Abstract
We provide an update regarding the differences between men and women in short-term postoperative mortality after coronary artery bypass grafting (CABG) and highlight the differences in postoperative risk of stroke, myocardial infarction, and new onset atrial fibrillation. We included 23 studies, with a total of 3,971,267 patients (70.7% men, 29.3% women), and provided results for groups of unbalanced studies and propensity matched studies. For short-term mortality, the pooled odds ratio (OR) from unbalanced studies was 1.71 (with 95% CI 1.69–1.74, I2 = 0%, p = 0.7), and from propensity matched studies was 1.32 (95% CI 1.14–1.52, I2 = 76%, p < 0.01). For postoperative stroke, the pooled effects were OR = 1.50 (95% CI 1.35–1.66, I2 = 83%, p < 0.01) and OR = 1.31 (95% CI 1.02–1.67, I2 = 81%, p < 0.01). For myocardial infarction, the pooled effects were OR = 1.09 (95% CI = 0.78–1.53, I2 = 70%, p < 0.01) and OR = 1.03 (95% CI = 0.86–1.24, I2 = 43%, p = 0.18). For postoperative atrial fibrillation, the pooled effect from unbalanced studies was OR = 0.89 (95% CI = 0.82–0.96, I2 = 34%, p = 0.18). The short-term mortality risk after CABG is higher in women, compared to men. Women are at higher risk of postoperative stroke. There is no significant difference in the likelihood of postoperative myocardial infarction in women compared to men. Men are at higher risk of postoperative atrial fibrillation after CABG.
Similar content being viewed by others
Introduction
Coronary artery bypass grafting (CABG) is the most frequently performed cardiac surgery worldwide1. Recent large-scale analyses have shown that after CABG, women are persistently at greater risk of operative death2,3,4,5. Although this disparity is generally attributed to preoperative and intraoperative differences between men and women, as well as to differences in biology and pathophysiology between the two sexes, its root causes have not yet been elucidated5. Considering women who undergo CABG are generally older, they also present higher comorbidities burden at the time of CABG, compared to men2,3. The meta-analysis of Bryce Robinson et al.2 focused on describing the differences between men and women following isolated CABG, by pooling results concerning operative mortality, late mortality, long-term myocardial infarction, stroke, repeat revascularization, and major adverse cardiac events (MACE). The authors concluded that females undergoing isolated CABG are not only at higher risk for operative and late mortality when compared with males, but also at higher risk of late non-fatal events including MACE, myocardial infarction, and stroke. Meta-regression analysis showed that the rate of preoperative myocardial infarction, smoking, renal failure and the number of grafts performed were associated with the operative mortality. In a meta-analysis which included 112 studies reporting unadjusted short-term mortality odds ratios and 25 studies which adjusted for various confounders, Shi et al.3 found a higher risk for death within 30-day after CABG in women, compared to men. Furthermore, to evaluate whether unmeasured confounding explains higher mortality, the authors performed a bias analysis simulation which considered the prevalence of prior cerebrovascular accident, heart failure, diabetes, and peripheral vascular disease. Under the assumption that the confounding effects on the risk of death were the same in men and as in women, after performing 10,000 simulations to correct for different degrees of association between each confounder and death, Shi et al. concluded that confounding is unlikely to account for the increased risk for mortality in women. Driven by the fact that observational studies suggest that females have poorer postoperative outcomes compared to males following cardiac surgery, Dixon et al.4 performed a systematic review and meta-analysis reporting short-term mortality (in-hospital/30 day), long-term mortality, postoperative stroke, sternal wound infection, or myocardial infarction in both sexes, after adjusting for baseline characteristics, following either CABG, heart valve surgery, or the two surgeries combined. The long-term mortality was found to be equivalent in both sexes, for all types of cardiac surgery. Females were found to be at a greater risk of short-term mortality and postoperative stroke than males following CABG or valve surgery combined with CABG, but not after isolated valve surgery. Finally, only a limited number of reviews summarize the published evidence regarding the risk difference between sexes of postoperative outcomes4,5.
The purpose of our meta-analysis was to evaluate whether differences between men and women could be better established in postoperative short-term outcomes after CABG, based on the evidence published in the past 5 years. For this, we analysed mortality after CABG, and also short-term outcomes, including the occurrence of stroke, myocardial infarction, and postoperative atrial fibrillation, which are all associated with significant morbidity and mortality6,7,8,9.
