. For instance, Jovarauskas et al.37 used LCA to determine the environmental impact of wheat production under various meteorological conditions. The results of LCA can help improve the production, strategic planning, or even policymaking related to the product of concern. The creation and improvement of the legal framework associated with the environmental impact of a product or process is particularly relevant during the current times, when animal husbandry is having an increasing impact on the environment. LCA has been widely used to assess the sustainability of hydroponics for food production. For example, hydroponically grown tomatoes, leafy greens and herbs (e.g., basil, cilantro, mint, and salad), and lettuce were evaluated by Anton et al.38, Martin et al.39, and Romeo et al.40, respectively. In addition, a study evaluated the components of the hydroponic system and determined the environmental impact of different growth substrates41. A recent study in Iran (Isfahan province) assessed the environmental impact of hydroponically grown10. However, similar research on HWF production is still lacking. A complete evaluation of hydroponic fodder should consider its environmental impact, as well as the spatial heterogeneity in the growth area. Therefore, the aim of this study is to determine the spatial heterogeneity of the quantitative and qualitative properties of HWF in the growth tray and to evaluate the environmental impact of such heterogeneity.
Materials and methods
Hydroponic growing stand and set-up of operational parameters
The experiment was conducted from 2021 to 2022 using a self-designed hydroponic growing and analysis stand (Fig. 1) located at the Agricultural Machines Technological Process Research Laboratory of Vytautas Magnus University Agriculture Academy, Lithuania.
General view of the hydroponic growing and analysis stand: 1—irrigation tank; 2—aluminum frame; 3—growth trays; 4—LED lighting; 5—irrigation piping; 6—irrigation fluid inlet, 7—irrigation fluid outlet, 8—air intake system, 9—air extraction system, 10–opaque polycarbonate curtains, 11–main control unit.
The stand was designed with an autonomous monitoring system that was capable of measuring changes in the weight of the cultivated plants within intervals of a few seconds. The stand’s aluminum frame (made from 40 × 40 mm profiles) measured 2150 × 1400 × 1400 mm. The stand was equipped with four polyvinyl chloride (PVC) growth trays (1000 × 225 × 75 mm), each with a growth area of 0.225 m2 and a total growth area of 0.9 m2. One end of each growth tray was open to allow the flow of irrigation fluid through the sprouted seeds (Fig. 2).
The irrigation system provided the nutrients whose composition, rate, and flow could be changed. The stand was also equipped with a microclimate monitoring and control system that controlled the following parameters: concentration of CO2 in the growing environment, temperature, and relative humidity. All of the system’s parameters could be monitored and changed remotely. All collected data were stored digitally.
Lighting and photoperiod
The hydroponic stand was equipped with a GC Bar 8 LED growing light system (Greenception GmbH, Germany) for professional indoor horticulture cultivation with capabilities to produce up to 1500 μmol m−2 s– 1 of luminous flux and able to change the light intensity, spectrum, and duration. This lighting system consisted of eight LED fixtures, i.e., two for each growth tray. The maximum luminous flux was concentrated in the center of the stand. Opaque polycarbonate curtains were used between each growth tray’s LED fixtures to equalize the light flow (Fig. 1). Both the intensity and spectrum of the light were adjustable to suit a particular genotype, growth phase, and growth goals. Compared to other traditional artificial light sources used to grow plants, e.g., fluorescence lighting, LEDs have significant advantages in terms of light conversion efficiency42 and low heat radiation. The GC Bar 8 LED lighting system can be used close to plants without damaging sprouts with heat radiation. In addition, LEDs are available in a variety of wavelengths, allowing light to be optimized to improve plant yield and its quality43,44. Because this study did not aim to assess the effects of lighting on plant productivity and quality, a full-spectrum (400–700 nm) lighting program was chosen to recreate natural sunlight. A luminous flux of 250 μmol m−2 s−1 was selected for each growth tray. A typical 12 h photoperiod45 was used to illuminate the growth trays. At 8:00 AM, the lights were turned on, and at 8:00 PM, they were switched off.
Irrigation and slope angle of the growth trays
It is not necessary to use a nutrient solution for the production of HWF; tap water is sufficient as an irrigation fluid15,46. In this study, we used tap water with an electrical conductivity of 0.20 ± 0.02 mS cm−1. Electrical conductivity was measured using HI98192 portable EC/TDS/Resistivity/Salinity meter (UAB Hanna Instruments Baltics, Lithuania) at an accuracy of 0.01 mS cm−1.
Plants were irrigated 4 times day−1 (starting at 9:00 AM and repeating every 6 h), consisting of 2 min of irrigation at a flow rate of 3.3 l min−115. The irrigation fluid flow rate was controlled using QTDS-100H Handheld ultrasonic water flow meter (Q&T Instrument, China) with an accuracy of ± 1% and set to the required value using hand-operated valves. To minimize the effect of various pathogens on yield and quality of HWF, the irrigation fluid was recycled and was discharged into the sewer.
The growth trays’ slope angle of 6.50% (3.72°)15 was set by using shims (Fig. 2) and a Bosch DNM 60 L electronic spirit level with an accuracy of ± 0.05°.
Microclimate
The plants were cultivated at 21.08 ± 0.13 °C and 49.93 ± 0.41% relative humidity. The concentration of CO2 and the vapor pressure deficit in the hydroponic stand were monitored (Fig. 3), but were neither controlled nor analyzed in this study.
The temperature in the hydroponic stand was monitored, and relative humidity (Fig. 4) controlled to between limits of 40–60% by using the stand’s ventilation system (8 and 9 in Fig. 1). All microclimatic measurements were performed using an IntelliClimate microclimate monitoring and control device (Autogrow Systems Ltd, New Zealand) with accuracies of ± 0.5 °C for temperature and ± 2% for relative humidity.
Seed preparation for cultivation
Winter wheat (Triticum aestivum L.) seeds of the Tobak variety were obtained from a farm located in Šėta, Kėdainiai district, Lithuania. The seeds were cleaned of debris and other foreign matter. Before the experiment, seeds were germinated on moistened tissue paper47, resulting in 95.75 ± 2.39% germination.
Before the seeds to be sprouted were placed in the growth trays of the hydroponic stand, they were first soaked48 for 12 h. The seeds were soaked at room temperature (20.43 ± 0.12 °C) in tap water. After soaking, the weight of the seeds increased by about 50.5%. The trays, PVC pipes, and the irrigation tank were sterilized with sodium hypochlorite (1% of chlorine) solution before each cycle of fodder growth. After sterilizing these components, the entire system was thoroughly rinsed with clean water to remove residual disinfectant solution.
We placed 1.00 ± 0.03 kg of cleaned dry wheat seeds per growth tray (equivalent to 5.46 ± 0.17 kg m−2), which is similar to the 4.5–8.0 kg m−2 seeds used in other studies17,49. The seeds were spread in a uniform layer 3.0 cm high in the growth trays.
