Abstract
The primary cause of anemia worldwide is due to poor diet and iron deficiency. Iron (Fe) enriched yeast can be the most effective way to manage anemia because of the capability for biotransformation of mineral to organic and bioavailable iron. To overcome the low richness of yeast, the use of siderophore as cellular iron carriers is a new approach. In this research, for the first time the potential of siderophore in increasing the Fe enrichment of Saccharomyces boulardii (S. boulardii), which is important because of its probiotic properties and resistance to different stresses, has been investigated to produce of potential iron supplements. For this purpose, siderophore was produced by Pseudomonas aeruginosa (P. aeruginosa). Siderophore impact, along with ten other independent process variables, has been studied on the efficiency of iron biotransformation by the Plackett–Burman design (PBD). The results showed that the highest biotransformation yield was 17.77 mg Fe/g dry cell weight (DCW) in the highest biomass weight of 9 g/l. Iron concentration is the most important variable, with contributions of 46% and 70.79% for biomass weight and biotransformation, respectively, followed by fermentation time, agitation speed, and KH2PO4 concentration. But increasing the level of siderophore and zinc led to a significant negative effect. siderophore inefficiency may be attributed to the absence of membrane receptors for pyoverdine (Pvd) and pyochelin (Pch) siderophores. Also, the steric hindrance of the cell wall mannan, the stickiness and sediment ability of the yeast, can create limitations in the absorption of elements. Such yeast can be used as a potential source of iron even for vegetarians and vegans in the form of medicinal and fortified food products to improve the treatment of anemia. It is recommended that further research be focused on increasing the iron enrichment of yeast by overcoming the structural barrier of the cell wall, investigating factors affecting membrane permeability and iron transport potential of other types of siderophores.
Similar content being viewed by others
Introduction
Iron is an essential element for life and plays a crucial role in various physiological processes. Its deficiency can lead to health consequences and must be supplied through the diet1,2. However, dietary iron intake alone often fails to meet the needs of specific groups such as pregnant women and children, making additional supplementation necessary3. Global studies have demonstrated that supplements and foods fortified with mineral iron can effectively control anemia in developed countries4,5. Despite these efforts, anemia and iron deficiency due to less solubility and bioavailability of iron salt is still a health concern. Consequently, there is a continuous need to explore innovative strategies to improve the efficacy of iron supplementation to address this issue.
Biological systems convert inorganic iron into an organic absorbable form with lower toxicity potential, which is the most appropriate nutritional form of iron6. As a result, organic iron production through biotransformation of biological sources is a promising method to overcome the problems of inorganic iron, manage anemia and improve health. Biotransformation in microorganisms, which is the most useful biological source compared to other alternatives, occurs by absorbing mineral elements under controlled conditions and combining them with cellular macromolecules7,8.
Health-promoting Saccharomyces with its high nutritional value and wide application in fermented products offers a nutritional approach to enrich foods with essential metals, e.g. Fe9,10, selenium (Se), and zinc (Zn). Studies have shown that iron enriched yeasts have proven more effective than salts in controlling anemia7,11, even may be the most suitable source of iron for vegetarians and vegans. S. boulardii besides its probiotic properties12 because of its cell wall structure, compared to Saccharomyces cerevisiae (S. cerevisiae), has a higher resistance to various stresses like acids and enzymes13 as a result preventing problems caused by the release of iron and protecting it until intestinal absorption12. Also, resistance to ethanol14 and temperature up to 60 °C15, which may lead to increased survival of iron during fermentation and thermal cooking processes.
The yeast enrichment depends on factors such as growth conditions and genetic patterns16, which can be increased by optimizing the culture medium and using iron-sensitive strains compared to resistant isolates. Furthermore, one significant obstacle in this process is the limited permeability of the yeast cell wall, which can hinder iron uptake. Recently, various strategies have been employed to overcome this issue, including protoplasts creation8, utilizing electric fields17,18, and siderophores7.
Siderophores are microbial secondary metabolites that play a crucial role in metal acquisition, particularly iron and form relatively stable complex with them. These metabolites are produced under iron-restricted conditions to transport Fe from the medium into cells19,20. This property makes them a promising method for increasing iron enrichment in yeast. Siderophore-mediated metal transport requires specific membrane proteins, which are different based on the type of siderophore21. Therefore, the effectiveness of a siderophore depends on the presence of the desired receptor for that siderophore in the target cell (Fig. 1).
Zhang et al.7, have demonstrated the potential of siderophores in enhancing intracellular iron content and affecting yeast growth. Yun et al.22, found that the expression of siderophore receptors in S. cerevisiae under iron deprivation conditions further supports their role in iron uptake. Jalal et al.23’s study showed the successful transfer of Fe (III) to various fungal species using labeled siderophores and their potential for increasing iron enrichment in yeasts. Studies have also shown iron enriched yeast with optimal concentrations maintains its fermentation power24 and exhibits better baking properties compared to the mineral form25, it is a suitable option for enhancing the iron content of bakery products. However, excessive enrichment may decrease fermentation activity17 and limit its application to medicinal supplements.
PBD is a two-level fractional factorial matrix that investigates “n” variables with at least experiments “n + 1”. Compared to other strategies, this technique is an uncomplicated and fast method, particularly useful when researchers face multiple independent variables and are unsure which are most significant26.
In this research, for the first time, the potential of effective siderophores in facilitating cellular iron absorption on increasing the iron enrichment of S. boulardii, an economic probiotic, was investigated. Our aim was to produce a potentially health-promoting biomass rich in bioavailable organic iron as a practical supplement to replace iron salts and improve the treatment of anemia. To achieve this goal, siderophore was extracted from P. aeruginosa, a microorganism known for its high siderophore production potential. Also PBD was used as a statistical experimental design to screen eleven variables and identify the most effective ones on iron biotransformation and biomass weight of the selected strain.
Material and method
Chemicals and media
All chemicals and Yeast Peptone Dextrose (YPD) used in this study were of analytical grade and purchased from Merck (German Company) with some exceptions; S. boulardii (ATCC 74068) from the Mycology Research Center at the Faculty of Veterinary Medicine, University of Tehran; P. aeruginosa (ATCC 10828) from the Iranian Biological Resource Center (IBRC); and Deferoxamine mesylate (DFO) from Ronak Pharmaceutical Co.