Results
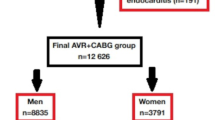
We included 23 studies, with a total of 3,971,267 patients (70.7% men, 29.3% women) (Table 1). Twenty-two of these studies analysed existent institutional, national, or multinational clinical registries and administrative databases. Being retrospective in nature, these studies covered evidence collected over a wide time span, some starting as early as 1998. One study analysed pooled individual patient data from four randomized controlled trials10, and one study performed a subgroup analysis of TiCAB randomized controlled trial11. Among the selected studies, 15 used the variable “sex” to differentiate between men and women, seven used the term “gender”, and one study used “sex” and “gender” interchangeably.
Postoperative mortality
We identified and pooled data from 13 studies reporting unadjusted short-term mortality (in hospital or at 30 days) in unbalanced groups, and from six studies comparing propensity matched groups of women and men. Our findings indicate that women undergoing CABG are at higher risk of short-term mortality compared to men (Fig. 1). For the unbalanced group, OR = 1.71 (95% CI 1.69–1.74), with low heterogeneity between studies (I2 = 0%, p = 0.70). In contrast with the unbalanced groups, the propensity matched groups showed significant heterogeneity (Fig. 1) and a pooled effect OR = 1.32 (95% CI 1.14–1.52, I2 = 76%, p < 0.01). The mixed effects analysis between the two groups indicated a statistically significant effect difference (p < 0.01) (Supplementary material, Figs. S1–S3).
Postoperative stroke
In the analysis of postoperative stroke, reported as either within hospital stroke or within 30 days from surgery, we included five studies that reported effects in propensity-matched groups of women and men, and 12 studies that reported unadjusted effects for postoperative stroke (both haemorrhagic or ischemic) after CABG (Fig. 2). Studies within both groups, propensity-matched and unbalanced, suffer from significant heterogeneity. The pooled effect estimates indicate in each case that women are at higher risk of stroke following CABG compared to men: OR = 1.50 (95% CI 1.35–1.66, I2 = 83%, p < 0.01), and OR = 1.31 (95% CI 1.02–1.67, I2 = 81%, p < 0.01). There was no statistically significant difference between the effects of the two groups (p = 0.32) (Supplementary material, Figs. S4–S6).
Forest plots showing pooled odds ratios (OR) for postoperative stroke (in hospital and 30-day results combined) following CABG, for each group, unbalanced and propensity-matched. When within-individual study outcomes were calculated separately by procedure, “_i” designates isolated CABG, and “_c” designates CABG concomitant with valve repair.
Postoperative myocardial infarction
We identified postoperative myocardial infarction in seven studies of the unbalanced group and in three studies of the propensity matched group. We included both ST-elevation myocardial infarction (STEMI) and non-ST elevation myocardial infarction (NSTEMI), as some studies did not specify the type of myocardial infarction. Our analysis indicated no statistically significant difference between women and men regarding the risk of myocardial infarction (Fig. 3). The pooled effects were OR = 1.09 (95% CI = 0.78–1.53, I2 = 70%, p < 0.01) and OR = 1.03 (95% CI = 0.86–1.24, I2 = 43%, p = 0.18), respectively. The I2 values indicate a moderate heterogeneity in each group. There was no statistically significant difference between the pooled effects of the two groups (p = 0.79) (Supplementary material, Figs. S7–S8).
Postoperative atrial fibrillation after CABG
We identified six unbalanced studies reporting postoperative atrial fibrillation after CABG (Fig. 4). The pooled effect estimate indicates that women are at lower odds of postoperative atrial fibrillation: OR = 0.89 (95% CI = 0.82–0.96, I2 = 34%, p = 0.18). I2 values showed low-to-moderate heterogeneity between the studies. In four of these studies women were significantly older than men (Vrancic et al., ter Woorst et al., Filardo et al., Gaudino et al.), while in two studies there was no significant difference in age between the two groups (Gurram et al., Matyal et al.). Sensitivity analysis revealed that the source of heterogeneity came from the study of Gaudino et al., which contributed with a heavy weight to the summary of effect and had narrow uncertainty intervals (Supplementary material—Figs. S9, S10). We identified one propensity matched study (Sajja et al.) which indicated an OR = 0.97 (95% CI 0.73–1.27, p = 0.836) for postoperative atrial fibrillation in women compared to men.
