Abstract
Given the global high prevalence of MASLD and its poor CVD prognosis, it is essential to perform risk stratification for MASLD patients. The specific impact of High Sensitivity Troponins (hs-cTn ) on mortality in MASLD patients remains unexplored. The NHANES databases from 1999 to 2004, which include data on high-sensitivity cardiac troponin (hs-cTn) levels and comorbidities, were linked with the most recent mortality dataset. Myocardial injury was determined using the 99th upper reference limits (URL) for hs-cTn. Our study included 3460 MASLD patients. The mean follow-up duration was 192 months, during which 1074 (23%) MASLD participants died from all-cause mortality, and 363 (7.3%) died from CVD mortality. Our findings indicate that MASLD patients with elevated levels of hs-cTnT (> 99th URL) exhibit increased risks of all-cause mortality [adjusted hazard ratio (aHR) = 1.93] and CVD mortality (aHR = 2.4). Similar results were observed for hs-cTnI, where the aHRs for all-cause mortality and CVD mortality were 2.03 and 2.97, respectively. Furthermore, we identified a nonlinear dose-response relationship between hs-cTn levels and the risk of mortality (P for nonlinearity < 0.001). Our findings suggest that hs-cTn can predict mortality risk in MASLD, aiding clinicians in risk-stratifying this population. Therefore, we recommend considering hs-cTn detection in individuals with MASLD to effectively assess their future mortality risk.
Similar content being viewed by others
Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD) is the most prevalent chronic liver disease worldwide 1. It is closely linked to common metabolic cardiovascular risk factors such as obesity, insulin resistance, type 2 diabetes (T2DM), metabolic syndrome, hypertension, and dyslipidemia 2. Therefore, MASLD is often linked to cardiovascular disease (CVD) due to shared cardiometabolic risk factors. Research indicates that CVD mortality is currently the leading cause of mortality among individuals with MASLD 3. However, MASLD patients do not necessarily have concurrent CVD. Therefore, identifying biomarkers to recognize MASLD patients with poor prognosis is of significant clinical importance. Additionally, early risk stratification and appropriate treatment can help guide therapy directed at improving the outcomes of MALSD patients.
Cardiovascular markers, such as N-terminal pro b-type natriuretic peptide (NT-proBNP) and troponin, play an increasingly important role in predicting CVD and mortality. Studies have found that NT-proBNP can predict future CVD events and mortality 4, 5 in both MASLD and non-MASLD patients. For decades, standard troponin assays have been approved for diagnosing acute coronary syndrome. Building upon this, the development of high-sensitivity assays has facilitated the detection of high-sensitivity cardiac troponin (hs-cTn) in the general population 6. Previous studies have shown that chronic myocardial injury (manifested as stable, mild increases in hs-cTn concentrations) is associated with a significantly increased risk of HF and CVD death 7. Moreover, similar conclusions have been drawn in healthy individuals 6, 8,9,10. Furthermore, in hospitalized COVID-19 patients with MASLD, troponin levels were significantly higher in those who died compared to survivors 11.
Given the global high prevalence of MASLD and its poor CVD prognosis, it is essential to perform risk stratification for MASLD patients. However, research in this area is currently limited. Therefore, we hypothesize that hs-cTn can predict mortality risk in the community-dwelling MASLD population. Our research aimed to explore the association between hs-cTn levels and both CVD and all-cause mortality in a nationally representative sample of adults with MASLD sourced from the National Health and Nutrition Examination Survey (NHANES 1999–2004).
Methods
Study design and participants
NHANES, conducted by the Centers for Disease Control and Prevention (CDC), is a nationwide survey designed to assess the health and nutritional status of the US population 12. The survey comprises a household interview and a medical examination conducted at a mobile examination center (MEC). NHANES study protocols were approved by the Research Ethics Review Board of the National Center for Health Statistics, and written informed consent was obtained from all participants. More detailed information about NHANES can be found on the official website. (https://www.cdc.gov/nchs/nhanes/index.htm).
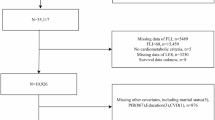
Until December 31, 2019, all subjects were prospectively followed up. Only participants from these three cycles were selected for this study, as hs-cTn information was available only for the NHANES 1999 to 2004 survey cycle, totaling 31,126 participants. Patients under 20 years of age, significant alcohol use (males with > 140 g/week or females > 70 g/week), those with incomplete laboratory data such as hs-cTn, and those lacking information about MASLD diagnosis were excluded. Figure 1 illustrates the study flowchart.
