Abstract
We investigated the effect of repeated sessions of anodal transcranial direct current stimulation (a-tDCS) on subjective and objective measures of recovery, cognitive and sport-specific performance in professional soccer players following a soccer match simulation (SMS). Sixteen soccer players participated in this randomized, crossover, and sham-controlled study. They completed baseline assessments of well-being, total quality recovery (TQR), electromyographic activity (EMG) of the thigh muscles, countermovement jump (CMJ), and cognitive and Loughborough soccer passing test (LSPT) skills. Then, the participants engaged in an SMS routine (2 × 45 min, 15-min intervals). There was no significant difference in rating of perceived exertion (RPE) during the SMS in the anodal (17.25 ± 0.85) and sham (16.93 ± 0.92) conditions (p = 0.19). Following the SMS, the participants were randomized to receive three sessions of a-tDCS (2 mA, 20 min, +F3/−F4) targeting the left dorsolateral prefrontal cortex (DLPFC) or sham immediately after, 24 h, and 48 h after the SMS. Finally, the same outcome measures were evaluated 24 and 48 h following the SMS. A two-way repeated-measures ANOVA showed that a-tDCS stimulation improved passing skills (decreased time to perform the LSPT and number of errors; all ps < 0.01; d = 0.56–2.9) and increased the feeling of well-being (p = 0.02; d = 2.8), with no effect on TQR, cognitive performance, CMJ performance, and EMG. Therefore, the results of the present study indicate, for the first time, that repeated a-tDCS could be used as an adjunct method to accelerate soccer players' well-being and technical performance recovery, particularly after congested matches and/or training sessions. These findings may also be applicable to other team sports with characteristics similar to soccer (e.g., futsal, handball, basketball, etc.).
Similar content being viewed by others
Introduction
Soccer is a team sport and it is considered one of the most popular sports in the world. Professional soccer players engage in intense competitions and training workouts every year1. Players who participate in two games per week and have less than four recovery days between matches are at a higher risk of injury and experience an increased perception of pain, residual fatigue, reduced vigilance, and lower motivation to exercise in the days after the competition2,3. The chronic effect of this perturbation in the stress/recovery ratio is the potential development of non-functional overreaching or overtraining syndrome, which considerably reduces athletic performance for weeks or even months4,5,6. Therefore, recovery monitoring and recovery-enhancing interventions have attracted the attention of a large number of researchers and coaches in this field7. The most common practices aiming to speed-up recovery include the use of ergogenic aids, hydrotherapy, active recovery, stretching, compression garments, and massage8. Nevertheless, most of the abovementioned recovery methods target the periphery (different muscle groups) and overlook the role of the central nervous system (CNS) in the recovery process.
Considering the adverse consequences of central and cognitive fatigue after the competition as well as the importance of psychological responses to exercise9, strategies that can enhance the sense of well-being and central recovery are of interest. Mental fatigue, deficits in attention, and poor decision-making have also been reported after playing soccer10,11. This is particularly important given that studies showed that mental fatigue increases the perception of effort during physical exertion and reduces soccer-specific running capacity, short passing, and shooting performance12, decreases the speed and accuracy of soccer-specific decision-making tasks13. It has also been demonstrated that mental fatigue affects technical but not physical performance in youth players during soccer game-based situations14. Interestingly, despite the popularity of soccer, there is limited scientific evidence for the validity of recovery interventions for the neural system.
Transcranial direct current stimulation (tDCS) is one of the most common non-invasive brain stimulation techniques, that has been shown to have positive effects on different aspects of sport-related performance15,16,17. Indeed, the positive effects of tDCS on neuromuscular and whole-body endurance, strength, power output, cognitive function, affective valence, and perceptual responses have been reported18,19,20,21,22. These promising outcomes paved the path for further investigations regarding the efficacy of tDCS in other important facets of exercise performance such as CNS recovery after performing exercise-related fatiguing tasks. In this regard, Moreira et al.23 showed for the first time that adding a single session of anodal tDCS (a-tDCS) targeting the left DLPFC to a normal recovery practice on the day after the official soccer match could slightly improve perceived well-being compared to using the normal recovery practice alone in world-class female soccer players. In another study, Moreira et al.24 similarly found that a single session of a-tDCS alone, targeting the left DLPFC, in the morning following official matches improved the well-being and parasympathetic autonomic markers during the submaximal exercise of professional male soccer players. They concluded that tDCS might be considered a new recovery approach but further research with rigorous research design is needed to corroborate its efficacy in real situations23,24.