Siderophore production
P. aeruginosa seed liquid preparation
P. aeruginosa was activated in Luria–Bertani (LB) agar (g/l: 5 yeast extract, 10 NaCl, 10 Tryptone, 15 agar) at 37 °C for 24 h. Then a single colony was transferred to the LB and incubated overnight under 37 °C, 200 rpm conditions. Inoculum was prepared in 0.5 McFarland standard (1.5 × 108 CFU/ml).
Siderophore qualitative detection
The amount of total siderophore secretion was measured by the Chrome azurol S (CAS) method, according to Schwyn and Neilands27 modified by Lakshmanan et al.28 method. Briefly, CAS-Fe complex was prepared by adding 10 ml Iron III solution (1 mM FeCl3∙6H2O + 10 mM HCl) to 50 ml CAS solution (60.5 mg CAS dissolved in 50 ml water) slowly and under stirring conditions. Prepared complex was added to 40 ml Cetyltrimethylammonium bromide (HDTMA) solution (72.9 mg HDTMA dissolved in 40 ml water) and autoclaved with glass beads as CAS dye solution. The final solution was added to 900 ml sterile LB agar medium (pH 6.8). 100 ul P. aeruginosa seed liquid was inoculated in the center well of the CAS agar plate and incubated at 37 °C for 6 days.
Siderophore production in broth culture
The siderophore was produced in a medium with adequate low iron according to Schwyn and Neilands27 method with modification on culture media. Briefly, 10 mM FeSO4∙7H2O was added to 10 ml sterile LB at room temperature through a membrane filter 0.22 um. Then 100 ul bacterial seed liquid was inoculated and incubated overnight at 37 °C and 200 rpm. 100 ul of prepared preculture was added to the same medium and incubated for 6 days (37 °C and 200 rpm). In the following, the culture was centrifuged (4500 × g, 10 min), and the supernatant was stored at – 20 °C until colorimetric testing.
Siderophore quantitative determination
Total siderophore was measured by CAS assay according to Schwyn and Neilands27. Briefly, 1.5 ml iron III solution (1 mM FeCl3∙6H2O + 10 mM HCl) was slowly added along the glass wall to 7.5 ml CAS dye solution 2 mM under stirring. Prepared CAS-Fe complex and buffer solution (4.307 g PIPES, adjusted at pH 5.6 by HCl) was added to 6 ml HDTMA 10 mM then was reached to 100 ml volume and autoclaved as CAS assay solution. Shuttle solution (0.5 ml CAS assay solution + 10 ul 5-sulfosalicylic acid 4 mM) added to 0.5 ml of both supernatant (sample) and same medium without inoculum (reference). In the presence of siderophore, the blue color of the solution decreases after 1 h incubation at room temperature. The absorbance (A) was measured at 630 nm (culture medium without inoculation was considered as blank). Total siderophore absorbance was calculated by the following formula:
Catechol and hydroxamate siderophore types were measured based on chemical properties. Catechol was measured according to the Arnow assay29. Briefly, three solutions of 1 ml HCl 0.5 N, 1 ml nitrite molybdate reagent (10 g sodium nitrite and 10 g sodium molybdate dissolved in 100 ml water) and 1 ml NaOH 1 N, were added to 1 ml supernatant respectively and vortexed at each stage. The final solution was incubated for 5 min at room temperature and the absorbance was read at 510 nm. Hydroxamate was measured according to the Csaky assay30. Briefly, The following solutions were added to 200 ul supernatant, in order: 200 ul H2SO4 6N (the solution was placed in a hot water bath for 6 h to hydrolyze siderophores), 600 ul buffer solution of sodium acetate 35%, 200 ul of sulfanilic acid solution (1 g sulfanilic acid dissolved in 100 ml acetic acid 30% v/v in a hot water bath), 100 ul iodine solution (1.3 g iodine dissolved in 100 ml glacial acetic acid), 200 ul sodium arsenite solution 2% v/v (incubated for 3–5 min at room temperature to disappear excess iodine) and 200 ul alpha-naphthylamine solution (3 g alpha-naphthylamine dissolved in 1 L acetic acid 30% v/v), finally the volume reached to 2 ml with distilled water. For color formation, the solution was incubated at room temperature for 20–30 min, then the absorbance was read at 526 nm. DFO, 2,3-dihydroxybenzoic acid (2,3-DHBA) and hydroxylamine hydrochloride were used as standards to detect total, catechol-type, and hydroxamate-type of siderophores, respectively.
Yeast preculture and Inoculum preparation
Yeast was pre-cultured based on Malairuang et al.31 with some modifications as described elsewhere32. Briefly, S. boulardii after activation was incubated in YPD broth for one night at 37 °C and 150 rpm.
Measurement of optical density (OD), DCW, and cell number of yeast for calibration curves
The grown yeast was centrifuged (4500 × g, 10 min) and serial dilution was prepared from the cell suspension. The predefined volume of each dilution: (1) was counted (CFU/ml) using a Thoma hemocytometer (Neubaur chamber)33 on a microscope 400 × (CETI; Medline Scientific Limited, Oxfordshire UK). (2) Was read at OD600 nm by spectrophotometer (PerkinElmer, USA). (3) Was centrifuged in the same condition and pellets dried until reaching a constant weight. (OD and DCW were measured based on Malairuang et al.31 with modifications on centrifuge speed as described on Tafazzoli et al.32).
Yeast growth curve
The cell growth curve was prepared according to Olivares-Marin et al.34 with modifications as described elsewhere32. Briefly, several YPD containing 10% v/v seed liquid were prepared and an aliquot of each media was centrifuged (1500 × g, 5 min) at specified intervals of incubation (37 °C, 150 rpm), then the OD was measured at λ 600 nm.