Definition of MASLD
MASLD was defined as steatosis combined with any one of the following cardiometabolic criteria 13. Due to the absence of liver transient elastography and histopathological data in the NHANES database from 1999 to 2004, steatosis assessment primarily relied on the Fatty Liver Index ≥ 60 (FLI). The FLI, validated as a reliable tool for assessing hepatic steatosis, was calculated using the formula 14: FLI= (e0.953×Ln (TG) + 0.139×BMI + 0.718×Ln (GGT) + 0.053×WC − 15.745)/ (1+ e0.953×Ln (TG) + 0.139×BMI + 0.718×Ln (GGT) + 0.053×WC − 15.745)×100. In this formula, TG, GGT, BMI, and WC represent triglycerides (mg/dL), glutamyl transpeptidase (U/L), body mass index (kg/m²), and waist circumference (cm), respectively. Cardiometabolic criteria were defined as meeting any of the following conditions: (1) BMI ≥ 25 kg/m2 or WC > 94 cm (for men) and > 80 cm (for women); (2) fasting plasma glucose ≥ 5.6 mmol/L or 2-hour postprandial plasma glucose ≥ 7.8 mmol/L or glycosylated hemoglobin (HbA1c) ≥ 5.7% or T2DM or hypoglycemic therapy; (3) blood pressure ≥ 130/85 mmHg or antihypertensive therapy; (4) triglyceride ≥ 1.70 mmol/L; (5) Plasma high-density lipoprotein cholesterol (HDL-C) ≤ 1.0mmol/L (for men) and ≤ 1.3 mmol/L (for women) or lipid-lowering therapy.
Measurement of cardiac biomarkers
NHANES participants from 1999 to 2004 had serum samples stored at the time of the survey, with consent for their use in future research. Between 2018 and 2020, laboratory testing was conducted on frozen specimens shipped to the Clinical Chemistry Research Laboratory at the University of Maryland (Baltimore, MD). High-sensitivity cardiac troponin T (measured using Roche Cobas e601) had 99th upper reference limits (URL) of 22 ng/L for males and 14 ng/L for females. High-sensitivity cardiac troponin I (measured using Ortho Vitros 3600) had 99th URL of 22 ng/L for males and 14 ng/L for females, respectively. Previous studies presumed that survey participants were free of acute myocardial ischemia during their participation because they were at home 15, and myocardial injury was defined as exceeding these 99th URL 16.
Assessment of covariates
Demographic data collected included information on sex, age, race, income-to-poverty ratio, and education level. Examination data encompassed measurements like BMI and WC. Laboratory data covered parameters including creatinine, triglycerides, glutamyl transpeptidase (GGT), total cholesterol, alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, and glycosylated hemoglobin (HbA1c). Questionnaire data included details on smoking status, alcohol consumption, hypertension, diabetes, heart failure (HF), coronary heart disease (CHD), angina, myocardial infarction, stroke, cancer, and physical activity (PA). These data were obtained from NHANES. Obesity was defined as having a BMI of 30 kg/m2 or higher. Race was categorized as Mexican American, other Hispanic, non-Hispanic white, non-Hispanic black or other races. Education level was categorized as less than high school, high school or general educational development, and more than high school. Smoking status was categorized as never-smoker, former smoker, and current smoker. The definition of hypertension status is met if any of the following criteria: (1) self-reported hypertension; (2) use of antihypertensive medication; (3) non-same-day randomized recordings of three occasions where systolic blood pressure was ≥ 140 mmHg or diastolic blood pressure was ≥ 90 mmHg. Diagnosed by a doctor, blood glucose levels of ≥ 11.1 mmol/L in a 2-hour oral glucose tolerance test or random blood glucose, fasting glucose levels of ≥ 7.0 mmol/L, or HbA1c levels > 6.5%, or use of diabetes medications or insulin, formed the diagnosis of diabetes. The definition of cancer, angina pectoris, heart attack, CHD, HF, and stroke relied on a self-reported history of disease. Diagnosis of CHD included self-reported CHD, angina pectoris, or myocardial infarction. CVD included angina pectoris, heart attack, CHD, HF, and stroke. Previous research has shown that PA is a robust predictor of both cardiovascular and all-cause mortality 17,18,19. In our study, PA was assessed by calculating responses to questions about the frequency and duration of light-moderate and vigorous activity 19. The original frequencies and durations were initially recorded in different time units but were subsequently recoded as weekly occurrences and minutes each time, respectively. Participants who reported inability to engage in activities at a certain intensity level were classified as non-participants at that level. Subsequently, the time per week (min/week) spent in light-moderate physical activity (MPA) and vigorous physical activity (VPA) was calculated. Overall, PA = MPA + 2 × VPA20. To mitigate over-reporting and reduce the impact of outliers on results, PA was capped at 1680 min per week 21. PA was categorized according to the 2020 World Health Organization (WHO) PA guidelines as follows: PA < 150 min/week or PA ≥ 150 min/week22. In the NHANES Methods and Analysis Guide, comprehensive information regarding variable collection methods is available (https://wwwn.cdc.gov/nchs/nhanes/AnalyticGuidelines).