Nevertheless, despite these encouraging findings, it must be emphasized that the application of tDCS as a tool to speed-up recovery is in its infancy, and accordingly, more studies are warranted to take all the aspects of the issue into account and bridge the gaps in the current literature. For instance, it has been reported that the after-effects of a single session of tDCS might last up to 90 min25. Let’s bear in mind that the ultimate goal of an efficient recovery practice is to improve athletic performance within the days following matches and training sessions and, hence, from the practical point of view, it raises the question of whether applying a single session of tDCS could contribute to performance for a longer time. Interestingly, recent findings have suggested that multi-session tDCS might result in a cumulative after-effect lasting for days or even weeks, depending on how many consecutive sessions of tDCS were applied26,27,28. So, the effectiveness of multi-session tDCS as a recovery practice is an interesting question to be addressed. In addition, in those few studies investigating the efficacy of tDCS as a recovery tool, the subjective measures of recovery and some markers of the autonomic nervous system were the only variables measured in response to the tDCS intervention while no sport-related performance was measured, which leaves a gap as whether tDCS could improve not only perceived recovery but also performance recovery. Finally, a single measurement was performed in those studies making uncertain the time course of the recovery response.
Taken together, exercise-induced fatigue may affect both peripheral- and central-related aspects of fatigue, such as increased perception of physical and mental fatigue, pain, soreness, tiredness, weakness, decreased vigilance, alertness, motivation, and cognitive performance, all of which have an impact on athletes’ performance in subsequent training and/or competition sessions2,3,12,13,14. In this context, brain areas involved in higher hierarchy processing of numerous physiological signals, as well as cognitive processing known to be involved in exercise performance29,30,31, could be targeted with a-tDCS to modulate central elements of fatigue and, potentially, improve recovery. Hence, considering recent findings in this field and also the abovementioned gaps in the literature, the present study aimed to investigate the efficacy of repeated sessions of a-tDCS after a Soccer Match Simulation (SMS) on subjective measures of recovery (i.e., well-being and perceived recovery), cognitive, physical (countermovement jump), and sport-specific performance (passing performance) as well as neurophysiological parameters (electromyography; EMG) in professional soccer players using a repeated-measure design. We hypothesized that applying three consecutive sessions of tDCS (within three days) would improve well-being, perceived recovery, cognitive function, neurophysiological responses, physical and soccer-specific performance at 24 and 48 h after performing an SMS.
Methods
General experimental design
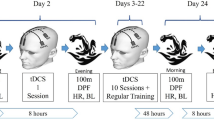
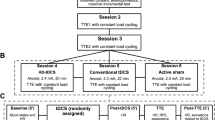
This was a randomized, crossover, counterbalanced, double-blinded, and sham-controlled study. Each participant came to the laboratory on eight different occasions. The first and second visits were designed to get participants acquainted with the study protocol and measure the baseline parameters, respectively. Then, in a crossover study design, the participants were randomly exposed to a-tDCS and sham tDCS conditions. Each condition consisted of three experimental sessions (six experimental sessions in total). During the first meeting, the participants were familiarized with the whole experimental procedure, performing the SMS, brain stimulation, and the measurements of the outcome variables. In the second visit, the baseline values of the study variables including the Well-Being Questionnaire (WBQ), Total Quality Recovery (TQR), cognitive function [color word Stroop test (CWST)], explosive power [countermovement jump (CMJ)], neurophysiological responses [EMG during maximal isometric voluntary contraction (MIVC) and CMJ], and soccer-specific performance [the Loughborough Soccer Passing Test (LSPT)] were measured in the respective sequence mentioned. At least 72 h after the baseline measures, the participants were randomly exposed to either anodal stimulation or sham stimulation conditions, each consisting three experimental sessions.
In the 1st session of each condition, participants first performed the SMS, and immediately after the SMS received 20 min of either anodal stimulation or sham stimulation. At 24 h after the SMS, participants returned to the lab for the 2nd experimental session and received 20 min of either anodal or sham stimulation again, according to the randomization. After the stimulation, the study variables were measured similar to the baseline measures. Finally, 48 h after the SMS, participants came to the lab for the 3rd experimental session. The procedure of the 3rd experimental session was the same as the 2nd experimental session including the respective tDCS followed by the variable measurements. A one-week wash-out was used between two experimental conditions. The efficacy of three consecutive sessions of tDCS and one week wash out period have already been proven scientific studies. Also, the time course of these three consecutive days was based on the common recovery approach in soccer in which the recovery process starts immediately after the match, and the first 2 days (+ 1, + 2) after the match is the recovery phase, the second 2 days (− 4, − 3) is the loading phase, and the last 2 days (− 2, − 1) before the next match is the tapering phase. Therefore, immediately after training, day + 1, and day + 2 are the most important days for recovery, and accordingly, we used these 3 days for our recovery strategy in the present study. After performing the SMS and during the 48-h phase of interventions and measurements, the participants did not take part in training sessions and also did not receive any other recovery interventions. During the one-week wash-out phase, they had their normal training routine. A 24-h paper-based dietary recall was used in the second session (baseline measures session) and the participants were instructed to follow the same diet 24 h before each experimental session. In addition, to prevent any confounding effects of circadian rhythm on the study variables, each participant came to the laboratory at the identical time of the day in a laboratory-controlled ambient condition (19–22 °C; 50–60% relative humidity) in all experimental sessions. The experimental procedure is depicted in Fig. 1.