Discussion
We identified discrepancies and similarities between women and men within the short-term period following CABG. Considering short-term mortality, our meta-analysis indicates that women are at higher risk of short-term mortality after CABG, and this finding is consistent with previously published reviews2,3,4. It has already been shown that short-term mortality risk is significantly and persistently higher in women, even after adjusting for age and co-morbidities2,3,4.
Sexual dimorphism influences aging-related alterations, leading to distinct cardiac and vascular phenotypes and outcomes. Disparities in myocardial and vascular biology between sexes have been documented, resulting in varied cardiovascular risk profiles throughout life. The diversity in sex specificities extends beyond genetic codes and expression, to encompass differences in stress response and aging trajectories33. In the general population, women are at higher risk of stroke, with more severe neurological deficits compared to men34,35. Our analysis confirms that after CABG, women are also at higher risk of stroke, whereas there is no significant difference in risk of myocardial infarction between men and women. The aetiology of stroke following CABG is diverse36, as is the aetiology of myocardial infarction—acute graft failure, technical factors related to graft manipulation, and concomitant valve surgery being the main contributors for the latter37. Although it is known that differences in anatomical features (smaller vessels, smaller volume of blood in women) explain why procedural factors and inflammatory responses to surgery are different in women compared to men, currently there is still no precise understanding of the degree to which these differences contribute to the different outcomes. Compared with the meta-analysis on the impact of sex on outcomes after cardiac surgery by Dixon et al.4, we chose to extract and summarize unadjusted effects of sex, and so we were able to include a larger number of, and more recent studies reporting on sex-specific post-operative outcomes for stroke and myocardial infarction. Interestingly, our findings regarding the post-operative stroke and myocardial infarction both in the propensity matched groups and in the unbalanced groups are similar to the findings of Dixon et al.4 in the analysis of studies reporting adjusted odds ratios.
We provide a summary of the unadjusted effect of postoperative atrial fibrillation, as reported in studies which analysed sex-specific characteristics and outcomes. Postoperative atrial fibrillation denotes new-onset atrial fibrillation in the postoperative hospitalization period38. Our results indicate that men are at higher risk of postoperative atrial fibrillation after CABG, even though women undergoing CABG are generally older and carry more co-morbidities. These results are consistent with previous studies. In their adjusted analysis, Filardo et al.16 found that the incidence of postoperative atrial fibrillation after CABG was higher in men compared to women, and also men had longer duration of their first atrial fibrillation episodes. Gaudino et al.10 also indicated that men after CABG were at higher risk of postoperative atrial fibrillation compared to women, with an adjusted odds ratio (men as reference) = 0.82 (95% CI 0.73–0.94). Conversely, neither the adjusted analysis by Matyal et al.21 nor the propensity matched study by Sajja et al.32 reported any statistically significant difference regarding postoperative atrial fibrillation between men and women.
Postoperative atrial fibrillation is an important outcome to be addressed in the postoperative setting. Atrial fibrillation is associated with graft failure39. Although our analysis indicates that women are not particularly at elevated risk of postoperative atrial fibrillation compared to men, this occurrence is still of concern because atrial fibrillation carries a higher risk of stroke in women compared to men34,40,41. Numerous studies elaborate predictive models for postoperative atrial fibrillation42,43,44,45,46,47 and analyse sex as co-variates. When reports do not consider the predictors for postoperative atrial fibrillation as being sex specific, the value of the findings can be offset. For example, the size of the left atrium (LA) is known to differ subtly between men and women48. The LA size is often identified as an independent predictor of postoperative atrial fibrillation after CABG49,50,51,52,53. Not analysing sex-dependent thresholds in LA size might hinder the validity and applicability of the findings49,50,51,52. In contrast, when sex-specific thresholds in the postoperative enlargement of LA are taken into account, they can be valuable for risk stratification53.