Assessment of mortality
Baseline NHANES data from 1999 to 2004 were linked with fatality data reported in the National Death Index as of December 31, 2019. This linkage was conducted to determine mortality status. The study endpoints included all-cause mortality and CVD mortality. All-cause mortality was defined as death from any cause. CVD mortality encompassed deaths attributed to heart diseases and cerebrovascular diseases, identified by ICD codes I00–I09, I11, I13, I20–I51, and I60–I69, which include conditions such as myocardial infarction, stroke, and related CVD events.
Statistical analysis
We conducted a secondary analysis of NHANES dataset. It is recommended to use sampling weights and design variables in all analyses. Neglecting these parameters may result in biased estimates and inflated significance levels 23. The sampling weights were calculated as follows: 1999–2002 weights were 2/3 × 4-year MEC weight, and 2003–2004 weights were 1/3 × 2 year MEC weight. Continuous variables were presented as mean ± standard deviation and compared using Student’s t-test, while categorical variables were displayed as frequency counts and percentages. Survival analysis using the Kaplan–Meier method was employed to compare survival times between the two groups, with differences analyzed using the log-rank test. Weighted Cox proportional hazard models were utilized to estimate hazard ratios (HR) for the association between myocardial injury and mortality. Three Cox regression models were constructed, each adjusting for different confounding factors. The confounding factors were defined based on clinical experience and previous study 24. Model 1 did not adjust for any confounding factors. Model 2 was adjusted for age, sex, family income, smoking status, education level, and alcohol consumption status. Model 3 was adjusted for age, sex, family income, smoking status, education level, alcohol consumption status, obesity, creatinine level, hypertension, diabetes, hyperlipidemia, HF, CHD, angina, heart attack, stroke, cancer, statin use, and PA. We performed Cox proportional hazards regression models with restricted cubic spline (RCS) analyses using four knots to investigate the correlation between hs-cTn and mortality. In the RCS model, we also adjusted for confounding factors: age, sex, family income, smoking status, education level, alcohol consumption status, obesity, creatinine level, hypertension, diabetes, hyperlipidemia, HF, CHD, angina, heart attack, stroke, cancer statin use, and PA. We also conducted subgroup analyses to evaluate the impact of hs-cTn on all-cause mortality and CVD mortality in various subgroups stratified by age (< 60 years, and ≥ 60 years), sex (male, and female), alcohol consumption status (Non-drinker, 1–5 drinks/month, and 5 + drinks/month), obesity, diabetes, cancer, CVD, and PA status (PA < 150 min/week and PA ≥ 150 min/week). All data in this study were analyzed using R, with statistical signifiance set at P < 0.05.
Ethics statement
NHANES study protocols were approved by the Research Ethics Review Board of the National Center for Health Statistics and written informed consent was obtained from all participants in this study. Therefore, ethical approval and participant consent were not needed.
Results
Characteristics of study participants
Figure 1 displays the screening process for the study population. This study included a total of 3,460 MASLD patients who met the inclusion criteria. The mean age of the enrolled patients was 48 years, with females comprising 40% of the cohort, and the prevalence of CVD was 11%. No statistically significant differences were observed between the myocardial injury group and the non-myocardial injury group in terms of sex, BMI, triglycerides, total cholesterol, or GGT levels. Patients in the myocardial injury group were older, had lower levels of education and household income, lower PA, higher WC, and a higher frequency of alcohol consumption. They also had higher rates of diabetes, hypertension, hyperlipidemia, heart failure, coronary artery disease, angina, cerebral infarction, cancer, and a higher rate of statin use compared to those in the non-myocardial injury group. Additionally, they exhibited higher all-cause and CVD mortality. Patient characteristics are presented in Table 1.