Participants
Sixteen male soccer players from Under-21 Iran’s Premier League (two teams) and First National League (four teams) participated in this crossover, double-blinded, and sham-controlled study. The sample size was calculated a priori using the G*Power (Version 3.1.9.2, Kiel, Germany) for a repeated-measure ANOVA within factors as follows: α = 0.05; power (1−β err prob): 0.80; effect size f: 0.4, which was based on the studies measuring the efficacy of tDCS in sport-specific performance and recovery17,23; the number of groups = 1; correlation among repeated measures = 0.5, and non-sphericity correction = 1. According to the software’s output, 12 participants were adequate for this study; however, to cope with the high dropout rate in such studies22,32, 16 participants were recruited and voluntarily took part in our study.
The sample included one central defender, one attacker, five fullbacks, five midfielders, and four wingers. This study was conducted at the pre-season (individual training phase, before beginning the re-building phase) in which the players had at least six training sessions per week including aerobic, anaerobic, resistance, explosive power, and soccer-specific technical training. They also participated in some preparation matches during this period. The participants’ characteristics are presented in Table 1. The inclusion criteria were: (1) playing soccer for an official soccer club in Iran for at least 3 years; (2) having no implanted medical devices or pacemakers in the body; (3) having no acute and chronic cardiovascular or orthopedic diseases; (4) no tobacco, drug, and alcohol consumption; (5) no poor or deficient color vision. The exclusion criteria were: (1) not completing the study protocol or abandoning the study; and (2) injury related or not to the study that could influence the outcome variables. After being familiarized with the study protocol, written informed consent was obtained from each participant. The whole experimental procedure was conducted under the declaration of Helsinki. This study was approved by the Research Ethics Committee of the Iran Sports Science Research Institute (Approval Number: IR.SSRC.REC.1401.144).
Randomization, allocation, and concealment
The order of receiving two experimental conditions (anodal vs sham tDCS) was randomized and counterbalanced by an individual out of the main research team using a publicly available website (www.randomization.com) so that half of the sample received a-tDCS first followed by sham and the other half in the opposite order. tDCS was applied by an experienced researcher with tDCS who was not involved in the other steps of the study (i.e., a different outcome assessor). To do so, in each tDCS session, when the stimulation was about to start the main investigator (outcome assessor) would leave the laboratory and not come back until the stimulation was finished, the tDCS device was turned off, and the electrodes had been removed from the participant’s head. In addition, to blind the participants regarding the experimental condition they were receiving, the tDCS device was placed out of their sight and also was covered so that the participants could not see the device and any information on its display21.
Soccer match simulation (SMS)
We used an SMS based on a validated exercise protocol that simulates a soccer match in terms of physiological demands and technical actions33,34. This decision was based on the fact that in an actual soccer match, we could not control the distance covered, exercise intensities, physical and technical actions neither between players in a match nor within players in different matches, which could confound any result of the outcome measures. The SMS was performed individually and included two 45-min halves of soccer-specific activities separated by a 15-min passive recovery break as the half-time. Briefly, the exercise protocol was composed of 4.5-min blocks of soccer-specific intermittent activity and skills. Each block consisted of three repeated cycles of three 20-m walks, one walk to the side, an alternating timed 15-m sprint or a 20-m dribble, 4-s passive recovery periods, 5 × 20-m jogging at a speed corresponding to 40% VO2max, one 20-m backward jogging at 40% VO2max and 2 × 20-m running at 85% VO2max. One minute of soccer passing followed by one minute of recovery was completed after all blocks. The participants performed seven blocks during each half of the exercise protocol. A total distance of 10.1 km was covered by the participants in which 93 on-the-ball actions including 56 passes, 16 shots, and 21 dribbles were completed. The frequency of on-the-ball activities was similar to the real soccer games in the English Football Association Premier League. Every 15 min during the SMS (minutes 15, 30, 45, 60, 75, and 90) the participants reported their rating of perceived exertion [RPE (6–20 Borg scale)]. The mean value of the RPE during the SMS in two different conditions was used for data analysis.