Statistical screening of the variables influencing the responses by S. boulardii using the Plackett–Burman matrix
In the current work, by reviewing the prior research (Table 1), eleven independent variables were chosen and coded at two levels high (+ 1) and low (− 1). Then they were screened by PBD in 12 runs with the aim of selecting the effective ones on S. boulardii iron biotransformation and biomass weight. According to the Table 2, the medium containing carbon, tween 80, MgCl2∙6H2O, KH2PO4, ZnSO4, NH4Cl and peptone was adjusted at pH 5.5 by HCl and autoclaved (121 °C, 15 min). Seed liquid volume included 13 × 105 CFU/ml S. boulardii was inoculated to the medium and incubated. FeSO4 and siderophore were added at the beginning of the logarithmic phase to increase iron enrichment. The connection between the independent factors and the result to predict the best response was modeled using the first-order polynomial Eq. (1):
where Y is the response, β0 is the coefficient of the model intercept, βi is the linear coefficient and Xi is the independent factors levels.
Dry weight of iron enriched biomass
CDW was calculation based on Gaensly et al.38 with modifications on centrifuge conditions as described on Tafazzoli et al.32. Briefly 10 ml of the fermented medium was centrifuged (4500 × g, 10 min and 4 °C) to remove medium and free iron. Pellets were dried at 60 °C until they reached a constant weight (g/l).
Determination of organic Fe content
The organic Fe content obtain by subtracting the mineral from the total, which were prepared base on Esmaeili et al.39 with some modifications as described elsewhere32. Briefly: To measure total Fe, 20 mg of biomass powder was digested by nitric acid 65–68% v/v and HCl 37% in heated. To determine inorganic Fe, 100 mg of biomass powder was resuspended, boiled for 1 h and centrifuged (8000 × g, 15 min), supernatant containing mineral iron. Prepared Fe solutions, passed through a 0.45u filter and stored in the freezer until the Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) (VISTA-PRO, USA) testing.
Statistical analysis
Design Expert software version 11 (Stat-Ease Inc., USA) created experimental matrices and analyzed them with an analysis of variance (ANOVA). All experiments were performed in random order, and each response was a repetition average of each experiment. The calculations and graphs were performed using Excel 2016. Also, graphical abstracts were designed by BioRender application.
Results and discussion
Produced siderophore investigation
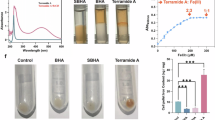
Siderophore was secreted from P. aeruginosa inoculated in the central well of blue agar medium containing the ternary complex indicator Fe-CAS-HDTMA in the form of a circular halo27. Due to the siderophore’s higher affinity for iron than the CAS metal chelator, breaking the complex changed the blue color of the medium to yellow-orange. The color change confirms siderophore secretion qualitatively (Fig. 2).
P. aeruginosa secreted a total of 5 µg/ml siderophore in the low-iron broth medium, while catechol and hydroxamate types were not detected. Standard serial dilutions and calibration curves for the investigated siderophore types were plotted on Fig. 3.
Examination of DCW, OD and cell number of growing yeast over time
Yeast calibration curve of S. boulardii based on OD, DCW and cell count was drawn in Fig. 4. Also, the growth curve (Fig. 5) showed that the yeast entered the growth phase at 11 h after inoculation and the maximum growth was observed at approximately 30 h, as explained elsewhere32.
Screening prosses of factors affecting on responses using PBD
Twelve runs were conducted for statistical screening of eleven independent variables and identifying the most effective ones on responses (Table 3). The results showed, the increase of iron in the culture media caused a significant positive effect in responses. The optimization process importance of other factors is demonstrated with noticeable variation in findings 19.73–160.12 mg/ml in biotransformation and 0.2–9 g/l in biomass weight, in the fixed iron range of 8 mg (Fig. 6). It was also observed that the color of dried biomass with higher iron content (more than 70 mg/l) is obviously darker than low-enriched, as Nowosad and Sujka18 has also mentioned this concept.
Although this study deals with the potential of Fe accumulation in S. boulardii for the first time, this potential for other elements has also been less explored. Research has shown this yeast has limited richness, as it was observed in Nie et al.40; Kitamura et al.41; González-Salitre et al.42, studies about Se accumulation. Hyrslova et al.43 also reported in optimal conditions, Se enrichment was low (0.2 mg/g DCW). This was in the case that other species, even S. cerevisiae, faced a greater accumulation of elements (Table 1). As in Hyrslova et al.43 study, the process of increasing enrichment with extended cultivation time in S. cerevisiae was not repeated for S. boulardii. In addition to the observed resistance to the accumulation of elements, S. boulardii also has a more resistance to different stresses than other species, which may be related to the hard wall structure. Despite the genomic relatedness, S. boulardii has greater overall cell wall thickness13 with a distinct oligosaccharide composition such as a higher mannan content compared to S. cerevisiae44. As the cell wall structure is shown in Fig. 7, the arrangement of glucans as polymer chains creates a space that leads to permeability and allows the diffusion of essential molecules. But increased monoproteins and their related mannans, by increasing the steric hindrance, may reduce the possibility of surface adsorption of elements45,46.
Also, S. boulardii has shown a high adhesion ability due to the special structure of the wall. Hyrslova et al.43 found the yeast adhesion and sedimentation rates have a positive correlation with the cell wall mannan content, as a result, S. boulardii showed a higher adhesion capacity than S. cerevisiae44. This it’s very important for colonization and sticking to the cells of the intestinal tract and as a result the survival and application of probiotic properties. Their studies also showed that adhesion ability is closely related to the hydrophobicity of the cell surface43. As a result, the hydrophobicity, adhesion, and sedimentability of S. boulardii may interfere with the absorption of elements13, which can also be a justification for the low enrichment capacity of S. boulardii.
Yeast extract was one of the culture ingredients, which we encountered with culture precipitation and turbidity. The reason is the presence of ZnSO4 and conventional yeast extract together. They are also problematic in beverages47. As a result, yeast extract was removed to prepare a rich medium. The causes of this phenomenon can be: (1) Zn precipitation due to CaO in yeast extract. The alkaline agents such as calcium hydroxide or oxide can lead to the precipitation of zinc sulfate solution salt in the form of zinc oxide or hydroxide chalk crystals in an acidic solution48. (2) Precipitation of yeast extract proteins with Zn49.
Subsequently, precipitation was again created after adjusting pH by NaOH, which the reason is MgCl2. MgCl2 produces solid Mg (OH)2 during the reaction with some alkaline compounds [MgCl2 + NaOH → NaCl + Mg (OH)2]. To solve the problem, all medium's pH was raised above 5.5 by an equal amount of peptone, then was adjusted with HCl.