Survival analysis
Details of the survival analysis results are shown in Table 1; Fig. 2. The mean follow-up duration was 192 months, during which 1,074 (23%) MASLD participants died from all-cause mortality and 363 (7.3%) died from CVD mortality. Kaplan–Meier survival curves indicate significant differences in all-cause mortality and CVD mortality among groups.
Kaplan–Meier survival curves for myocardial injury and mortality. (A) Kaplan–Meier survival curve by hs-cTn T (all-cause mortality), (B) Kaplan–Meier survival curve by hs-cTn I (all-cause mortality), (C) Kaplan–Meier survival curve by hs-cTn T (CVD mortality), (D) Kaplan–Meier survival curve by hs-cTn I (CVD mortality).
hs-cTnT
After adjusting for age, sex, family income, smoking status, education level, and alcohol consumption status using a Cox regression model, individuals with myocardial injury had an adjusted hazard ratio (aHR) of 2.16 (95% CI 1.67–2.79, P < 0.001) for all-cause mortality and 2.9 (95% CI 2.14–3.94, P < 0.001) for CVD mortality compared to those without myocardial injury. Furthermore, after adjusting for age, sex, race, family income, education level, obesity, creatinine level, alcohol consumption status, smoking status, hypertension, diabetes, heart failure, coronary heart disease, angina, heart attack, stroke, cancer statin use, and PA, individuals with myocardial injury had an aHR of 1.93 (95% CI 1.5–2.47, P < 0.001) for all-cause mortality and 2.4 (95% CI 1.76–3.28, P < 0.001) for CVD mortality compared to those without myocardial injury. Details are provided in Table 2; Fig. 4.
Association between hs-cTn and mortality of individuals with MASLD. (A) Association between hs-cTn T and all-cause mortality of individuals with MASLD, (B) association between hs-cTn I and all-cause mortality of individuals with MASLD, (C) association between hs-cTn T and CVD mortality of individuals with MASLD, (D) association between hs-cTn I and CVD mortality of individuals with MASLD.
hs-cTnI
After adjusting for age, sex, family income, smoking status, education level, and alcohol consumption status using a Cox regression model, individuals with myocardial injury had an aHR of 2.29 (95% CI 1.77–2.95, P < 0.001) for all-cause mortality and 3.63 (95% CI 2.36–5.59, P < 0.001) for CVD mortality compared to those without myocardial injury. Furthermore, after adjusting for age, sex, race, family income, education level, obesity, creatinine level, alcohol consumption status, smoking status, hypertension, diabetes, heart failure, coronary heart disease, angina, heart attack, stroke, cancer statin use, and PA, individuals with myocardial injury had an aHR of 2.03 (95% CI 1.48–2.79, P < 0.001) for all-cause mortality and 2.97 (95% CI 1.69–5.22, P < 0.001) for CVD mortality compared to those without myocardial injury. Details are provided in Table 2; Fig. 5.
Dose-response of hs-cTn and mortality risk
To enhance the robustness of the findings, we explored the presence of a nonlinear association between hs-cTn and mortality risk. As depicted in Fig. 3, we identified a nonlinear dose-response relationship between hs-cTn and mortality risk (P for nonlinearity < 0.001).
Subgroup analyses
Subgroup analysis results, adjusted for age, sex, race, family income, education level, obesity, creatinine level, alcohol consumption status, smoking status, hypertension, diabetes, heart failure, coronary heart disease, angina, heart attack, stroke, cancer, and PA, are presented in Figs. 4 and 5. Subgroup analysis revealed that myocardial injury was significantly associated with all-cause /CVD mortality in MASLD patients across various categories, including age (< 60 years and ≥ 60 years), sex (male and female), alcohol consumption status (non-drinker and 1–5 drinks/month), obesity (present or absent), diabetes (present or absent), cancer (absent), CVD (present or absent), and physical activity status (PA < 150 min/week and PA ≥ 150 min/week).
Discussion
To the best of our knowledge, this study is the first to evaluate the association between hs-cTn and both all-cause and CVD mortality in patients with MASLD. In the general population of US adults with MASLD, we observed strong and independent associations of hs-cTn with both all-cause and CVD mortality. These predictive associations remained consistent across population subgroups defined by age, sex, obesity, PA, diabetes, without cancer, and CVD. Our findings suggest that hs-cTn may be a predictor of mortality risk in MASLD patients, providing valuable insights for clinicians in the risk stratification of this population. The measurement of hs-cTn has the potential to enhance risk assessment beyond traditional factors.