Transcranial direct current stimulation (tDCS)
tDCS (NeuroStim 2, Medina Tebgostar, Tehran, Iran) was applied targeting to produce anodal stimulation over the left DLPFC (+ F3/− F4 montage)23,24. tDCS was administered through a pair of electrodes with a current intensity of 2 mA for 20 min with a 30-s ramp up/down, using a battery-driven constant electric stimulator. For stimulation, we used two silicone conductive electrodes (7 × 5 cm, 35 cm2; 0.057 mA/cm2) inside a sponge soaked in saline solution. The anode electrode was positioned vertically over F3 and the cathode over F4, based on the 10–20 international EEG system34,35. This montage was based on a computational modeling of the current induced by tDCS in the target region (see description below). The sham tDCS was applied using the same electrode placement but the current was turned off after 30 s of stimulation, with a charging period of 30 s at the beginning and end of the stimulation. We conducted the blind stimulation so that the players did not know what type of stimulation was being applied. Anodal tDCS was applied three times, immediately, 24 h, and 48 h after the SMS.
tDCS-induced electric field simulation
The brain current flow during tDCS was calculated using a finite element model following the standard pipeline in SimNIBS 4.0.036. The magnetic resonance imaging (MRI) MNI 152 head model available in the software was used. MRI data were segmented into surfaces corresponding to the white matter (WM), gray matter (GM), cerebrospinal fluid (CSF), skull, and skin. The electrical conductivities of each segment were determined according to values previously established as follows: WM = 0.126 Siemens/meter (S/m), GM = 0.275 S/m, CSF = 1.654 S/m, bone = 0.010 S/m, and skin/scalp = 0.465 S/m37, rubber electrode = 29.4 S/m, and saline-soaked sponges = 1.000 S/m. All information concerning the respective tDCS montages was entered into the software: current intensity = 2 mA; electrode position (+ F3/− F4); electrode and sponge sizes (5 × 7 cm); electrode thickness = 1 mm; sponge thickness = 5 mm. The results of the simulations are presented in Fig. 2, in terms of the electric field magnitude and radial electric field (normal to the cortical surface), both of which are the most important variables for tDCS to exert its neuromodulatory effects38. As can be seen in Fig. 2, the montage targeting the left DLPFC reached our nominal target with enough electric field magnitude (Fig. 2A–E) with inward current (Fig. 2F–H), which is expected to induce neuromodulatory effect39. The DLPFC tDCS montage also reached other prefrontal areas between the electrodes such as the bilateral frontopolar, ventromedial, and ventrolateral PFC, and right DLPFC (Fig. 2).
The magnitude and radial component of the electric field induced by tDCS. Analysis of tDCS-induced strength and radial component (normal to the cortical surface) of the electric field (EF) using a head model developed from magnetic resonance imaging (MNI152). Electrode montages targeting the left dorsolateral prefrontal cortex with anodal tDCS. Anodal (red rectangle; 5 × 7 cm) and cathodal (blue rectangle; 5 × 7 cm) electrodes were placed over the scalp (A and E). The magnitude of the EF is shown in panels (B–D) with hot colors (e.g., red) representing stronger EF and cold colors (e.g., blue) representing weaker EF. Panels (F–H) depict the radial EF, with red representing the electric current flowing into the cortex (i.e., inducing excitatory effects) and blue representing the electric current flowing out of the cortex (i.e., inducing inhibitory effects). Panels (D) show that the tDCS montage reached the target area with enough electric current magnitude to generate a neuromodulatory effect (blue circle roughly indicating the target location). Furthermore, as shown in panel (H), the target area was stimulated with the desired polarity (i.e., anodal current) to elicit excitatory effects in the target regions (blue circle roughly indicating the target areas).
Blinding assessment—tDCS-induced sensations
Participants completed a questionnaire provided by Fertonani et al.40 after each session, listing the sensations and level of intensity experienced during the stimulation. Itching, pain, burning, warmth/heat, pitching, metallic/iron taste, fatigue, and other sensations (open questions) were all listed on the questionnaire. The degrees were none (0), mild (1), moderate (2), considerable (3), and strong (4). Participants also indicated whether these sensations affected their ability to perform the exercise (1 = not at all; 2 = slightly; 3 = considerably; 4 = much; 5 = very much); when the discomfort started (1 = beginning; 2 = at about the middle; 3 = towards the end); and when it stopped (1 = stopped quickly; 2 = stopped in the middle; 3 = stopped at the end). The “discomfort” generated by tDCS was computed as the total of the strength scores recorded for all sensations so that the discomfort variable ranged from 0 (lack of discomfort) to 28 (maximum discomfort). In the last experimental session, participants were asked to answer the question pertaining to each session: “Do you think you received a real or placebo stimulation?” with response options of “real”, “placebo” or “I don’t know.” Considering that the end of the study correct guess rate (% of participants that successfully guessed their experimental condition) might result in a misleading interpretation of blinding effectiveness41,42, we reported the “active stimulation guess rate” (% of participants who guessed they received the active treatment) and consider the latter as the measure of blind effectiveness41.