Moreover, when adding blueish-greenish FeSO4 solution to medium, encountered yellow color. It is due to chlorine produced from ammonium chloride [2NH4Cl → 2NH4 + Cl2] and HCl, which led to the conversion of ferrous sulphate to ferric chloride [2FeSO4 + 4HCl + Cl2 → 2H2SO4 + 2FeCl3]. After filtration, the blue color of the media returned and calculations showed that the amount of FeSO4 lost was not significant and can be ignored.
The estimated effect of variables
Table 4 and Fig.8, illustrate the estimated effect of variables on responses. A high estimated effect (positive or negative) has a significant, while low effects are ineffective, also the signs of the variables indicate the type of effect. The contribution percentage and T-stat of |effect| indicate the factor importance regardless of a sign type, unlike the regression coefficient and main effect. Main effect (Fig. 8A,B) enables the influence estimation of each factor on responses. Moreover, the Pareto chart (Fig. 8C,D) shows the factors affecting response in relative importance order. This chart reproduces the relation between t-value (the absolute values of the estimated effects divided by the standard error) vs. ranks. As well, it draws two horizontal reference lines on the chart. Bonferroni limit is threshold of the significant variables means any effect that extends from that is potentially important. While the Effects that lie between these lines are considered moderately important50.
The above results indicate that six of the eleven variables have a major influence on responses. Moreover, iron concentration emerged as the most significant with a positive effect. Iron’s main effect, coefficient estimate and contribution percentage are 4.2, 2.10, 46 for biomass weight and 97.76, 48.88, 70.79 for biotransformation, respectively, followed by fermentation time, agitation speed and KH2PO4 concentration.
The significant effect of iron in stimulating cell growth can be due to the vital role of this micronutrient in many biological processes such as carbon pathways and cellular respiration51. The constituent elements of KH2PO4 also play an important role in metabolic pathways and cellular structures for example, phosphate is a cofactor required by some enzymes52 and can lead to improved growth by affecting enzyme activity and metabolism. On the other hand, iron and KH2PO4 can increase the solubility of iron (Fe2+) by stimulating cell growth and as a result of the rapid reduction of the initial pH of fermentation caused by the excretion of acidic compounds, and indirectly contribute to further enrichment of iron and cell growth53.
A longer cultivation period had a significant effect on growth and increasing organic iron content, as Hyrslova et al.43 also, reported on selenium uptake in S. boulardii. Also the agitation speed can be effective in increasing the enrichment by increasing the probability of passing through the mannan filaments and iron hitting the cell surface.
In addition, siderophore concentration with an enormous difference, followed by ZnSO4, had a major negative effect on the result. This finding was contradicts the studies conducted by Zhang et al.7, Yun et al.22, Jalal et al.23, which demonstrated the potent potential of siderophore for clearing iron storage in media and transferring it to cells. Probably, since each siderophore has a unique membrane receptor21, the inability of the complex to transport iron may be due to the absence of P. aeruginosa siderophore mainly Pvd and Pch receptors, in S. boulardii54. As a result, by increasing the concentration of siderophore, more iron was trapped in the media, and the possibility of free iron entering the cell is also reduced, preventing iron enrichment and biomass growth. A negative effect of high Zn concentration on yeast growth and fermentation activity was also observed in Sheykhi et al.55, Shet et al.56, Liu et al.57 studies, which may be due to toxicity and reduced cell division.
In this study, surfactant had a positive but negligible effect on growth and enrichment. Many studies have shown that Tween 80 is effective in increasing microbial biosynthesis58,59,60,61,62,63,64 and biomass growth58,61, but their mechanism of action is not completely clear. As in Cui et al.65 research, Tween 80 formed hydrophobic micelles with iron, here too, the positive effect of Tween 80 on the responses is probably due to facilitating the transfer of iron enclosed in micelles with external non-polar groups, from hydrophobic mannans of yeast. Like Xu et al.66 studies, that by dissolving the desired compound in micelles of Tween 80, they succeeded in greatly improving the bioavailability and as a result, the biotransformation of the compound in the microorganism. The insignificant effect of the surfactant on the responses can be caused by the low concentration of Tween 80. However, the high concentration of Tween 80 limits the transfer of substances to microorganisms by creating a more compact micellar structure with an inhibitory effect on surfactant biodegradability66,67,68. Also, Tween 80 may have increased the absorption of nutrients and cell growth by increasing the homogeneity of the culture medium and membrane permeability. As Zhang and Cheung58, Zhang et al.59, Liang et al.60, Li et al.61, Sheng et al.62 and Yin et al.64 reported that Tween 80, by integrating into the cell membrane, increased the ratio of unsaturated to saturated fatty acids and consequently the permeability of the lipid bilayer membrane, which led to a significant increase in the cell's ability to absorb essential elements. However, the addition of Tween-80 at higher concentrations inhibited cell growth60.
Analysis of variance (ANOVA) of PBD
To validate the obtained results of the experimental design, ANOVA was performed (Table 5). Significance of model and each variable was verified by F-value, P-values (and student’s t-test in Table 4). Large F-value and t-value as well low P-value (P < 0.05) or high confidence levels (Cl ≥ 95%) indicate a real effect on response69. Some investigators reported that confidence levels higher than 70% are also acceptable70. The analysis, according to low probability value ≥ 0.0001 for both response and high F-value 41.87 for biomass weight and 204.31 for biotransformation, demonstrated that the model was highly significant. For biomass weight, the most significant variables were Iron concentration (G), fermentation time (B), agitation speed (A) and siderophore concentration (L). Their F-value is 217.41, 99.13, 60.77, 39.41 and t-value are 14.74, 9.95, 7.79, 6.27, respectively. All of them had a P-value of 0.0001 with coefficient level 99.99%. Follow by ZnSO4 (K) and KH2PO4 (J) with coefficient level 99.93% and 99.37% respectively. For biotransformation, the most significant variables were Iron concentration (G), fermentation time (B), siderophore (L) and KH2PO4 (J) concentration and agitation speed (A). They include F-value1599.56, 190.02, 161.73, 90.29, 82.44 and t-value 39.99, 13.78, 12.71, 9.5, 9.07 respectively. All of them had a P-value of 0.0001 with coefficient level 99.99%. The data revealed seed liquid volume (C), tween 80 (F), carbon (E) and NH4Cl (H) concentration are four non-significant independent factors (P˃0.05) in the answers.