Criteria for evaluating HWF
The following quantitative properties were examined in this study: dry matter (DM, %), yield productivity (YP, kg kg−1), and their distribution in the growth tray area. Qualitative properties were crude protein (CP, %), ether extract (EE, %), crude fiber (CF, %), and starch content (SC, %).
Quantitative properties
YP determination The means of yield productivity of the germinated seeds were calculated using the following equation:
where Wt is the total weight of HWF, kg; WS is the weight of soaked wheat seeds that were spread in the growth tray, kg; W is the weight of the empty growth tray, kg; n is the number of repetitions, (n = 4); ki is the coefficient of imbibition, which was calculated as follows:
where WSd is the weight of total dry wheat seeds that were soaked, kg; WSimb is the weight wheat seeds after soaking, kg.
During the growth cycle, the weight (Wt) of each growth tray was measured in 1 Hz frequency using a Flintec PB7.5 (C3 accuracy (0.023%) class) load cells (Flintec Inc, US) (Fig. 2). Sensors signals were amplified using Laumas TLB485 digital-analog weight transmitter (24-bit analog to digital converter, 4.8 kHz) (Laumas, Italy).
Dry matter determination Triplicate 75 × 75 mm samples (80–230 g) were placed in the aluminum dishes and weighed (weighing accuracy of ± 1 mg). The dishes were placed in the oven, dried at 105 °C, and weighed periodically until the sample weight became constant (± 2 mg). The drying time ranged from 2 to 12 h or longer, depending on the moisture of the samples.
After drying, the dishes were removed from the oven, covered, and placed in a desiccator, which was cooled to room temperature. After cooling, the dishes with contents were weighed (weighing accuracy of ± 1 mg). The dry matter (%) of the sample was calculated as follows (LST ISO 712:2010):
where TS1 is the mass of the test sample before drying, g; TS2 is the mass of the test sample after drying, g.
The arithmetic mean was calculated from the results of three samples.
Qualitative properties
Crude protein was determined using the Kjeldahl method (ISO 20483:2006). Crude fiber was determined using the Weender method (ISO 6865:2000). Ether extract was determined using the Soxhlet extraction method (AOAC 920.39-1920/LST ISO 1443), and starch content was determined using the polarimetric method (LST EN ISO 10520:2000).
Modeling of environmental performance of HWF based on factory LCA
The objective of this part of the work was to assess the environmental impact of an insulated 100 m2 building (10 × 10 m) with an HWF production system located in Eastern Europe (Lithuania). This system consists of 10 aluminum frames, each with 5 levels, each level with 4 growth trays (4.0 × 0.225 m). There were 20 growth trays in one frame. The total growth area of the factory is 200 m2. The room was heated using air conditioning, and ventilation was performed using a recuperation system. The irrigation fluid supply system consisted of PVC piping, water pumps, and a tank. The irrigation fluid was not recycled but was simply discharged into the sewer.
The microclimatic conditions (temperature of 21.08 ± 0.13 °C and relative humidity of 49.93 ± 0.41%), slope angle (6.5%), irrigation frequency (4 times day−1 at a flow rate of 3 l min−1 for 2 min) and photoperiod (12–hour photoperiod with luminous flux of 250 μmol m−2 s−1 of full spectrum) corresponds to the conditions we used in our experiment. Modelling of the factory used the results of our HWF yield determination study.
The environmental impact of HWF production was assessed by LCA. The LCA methodology followed the procedure given in ISO 14040/44:200650. The sustainability of HWF production took into account all possible environmental factors affecting the whole life cycle of HWF. The functional unit (FU) was defined as 1000 kg of HWF. The environmental impact assessment was conducted by SimaPro 9.3 process modelling software. The data on raw materials, transportation, and equipment were obtained from the Ecoinvent V3 database51. The environmental performance of the hydroponic production system was determined based on their midpoint impacts (CML-IA baseline V3.06 / EU25). Environmental impacts were characterized as the following: non-fossil fuel abiotic depletion (AD non-fossil fuels, kg Sbeq), fossil fuel abiotic resource depletion (AD, kg Sbeq), global warming potential (GWP, for 100-year time horizon, kg CO2eq), ozone depletion potential (ODP, kg CFC-11eq), human toxicity (HTP, kg 1.4-DBeq), freshwater aquatic ecotoxicity (FAE, kg 1.4-DBeq), marine aquatic ecotoxicity (MAE, kg 1.4-DBeq), terrestrial ecotoxicity (TE, kg kg 1.4-DBeq), photochemical oxidation (PO, kg C2H4eq), acidification potential (AP, kg SO2eq), and eutrophication potential (EP, kg \({\text{PO}}_{4}^{3}\)eq).
Our main goal was to create an environmental model and to assess the environmental impact of an industrial HWF production facility by quantifying all inputs and outputs of material flows (Fig. 5) and assessing how these material flows affect the environment.
The life cycle inventory of hydroponic systems for a 1 month operating period is shown in Table 1.
Statistical analysis
All experiments in this study were repeated four times. Differences among means were compared by the one-way analysis of variance (ANOVA) module of the statistical software Statistica 10.0 and by performing Tukey HSD tests. A probability level of 0.05 was used as the criterion for tests of significance throughout the data analysis52.
Results and discussion
Quantitative and qualitative properties of HWF
Our hypothesis was that the YP of HWF and its qualitative and environmental impact properties are not evenly distributed throughout the area of the growth tray. The results of our experiment confirmed this hypothesis, and it was found that the main reason for the spatial heterogeneity of the above properties is the uneven watering of the seeds due to the increasingly dense root system. Consequently, watering became less and less optimal throughout the entire length of the growth tray during the HWF growth cycle.
Seed productivity in the growth area was divided into four characteristic YP zones (Fig. 6): high (zone A), medium (zone B), low (zone C), and very low (zone D). The most fertile part, where the YP varied from 6.15 to 7.20 kg kg−1 (Fig. 6, zone A), accounted for only 22.3 ± 4.2% of the entire growth tray area (Fig. 7), and is located in the central part of the growth tray, where optimal germination moisture was maintained. Zone B accounted for 44.7 ± 3.97% of the total cultivated area (YP range: 5.10 to 6.15 kg kg−1). The fodder growing in this zone did not show any obvious signs of either a lack or excess access to moisture. However, in this characteristic zone, YP could benefit from optimizing water flow and homogeneity of HWF. Growth zones C and D posed various problems for fodder production, in terms of quality and quantity. Therefore, growers should focus on producing fodder as homogenous as possible across the growth area.
Implementation of the NFT means that irrigation fluid flowed from the highest to the lowest part of the growth tray. Grigas et al.15 found that the slope angle determined the efficiency of seed irrigation. Accordingly, we chose a slope angle of 6.5% of the growth tray to cultivate HWF. However, this slope angle did not produce homogeneous harvest or fodder quality throughout the area of the growth tray. An even distribution of the values of the quantitative and qualitative properties reflects the homogeneity of the cultivated fodder. We noted several problems during the experiment: first, seeds that germinated at the highest point of the tray dried out, while those at the lowest point were soaked. This was due to the increasingly dense root system, which affected the water supply towards the bottom of the growth tray. Additionally, the potential energy of the water that hydrates the seeds decreased toward the bottom of the tray. A given volume or mass of water tends to move from a region of higher potential energy to a region of lower potential energy53.