Well-Being Questionnaire (WBQ) and Total Quality Recovery Scale (TQR)
The TQR scale was used to monitor the participants’ quality of the recovery subjectively23. The participants were instructed to choose a value between 6 (“very, very poor recovery”) and 20 (“very, very good recovery”) to assess their perceived level of recovery. The higher score on the TQR scale indicated a better quality of the recovery. We also used the WBQ in this study to assess general indicators of the participants’ fatigue, sleep quality, general muscle pain, stress level, and mood24. The WBQ consisted of five questions, each answerable on a five-point scale. The total WBQ score was determined by summing the scores of these five questions, with higher values representing better well-being24,43. The participants were familiarized with the TQR and WBQ during the familiarization session. They were asked to complete the TQR and WBQ at baseline, 24 h after the SMS, and 48 h after the SMS in a quiet room, by themselves to avoid any influence on their responses. At post-24 and post-48 after the SMS, the participants completed the questionnaire as soon as they came to the lab (before receiving the respective tDCS session post-24 and post-48 h following the SMS).
Color Word Stroop test
A computerized version of the Stroop test was used to assess cognitive performance44. In the familiarization session, the participants were acquainted with the test procedure, how to perform it, and the location of the keys on the keyboard. In this version of the Stroop test, four words (Yellow, Blue, Green, and Red) were displayed either in congruent color (e.g., the word “Blue” written in the blue color) or in incongruent color (e.g., the word “Green” written in the yellow color). A total of 96 stimuli including 48 congruent and 48 incongruent stimuli were intermixed and randomly displayed on the monitor. Each stimulus was presented for 2 s, with an interstimulus interval of around ~ 800 ms. On the keyboard, there were four buttons corresponding to four colors (Yellow, Blue, Green, and Red) used in the test. The participants were instructed to press the button corresponding to the color of the word displayed on the screen (ignoring the meaning of that word). For example, if the word “Blue” was displayed in “Green” color, the participants had to press the green button. This test measured selective attention and response inhibition. The interference effect was calculated by subtracting the score of the correct number of incongruent stimuli from the score of the correct number of congruent stimuli45. Higher scores indicated higher interference effects and poor response inhibition (inability to deliberately inhibit automatic or prepotent responses).
Countermovement Jump (CMJ)
All participants performed three maximal effort CMJ with a five-minute interval. The participants were asked to start from an upright standing position, make a preliminary downward movement by flexing at the knees and hips, and then immediately extend the knees and hips again to jump vertically up off the ground. The My Jump 2 IOS app was used to measure the height of a jump. The My Jump is a valid and reliable tool for measuring the height of different types of jumps46,47. The app was installed on an iPad.Pro 2021 and used to calculate the flight time of the jump by recording the take-off and landing frames of the video and then transforming it into a jump height47. The best measure was chosen for further analysis48.
EMG
The surface EMG signals were strictly collected according to the recommended standards49,50. An EMG device (Biometrics Datalite, UK) with a 1000 Hz frequency was used to determine the effectiveness of interventions on neuromuscular fatigue caused by SMS and also to record electromyography activity during jumping. After shaving and cleaning the electrode placement site on the skin with alcohol and sandpaper to reduce resistance, the electrode was put on the skin. The electrode and cables were fixed on the skin to prevent interference with the subject's movement. The electrode used to record the electric waves of the muscles during the jump was situated in the same place. In addition, electrode placement and EMG recording of the rectus femoris (RF) and vastus lateralis (VL) (showing the activity of the quadriceps muscle group) were performed based on the European protocol of non-invasive assessment of muscle activity using surface electromyography (SENIAM.ORG). The subject lies supine on the bed and bends his knee slightly using a pillow. In this position, using the meter, the electromyography sensor was placed at a distance of 50% between the upper part of the patella bone and the anterior superior iliac spine (ASIS) and in the direction of the muscle fibers for the RF muscle. The electromyography sensor for the VL muscle was positioned on the lower third of the connecting line between the outer part of the patella bone and ASIS, in the direction of the muscle fibers. EMG activity was recorded while performing the jump. The electromyography index was determined using the RMS/MVC ratio. MegaWin software (version 3.0.1) was utilized to analyze electromyography data.
MIVC
In each experimental session (baseline, post-24, and post-48), after cognitive testing and filling out the questionnaires, participants performed 3–5 s knee extension MIVC three times with a 150-s rest in between on a custom-made chair with knee and hip fixed at 90° as recommended for Vastus Lateralis (VL) and Rectus Femoris (RF) muscles MIVC test51. Verbal encouragement was provided to each participant during the test. During the MIVC test, EMG signals of the VL, and RF muscles were recorded. The results corresponding to the best MIVC were used for normalizing the EMG signals of that session.