A good correlation between the experimental and predicted values was indicated by a higher correlation coefficient (R), like here 0.9872. The coefficient of determination (R2) is in the range of 0 to 1, and a regression model with R2 larger than 0.9 indicates a high correlation and prediction power71. In the present study, the R2-value are R1 = 0.9746, R2 = 0.9947 means only 1.21% and 0.52% of the total changes are not explained for biomass weight and biotransformation, respectively. High value of adjusted determination coefficient (Adj. R2), like here 0.9938, refers to a high accuracy of the relationships between the studied experimental factors and response69. The higher Predicted R-Squared (Pred-R2) like here 95.94% implies that the model with more power predicts responses between the factors tested. The Pred-R2 and Adj-R2 must be close and within 20% of each other, which this confirms model’s statistical reliability. Adequate Precision with ratio greater than 4 like here 40.14, estimates the desirable signal-to-noise ratio, and indicates the good fit of the model. The lower value of coefficient of variation (C.V.%) like here 11.09%, indicates the greater reliability of the experiment. A smaller predicted residual sum of squares (PRESS) like here 9.3473, is the display criterion the better fits of each point in the design with the model.
According to the analysis, there are four significant variables with a positive effect on the responses. Iron concentration (G) is the most significant variable with an enormous difference compared to the others, with P-value 0.0001 for both responses, followed by fermentation time (B), agitation speed (A) and KH2PO4 (J) concentration with a confidence above 99%.
Regression equation
The regression coefficients (Table 4) were fitted to the first-order polynomial equation to represent the optimum responses as a function of the independent factors. By ignoring the insignificant factors (P > 0.1), the following equation of regression in terms of coded variables was obtained:
Model adequacy checking
The model’s correctness and accuracy were confirmed with the following graphs:
-
1.
The normal probability plot (NPP) of the studentized residuals versus the model’s expected response normal values is given in Fig. 9A, B for responses. This is an important diagnostic technique for visualizing the residuals distribution. So that the deviation from the straight line indicates systematic departures from the normality72. Here, the residual points are closer to the diagonal reference line with a smaller value, which indicates a good agreement between the model with the experimental results.
-
2.
Fig. 9C,D indicates a scatter plot of studentized residuals against predicted response values. The in range, uniform and random distribution of the residuals indicate their constant variance and the model’s accuracy.
-
3.
Fig. 9E, F presents the relationship between predicted and actual values of responses. In this plot, points gathered around the diagonal line indicating a significant correlation.
-
4.
Box–Cox plot of model transformation of the response (Fig. 9G,H), represents a potential best practice for selecting a better power transformation to further improve the model. In this graph, the blue line represents the current transformation confidence interval value (current λ = 1). The green line indicates the best lambda value (best λ) for R1 = 0.02 and R2 = 0.81. And the red lines represent the confidence intervals values, in ranges of minimum (low C.I. = 0.43, 0.51) and the maximum (high C.I. = 1.38, 1.11) for biotransformation and biomass weight, respectively. If the blue and green lines fell within the red lines, the model will be in the optimal zone. And the current λ value quite close to the best λ indicates the model fits the experimental data well and no data transformation is needed, as they are here.
The normal probability plot of the studentized residuals (A, B). plot of internally studentized residuals against predicted values (C, D). Plot of the correlation between predicted versus observed values (E, F) and Box-Cox Plot for Power Transforms (G, H) for biotransformation and biomass weight determined by the first-order polynomial equation.
This S. boulardii yeast type rich in organic iron is a potential resource for managing and controlling the problem of anemia in the world, even for vegetarians and vegans. Because iron with high bioavailability and low toxicity is placed next to a resistant probiotic with health-giving properties. Which it can create a revolution in the production of the most bioavailable iron in the form of pharmaceutical supplements, fortified foods, fermented products, and also providing iron lost in bread flour. However, several opportunities and challenges are revealed after analyzing the results. It is hoped that future research will increase the potential of iron enrichment in Bullardi. For this purpose, the most important action is to discover conditions that increase the absorption power of the S. boulardii wall or transfer iron to the yeast in a more targeted way. Also, the ability to transport iron of other types of siderophores should be investigated.
Conclusion
In this research, the PBD statistical method was used to screen eleven variables and identify the most effective ones in increasing biomass weight and iron biotransformation of S. boulardii. The results showed the highest biotransformation was 17.77 mg Fe/g DCW in the highest biomass weight of 9 g/L. The Iron concentration had the most positive important effect, with P-value 0.0001 for both responses and main effects 4.2 and 97.76 for biomass weight and biotransformation, respectively, and followed by fermentation time, agitation speed and KH2PO4 concentration. Siderophores, despite the powerful potential of combining with iron and transferring it to the cell, showed a significant negative influence on Fe biotransformation in this yeast strain (p ≤ 0.05). Siderophore inefficiency in transporting iron into the cell may be due to the lack of receptors for Pvd and Pch siderophores in the membrane. Also, the steric hindrance of the oligosaccharide structure of the cell wall, stickiness and sedimentability of yeasts can also be an important factor in the limited membrane permeability and low richness. However, this yeast has the potential to be used as an effective source of iron in foods, fermented products, and supplements even for vegetarians and vegans, thus improving the treatment of anemia.
Siderophore can contribute to the development of effective strategies to increase iron enrichment in S. boulardii so in future studies it is suggested to investigate the potential of other types of siderophore in transferring iron to this yeast. Also, factors affecting membrane permeability and overcoming the structural barrier of the cell wall to produce organic iron rich biomass should be studied.
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Ramos-Alonso, L. et al. Molecular strategies to increase yeast iron accumulation and resistance. Metallomics 10, 1245–1256. https://doi.org/10.1039/c8mt00124c (2018).
Gafter-Gvili, A., Schechter, A. & Rozen-Zvi, B. Iron deficiency anemia in chronic kidney disease. Acta Haematol. 142, 44–50. https://doi.org/10.1159/000496492 (2019).