In the upper part of the growth tray, there were noticeable signs of water stress in the germinating wheat seeds. Under water stress, the low water potential inhibits seed germination54,55. Increasing water stress levels reduced the activities of enzymes, resulting in negative effects on the metabolism of carbohydrates, which prevented water uptake. Moreover, reducing the water potential reduced the levels of necessary nutrients such as calcium and potassium, thus changing the activation of enzymes and seed hormones during the germination phases56. Khayatnezhad et al.57 reported that the decreases in shoot and root length, and in fresh and dry weights due to increasing water stress results in physiological disturbance that reduces the processes of water absorption and plant expansion, resulting in the map shown in Fig. 6.
In the lower part of the growth tray, we observed a similar decrease in seed productivity (Fig. 6), although this decrease was due to anoxia. The immersion of seeds in water directly affects the diffusion of oxygen to plant tissues, thus restricting oxygen exchange and mitochondrial respiration, which had negative effects on the physiological and biochemical processes of the plants58,59,60. Cereals such as barley and wheat are unable to germinate under anoxic conditions61. Moreover, prolonged seed imbibition under submergence induces the leakage of potassium and phosphate, as well as the diffusion of carbohydrates and amino acids, causing seedling starvation and the proliferation of pathogenic microorganisms62. These problems are particularly acute in recirculating irrigation systems. A uniform, high quality harvest throughout the entire growth area of HWF requires solving these cultivation problems.
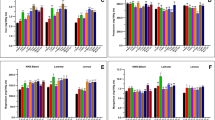
Figure 8 shows that in the lower part of the growth tray, where YP was the lowest and growing conditions were the worst, produced 24.9% of the dry matter (Fig. 6, zone D). According to Fraś et al.63, the dry matter of wheat seeds consists of 60.8% to 67.6% starch and 11.8% to 15.2% crude protein. Furthermore, wheat seeds contain 1.71% of ether extract, 0.41% of sugars64, and 3.4% of crude fiber65. Therefore, dry matter (Fig. 8) is closely related to starch degradation (Fig. 10).
Sugars are a readily available source of energy and are one of the main components needed to maintain metabolism66. However, sugar levels are extremely low in wheat seeds Tacer-Caba et al.64. To replenish the reserves of sugar needed for germination, the starch in the seeds must be hydrolyzed by enzymes such as α-amylase, β-amylase, α-glucosidase, and debranching enzyme. However, only α-amylase plays a major role in starch degradation67. α-Amylase is not produced in anoxia-susceptible cereals, such as wheat and barley66. Therefore, grains immersed in the lower part of the tray lacked both oxygen and germination energy, as starch could not be converted into sugar. In addition, as soon as oxygen availability decreases below a threshold value (usually below 1% O2), sugars such as glucose and sucrose are rapidly channeled to fermentative66.
We conclude that the observed loss of dry matter was mainly due to the degradation of starch (which accounted for almost 3/4 of the total dry matter) via hydrolysis (Fig. 9).
Optimal germination was observed in the central and upper part of the growth tray. Dry matter that was lost during germination was due to the oxidation of starch and saccharides during respiration (Figs. 8, 9), to provide the energy required for grain germination; the CO2 released during the process was easily removed from the grain68. The amount of dry matter decreased (or degraded) not only due to starch hydrolysis, but also due to water imbibition during soaking. Figure 10 clearly shows that dry matter decreased from 20 to 10% as did the starch content (by 70%).
Our results indicate that starch loss is inevitable, even when HWF production is efficient and sustainable. Many studies have shown that starch digestibility increases because of sprouting69, and sprouted cereals are generally more digestible because of their enzymatically damaged starch granules, thin cell walls, and higher content of readily available sugars69. Brown rice sprouted for 2 days at 30 °C and then boiled contains 20% to 30% more rapidly digestible starch than its non-sprouted counterpart70,71.
Figure 8 shows that while the central part of the growth tray had the lowest proportion of dry matter (zones A and B), the absolute quantity of dry matter was still similar to or higher than those in zones B and C (Fig. 6).
Crude fiber makes up about 3.4% of all dry matter in in wheat seeds65. The amount of crude fiber in HWF was significantly higher than that in the seeds (Fig. 11). Similarly, a germinated wheat study showed that at 168 h after germination, the total amount of crude fiber in germinated wheat was higher than that in non-germinated wheat72. Malleshi and Desikachar73 suggested that this increase in crude fiber content is due mainly to the degradation of starch. There is no indication that fiber components such as cellulose and lignin are synthesized from carbohydrates during sprouting74.
The decrease in crude fiber content can be explained by cell wall degradation during sprouting. For example, the endosperm cell wall of sorghum undergoes changes in composition and solubility patterns during sprouting75. In sprouted cereals, the length of the soaking and sprouting periods and the inclusion or exclusion of rootlet and shoots during drying and milling of sprouts also appear to influence the crude fiber content of the resulting meal74.
The causes of crude fiber (Fig. 11) and crude protein heterogeneity in the growth area are similar (Fig. 12). In general, the total crude protein content of cereals changes during germination74,76,77. Compared to seeds, the content and quality of protein of hydroponic fodder are higher, and its amino acid composition is more favorable to livestock78,79,80,81.
The crude protein content of HWF increased due to the loss of dry matter resulting from the hydrolysis and metabolism of carbohydrates and fats as sources of energy for microorganisms82. Even when the crude protein content does not increase, germination improves the biological value of proteins82. The activation of proteases during seed germination breaks down complex proteins into simple peptides, albumins, globulins, and amino acids74. Therefore, the quantity, quality, and composition of crude protein are all very important.
We observed an increase in ether extract after the germination cycle, and their distribution in the growth tray area is shown in Fig. 13. The highest level of ether extract was observed in the center of the growth tray, where growth of HWF was most optimal. At the bottom and top of the tray, the amount of ether extract changed less rapidly due to the slower growth processes. The strong correlation (R2 = 0.79) between the levels of dry matter and ether extract indicates the same (Fig. 14).
The levels of ether extract in cereals increase slightly during the water imbibition stage during soaking, and then later declines during the germination stage, as lipids are used for respiration83. Ether extract levels increase as plants grow because of increases in the levels of structural lipids and chlorophyll. However, ether extract levels do not change during seed germination84,85.
In this study, we observed a strong correlation (R2 = 0.72) between dry matter and crude protein (Fig. 14). Moreover, as the proportion of dry matter decreased from 20 to 10%, the proportions of crude protein and ether extract increased by 6% and 13%, respectively.