Loughborough Soccer Passing Test (LSPT)
The LSPT test is a valid test that evaluates football technical performance52. This test involved 16 passes performed within a circuit of cones and grids in clockwise order as quickly and accurately as possible. The participants began with the ball in the central cone, and the first examiner recorded timing using a hand-held stopwatch when the ball was touched forward out of the inner rectangle. The second examiner was involved in determining the color of the next target (i.e. order of passes). The next target was called right before the participant finished the current pass. The same examiner was used in each role in all assessments so as to eliminate inter-experimenter variability. The order of passes was determined by one of four trial orders that were randomly generated by the investigators so that each trial consisted of eight long (green and blue) and eight short (white and red) passes. Participants were informed that passes could only be executed from within the passing area, between the marked lines. The ball had to cross two of the inner marked lines after retrieving from the previous pass before trying the next pass. Players were informed that to achieve the best performance on the LSPT, they must perform the test as quickly as possible while making the least number of errors. The second examiner paused the stopwatch at the last pass when it contacted the target area. No feedback was provided to the players during the test. Recording penalty time points that were accrued during the trials was the duty of the second examiner. Consequently, the examiner was in such a position that the four target sectors could be seen. These errors were penalized: 5 s for missing the bench completely or passing to the wrong bench; 3 s for missing the target area; 3 s for handling the ball; 2 s for passing the ball from outside of the designated area; 2 s if the ball touched any cone; 1 s for every second taken over the allocated 43 s to complete the test; and 1 s was deducted from the total time if the ball had hit the 10-cm strip in the middle of the target. Lastly, three indicators were utilized for statistical analysis. Movement time (the time to complete the test), number of errors, and total time. The total time encompasses both the actual time and the mentioned penalty times.
Rating of perceived exertion (RPE)
The 6–20 Borg scale was used to measure the RPE every 15 min during the SMS in two different conditions. The average of the RPEs during each SMS was used to compare the perceived intensity of each SMS. The RPE was also measured in three experimental sessions (baseline, post-24, and post-48) at the end of the LSPT test. Participants were familiarized with the Borg scale and received instruction on how to rate their perceived exertion. The subjects' answers were recorded individually, without the presence of other players. Their responses were used to calculate average RPE responses for each experimental condition and used for statistical analyses53.
Statistical analysis
Data are presented as the mean ± standard deviation (M ± SD). The Shapiro–Wilk test was used to evaluate the normal distribution of each data set. Two-way repeated measures ANOVA (2 × 3 factorial design; 2 stimulation conditions and 3-time points) was used to analyze TQR, CMJ, EMG, LSPT, and RPE during the LSPT. In the case of significant ‘condition × time’ interaction effect, the simple main effect of condition and time was analyzed and when necessary, the Bonferroni post hoc test was used for the pairwise comparisons. In case of a violation in the assumption of sphericity, the Greenhouse–Geisser epsilon correction was applied. When the assumption of normality was not met (WBQ and CWST), the Wilcoxon signed-rank test was used for comparing two conditions at each time point, and the Friedman test was adopted for comparing 3 time points within each condition. If significant results were obtained in the Friedman test, the adjusted Bonferroni correction was used for pairwise comparisons. Moreover, the Wilcoxon signed-rank test was used to compare the mean value of RPE between two conditions during the SMS. Partial eta squared (ɳ2p) was used as a measure of the effect size for the ANOVAs and interpreted as small (0.01–0.059), medium (0.06–0.139), or large (≥ 0.14). Cohen’s dav calculation of the effect size was also used for pairwise comparison and interpreted as small (0.20–0.49), medium (0.50–0.79), or large (≥ 0.80)54. The Friedman test was used to compare tDCS-induced sensations, followed by the Durbin-Conover test for multiple comparisons. The statistical analyses were performed using SPSS 23 (SPSS Inc., Chicago, IL, USA) and JAMOVI 2.4.11. A p ˂ 0.05 was adopted.
Results
tDCS-induced sensations and blinding
All 16 participants received the experimental conditions according to the randomization. There were no serious side or adverse effects reported. The most common sensations reported were itching and burning. Warmth/heat was reported but at a low frequency (< 20%) and one participant (6%) reported fatigue in one of the sessions of active tDCS. No other sensation beyond the ones reported in the questionnaire was reported. The location of the sensations was on the head for all participants, starting at the beginning of the stimulation and stopping quickly after it (Table 2). No statistically significant difference between conditions and session was found for any of the sensations (all ps > 0.24). All participants reported these sensations to affect their performance positively in all sessions. The percentage of correct guesses regarding the tDCS condition differed among conditions (χ2(5) = 53.0; p < 0.001), with all sessions of a-tDCS (81.3%, 87.5%, and 100%) presenting higher correct guesses than all sham sessions (18.8%, 6.3%; 12.5%; all ps < 0.001). This was because most participants (> 80%) thought they received a-tDCS (active stimulation guess rates) in all sessions, without difference among them (χ2(5) = 4.02; p = 0.547). Hence, considering the similar tDCS-induced sensations and active guess rate it can be assumed that the study blinding protocol was effective.