We enrolled 3,460 MASLD patients in the United States diagnosed between 1999 and 2004, with a median follow-up period of 192 months. Our findings revealed a nonlinear association between hs-cTn levels and both all-cause mortality and CVD mortality. Patients with higher levels of hs-cTnT (> 99th URL) in MASLD exhibit elevated risks of all-cause mortality (aHR = 1.93) and CVD mortality (aHR = 2.4). Furthermore, similar results were found with hs-cTnI, with aHR of all-cause mortality and CVD mortality being 2.03 and 2.97, respectively. Hs-cTn was identified as strongly associated with a poor prognosis in patients with cardiovascular disease. Aimo et al. conducted a meta-analysis involving 9,289 patients with chronic heart failure, revealing that hs-cTn T is a robust and independent predictor of cardiovascular mortality (HR = 1.48, 95% CI 1.41–1.55), all-cause mortality (HR = 1.40, 95% CI 1.33–1.48), and CVD hospitalization (HR = 1.42, 95% CI 1.36–1.49) 25. Furthermore, previous studies in general community populations have reached similar conclusions. McEvoy et al. studied 8,838 participants with biracial representation from the Atherosclerosis Risk in Communities Study who were initially free of CHD and HF and had hs-cTnT measured. They found that incident detectable hs-cTnT (baseline, < 0.005 ng/mL; follow-up, ≥ 0.005 ng/mL) was independently associated with subsequent CHD (HR = 1.4, 95% CI 1.2–1.6), HF (HR = 2.0, 95% CI 1.6–2.4), and death (HR = 1.5, 95% CI 1.3–1.7) compared to an hs-cTnT level less than 0.005 ng/mL at both visits 26. Forlano et al. analyzed the difference between COVID-19 patients with MASLD who died and those who were discharged. They found that troponin levels were significantly higher in the group that died compared to the discharged patients (34 ng/mL vs. 10 ng/mL, P = 0.02)11. Our study further supports the notion that hs-cTn provides prognostic value for the general population and extends this finding to individuals with MASLD. In our research, CVD accounted for 11% of patients with MASLD. Furthermore, we conducted a subgroup analysis based on the presence or absence of CVD in MASLD patients, which further reinforced our conclusion that elevated hs-cTn offers prognostic value for all-cause mortality and CVD-related mortality in both MASLD patients with and without CVD.
At present, the non-cardiovascular mechanisms behind elevated hs-cTn levels remain unclear. Hs-cTn is a sensitive indicator that can be influenced by many factors. Therefore, when calculating the 99th URL for hs-cTn, various factors should be taken into consideration. First, demographic characteristics (such as age, gender) influence the concentration of hs-cTn. Research suggests that elevated hs-cTn levels in older adults are due to age-related changes rather than underlying cardiovascular disease 27. In different age cohorts, the 99th URL level increases with advancing age. Additionally, the 99th URL in healthy older men is significantly higher than in older women. High-sensitivity assessments have demonstrated that due to physiological sex differences, hs-cTn levels are higher in men than in women 28. Therefore, the use of a single hs-cTn threshold may lead to underestimation of myocardial injury risk in women and overdiagnosis in men and older adults 29, 30. In clinical practice, age and gender should be taken into account. In our analysis, we set different myocardial injury thresholds based on gender. Additionally, we performed a stratified analysis by age and gender, and the results were consistent, further reinforcing our conclusions. Secondly, different timing of measurements and physical activity may influence hs-cTn levels. Research indicates that hs-cTn levels exhibit diurnal variation 31. A study measuring hs-cTnT levels throughout the day found that hs-cTnT peaks in the morning, then decreases, and rises again at night 32. Additionally, research indicates that physical activity can increase hs-cTn levels 33. Thirdly, many non-cardiovascular diseases can also affect hs-cTn levels. Studies have shown that non-cardiac causes of chronic myocardial injury include diabetes, chronic obstructive pulmonary disease, pulmonary hypertension, toxins, chronic kidney disease, and anemia 34,35,36,37. Finally, various factors may lead to false-positive elevations in hs-cTn. Common causes of false elevations in cardiac-specific troponin include rheumatoid factor, heterophile antibodies, anti-troponin antibodies, large troponin complexes, hemolysis, and fibrin clots 38, 39. These factors can occur in both healthy individuals and patients with heart disease, potentially leading to false positives and affecting clinical interpretation and medical management. Therefore, when assessing myocardial injury in MASLD patients, it is essential to consider baseline characteristics, medical history, treatment, and diagnostic, as these variables may influence hs-cTn levels.