In summary, the distributions of yield and the quality of HWF in growth trays indicate that zones C and D (Fig. 6) must be significantly reduced or eliminated altogether. If these zones are eliminated, then one kg of dry wheat grains will be able to produce about 6–7 kg of HWF (consisting of 10.7–12.4% dry matter, 17.3–17.5% crude protein, 1.8–2.3% starch, 13.1–14.4% crude fiber, and 4.5–4.6% ether extract).
Efficient and sustainable fodder production requires an assessment of the impact of HWF production on the environment. In addition, it is important to determine the significance of the heterogeneity of HWF yield on the environment.
Life cycle inventory analysis
During life cycle inventory analysis, we quantified the relevant inputs and outputs of a hydroponic cultivation system consisting of many complex components. We divided the entire life cycle inventory into five main points:
-
1.
Tap water, which is used not for plant irrigation and soaking. Soaking is not carried out on growth trays but in a separate container
-
2.
Electricity for lighting, irrigation, and microclimate management
-
3.
Raw materials (grains and sodium hypochlorite)
-
4.
Even if the required raw materials are stored close to the growth facility, the environmental impact of the equipment used for transporting the raw materials from the supplier to the factory should be assessed
-
5.
The environmental impact of all production equipment was calculated over the entire life cycle of the equipment, because the equipment is not consumed during each production cycle.
The yields after 30 days of cultivation were compared by evaluating different yield levels, i.e., 3, 4, 5, 6, and 7 kg of HWF from 1 kg of dry seeds (Table 1). Each yield level produced 14.27, 19.02, 23.78, 28.54, and 33.29 t of HWF, respectively. The growth cycle and other growth parameters were chosen in a similar manner so that the yield after 30 days of growth could be objectively evaluated. Each yield level differed by the number of seed required to produce one FU. For example, growing HWF at a YP of 3 kg kg−1 will consume ~ 333 kg of dried wheat seeds, while at a YP of 7 kg kg−1 consumes 43% less seeds, i.e., 142.86 kg of seeds.
Environmental impact
Assessment of global warming potential heterogeneity in factory modelling
The first part of this study showed that the YP of fodder and the values of the qualitative and quantitative properties of fodder were not homogenous throughout the area of the growth tray. Similarly, we hypothesize that the environmental impact of different areas of the growth tray will also be heterogeneous. The values of YP obtained in the first part of this study were used to model the global warming potential of a HWF production facility (factory).
We modeled the heterogeneity of the global warming potential of the area of a growth tray (4 m long and 0.225 m wide) (Fig. 15). The tray area supporting optimal fodder growth produced global warming potentials varying from 94 to 105 kg CO2eq t−1 of feed, while in tray areas that did not maintain optimum fodder growth, the global warming potentials were almost twice as high (between 200 and 220 kg CO2eq t−1 of feed).
YP and dry matter are directly related. Therefore, after determining the YP, it is possible to determine the relation between dry matter and global warming potential (Fig. 16). Dry matter and global warming potential are strongly correlated (R2 = 0.80; Fig. 16). Therefore, controlling the level of dry matter in hydroponic systems through changes in technical or design parameters will also have a positive impact on the environment. We conclude that growing HWF under the most optimal conditions will lower the system’s negative impact on the environment.
Because less than optimal conditions have a negative impact on the environment, soaking or drying of seeds in the front and back side of the growth tray, respectively, can almost double the impact on the environment.
Environmental impact assessment of different YP by factory modelling
The hydroponic cultivation of different food crops has already been studied86,87, however, the environmental impact of HWF production has not been well-studied. Our study indicates that HWF yields in growth trays are not as homogeneous as previously thought. The heterogeneous conditions in growth trays pose particularly serious problems not only economically and qualitatively, but also in environmental terms. An initial economic evaluation15 has already been carried out, highlighting the need for further detailed analysis to develop effective solutions. It is therefore essential to assess the environmental impact of HWF grown using current technology and to recommend design or technological changes to improve sustainable fodder production.
Eleven midpoint environmental impacts were characterized using data collected from our experiment. The results of LCA show that HWF with a YP of 3 kg kg−1 (corresponding to Zone D in Fig. 6) has the worst impact on the environment (Table 2). HWF grown under the best growing conditions (corresponding to Zone A in Fig. 6) and had the least environmental impact. However, based on our analysis, the goal should not be to maximize the yield of HWF in certain areas of the growth tray, but rather to make it as uniform as possible.
Results of LCA show not only that 7 kg kg–1 YP (dry matter 11.5%, Fig. 8) has the least overall impact on the environment, based on various impact categories (Table 2) that were changed most significantly. The four main impact categories affected were: marine aquatic ecotoxicity, abiotic depletion (fossil fuels), global warming potential, and freshwater aquatic ecotoxicity.
Table 2 shows that eutrophication, which is a major global environmental problem88, did not change much in response to YP. The reason for this is that no fertilizers were used when growing HWF.
Marine aquatic ecotoxicity is the most important category when assessing the impact on the environment89, and this impact category was affected most by electricity consumption (87.7%), as shown in Table 3. Similar results were obtained from hydroponically grown barley in Iran10. According to the Lithuanian Department of Statistics90, Lithuania depends on energy imports, as domestic production covered only one-quarter of total energy supply (TES) in 2019. Two-thirds of TES comes from oil and natural gas and a quarter from renewables. All ecotoxicity-related impacts (freshwater, marine and terrestrial) are caused mainly by oil and natural gas generated power. This explains why the impact of HWF on marine aquatic ecotoxicity is so high compared to other impact categories. Therefore, it is particularly important to find a balance between fodder production that is both sustainable and efficient.
Electricity consumption in hydroponic systems is not the only impact category that poses a problem for marine aquatic ecotoxicity. More than half (58.6%) of total abiotic depletion is due to electricity consumption and its source (Table 3). Seeds used for germination accounted for 34.2% of total abiotic depletion, and this is mainly due to the energy consumed during their production, mostly from diesel burned in machinery during production. In terms of global warming potential, seed production accounted for half (50.4%) of the total CO2 footprint. Within the seed production CO2 footprint, at least 51.9% was from seeds needed for crop cultivation. Of the remaining footprint, 20.8% was from fuel used for agricultural machinery; 10.5% was from the use of ammonium nitrate; and 4.3% was from the use of complex NPK fertilizers. Seeds for fodder use accounted for 37.4% of the total footprint. Interestingly, 86.1% of the impact on freshwater aquatic ecotoxicity was caused by insecticides. Similar to its impact on marine aquatic ecotoxicity, electricity also had the greatest importance in this category of influence (50.2%).
Our analysis indicates that electricity consumption has the biggest impact on the environment when growing HWF. In the hydroponic system that we analyzed, lighting accounted for 57.1% of electricity consumption. Grigas et al.15 showed that using only indoor fluorescent lamps for the cultivation of HWF can result in a sufficiently high YP (5.7 kg kg−1). Therefore, giving up the lighting on each level of the cultivation, but leaving only the lighting of the growth room, electricity consumption can be reduced by almost 80%. However, the impact of electricity consumption on the environment depends on the main source of electricity generation, which varies from country to country91. Hydroponic fodder production mainly results from electricity which can be decreased using renewable energy technologies10. In Lithuania, renewable energy provides at least a quarter of the country’s electricity90 and therefore, cultivation of HWF is sufficiently sustainable.