The overall results of the study are presented in Table 3.
Well-Being Questionnaire (WBQ) and Total Quality Recovery Scale (TQR)
There was no significant ‘condition × time’ interaction effect (F(2,30) = 2.63, p = 0.08, ɳ2p = 0.15, Power = 0.484) or main effect of condition (F(1,15) = 1.46, p = 0.24, ɳ2p = 0.089, Power = 0.206) and time (F(1.07,16.1) = 2.8, p = 0.11, ɳ2p = 0.157, Power = 0.361) on the TQR; Fig. 3A. There was no significant difference in the WBQ between the two conditions at baseline (z = − 1.47, p = 0.14) while the WBQ scores were significantly higher under anodal stimulation at post-24 (z = − 2.96, p = 0.003) and post-48 (z = − 3.55, p˂0.001) compared to the sham. Our results also demonstrated a significant difference in the WBQ over time (from baseline to post-48) under anodal stimulation (χ2(2) = 7.46, p = 0.024). Pairwise comparisons revealed that the WBQ was significantly higher at post-48 compared to the baseline (p = 0.017, dav = 2.8). Inversely, there was no significant difference in the WBQ under sham conditions over time (χ2(2) = 2.71, p = 0.25); Fig. 3B.
Comparison of perceived recovery (A), well-being (B), and cognitive performance (C) at baseline, 24 and 48 h after a soccer match simulation in the anodal and sham Transcranial Direct Current Stimulation conditions. * = statistically significant compared to sham at the same timepoint (p < 0.05); a = statistically significant compared to baseline within the same condition (p < 0.05).
Color Word Stroop Test (CWST)
There was no significant difference in the interference score of the CWST between the two conditions at baseline (z = − 0.45, p = 0.64), post-24 (z = − 1.64, p = 0.11), and also post-48 (z = − 0.81, p = 0.41). Likewise, no significant changes were observed in the interference score of the CWST over time (from baseline to post-48) neither in the anodal (χ2(2) = 3.82, p = 0.14) nor sham conditions (χ2(2) = 0.89, p = 0.64); Fig. 3C.
Countermovement Jump (CMJ)
The results showed that there was no significant ‘condition × time’ interaction effect (F(2,30) = 2.34, p = 0.11, ɳ2p = 0.135, Power = 0.438), no significant main effect of condition (F(1,15) = 0.09, p = 0.76, ɳ2p = 0.006, Power = 0.06), and no significant main effect of time (F(2,30) = 0.87, p = 0.42, ɳ2p = 0.055, Power = 0.187) on the CMJ; Fig. 4A.
EMG
There was a significant main effect of condition (F(1,15) = 21.38, p ˂ 0.001, ɳ2p = 0.588, Power = 0.991) and time (F(2,30) = 9.47, p = 0.001, ɳ2p = 0.387, Power = 0.967) on the EMG of the VL muscle, with no significant ‘condition × time’ interaction effect (F(2,30) = 2.63, p = 0.08, ɳ2p = 0.15, Power = 0.484). Pairwise comparisons showed a higher normalized EMG amplitude under anodal condition compared to the sham (p˂0.001, dav = 0.59) and a higher EMG amplitude at post-24 compared to baseline (p = 0.01, dav = 0.74); Fig. 4B.
As for the EMG of the RF muscle, there was no significant main effect of time (F(2,30) = 0.51, p = 0.6, ɳ2p = 0.033, Power = 0.126) and ‘condition × time’ interaction (F(2,30) = 0.36, p = 0.69, ɳ2p = 0.024, Power = 0.104). However, a significant main effect of condition (F(1,15) = 8.33, p = 0.011, ɳ2p = 0.357, Power = 0.770) was observed showing a higher normalized EMG amplitude in anodal condition compared to the sham (p = 0.011); Fig. 4(C).
Loughborough Soccer Passing Test (LSPT)
There was a significant main effect of condition (F(1,15) = 5.55, p = 0.028, ɳ2p = 0.282, Power = 0.622) and time (F(2,30) = 9.23, p = 0.001, ɳ2p = 0.381, Power = 0.964), with no significant ‘condition × time’ interaction effect (F(2,30) = 2.51, p = 0.09, ɳ2p = 0.143, Power = 0.464) on the movement time of the LSPT. The main effect of the condition indicated that the movement time of the LSPT was significantly lower under anodal condition compared to the sham (p = 028). Moreover, pairwise comparisons revealed that the movement time of the LSPT was significantly lower at post-24 and post-48 compared to the baseline (p = 0.027, dav = 0.58, p = 0.003, dav = 0.56, respectively); Fig. 5A.