Halterman, J. S., Kaczorowski, J. M., Aligne, C. A., Auinger, P. & Szilagyi, P. G. Iron deficiency and cognitive achievement among school-aged children and adolescents in the United States. Pediatrics. 107, 1381–1386. https://doi.org/10.1542/peds.107.6.1381 (2001).
World Health Organization. Anaemia in women and children [Internet]. https://www.who.int/data/gho/data/themes/topics/anaemia_in_women_and_children (2023).
World Health Organization. The Global Prevalence of Anaemia in 2011 43 (World Health Organization, 2015).
Amils, R. C. Encyclopedia of Astrobiology 293 (Springer, 2011). https://doi.org/10.1007/978-3-642-11274-4.
Zhang, X. G. et al. Preparation of S-iron-enriched yeast using siderophores and its effect on iron deficiency anemia in rats. Food Chem. 365, 130508. https://doi.org/10.1016/j.foodchem.2021.130508 (2021).
Yuan, Y., Guo, X., He, X., Zhang, B. & Liu, S. Construction of a high-biomass, iron-enriched yeast strain and study on distribution of iron in the cells of Saccharomyces cerevisiae. Biotechnol. Lett. 26, 311–315. https://doi.org/10.1023/b:bile.0000015449.30186.90 (2004).
Gaudreau, H., Tompkins, T. A. & Champagne, C. P. The distribution of iron in iron-enriched cells of Saccharomyces cerevisiae. Acta Aliment. 30, 355–356. https://doi.org/10.1556/AAlim.30.2001.4.4 (2001).
Paš, M., Piškur, B., Šuštarič, M. & Raspor, P. Iron enriched yeast biomass—a promising mineral feed supplement. Bioresour. Technol. 98, 1622–1628. https://doi.org/10.1016/j.biortech.2006.06.002 (2007).
Pirman, T. & Orešnik, A. Fe bioavailability from Fe-enriched yeast biomass in growing rats. Animal 6, 221–226. https://doi.org/10.1017/S1751731111001546 (2012).
McFarland, L. V. Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J. Gastroenterol. 16, 2202–2222. https://doi.org/10.3748/wjg.v16.i18.2202 (2010).
Hudson, L. E. et al. Characterization of the probiotic yeast Saccharomyces boulardii in the healthy mucosal immune system. PLoS One. 11, e0153351. https://doi.org/10.1371/journal.pone.0153351 (2016).
Ramírez-Cota, G. Y., López-Villegas, E. O., Jiménez-Aparicio, A. R. & Hernández-Sánchez, H. Modeling the ethanol tolerance of the probiotic yeast Saccharomyces cerevisiae var. boulardii CNCM I-745 for its possible use in a functional beer. Probiot. Antimicrob. 13, 187–194. https://doi.org/10.1007/s12602-020-09680-5 (2021).
Graff, S., Chaumeil, J. C., Boy, P., Lai-Kuen, R. & Charrueau, C. Formulations for protecting the probiotic Saccharomyces boulardii from degradation in acidic condition. Biol. Pharm. Bull. 31, 266–272. https://doi.org/10.1248/bpb.31.266 (2008).
Martínez-Garay, C. A., De Llanos, R., Romero, A. M., Martínez-Pastor, M. T. & Puig, S. Responses of Saccharomyces cerevisiae strains from different origins to elevated iron concentrations. Appl. Environ. Microbiol. 82, 1906–1916. https://doi.org/10.1128/AEM.03464-15 (2016).
Nowosad, K., Sujka, M., Pankiewicz, U., Miklavčič, D. & Arczewska, M. Pulsed electric field (Pef) enhances iron uptake by the yeast Saccharomyces cerevisiae. Biomolecules. 11, 850. https://doi.org/10.3390/biom11060850 (2021).
Nowosad, K. & Sujka, M. The use of iron-enriched yeast for the production of flatbread. Molecules 26, 5204. https://doi.org/10.3390/molecules26175204 (2021).
De Serrano, L. O. Biotechnology of siderophores in high-impact scientific fields. Biomol. Concepts 8, 169–178. https://doi.org/10.1515/bmc-2017-0016 (2017).
Soares, E. V. Perspective on the biotechnological production of bacterial siderophores and their use. Appl. Microbiol. Biotechnol. 106, 3985–4004. https://doi.org/10.1007/s00253-022-11995-y (2022).
Miethke, M. & Marahiel, M. A. Siderophore-based iron acquisition and pathogen control. Microbiol. Mol. Biol. Rev. 71, 453–551. https://doi.org/10.1128/MMBR.00012-07 (2007).
Yun, C. W., Tiedeman, J. S., Moore, R. E. & Philpott, C. C. Siderophore-iron uptake in Saccharomyces cerevisiae: Identification of ferrichrome and fusarinine transporters. J. Biol. Chem. 275, 16354–16359. https://doi.org/10.1074/jbc.M001456200 (2000).
Jalal, M. A. F., Love, S. K. & Van Der Helm, D. Siderophore mediated iron(III) uptake in Gliocladium virens 2. Role of ferric mono- and dihydroxamates as iron transport agents. J. Inorg. Biochem. 29, 259–267. https://doi.org/10.1016/0162-0134(87)80033-8 (1987).
Gaensly, F., Wille, G. M. F. C., Brand, D. & Bonfim, T. M. B. Iron enriched Saccharomyces cerevisiae maintains its fermenting power and bakery properties. Food Sci. Technol. 31, 980–983 (2011).
Kyyaly, M. A., Powell, C. & Ramadan, E. Preparation of iron-enriched baker’s yeast and its efficiency in recovery of rats from dietary iron deficiency. Nutrition. 31, 1155–1164. https://doi.org/10.1016/j.nut.2015.04.017 (2015).
Strobel, R. J. & Sullivan, G. R. Experimental design for improvement of fermentations. In Manual of Industrial Microbiology and Biotechnology (eds. Demain, A. L. and Davies, J. E.) 80–93 (ASM, 1999).
Schwyn, B. & Neilands, J. B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 160, 47–56. https://doi.org/10.1016/0003-2697(87)90612-9 (1987).