We evaluated the environmental impact of one FU of hydroponically grown fodder to that of another concentrated fodder, i.e., wheat seeds (1 FU = 1 tonne). The results (Fig. 17) show that HWF outperforms wheat seeds in three of the four main impact categories.
Notably, the global warming potential (expressed in CO2eq t−1) of HWF is more than four times lower than that of wheat seeds as concentrated feed.
Conclusions
The NFT has many advantages when growing wheat fodder hydroponically. Our results show that seeds germinated at the highest point of the growth tray dry out, while those at the lowest point are soaked. Differences in the potential energy of the water that hydrates the seeds are the main reason for the non-uniform growth of HWF. A uniform, high-quality harvest throughout the entire HWF growth tray requires first solving these cultivation problems.
Our results demonstrate that seed productivity in the growth area is divided into four characteristic yield zones A, B, C, and D. The most fertile zone A accounted for only 22.3 ± 4.2% of the entire growth tray area; and zones B, C, and D accounted for 44.7 ± 4.0%, 28.0 ± 1.3%, and 5.0 ± 0.3% of the growth area, respectively. Zones C and D, which are produced in the upper and lower parts of the growth tray due to uneven watering of the seeds through the NFT, pose various problems for forage production in terms of quantity, quality, and environmental impact. Areas of the tray that are the least fertile, of poor quality, and had the worst impact on the environment must be significantly reduced or eliminated altogether. If these areas are eliminated, then one kg of dry wheat grains (zones A and B) will yield about 6–7 kg of HWF (consisting of 10.7–12.4% dry matter, 17.3–17.5% crude protein, 1.8–2.3% starch, 13.1–14.4% crude fiber, and 4.5–4.6% ether extract). Results of LCA show that HWF with YP of 3–5 kg from one kg of dry grains (zones C and D) has the most adverse impact on the environment (150 and 220 kg CO2eq t−1). In zone A, where the conditions for fodder properties production were the most optimal, CO2eq varied from 94 to 115 kg CO2eq t−1 of feed.
We found that increasing DM from 11 to 13% (zones A and B) to 18–22% (zones C and D) resulted in increases in CO2eq and SC by ~ 20% (from ~ 100 to 120 kg CO2eq t−1 of feed) and 70% (from 1.5% to 4.5%), respectively. In contrast, other qualitative parameters decreased: CF, CP, and EE decreased by ~ 50% (from 15.5 to 7.5%), ~ 6% (from 17.5 to 16.5%), and ~ 13% (from 3.5 to 2.7%), respectively.
HWF yields in the growth area are not as homogeneous as initially believed, which poses particularly serious problems economically, qualitatively, and environmentally. The environmental performance of hydroponic production system was assessed using SimaPro 9.3 process modelling software and the data collected during the experimental part of the study. The results of the LCA show that HWF with YP of 3–5 kg from one kg of dry grains (zones C and D) has the worst impact on the environment, while HWF grown under the best growing conditions had the lowest environmental impact. One tonne of HWF at a YP of 7 kg kg−1 had the most impact on marine aquatic ecotoxicity (41,665.50 1.4-DBeq), fossil fuel abiotic resource depletion (1025.5 MJ), global warming potential (100.68 kg CO2eq), and freshwater aquatic ecotoxicity (26.23 kg 1.4-DBeq). The main source of environmental concern from growing HWF is electricity consumption, which can be reduced by 80% by giving up lighting for every floor of cultivation.
Future research should focus on the influence of the shape of the bottom part of the growth tray on the efficiency of HWF production. After assessing the problem and proposing a solution it is likely that HWF can be produced with uniform quantity and quality, as well as reducing the spread of various pathogens. Also, while our current findings provide valuable insights into the biological and operational aspects, there is a recognized need for a more detailed economic analysis in future research. Specifically, future studies should focus on a comprehensive economic evaluation of the input costs, total yield costs, and the comparative analysis of the cost of wheat seeds (DM basis) against the wheat fodder. Such an analysis will be crucial for assessing the overall economic viability and sustainability of the wheat fodder production system.
Data availability
The authors declare that the data generated or analysed during this study are available within the paper.
References
Bonny, S. P. F., Gardner, G. E., Pethick, D. W. & Hocquette, J.-F. What is artificial meat and what does it mean for the future of the meat industry?. J. Integr. Agric. 14, 255–263. https://doi.org/10.1016/S2095-3119(14)60888-1 (2015).
McGregor, A. & Houston, D. Cattle in the anthropocene: Four propositions. Trans. Inst. Br. Geogr. 43, 3–16. https://doi.org/10.1111/tran.12193 (2018).
Leip, A. et al. Impacts of European livestock production: nitrogen, sulphur, phosphorus and greenhouse gas emissions, land-use, water eutrophication and biodiversity. Environ. Res. Lett. 10, 115004. https://doi.org/10.1088/1748-9326/10/11/115004 (2015).
Alexandratos, N. & Bruinsma, J. World agriculture towards 2030/2050: The 2012 revision. ESA Work. Pap. (2012). https://doi.org/10.22004/ag.econ.288998
Saidul, M., Mukhtar, S., Capareda, S. & Rahm, S. Greenhouse gas emissions from housing and manure management systems at confined livestock operations. In Waste Management—An Integrated Vision (ed. Rebellon, L. F. M.) (InTech, 2012). https://doi.org/10.5772/51175.
Sejian, V. et al. Livestock as sources of greenhouse gases and its significance to climate change. In Greenhouse Gases (eds Moya, B. L. & Pous, J.) (InTech, 2016). https://doi.org/10.5772/62135.
Zhou, J. B., Jiang, M. M. & Chen, G. Q. Estimation of methane and nitrous oxide emission from livestock and poultry in China during 1949–2003. Energy Policy 35, 3759–3767. https://doi.org/10.1016/j.enpol.2007.01.013 (2007).
Opio, C., Gerber, P. & Steinfeld, H. Livestock and the environment: Addressing the consequences of livestock sector growth. Adv. Anim. Biosci. 2, 601–607. https://doi.org/10.1017/S204047001100286X (2011).
Sakadevan, K. & Nguyen, M.-L. Livestock production and its impact on nutrient pollution and greenhouse gas emissions. Adv. Agron. 141, 147–184. https://doi.org/10.1016/bs.agron.2016.10.002 (2017).
Ghasemi-Mobtaker, H., Sharifi, M., Taherzadeh-Shalmaei, N. & Afrasiabi, S. A new method for green forage production: Energy use efficiency and environmental sustainability. J. Clean. Prod. 363, 132562. https://doi.org/10.1016/j.jclepro.2022.132562 (2022).