Our study indicates that MASLD patients with high hs-cTn levels, regardless of the presence of CVD, may have an increased risk of all-cause mortality and CVD mortality. Therefore, this provides a new approach to managing MASLD patients. Identifying a reliable method to address sustained myocardial injury detected by persistently elevated hs-cTn levels could potentially reduce mortality in MASLD patients. However, there is a lack of specific medications that clearly reduce hs-cTn levels, and there are few studies comparing the impact of hs-cTn levels before and after treatment on prognosis. Defilippi et al. found that regular physical activity reduced the age-related increase in hs-cTnT and also modified the relationship between hs-cTnT changes and the risk of heart failure 40. Packer et al. found that angiotensin-neprilysin inhibitors can lower circulating cardiac troponin levels and reduce overall mortality in heart failure patients 41. Everett et al. found that rosuvastatin can reduce cardiovascular events and all-cause mortality in patients without underlying cardiovascular disease, with benefits increasing as hs-cTnI levels rise. However, the lack of follow-up data on hs-cTnI prevented them from confirming whether rosuvastatin lowers hs-cTnI levels 42. Gravning et al. found that after 3 months of rosuvastatin use in heart failure patients, there was no significant change in hs-cTnT levels 43. Murphy et al. conducted a 12-month prospective study on 715 patients with reduced ejection fraction heart failure. They found that after starting treatment with sacubitril/valsartan, hs-cTnT levels significantly decreased (mean change of 6.7%). Additionally, they observed that changes in hs-cTnT were associated with left atrial and left ventricular reverse remodeling 44. Although hs-cTn can indicate the presence of myocardial injury, it cannot determine the underlying cause of the damage. Therefore, whether treatment or intervention can improve the prognosis of patients with elevated hs-cTn levels may depend on whether it targets the pathophysiological mechanisms underlying hs-cTn abnormalities. It is recommended to manage cardiometabolic factors common to both MASLD and CVD to reduce the risk of CVD and CVD mortality 45. MASLD patients have different cardiometabolic factors such as hypertension, diabetes, dyslipidemia, and obesity 2. This means that an intervention effective for one patient may be ineffective for another. There are various treatment approaches to improve the prognosis of MASLD patients, including lifestyle changes, dietary control, exercise, weight loss surgery, lipid-lowering agents, and glucose-lowering medications 45. However, whether these interventions improve prognosis by reducing hs-cTn levels, and the extent to which lowering hs-cTn contributes to prognosis improvement, still requires further research to confirm.
As far as we know, this is the first study to explore the association between hs-cTn and mortality risk in MASLD patients. Our findings may provide valuable insights into mortality and cardiovascular risk in this population. However, our study has several limitations. Firstly, many variables in NHANES relied on self-reported physician diagnoses, potentially affecting the reliability of our findings due to the less accurate nature of questionnaires compared to objective records. Secondly, we defined hepatic steatosis using FLI due to the absence of imaging and histologic data in the NHANES database from 1999 to 2004. However, FLI is considered a reliable criterion for diagnosing hepatic steatosis 14. Thirdly, the hs-cTn measurements in our study were derived from serum samples stored for an extended period. Despite this, the inter-assay coefficients of variation for these hs-cTn assays were excellent. Previous studies have demonstrated their accuracy and reliability in long-term stored samples 46. Additionally, our study lacks long-term follow-up data on hs-cTn, making it difficult to explore the impact of hs-cTn changes on the prognosis of MASLD patients. Finally, the recent acknowledgment of statins’ benefits in MASLD has resulted in their prescription to these patients, whereas previously they were avoided due to concerns about liver injury 47. Our study, based on a population from 1999 to 2004, found that the deceased group exhibited a higher rate of statin use (6.7% vs. 2.2%, P < 0.001). This limitation should be addressed in future prospective studies.
Conclusion
Our findings suggest that hs-cTn may predict mortality risk in MASLD, aiding clinicians in risk-stratifying this population. Therefore, we recommend considering hs-cTn detection in individuals with MASLD to effectively assess their future mortality risk.
Data availability
The datasets analyzed during the current study are available in the National Health and Nutrition Examination Survey repository (http://www.cdc.gov/nchs/nhanes.htm).
References
Zhu, J. Z. et al. Prevalence of nonalcoholic fatty liver disease and economy. Dig. Dis. Sci. 60 (11), 3194–3202 (2015).
Francque, S. M., van der Graaff, D. & Kwanten, W. J. Non-alcoholic fatty liver disease and cardiovascular risk: pathophysiological mechanisms and implications. J. Hepatol. 65 (2), 425–443 (2016).