Given the complex differences that delineate the female patient population from the male patient population in the context of CABG, it is necessary to develop studies in which the baseline and intra-operative characteristics are analysed separately, in a sex-specific and age-dependent manner. The post-operative outcomes could be associated with sex different thresholds. For example, Khalagy et al.54 indicated that the levels of preoperative haemoglobin associated with in hospital mortality after CABG are different in men compared to women. Developing more studies that provide a differentiated analysis for male and female patients is crucial. This approach will increase the chance of obtaining accurate predictive models separately for men and women, meaningful in clinical practice and particularly in secondary cardiovascular prevention.
Limitations of our study stem from the following: first, we included studies which combine outcomes for isolated CABG and CABG concomitant with valve surgery. Second, it was not possible to perform a separate analysis for elective and urgent CABG, nor for procedural factors (such as on pump and off pump CABG, number of grafts, and others). Third, the studies with a propensity matched design use different criteria for matching the cohorts of men with the cohorts of women. In addition, for short-term mortality and postoperative stroke, the propensity-matched group and the unbalanced studies might have had overlapping databases. Fourth, we could not make the difference between haemorrhagic or ischemic post operative stroke as in some studies that was not specified. Fifth, the criteria used to define myocardial infarction differed among the studies, which could have contributed to the observed heterogeneity, and we could not differentiate between STEMI or NSTEMI. Sixth, in several studies, there is no mentioned definition for postoperative atrial fibrillation in terms of duration or assessment method. Also, in most of the studies we selected for analysis, it was not possible to obtain information regarding the incidence of preoperative atrial fibrillation in men, compared to women.
Conclusion
In our meta-analysis, the summaries of effects from unbalanced groups and propensity matched groups indicate that the gap between women and men regarding the short-term postoperative mortality risk persists, and that women are at higher risk of postoperative stroke. The available evidence does not indicate that the likelihood of postoperative myocardial infarction is different in women compared to men. Despite women undergoing CABG being generally older and having more co-morbidities compared to men, men remain at higher risk of postoperative atrial fibrillation.
Methods
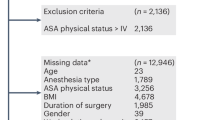
This meta-analysis was based on data extracted from previously published research and the data and study materials are available in the public domain. We performed a systematic literature search in PubMed database from January 1, 2019, to November 14, 2023. We used a combination of search terms (CABG, sex, gender, men, women, male, female, differences) and Boolean operators (“OR” for combining search terms for similar concepts, in parentheses, and “AND” to combine different concepts). Titles and abstracts were screened against the inclusion and exclusion criteria. Studies were included in the meta-analysis if they were observational studies or randomized clinical trials; published in English; presenting data regarding the following short-term outcomes after CABG: 30-day mortality, in hospital mortality, postoperative stroke (cardiovascular accidents), postoperative myocardial infarction (perioperative, early, in-hospital, and 30-day myocardial infarction), postoperative atrial fibrillation; reporting characteristics (baseline, intraoperative) and outcomes independently for males and females, following isolated CABG or CABG concomitant with other type of surgery; reporting effects unadjusted for baseline characteristics, or outcomes for propensity matched groups. Studies were excluded if they were protocols, reviews, case series, or referring to minimally invasive CABG. When source databases overlapped between the studies reporting the same category of outcomes, we selected and included in the statistical analysis only the study with the largest sample size. The flow chart depicting the study selection process is shown in Fig. 5.
The study was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for abstracting data and assessing data quality and validity55. Two independent reviewers (DDL and ET) selected studies by screening titles, abstracts, and full texts of articles identified in this search. For eligible studies, data extraction was performed by two reviewers (DL and PR). The quality of the studies was assessed with the Newcastle-Ottawa Scale56 (Supplementary file, Table S1).