Mottet, A. et al. Climate change mitigation and productivity gains in livestock supply chains: insights from regional case studies. Reg. Environ. Change 17, 129–141. https://doi.org/10.1007/s10113-016-0986-3 (2017).
Pikaar, I. et al. Decoupling livestock from land use through industrial feed production pathways. Environ. Sci. Technol. 52, 7351–7359. https://doi.org/10.1021/acs.est.8b00216 (2018).
Arnell, N. Climate change and global water resources. Glob. Environ. Change 9, S31–S49. https://doi.org/10.1016/S0959-3780(99)00017-5 (1999).
Adjesiwor, A. T. & Islam, M. A. Rising nitrogen fertilizer prices and projected increase in maize ethanol production: The future of forage production and the potential of legumes in forage production systems. Grassl. Sci. 62, 203–212. https://doi.org/10.1111/grs.12130 (2016).
Grigas, A., Kemzūraitė, A., Steponavičius, D., Steponavičienė, A. & Domeika, R. Impact of slope of growing trays on productivity of wheat green fodder by a nutrient film technique system. Water 12, 3009. https://doi.org/10.3390/w12113009 (2020).
Alipio, M. I., dela Cruz, A. E. M., Doria, J. D. A. & Fruto, R. M. S. On the design of nutrient film technique hydroponics farm for smart agriculture. Eng. Agric. Environ. Food 12, 315–324. https://doi.org/10.1016/j.eaef.2019.02.008 (2019).
Fazaeli, H., Golmohammadi, H. A., Shoayee, A. A., Montajebi, N. & Mosharraf, S. H. Performance of feedlot calves fed hydroponics fodder barley. J. Agric. Sci. Technol. 13, 367–375 (2011).
Jordan-Meille, L. & Pellerin, S. Shoot and root growth of hydroponic maize (Zea mays L.) as influenced by K deficiency. Plant Soil 304, 157–168. https://doi.org/10.1007/s11104-007-9534-8 (2008).
Urbina, M. A., Correa, F., Aburto, F. & Ferrio, J. P. Adsorption of polyethylene microbeads and physiological effects on hydroponic maize. Sci. Total Environ. 741, 140216. https://doi.org/10.1016/j.scitotenv.2020.140216 (2020).
Khorshidi, M. B., Khorshidi, M. B., Yarnia, M. & Hassanpanah, D. Salinity effect on nutrients accumulation in alfalfa shoots in hydroponic condition. J. Food Agric. Environ. 7, 787–790 (2009).
Adekeye, A. B., Onifade, O. S., Amole, G. T., Aderinboye, R. Y. & Jolaoso, O. A. Water use efficiency and fodder yield of maize (Zea mays) and wheat (Triticum aestivum) under hydroponic condition as affected by sources of water and days to harvest. Afr. J. Agric. Res. 16, 909–915. https://doi.org/10.5897/AJAR2019.14503 (2020).
Al-Karaki, G. N. & Al-Hashimi, M. Green fodder production and water use efficiency of some forage crops under hydroponic conditions. ISRN Agron. 1–5, 2012. https://doi.org/10.5402/2012/924672 (2012).
Gruda, N., Bisbis, M. & Tanny, J. Impacts of protected vegetable cultivation on climate change and adaptation strategies for cleaner production - A review. J. Clean. Prod. 225, 324–339. https://doi.org/10.1016/j.jclepro.2019.03.295 (2019).
Folinas, D., Manikas, I. & Manos, B. Traceability data management for food chains. Br. Food J. 108, 622–633. https://doi.org/10.1108/00070700610682319 (2006).
Halbert-Howard, A., Häfner, F., Karlowsky, S., Schwarz, D. & Krause, A. Evaluating recycling fertilizers for tomato cultivation in hydroponics, and their impact on greenhouse gas emissions. Environ. Sci. Pollut. Res. 28, 59284–59303. https://doi.org/10.1007/s11356-020-10461-4 (2021).
Botonaki, A., Polymeros, K., Tsakiridou, E. & Mattas, K. The role of food quality certification on consumers’ food choices. Br. Food J. 108, 77–90. https://doi.org/10.1108/00070700610644906 (2006).
Pourmohammadi, K. & Abedi, E. Hydrolytic enzymes and their directly and indirectly effects on gluten and dough properties: An extensive review. Food Sci. Nutr. 9, 3988–4006. https://doi.org/10.1002/fsn3.2344 (2021).
Kide, W., Desai, B. & Dhekale, J. Feeding effects of maize and barley hydroponic fodder on dry matter intake, nutrient digestibility and body weight gain of Konkan Kanyal goats. Life Sci. Int. Res. J. 2, 96–101 (2015).
Kazlauskas, M., Bručienė, I., Jasinskas, A. & Šarauskis, E. Comparative analysis of energy and GHG emissions using fixed and variable fertilization rates. Agronomy 11, 138. https://doi.org/10.3390/agronomy11010138 (2021).
Monteiro, A., Santos, S. & Gonçalves, P. Precision agriculture for crop and livestock farming - Brief review. Animals 11, 2345. https://doi.org/10.3390/ani11082345 (2021).
Roy, P. et al. A review of life cycle assessment (LCA) on some food products. J. Food Eng. 90, 1–10. https://doi.org/10.1016/j.jfoodeng.2008.06.016 (2009).
Sieverding, H. et al. A life cycle analysis (LCA) primer for the agricultural community. Agron. J. 112, 3788–3807. https://doi.org/10.1002/agj2.20279 (2020).
Alhashim, R., Deepa, R. & Anandhi, A. Environmental impact assessment of agricultural production using LCA: A review. Climate 9, 164. https://doi.org/10.3390/cli9110164 (2021).
Brentrup, F., Küsters, J., Lammel, J., Barraclough, P. & Kuhlmann, H. Environmental impact assessment of agricultural production systems using the life cycle assessment (LCA) methodology II. The application to N fertilizer use in winter wheat production systems. Eur. J. Agron. 20, 265–279. https://doi.org/10.1016/S1161-0301(03)00039-X (2004).
Fallahpour, F., Aminghafouri, A., Ghalegolab Behbahani, A. & Bannayan, M. The environmental impact assessment of wheat and barley production by using life cycle assessment (LCA) methodology. Environ. Dev. Sustain. 14, 979–992. https://doi.org/10.1007/s10668-012-9367-3 (2012).
Holka, M. Life cycle assessment (LCA) of winter wheat in an intensive crop production system in Wielkopolska region (Poland). Appl. Ecol. Environ. Res. 14, 535–545. https://doi.org/10.15666/aeer/1403_535545 (2016).
Jovarauskas, D., Steponavičius, D., Kemzūraitė, A., Zinkevičius, R. & Venslauskas, K. Comparative analysis of the environmental impact of conventional and precision spring wheat fertilization under various meteorological conditions. J. Environ. Manag. 296, 113150. https://doi.org/10.1016/j.jenvman.2021.113150 (2021).