Lakshmanan, V. A natural rice rhizospheric bacterium abates arsenic accumulation in rice (Oryza sativa L.). Planta. 242, 1037–1050. https://doi.org/10.1007/s00425-015-2340-2 (2015).
Arnow, L. E. Colorimetric determination of the components of 3,4-dihydroxyphenylalanine-tyrosine mixtures. J. Biol. Chem. 118, 531–537. https://doi.org/10.1016/S0021-9258(18)74509-2 (1937).
Payne, S. M. Detection, isolation, and characterization of siderophores. Meth. Enzymol. 235, 329–344. https://doi.org/10.1016/0076-6879(94)35151-1 (1994).
Malairuang, K., Krajang, M., Sukna, J., Rattanapradit, K. & Chamsart, S. High cell density cultivation of Saccharomyces cerevisiae with intensive multiple sequential batches together with a novel technique of fed-batch at cell level (FBC). Processes. 8, 1–26. https://doi.org/10.3390/pr8101321 (2020).
Tafazzoli, K., Ghavami, M. & Khosravi-Darani, K. Production of iron enrisched Saccharomyces boulardii: Impact of process variables. Sci. Rep. 14, 4844. https://doi.org/10.1038/s41598-024-55433-7 (2024).
Wang, R., Lorantfy, B., Fusco, S., Olsson, L. & Franzén, C. J. Analysis of methods for quantifying yeast cell concentration in complex lignocellulosic fermentation processes. Sci. Rep. 11, 11293. https://doi.org/10.1038/s41598-021-90703-8 (2021).
Olivares-Marin, I. K., González-Hernández, J. C., Regalado-Gonzalez, C. & Madrigal-Perez, L. A. Saccharomyces cerevisiae exponential growth kinetics in batch culture to analyze respiratory and fermentative metabolism. J. Vis. Exp. 2018, 58192. https://doi.org/10.3791/58192 (2018).
Zhang, X. G. et al. Effects of Fe-YM1504 on iron deficiency anemia in rats. Food Funct. 7, 3184–3192. https://doi.org/10.1039/C6FO00423G (2016).
Zhang, X. G., Peng, Y. N., Li, X. R., Ma, G. D. & Chen, X. Q. Screening of iron-enriched fungus from natural environment and evaluation of organically bound iron bioavailability in rats. Food Sci. Technol. Int. 35, 58–66. https://doi.org/10.1590/1678-457X.6454 (2015).
Sabatier, M. et al. Iron bioavailability from fresh cheese fortified with iron-enriched yeast. Eur. J. Nutr. 56, 1551–1560. https://doi.org/10.1007/s00394-016-1200-6 (2017).
Gaensly, F., Picheth, G., Brand, D. & Bonfim, T. M. B. The uptake of different iron salts by the yeast Saccharomyces cerevisiae. Braz. J. Microbiol. 45, 491–494. https://doi.org/10.1590/s1517-83822014000200016 (2014).
Esmaeili, S., Khosravi-Darani, K., Pourahmad, R. & Komeili, R. An experimental design for production of selenium-enriched yeast. World Appl. Sci. J. 19, 31–37. https://doi.org/10.5829/idosi.wasj.2012.19.01.2634 (2012).
Nie, X. et al. ARTP mutagenesis promotes selenium accumulation in Saccharomyces boulardii. LWT. 168, 113916. https://doi.org/10.1016/j.lwt.2022.113916 (2022).
Kitamura, D. H. et al. Selenium-enriched probiotic Saccharomyces boulardii CCT 4308 biomass production using low-cost sugarcane molasses medium. Braz. Arch. Biol. Technol. 64, e21200658. https://doi.org/10.1590/1678-4324-75years-2021200658 (2021).
González-Salitre, L. et al. Physicochemical and microbiological parameters during the manufacturing of a beer-type fermented beverage using selenized Saccharomycesboulardii. Heliyon. 9, e21190. https://doi.org/10.1016/j.heliyon.2023.e21190 (2023).
Hyrslova, I. et al. In vitro digestion and characterization of selenized Saccharomyces cerevisiae, Pichia fermentans and probiotic Saccharomyces boulardii. J. Trace Elem. Med. Biol. 83, 127402. https://doi.org/10.1016/j.jtemb.2024.127402 (2024).
Kwak, S. et al. Dissection and enhancement of prebiotic properties of yeast cell wall oligosaccharides through metabolic engineering. Biomaterials 282, 121379. https://doi.org/10.1016/j.biomaterials.2022.121379 (2022).
Graus, M. S. et al. Mannan molecular substructures control nanoscale glucan exposure in Candida. Cell Rep. 24, 2432–2442. https://doi.org/10.1016/j.celrep.2018.07.088 (2018).
Gow, N. A. R. & Hube, B. Importance of the Candida albicans cell wall during commensalism and infection. Curr. Opin. Microbiol. 15, 406–412. https://doi.org/10.1016/j.mib.2012.04.005 (2012).
Kodoi, R., Takahashi, T., Oka, O. & Urasaki, H. Inventors; Oriental Yeast Co Ltd, assignee. Method for producing high-zinc-content yeast extract, high-zinc-content yeast extract, foods, and agent for maintaining and restoring green color of vegetables. United States patent US20140212571A1. https://patents.google.com/patent/US20140212571A1/en (2014).
Choi, Y. Inventor; Barrick Gold Corp, assignee. Production of zinc sulphate concentrates from a dilute zinc sulphate solution. United States patent US20110165059A1. https://patents.google.com/patent/US20110165059A1/en (2011).
Polson, C., Sarkar, P., Incledon, B., Raguvaran, V. & Grant, R. Optimization of protein precipitation based upon effectiveness of protein removal and ionization effect in liquid chromatography-tandem mass spectrometry. Chromatogr. B Ana Technol. Biomed. Life Sci. 785, 263–275. https://doi.org/10.1016/s1570-0232(02)00914-5 (2003).
El-Neggar, N. E. et al. Process development for scale-up production of a therapeutic L-asparaginase by Streptomyces brollosae NEAE-115 from shake flasks to bioreactor. Sci. Rep. 9, 13571. https://doi.org/10.1038/s41598-019-49709-6 (2019).