Konyn, P., Ahmed, A. & Kim, D. Causes and risk profiles of mortality among individuals with nonalcoholic fatty liver disease. Clin. Mol. Hepatol. 29 (Suppl), S43-s57 (2023).
Younossi, Z. M. et al. Serum biomarkers are associated with atherosclerotic cardiovascular disease among patients with nonalcoholic fatty liver disease undergoing elective angiography. Clin. Gastroenterol. Hepatol. 20 (5), e1149–e1156 (2022).
Maechler, M. et al. 472-P: Pro-B-type natriuretic peptide strongly predicts future cardiovascular events in cardiovascular disease patients with NAFLD as well as in those without NAFLD. Diabetes 72 (Supplement_1) (2023). .
Welsh, P. et al. Cardiac troponin T and troponin I in the general population. Circulation 139 (24), 2754–2764 (2019).
Omland, T. et al. Prognostic value of cardiac troponin I measured with a highly sensitive assay in patients with stable coronary artery disease. J. Am. Coll. Cardiol. 61 (12), 1240–1249 (2013).
Jia, X. et al. High-sensitivity troponin I and incident coronary events, stroke, heart failure hospitalization, and mortality in the ARIC study. Circulation. 139 (23), 2642–2653 (2019).
McEvoy, J. W. et al. High-sensitivity troponins and mortality in the general population. Eur. Heart J.44 (28), 2595–2605 (2023).
Das, S. R. et al. 2020 Expert consensus decision pathway on novel therapies for cardiovascular risk reduction in patients with type 2 diabetes: A report of the American College of Cardiology solution set oversight committee. J. Am. Coll. Cardiol.76 (9), 1117–1145 (2020).
Forlano, R. et al. In-hospital mortality is associated with inflammatory response in NAFLD patients admitted for COVID-19. PLoS One. 15 (10), e0240400 (2020).
Zipf, G. et al. National Health and Nutrition Examination Survey: Plan and operations, 1999–2010. Vital Health Stat.1 (56), 1–37 (2013).
Rinella, M. E. et al. A multisociety Delphi Consensus Statement on new fatty liver disease nomenclature. Hepatology. 78 (6), 1966–1986 (2023).
Bedogni, G. et al. The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol.6, 33 (2006).
Bashar, H. et al. Association of myocardial injury with adverse long-term survival among cancer patients. Eur. J. Prev. Cardiol. (2024).
Thygesen, K. et al. Fourth universal definition of myocardial infarction (2018). Circulation. 138 (20), e618–e651 (2018).
Franklin, B. A. et al. Physical activity and cardiorespiratory fitness as modulators of health outcomes: A compelling research-based case presented to the medical community. Mayo Clin Proc. 98(2), 316–331 (2023).
Laukkanen, J. A., Isiozor, N. M. & Kunutsor, S. K. Objectively assessed cardiorespiratory fitness and all-cause mortality risk: An updated meta-analysis of 37 cohort studies involving 2,258,029 participants. Mayo Clin Proc. 97(6), 1054–1073 (2022).
Cabanas-Sánchez, V. et al. Physical activity and cause-specific cardiovascular mortality among people with and without cardiovascular disease: A cohort study of 0.6 million US adults. Mayo Clin Proc. 99(4), 564–577 (2024).
Gebel, K. et al. Effect of moderate to vigorous physical activity on all-cause mortality in middle-aged and older Australians. JAMA Intern. Med.175 (6), 970–977 (2015).
Stamatakis, E. et al. Sitting time, physical activity, and risk of mortality in adults. J. Am. Coll. Cardiol.73 (16), 2062–2072 (2019).
Bull, F. C. et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med.54 (24), 1451–1462 (2020).
Johnson, C. L. et al. National Health and Nutrition Examination Survey: Analytic guidelines, 1999–2010. Vital Health Stat. 2(161), 1–24 (2013).
McEvoy, J. W. et al. Four high sensitivity troponin assays and mortality in US adults with cardiovascular disease: The National Health and Nutrition Examination Survey, 1999–2004. Am. J. Prev. Cardiol.17, 100631 (2024).
Aimo, A. et al. Prognostic value of high-sensitivity troponin T in chronic heart failure: An individual patient data meta-analysis. Circulation. 137 (3), 286–297 (2018).
McEvoy, J. W. et al. Six-year change in high-sensitivity cardiac troponin T and risk of subsequent coronary heart disease, heart failure, and death. JAMA Cardiol.1 (5), 519–528 (2016).