The extracted data were baseline characteristics (being a man or woman, age at the time of CABG), procedural characteristics (elective or urgent CABG), and short-term outcomes: in hospital mortality, 30-day mortality, and 30-day or in hospital stroke, myocardial infarction, and postoperative atrial fibrillation. Criteria used in each selected study for defining myocardial infarction are presented in Supplementary file, Table S2. Except for one study (Filardo et al.16), no explicit criteria were used for defining postoperative atrial fibrillation, Table S3. After extracting the number of events from the individual studies, we computed the pooled odds ratios (OR) for women vs. men, with 95% confidence intervals (CI), using the generic inverse variance method. Group analysis was performed by study design type (unbalanced groups, propensity matched groups of women compared to men). For investigating variance between the groups, for each outcome, we used the mixed effects model, consisting in analysis within the group using the random effect model, and analysis between the groups using the fixed effects model. Heterogeneity was considered low if I2 < 25%, and significant if I2 > 75%. For groups with low heterogeneity, we performed a separate analysis using the fixed effect model. When studies overlapped in terms of source database, we included in the group only the study with a larger sample size (narrower uncertainty interval). Statistical significance was considered for p < 0.05. We used the R ’meta’ and ‘dmetar’ packages (R version 4.3.2, R Foundation for Statistical Computing, Vienna, Austria)57 and ‘metabin’ function58. To assess the robustness of our study, we conducted sensitivity analysis by leaving out studies with too broad or too narrow selection criteria, or outliers (Supplementary file). Funnel plots and Egger’s test were used to identify the publication bias (Supplementary file—Fig. S11).
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Melly, L., Torregrossa, G., Lee, T., Jansens, J. L. & Puskas, J. D. Fifty years of coronary artery bypass grafting. J. Thorac. Dis. 10(3), 1960–1967. https://doi.org/10.21037/jtd.2018.02.43 (2018).
Bryce Robinson, N. et al. Sex differences in outcomes following coronary artery bypass grafting: A meta-analysis. Interact. Cardiovasc. Thorac. Surg. 33(6), 841–847. https://doi.org/10.1093/icvts/ivab191 (2021).
Shi, D. et al. Higher mortality in women after coronary artery bypass: Meta-analysis and bias analysis of confounding. Ann. Thorac. Surg. 113(2), 674–680. https://doi.org/10.1016/j.athoracsur.2020.11.039 (2022).
Dixon, L. K. et al. Impact of sex on outcomes after cardiac surgery: A systematic review and meta-analysis. Int. J. Cardiol. 343, 27–34. https://doi.org/10.1016/j.ijcard.2021.09.011 (2021).
Harik, L. et al. Sex differences in coronary artery bypass graft surgery outcomes: A narrative review. J. Thorac. Dis. 15(9), 5041–5054. https://doi.org/10.21037/jtd-23-294 (2023).
Wagner, B. D. et al. Factors associated with long-term survival in patients with stroke after coronary artery bypass grafting. J. Int. Med. Res. 48(7), 300060520920428. https://doi.org/10.1177/0300060520920428 (2020).
Litwinowicz, R. et al. Long-term survival following postoperative myocardial infraction after coronary artery bypass surgery. J. Thorac. Dis. 14(1), 102–112. https://doi.org/10.21037/jtd-21-1279 (2022).
Lin, M. H. et al. Perioperative/postoperative atrial fibrillation and risk of subsequent stroke and/or mortality: A meta-analysis. Stroke 50(6), 1364–1371. https://doi.org/10.1161/STROKEAHA.118.023921 (2019).
Alex Chau, Y. L. et al. The impact of post-operative atrial fibrillation on outcomes in coronary artery bypass graft and combined procedures. J. Geriatr. Cardiol. JGC 18(5), 319–326. https://doi.org/10.11909/j.issn.1671-5411.2021.05.005 (2021).
Gaudino, M. et al. Sex differences in outcomes after coronary artery bypass grafting: A pooled analysis of individual patient data. Eur. Heart J. 43(1), 18–28. https://doi.org/10.1093/eurheartj/ehab504 (2021).
Sandner, S. et al. Sex differences among patients receiving ticagrelor monotherapy or aspirin after coronary bypass surgery: A prespecified subgroup analysis of the TiCAB trial. Int. J. Cardiol. 370, 129–135. https://doi.org/10.1016/j.ijcard.2022.10.166 (2023).
Gurram, A. et al. Female gender is not a risk factor for early mortality after coronary artery bypass grafting. Ann. Card. Anaesth. 22(2), 187–193. https://doi.org/10.4103/aca.ACA_27_18 (2019).
Johnston, A., Mesana, T. G., Lee, D. S., Eddeen, A. B. & Sun, L. Y. Sex differences in long-term survival after major cardiac surgery: A population-based cohort study. J. Am. Heart Assoc. 8(17), e013260. https://doi.org/10.1161/JAHA.119.013260 (2019).