Anton, A., Montero, J. I., Munoz, P. & Castells, F. LCA and tomato production in Mediterranean greenhouses. Int. J. Agric. Resour. Gov. Ecol. 4, 102–112. https://doi.org/10.1504/IJARGE.2005.007192 (2005).
Martin, M., Poulikidou, S. & Molin, E. Exploring the environmental performance of urban symbiosis for vertical hydroponic farming. Sustainability 11, 6724. https://doi.org/10.3390/su11236724 (2019).
Romeo, D., Vea, E. B. & Thomsen, M. Environmental impacts of urban hydroponics in Europe: A case study in Lyon. Procedia CIRP 69, 540–545. https://doi.org/10.1016/j.procir.2017.11.048 (2018).
Vinci, G. & Rapa, M. Hydroponic cultivation: life cycle assessment of substrate choice. Br. Food J. 121, 1801–1812. https://doi.org/10.1108/BFJ-02-2019-0112 (2019).
Cocetta, G. et al. Light use efficiency for vegetables production in protected and indoor environments. Eur. Phys. J. Plus 132, 43. https://doi.org/10.1140/epjp/i2017-11298-x (2017).
Morrow, R. C. LED lighting in horticulture. HortScience 43, 1947–1950. https://doi.org/10.21273/HORTSCI.43.7.1947 (2008).
Yeh, N. & Chung, J.-P. High-brightness LEDs - Energy efficient lighting sources and their potential in indoor plant cultivation. Renew. Sustain. Energy Rev. 13, 2175–2180. https://doi.org/10.1016/j.rser.2009.01.027 (2009).
Harun, A. N., Ahmad, R. & Mohamed, N. Plant growth optimization using variable intensity and Far Red LED treatment in indoor farming, in 2015 International Conference on Smart Sensors and Application (ICSSA), 92–97 (IEEE, 2015). https://doi.org/10.1109/ICSSA.2015.7322517
Dung, D. D., Godwin, I. R. & Nolan, J. V. Nutrient content and in Sacco degradation of hydroponic barley sprouts grown using nutrient solution or tap water. J. Anim. Vet. Adv. 9, 2432–2436. https://doi.org/10.3923/javaa.2010.2432.2436 (2010).
Zeng, Y. J., Qi, X. N., Li, Y. Z., Wang, Y. Q. & Wang, Y. R. Optimising the preparation method of seeds of Agriophyllum squarrosum prior to staining in tetrazolium solution. Seed Sci. Technol. 42, 300–306. https://doi.org/10.15258/sst.2014.42.2.19 (2014).
Koneva, M. S. et al. Optimization of soaking stage in technological process of wheat germination by hydroponic method when objective function is defined implicitly. J. Phys. Conf. Ser. 1015, 032018. https://doi.org/10.1088/1742-6596/1015/3/032018 (2018).
Ajmi, A., Salih, A., Kadim, I. & Othman, Y. Yield and water use efficiency of barley fodder produced under hydroponic system in GCC countries using tertiary treated sewage effluents. J. Phytol. 1, 342–348 (2009).
Mousavi-Avval, S. H. et al. Use of LCA indicators to assess Iranian rapeseed production systems with different residue management practices. Ecol. Indic. 80, 31–39. https://doi.org/10.1016/j.ecolind.2017.04.045 (2017).
Wernet, G. et al. The ecoinvent database version 3 (part I): overview and methodology. Int. J. Life Cycle Assess. 21, 1218–1230. https://doi.org/10.1007/s11367-016-1087-8 (2016).
Olsson, U., Engstrand, U. & Rupšys, P. Statistical Methods SAS and MINITAB (LŽŪU Press Center, 2000). (in Lithuanian).
Campbell, G. S. & Campbell, C. S. Water content and potential, measurement, in Reference Module in Earth Systems and Environmental Sciences. https://doi.org/10.1016/B978-0-12-409548-9.05333-1 (2013).
Jungang, W., Guocang, C. & Chenglie, Z. The effects of water stress on soluble protein content, the activity of SOD, POD and CAT of two ecotypes of reeds (Phragmites communis). Acta Botanica Boreali-Occidentalia Sinica 22, 561–565 (2002).
Shen, L., Orcutt, D. M. & Foster, J. G. Influence of drought on the concentration and distribution of 2,4-diaminobutyric acid and other free amino acids in tissues of flatpea (Lathyrus sylvestris L.). Environ. Exp. Bot. 30, 497–504. https://doi.org/10.1016/0098-8472(90)90030-8 (1990).
Zeid, I. M. & Shedeed, Z. A. Response of alfalfa to putrescine treatment under drought stress. Biol. Plant. 50, 635–640. https://doi.org/10.1007/s10535-006-0099-9 (2006).
Khayatnezhad, M., Gholamin, R., Jamaatie-Somarin, S. H. & Zabihi-Mahmoodabad, R. Effects of peg stress on corn cultivars (Zea mays L.) at germination stage. World Appl. Sci. J. 11, 504–506 (2010).
Liu, Z., Kumari, S., Zhang, L., Zheng, Y. & Ware, D. Characterization of miRNAs in response to short-term waterlogging in three inbred lines of Zea mays. PLoS One 7, e39786. https://doi.org/10.1371/journal.pone.0039786 (2012).
Xu, K. et al. Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature 442, 705–708. https://doi.org/10.1038/nature04920 (2006).
Zhou, W. et al. Plant waterlogging/flooding stress responses: From seed germination to maturation. Plant Physiol. Biochem. 148, 228–236. https://doi.org/10.1016/j.plaphy.2020.01.020 (2020).
Perata, P., Guglielminetti, L. & Alpi, A. Anaerobic carbohydrate metabolism in wheat and barley, two anoxia-intolerant cereal seeds. J. Exp. Bot. 47, 999–1006. https://doi.org/10.1093/jxb/47.8.999 (1996).
Menegus, F., Cattaruzza, L., Mattana, M., Beffagna, N. & Ragg, E. Response to anoxia in rice and wheat seedlings. Plant Physiol. 95, 760–767. https://doi.org/10.1104/pp.95.3.760 (1991).
Fraś, A. et al. Variability in the chemical composition of triticale grain, flour and bread. J. Cereal Sci. 71, 66–72. https://doi.org/10.1016/j.jcs.2016.06.016 (2016).
Tacer-Caba, Z., Nilufer-Erdil, D. & Ai, Y. Chemical composition of cereals and their products. In Handbook of Food Chemistry 301–329 (Springer, 2015).
Ali, H. S., Miah, A. G., Sabuz, S. H., Asaduzzaman, M. & Salma, U. Dietary effects of hydroponic wheat sprouted fodder on growth performance of turkey. Res. Agric. Livest. and Fish. 6, 101–110. https://doi.org/10.3329/ralf.v6i1.41392 (2019).
Loreti, E., Alpi, A. & Perata, P. α-Amylase expression under anoxia in rice seedlings: An update. Russ. J. Plant Physiol. 50, 737–743. https://doi.org/10.1023/B:RUPP.0000003271.64810.16 (2003).