Cho, H. et al. Frequency histograms of three high-sensitivity cardiac troponin assays in a reference population. J. Clin. Lab. Anal.36 (6), e24432 (2022).
Eggers, K. M. et al. Cardiac troponin I levels in an elderly population from the community–The implications of sex. Clin. Biochem.48 (12), 751–756 (2015).
Gore, M. O. et al. Age- and sex-dependent upper reference limits for the high-sensitivity cardiac troponin T assay. J. Am. Coll. Cardiol.63 (14), 1441–1448 (2014).
McRae, A. et al. Sex-specific, high-sensitivity cardiac troponin T cut-off concentrations for ruling out acute myocardial infarction with a single measurement. Cjem. 21 (1), 26–33 (2019).
Michailovich Chaulin, A. Diagnostic role and methods of detection of cardiac troponins: An opinion from historical and current points of view. Curr. Cardiol. Rev.19 (1), e100622205865 (2023).
Klinkenberg, L. J. et al. Circulating cardiac troponin T exhibits a diurnal rhythm. J. Am. Coll. Cardiol.63 (17), 1788–1795 (2014).
Vroemen, W. H. M. et al. Cardiac troponin T: Only small molecules in recreational runners after marathon completion. J. Appl. Lab. Med.3 (5), 909–911 (2019).
Eggers, K. M. & Lindahl, B. Application of cardiac troponin in cardiovascular diseases other than acute coronary syndrome. Clin. Chem.63 (1), 223–235 (2017).
Parikh, N. I. et al. Cardiovascular disease risk factors in chronic kidney disease: Overall burden and rates of treatment and control. Arch. Intern. Med.166 (17), 1884–1891 (2006).
Sedighi, S. M. et al. Elevated cardiac troponin levels in geriatric patients without ACS: Role of comorbidities. CJC Open.3 (3), 248–255 (2021).
Wan Nur Aimi, W. M. Z. et al. Elevation of highly sensitive cardiac troponin T among end-stage renal disease patients without acute coronary syndrome. Malays J. Med. Sci.28 (5), 64–71 (2021).
Chaulin, A. & Duplyakov, D. The main causes and mechanisms of the formation of false positive results of troponin immunotests. East. Eur.11, 132–147 (2022).
Salaun, E. et al. Role of antitroponin antibodies and macrotroponin in the clinical interpretation of cardiac troponin. J. Am. Heart Assoc.13 (12), e035128 (2024).
deFilippi, C. R. et al. Physical activity, change in biomarkers of myocardial stress and injury, and subsequent heart failure risk in older adults. J. Am. Coll. Cardiol.60 (24), 2539–2547 (2012).
Packer, M. et al. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation. 131 (1), 54–61 (2015).
Everett, B. M. et al. High-sensitivity cardiac troponin I and B-type natriuretic peptide as predictors of vascular events in primary prevention: Impact of statin therapy. Circulation. 131 (21), 1851–1860 (2015).
Gravning, J. et al. Prognostic effect of high-sensitive troponin T assessment in elderly patients with chronic heart failure: Results from the CORONA trial. Circ. Heart Fail.7 (1), 96–103 (2014).
Murphy, S. P. et al. Association between angiotensin receptor-neprilysin inhibition, cardiovascular biomarkers, and cardiac remodeling in heart failure with reduced ejection fraction. Circ. Heart Fail.14 (6), e008410 (2021).
Cheng, P. N. et al. Taiwan Association for the Study of the Liver-Taiwan Society of Cardiology Taiwan Position Statement for the management of metabolic dysfunction-associated fatty liver disease and cardiovascular diseases. Clin. Mol. Hepatol.30 (1), 16–36 (2024).
Kavsak, P. A. et al. Macrocomplexes and high-sensitivity cardiac troponin assays in samples stored for over 15 years. Clin. Chim. Acta. 505, 6–8 (2020).
Ho, A. et al. Effect of statin use on liver enzymes and lipid profile in patients with non-alcoholic fatty liver disease (NAFLD). J. Clin. Lipidol. (2024).
Author information
Authors and Affiliations
Contributions
JJ Hu: Data curation, Writing – original draft. YT Du: Data analysis. YD Zhou: Editing. HY Wang: Article proofreading and language polishing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, J., Du, Y., Zhou, Y. et al. High sensitivity troponins and mortality in the population with metabolic dysfunction-associated steatotic liver disease. Sci Rep 14, 19541 (2024). https://doi.org/10.1038/s41598-024-70645-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70645-7