Ter Woorst, J. F., van Straten, A. H. M., Houterman, S. & Soliman-Hamad, M. A. Sex difference in coronary artery bypass grafting: Preoperative profile and early outcome. J. Cardiothorac. Vasc. Anesth. 33(10), 2679–2684. https://doi.org/10.1053/j.jvca.2019.02.040 (2019).
Vrancic, J. M. et al. Is sex a risk factor for death in patients with bilateral internal thoracic artery grafts?. J. Thorac. Cardiovasc. Surg. 158(5), 1345-1353.e1. https://doi.org/10.1016/j.jtcvs.2019.01.025 (2019).
Filardo, G. et al. Postoperative atrial fibrillation: Sex-specific characteristics and effect on survival. J. Thorac. Cardiovasc. Surg. 159(4), 1419-1425.e1. https://doi.org/10.1016/j.jtcvs.2019.04.097 (2020).
Mahowald, M. K., Alqahtani, F. & Alkhouli, M. Comparison of outcomes of coronary revascularization for acute myocardial infarction in men versus women. Am. J. Cardiol. 132, 1–7. https://doi.org/10.1016/j.amjcard.2020.07.014 (2020).
Mohamed, W. et al. Trends in sex-based differences in outcomes following coronary artery bypass grafting in the United States between 2004 and 2015. Int. J. Cardiol. 320, 42–48. https://doi.org/10.1016/j.ijcard.2020.07.039 (2020).
Eris, C., Engin, M., Sunbul, S. A., As, A. K. & Erdolu, B. Early postoperative results of on-pump coronary endarterectomy: Is gender a risk factor?. Heart Surg. Forum 24(4), E662–E669. https://doi.org/10.1532/hsf.3929 (2021).
Kytö, V., Sipilä, J., Rautava, P. & Gunn, J. Sex differences in outcomes following acute coronary syndrome treated with coronary artery bypass surgery. Heart Lung Circ. 30(1), 100–107. https://doi.org/10.1016/j.hlc.2020.02.009 (2021).
Matyal, R. et al. Update: Gender differences in CABG outcomes—Have we bridged the gap?. PLoS ONE 16(9), e0255170. https://doi.org/10.1371/journal.pone.0255170 (2021).
Chang, F. C. et al. Sex differences in risks of in-hospital and late outcomes after cardiac surgery: A nationwide population-based cohort study. BMJ Open 12(2), e058538. https://doi.org/10.1136/bmjopen-2021-058538 (2022).
Dixon, L. K. et al. Females have an increased risk of short-term mortality after cardiac surgery compared to males: Insights from a national database. J. Card. Surg. 37(11), 3507–3519. https://doi.org/10.1111/jocs.16928 (2022).
Gerfer, S. et al. Gender-related propensity score match analysis of ECMO therapy in postcardiotomy cardiogenic shock in patients after myocardial revascularization. Perfusion 37(5), 470–476. https://doi.org/10.1177/02676591211004363 (2022).
Moroni, F. et al. Sex differences in outcomes after percutaneous coronary intervention or coronary artery bypass graft for left main disease: From the DELTA Registries. J. Am. Heart Assoc. 11(5), e022320. https://doi.org/10.1161/JAHA.121.022320 (2022).
Ter Woorst, J. F., Olsthoorn, J. R., Houterman, S., van Straten, B. H. M. & Soliman-Hamad, M. A. Sex difference in long-term survival after coronary artery bypass grafting is age-dependent. J. Cardiothorac. Vasc. Anesth. 36(5), 1288–1295. https://doi.org/10.1053/j.jvca.2021.08.104 (2022).
Qu, W. W., Wei, J. W., Binongo, J. N. & Keeling, W. B. Sex differences in failure-to-rescue after coronary artery bypass grafting. Ann. Thorac. Surg. 114(5), 1596–1602. https://doi.org/10.1016/j.athoracsur.2021.09.070 (2022).
Ram, E. et al. Coronary artery bypass grafting following acute coronary syndrome: Impact of gender. Semin. Thorac. Cardiovasc. Surg. 34(3), 920–929. https://doi.org/10.1053/j.semtcvs.2021.07.015 (2022).