Sun, Z. & Henson, C. A. A quantitative assessment of the importance of barley seed α-amylase, β-amylase, debranching enzyme, and α-glucosidase in starch degradation. Arch. Biochem. Biophys. 284, 298–305. https://doi.org/10.1016/0003-9861(91)90299-X (1991).
Mbithi-Mwikya, S., van Camp, J., Yiru, Y. & Huyghebaert, A. Nutrient and antinutrient changes in finger millet (Eleusine coracan) during sprouting. LWT-Food Sci. Technol. 33, 9–14. https://doi.org/10.1006/fstl.1999.0605 (2000).
Yan, S. et al. Properties of field-sprouted sorghum and its performance in ethanol production. J. Cereal Sci. 51, 374–380. https://doi.org/10.1016/j.jcs.2009.12.006 (2010).
Chung, H.-J., Cho, D.-W., Park, J.-D., Kweon, D.-K. & Lim, S.-T. In vitro starch digestibility and pasting properties of germinated brown rice after hydrothermal treatments. J. Cereal Sci. 56, 451–456. https://doi.org/10.1016/j.jcs.2012.03.010 (2012).
You, S.-Y. et al. Impact of germination on the structures and in vitro digestibility of starch from waxy brown rice. Int. J. Biol. Macromol. 82, 863–870. https://doi.org/10.1016/j.ijbiomac.2015.11.023 (2016).
Koehler, P., Hartmann, G., Wieser, H. & Rychlik, M. Changes of folates, dietary fiber, and proteins in wheat as affected by germination. J. Agric. Food Chem. 55, 4678–4683. https://doi.org/10.1021/jf0633037 (2007).
Malleshi, N. G. & Desikachar, H. S. R. Nutritive value of malted millet flours. Qual. Plant. Plant Foods Hum. Nut. 36, 191–196. https://doi.org/10.1007/BF01092036 (1986).
Chavan, J. K., Kadam, S. S. & Beuchat, L. R. Nutritional improvement of cereals by sprouting. Crit. Rev. Food Sci. Nutr. 28, 401–437. https://doi.org/10.1080/10408398909527508 (1989).
Aisien, A. O. Enzymic modification of sorghum endosperm during seedling growth and malting. J. Sci. Food Agric. 33, 754–759. https://doi.org/10.1002/jsfa.2740330811 (1982).
Hamad, A. M. & Fields, M. L. Evaluation of the protein quality and available lysine of germinated and fermented cereals. J. Food Sci. 44, 456–459. https://doi.org/10.1111/j.1365-2621.1979.tb03811.x (1979).
Muckle, T. B. & Stirling, H. G. Review of the drying of cereals and legumes in the tropics. Trop. Stored Prod. Inform. 22, 11–30 (1971).
Abdallah, M. M. F. Seed sprouts, a pharaoh’s heritage to improve food quality. Arab Univ. J. Agric. Sci. 16, 469–478. https://doi.org/10.21608/ajs.2008.15018 (2008).
Rozan, P., Kuo, Y.-H. & Lambein, F. Amino acids in seeds and seedlings of the genus Lens. Phytochemisty 58, 281–289. https://doi.org/10.1016/S0031-9422(01)00200-X (2001).
Urbano, G. et al. Effects of germination on the composition and nutritive value of proteins in Pisum sativum L.. Food Chem. 93, 671–679. https://doi.org/10.1016/j.foodchem.2004.10.045 (2005).
Wang, K.-H. et al. Germination of peanut kernels to enhance resveratrol biosynthesis and prepare sprouts as a functional vegetable. J. Agric. Food Chem. 53, 242–246. https://doi.org/10.1021/jf048804b (2005).
Nkhata, S. G., Ayua, E., Kamau, E. H. & Shingiro, J.-B. Fermentation and germination improve nutritional value of cereals and legumes through activation of endogenous enzymes. Food Sci. Nutr. 6, 2446–2458. https://doi.org/10.1002/fsn3.846 (2018).
Traoré, T., Mouquet, C., Icard-Vernière, C., Traoré, A. S. & Trèche, S. Changes in nutrient composition, phytate and cyanide contents and α-amylase activity during cereal malting in small production units in Ouagadougou (Burkina Faso). Food Chem. 88, 105–114. https://doi.org/10.1016/j.foodchem.2004.01.032 (2004).
Dhillon, B., Choudhary, G. & Sodhi, N. S. A study on physicochemical, antioxidant and microbial properties of germinated wheat flour and its utilization in breads. J. Food Sci. Technol. 57, 2800–2808. https://doi.org/10.1007/s13197-020-04311-x (2020).
Singkhornart, S. & Ryu, G.-H. Effect of soaking time and steeping temperature on biochemical properties and γ-aminobutyric acid (GABA) content of germinated wheat and barley. Prev. Nutr. Food Sci. 16, 67–73. https://doi.org/10.3746/jfn.2011.16.1.067 (2011).
Chen, P., Zhu, G., Kim, H.-J., Brown, P. B. & Huang, J.-Y. Comparative life cycle assessment of aquaponics and hydroponics in the Midwestern United States. J. Clean. Prod. 275, 122888. https://doi.org/10.1016/j.jclepro.2020.122888 (2020).
Maucieri, C. et al. Life cycle assessment of a micro aquaponic system for educational purposes built using recovered material. J. Clean. Prod. 172, 3119–3127. https://doi.org/10.1016/j.jclepro.2017.11.097 (2018).
Helmes, R. J. K., Huijbregts, M. A. J., Henderson, A. D. & Jolliet, O. Spatially explicit fate factors of phosphorous emissions to freshwater at the global scale. Int. J. Life Cycle Assess. 17, 646–654. https://doi.org/10.1007/s11367-012-0382-2 (2012).
Ferreira, J., Esteves, B., Cruz-Lopes, L., Evtuguin, D. V. & Domingos, I. Environmental advantages through producing energy from grape stalk pellets instead of wood pellets and other sources. Int. J. Environ. Stud. 75, 812–826. https://doi.org/10.1080/00207233.2018.1446646 (2018).
IEA. IEA World Energy Statistics and Balances (accessed 8 January 2020); www.iea.org/statistics (2019).
Renou, S., Thomas, J. S., Aoustin, E. & Pons, M. N. Influence of impact assessment methods in wastewater treatment LCA. J. Clean. Prod. 16, 1098–1105. https://doi.org/10.1016/j.jclepro.2007.06.003 (2008).
Author information
Authors and Affiliations
Contributions
A.G.: Methodology, Investigation, Visualization, Writing–original draft; D.S.: Writing–original draft, Writing–review & editing, Supervision; A.K.: Writing–original draft, Writing–review and editing; Ž.T.: Methodology, Writing–review and editing; R.D.: Formal analysis, Writing–review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Grigas, A., Steponavičius, D., Kemzūraitė, A. et al. Spatial heterogeneity in the properties of hydroponic wheat fodder and its sustainability. Sci Rep 14, 19312 (2024). https://doi.org/10.1038/s41598-024-70128-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70128-9