The oxygen-transferring ferment of respiration. Nobel Lecture. https://www.nobelprize.org/uploads/2018/06/warburg-lecture.pdf (1931).
Hazra, A. et al. Coenzyme and prosthetic group biosynthesis. Encycl. Microbiol. 79–88, 2009. https://doi.org/10.1016/B978-012373944-5.00069-9 (2009).
Coote, N. & Kirsop, B. H. Factors responsible for the decrease in ph during beer fermentations. J. Inst. Brew. 82, 149–153. https://doi.org/10.1002/j.2050-0416.1976.tb03739.x (1976).
Cox, C. D. & Adams, P. Siderophore activity of pyoverdin for Pseudomonas aeruginosa. Infect. Immun. 48, 130–138. https://doi.org/10.1128/iai.48.1.130-138.1985 (1985).
Sheykhi, F., Ahmadifard, N., Samadi, N. & Nematzadeh, K. The effect of different concentrations of organic and inorganic zinc on the growth and zinc content in yeast (Saccharomyces Cerevisiae). BJM 7, 103–109 (2019).
Shet, A. R., Patil, L., Hombalimath, V. S., Yaraguppi, D. & Udapudi, B. B. Enrichment of Saccharomyces cerevisiae with zinc and their impact on cell growth. Biotechnol. Bioinf. Bioeng. 1, 523–527 (2011).
Liu, X. F., Supek, F., Nelson, N. & Culotta, V. C. Negative control of heavy metal uptake by the Saccharomyces cerevisiae BSD2 gene. J. Biol. Chem. 272, 11763–11769. https://doi.org/10.1074/jbc.272.18.11763 (1997).
Zhang, B. B. & Cheung, P. C. K. A mechanistic study of the enhancing effect of Tween 80 on the mycelial growth and exopolysaccharide production by Pleurotus tuber-regium. Bioresour. Technol. 102, 8323–8326. https://doi.org/10.1016/j.biortech.2011.06.021 (2011).
Zhang, B. B., Chen, L. & Cheung, P. C. K. Two-dimensional gel electrophoresis analysis of mycelial cells treated with tween 80: Differentially expressed protein related to enhanced metabolite production. J. Agric. Food. Chem. 60, 10585–10591. https://doi.org/10.1021/jf303570d (2012).
Liang, Y. et al. Influence of Tween-80 on the production and structure of water-insoluble curdlan from Agrobacterium sp.. Int. J. Biol Macromol. 106, 611–619. https://doi.org/10.1016/j.ijbiomac.2017.08.052 (2018).
Li, Q., Lei, Y., Hu, G., Lei, Y. & Dan, D. Effects of Tween 80 on the liquid fermentation of Lentinus edodes. Food Sci. Biotechnol. 27, 1103–1109. https://doi.org/10.1007/s10068-018-0339-8 (2018).
Sheng, L., Zhu, G. & Tong, Q. Mechanism study of Tween 80 enhancing the pullulan production by Aureobasidium pullulans. Carbohydr. Polym. 97, 121–123. https://doi.org/10.1016/j.carbpol.2013.04.058 (2013).
Tu, G., Wang, Y., Ji, Y. & Zou, X. The effect of Tween 80 on the polymalic acid and pullulan production by Aureobasidium pullulans CCTCC M2012223. World J. Microbiol. Biotechnol. 31, 219–226. https://doi.org/10.1007/s11274-014-1779-9 (2015).
Yin, H. et al. Evaluation of surfactant effect on β-poly (L-malic acid) production by Aureobasidium pullulans. Biotechnol. Biotechnol. Equip. 33, 954–966. https://doi.org/10.1080/13102818.2019.1631718 (2019).
Cui, J. et al. Tween 80 micelles loaded with Fe3O4 nanoparticles and artemisinin for combined oxygen-independent ferroptosis therapy of cancer. Pharmaceutics. 16, 639. https://doi.org/10.3390/pharmaceutics16050639 (2024).
Xu, H. X. et al. Degradation of fluoranthene by a newly isolated strain of Herbaspirillum chlorophenolicum from activated sludge. Biodegradation 22, 335–345. https://doi.org/10.1007/s10532-010-9403-7 (2011).
Jin, D., Jiang, X., Jing, X. & Ou, Z. Effects of concentration, head group, and structure of surfactants on the degradation of phenanthrene. J. Hazard Mater. 144, 215–221. https://doi.org/10.1016/j.jhazmat.2006.10.012 (2007).
Li, J. L. & Chen, B. H. Effect of nonionic surfactants on biodegradation of phenanthrene by a marine bacteria of Neptunomonas naphthovorans. J. Hazard Mater. 162, 66–73. https://doi.org/10.1016/j.jhazmat.2008.05.019 (2009).
Akhnazarova, S., Kafarov, V., Repyev, A. P. & Matskovsky, V. M. Experiment Optimization in Chemistry and Chemical Engineering (Mir, 1982).
Stowe, R. A. & Mayer, R. P. Efficient screening of process variables. J. Ind. Eng. Chem. 58, 36–40. https://doi.org/10.1021/IE50674A007 (1966).
El-Naggar, N. E., Haroun, S. A., El-Weshy, E. M., Metwally, E. A. & Sherief, A. A. Mathematical modeling for bioprocess optimization of a protein drug, uricase, production by Aspergillus welwitschiae strain 1–4. Sci. Rep. 9, 12971. https://doi.org/10.1038/s41598-019-49201-1 (2019).
Montgomery, D. C. Design and Analysis of Experiments 3rd edn. (Wiley, Uk, 1991).
Acknowledgements
All contributors are listed as authors.
Author information
Authors and Affiliations
Contributions
Conceptualization: KKD; Methodology: KT, KKD; Software: MG, KKD; Validation: KT, KKD; Analysis: KT, KKD; Investigation, KT; Data curation: KT, KKD; Writing original draft: KT, KKD, Writing-review and editing; KT, MG, KKD; Supervision; KKD, MG; Project administration, KKD.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tafazzoli, K., Ghavami, M. & Khosravi-Darani, K. Investigation of impact of siderophore and process variables on production of iron enriched Saccharomyces boulardii by Plackett–Burman design. Sci Rep 14, 22813 (2024). https://doi.org/10.1038/s41598-024-70467-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70467-7