Wang, E., Wang, Y., Hu, S. & Yuan, S. Impact of gender differences on hemostasis in patients after coronary artery bypass grafts surgeries in the context of tranexamic acid administration. J. Cardiothorac. Surg. 17(1), 123. https://doi.org/10.1186/s13019-022-01874-y (2022).
Daoulah, A. et al. Does gender affect the outcomes of myocardial revascularization for left-main coronary artery disease?. Angiology 75(2), 182–189. https://doi.org/10.1177/00033197231162481 (2023).
Gaudino, M. et al. Operative outcomes of women undergoing coronary artery bypass surgery in the US, 2011 to 2020. JAMA Surg. 158(5), 494–502. https://doi.org/10.1001/jamasurg.2022.8156 (2023).
Sajja, L. R., Mannam, G., Kamtam, D. N. & Balakrishna, N. Female gender does not have any significant impact on the early postoperative outcomes after coronary artery bypass grafting: A propensity-matched analysis. Indian J. Thorac. Cardiovasc. Surg. 39(3), 231–237. https://doi.org/10.1007/s12055-022-01465-5 (2023).
Ji, H. et al. Sex differences in myocardial and vascular aging. Circ. Res. 130(4), 566–577. https://doi.org/10.1161/CIRCRESAHA.121.319902 (2022).
Lang, C. et al. Do women with atrial fibrillation experience more severe strokes?: Results from the Austrian Stroke Unit Registry. Stroke 48(3), 778–780. https://doi.org/10.1161/STROKEAHA.116.015900 (2017).
Yoon, C. W. & Bushnell, C. D. Stroke in women: A review focused on epidemiology, risk factors, and outcomes. J. Stroke 25(1), 2–15. https://doi.org/10.5853/jos.2022.03468 (2023).
Gaudino, M. et al. Stroke after coronary artery bypass grafting and percutaneous coronary intervention: Incidence, pathogenesis, and outcomes. J. Am. Heart Assoc. 8(13), e013032. https://doi.org/10.1161/JAHA.119.013032 (2019).
Gaudino, M. et al. Considerations on the management of acute postoperative ischemia after cardiac surgery: A scientific statement from the American Heart Association. Circulation 148(5), 442–454. https://doi.org/10.1161/CIR.0000000000001154 (2023).
Perezgrovas-Olaria, R. et al. Differences in postoperative atrial fibrillation incidence and outcomes after cardiac surgery according to assessment method and definition: A systematic review and meta-analysis. J. Am. Heart Assoc. 12(19), e030907. https://doi.org/10.1161/JAHA.123.030907 (2023).
Han, Z., Zhang, G. & Chen, Y. Early asymptomatic graft failure in coronary artery bypass grafting: A study based on computed tomography angiography analysis. J. Cardiothorac. Surg. 18(1), 98. https://doi.org/10.1186/s13019-023-02199-0 (2023).
Emdin, C. A. et al. Atrial fibrillation as risk factor for cardiovascular disease and death in women compared with men: Systematic review and meta-analysis of cohort studies. BMJ 532, h7013. https://doi.org/10.1136/bmj.h7013 (2016).
Cove, C. L. et al. Female sex as an independent risk factor for stroke in atrial fibrillation: Possible mechanisms. Thromb. Haemost. 111(3), 385–391. https://doi.org/10.1160/TH13-04-0347 (2014).
Chen, Y. C. et al. Systemic immune-inflammation index for predicting postoperative atrial fibrillation following cardiac surgery: A meta-analysis. Front. Cardiovasc. Med. 11, 1290610. https://doi.org/10.3389/fcvm.2024.1290610 (2024).
Mekonen Gdey, M. et al. Predictors of developing postoperative atrial fibrillation in patients undergoing coronary artery bypass graft: A systematic review and meta-analysis. Cureus 15(12), e51316. https://doi.org/10.7759/cureus.51316 (2023).
Seo, E. J., Hong, J., Lee, H. J. & Son, Y. J. Perioperative risk factors for new-onset postoperative atrial fibrillation after coronary artery bypass grafting: A systematic review. BMC Cardiovasc. Disord. 21(1), 418. https://doi.org/10.1186/s12872-021-02224-x (2021).
Zhang, R. J. Z. et al. A prediction model for new-onset atrial fibrillation following coronary artery bypass graft surgery: A multicenter retrospective study. Heliyon 9(3), e14656. https://doi.org/10.1016/j.heliyon.2023.e14656 (2023).
Yang, Y. et al. Metabolic signatures in pericardial fluid and serum are associated with new-onset atrial fibrillation after isolated coronary artery bypass grafting. Transl. Res. 256, 30–40. https://doi.org/10.1016/j.trsl.2023.01.001 (2023).
Serafim, K. R. et al. The accuracy of the Stroke Risk Analysis (SRA) system for predicting atrial fibrillation in patients in the postoperative period of myocardial revascularization. PLoS ONE 18(3), e0282565. https://doi.org/10.1371/journal.pone.0282565 (2023).
Zemrak, F. et al. Left atrial structure in relationship to age, sex, ethnicity, and cardiovascular risk factors: MESA (Multi-Ethnic Study of Atherosclerosis). Circ. Cardiovasc. Imaging 10(2), e005379. https://doi.org/10.1161/CIRCIMAGING.116.005379 (2017).
Fan, K. et al. Predicting new-onset postoperative atrial fibrillation following isolated coronary artery bypass grafting: Development and validation of a novel nomogram. Int. J. Gen. Med. 15, 937–948. https://doi.org/10.2147/IJGM.S346339 (2022).
Rubanenko, O., Rubanenko, A. & Davydkin, I. Comprehensive analysis of factors associated with new episode of postoperative atrial fibrillation after coronary artery bypass graft surgery. Life 13(10), 2035. https://doi.org/10.3390/life13102035 (2023).
Zhang, H. et al. Development and validation of a diagnostic model based on left atrial diameter to predict postoperative atrial fibrillation after off-pump coronary artery bypass grafting. J. Thorac. Dis. 15(7), 3708–3725. https://doi.org/10.21037/jtd-22-1706 (2023).
Zhu, S., Che, H., Fan, Y. & Jiang, S. Prediction of new onset postoperative atrial fibrillation using a simple Nomogram. J. Cardiothorac. Surg. 18(1), 139. https://doi.org/10.1186/s13019-023-02198-1 (2023).
Karimi, A. et al. Left atrial size; a missing component in scoring systems for predicting atrial fibrillation following coronary artery bypass surgery. Acta Cardiol. Sin. 36(5), 456. https://doi.org/10.6515/ACS.202009_36(5).20181023A (2020).
Khalaji, A. et al. Mortality across the spectrum of hemoglobin level in patients undergoing surgical coronary revascularization. Clin. Cardiol. 46(5), 535–542. https://doi.org/10.1002/clc.24004 (2023).
Moher, D. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. Ann. Intern. Med. 151(4), 264. https://doi.org/10.7326/0003-4819-151-4-200908180-00135 (2009).
https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2023). https://www.R-project.org/.
RStudio Team. RStudio: Integrated Development for R (RStudio, PBC, 2020). http://www.rstudio.com/.
Funding
The study was supported by Valentine Gerbex-Bourget Foundation.
Author information
Authors and Affiliations
Contributions
D.D.L.: conceptualization of the study, screening and selection of the studies, data extraction, data analysis, wrote the original draft, reviewing and editing the manuscript. E.L.: screening and selection of the studies, contributions to the original draft, reviewing and editing the manuscript. P.R.: data extraction, reviewing and editing the manuscript. F.M.: reviewing and editing the manuscript. C.H.: conceptualization of the study, reviewing and editing the manuscript, acquired funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dumitriu LaGrange, D., Tessitore, E., Reymond, P. et al. A systematic review and meta-analysis of differences between men and women in short-term outcomes following coronary artery bypass graft surgery. Sci Rep 14, 20682 (2024). https://doi.org/10.1038/s41598-024-71414-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71414-2
Keywords
This article is cited by
-
Postoperative bleeding in coronary surgery: any differences associated with sex?
Indian Journal of Thoracic and Cardiovascular Surgery (2025)
-
Coronary artery surgery outcome differences by sex
Indian Journal of Thoracic and Cardiovascular Surgery (2025)
-
Relationship between gender and perioperative clinical features in lung cancer patients who underwent VATS lobectomy
Journal of Cardiothoracic Surgery (2024)