Comparison of performance in the Loughborough Soccer Passing Test (LSPT) in terms of movement time (A), total time (B), errors committed (C), and Ratings of Perceived Exertion (D) at baseline, 24 and 48 h after a soccer simulated match in the anodal and sham Transcranial Direct Current Stimulation conditions. * = statistically significant compared to sham at the same time point (p < 0.05); a = statistically significant compared to baseline within the same condition (p < 0.05).
Concerning the errors during the LSPT, there was a significant main effect of condition (F(1,15) = 56.6, p ˂ 0.001, ɳ2p = 0.791, Power = 1.000) and ‘condition × time’ interaction (F(1.3,20.04) = 7.47, p = 0.002, ɳ2p = 0.332, Power = 0.918), with no significant main effect of time (F(2,30) = 1.98, p = 0.15, ɳ2p = 0.117, Power = 0.378). According to the significant ‘condition × time’ interaction effect, simple main effect of condition and time was analyzed separately and the pairwise comparisons (comparing the errors during the LSPT between two conditions) showed that there was no significant difference at baseline (p = 0.07, dav = 0.71) while the number of errors was significantly lower at post-24 (p ˂ 0.001, dav = 2.55) and post-48 (p ˂ 0.001, dav = 2.88) under anodal condition compared to the sham. Analyzing the simple effect of time revealed that there was no significant difference in errors during the LSPT from baseline to post-48 under the sham condition while the number of errors was significantly lower at post-48 compared to the baseline (p = 0.003, dav = 1.58) and also post-24 (p = 0.024, dav = 1.05) under the anodal condition; Fig. 5B.
Concerning the total time of the LSPT, there was a significant main effect of time (F(1.3,20.2) = 8.49.6, p = 0.005, ɳ2p = 0.361, Power = 0.864), condition (F(1,15) = 28.28, p ˂ 0.001, ɳ2p = 0.653, Power = 0.999), and ‘condition × time’ interaction (F(1.4,21.9) = 13.3, p ˂ 0.001, ɳ2p = 0.470, Power = 0.995). According to the significant ‘condition × time’ interaction effect, the simple main effect of condition and time was analyzed separately and the pairwise comparisons (comparing the total time of the LSPT between two conditions) revealed that there was no significant difference at baseline (p = 0.053, dav = 0.75) while the total time of the LSPT was significantly lower at post-24 (p˂0.001, dav = 1.83) and post-48 (p ˂ 0.001, dav = 2.9) under anodal condition compared to the sham. Analyzing the simple effect of time indicated that there was no significant difference in total time of the LSPT from baseline to post-48 under the sham condition while the total time was significantly lower at post-24 (p = 0.001, dav = 0.85) and post-48 (p˂0.001, dav = 1.18) compared to the baseline under the anodal condition; Fig. 5C.
Rating of perceived exertion (RPE)
There was no significant difference in the average RPE during the SMS under the anodal (mean ± SD: 17.25 ± 0.85) and sham (mean ± SD: 16.93 ± 0.92) conditions (z = –1.29, p = 0.19).
The results showed no significant ‘condition × time’ interaction effect (F(2,30) = 1.12, p = 0.33, ɳ2p = 0.07, Power = 0.230) and the main effect of condition (F(1,15) = 1.29, p = 0.27, ɳ2p = 0.08, Power = 0.187) while there was a significant main effect of time (F(2,30) = 38.17.6, p˂0.005, ɳ2p = 0.718, Power = 1.000) on the RPE during the LSPT. Pairwise comparisons showed the RPE during the LSPT was lower at post-24 (p˂0.005, dav = 1.36) and also post-48 (p˂0.005, dav = 1.6) compared to the baseline; Fig. 5D.
Discussion
This study was conducted to determine the effect of repeated sessions of a-tDCS targeting the left DLPFC on subjective recovery (TQR and WBQ), neuromuscular recovery (CMJ and EMG), cognitive (Stroop test) and sport-specific technical performance (LSPT) of male soccer players after SMS. The main findings were that repeated sessions a-tDCS led to improvements in well-being and sport-specific technical performance, with no significant effects on perceived recovery, cognitive performance, jump height, and EMG of the thigh muscles. The findings indicate, for the first time, that repeated short-term stimulation of the left DLPFC can improve the technical performance and psychological state of professional soccer players after a soccer match.
We opted to use an SMS to have better control of the player's external load (i.e., distance covered, speed, soccer-specific technical actions), which could not be controlled for in actual soccer math, once it is a dynamic game, and it could vary between and within players. The protocol used in the present study simulates a soccer match in terms of physiological demands and technical actions33. In this way, we could ensure that all players would perform the same amount of exertion in both conditions. In fact, the RPE for the SMS was similar between the anodal (17.25 ± 0.85) and sham (16.93 ± 0.92) conditions, indicative of a perceived exertion of “hard/heavy” to “very hard”. Moreover, the standard deviation for both sessions indicates that the RPE response was consistent among players. This confirms that the protocol employed allows for an adequate analysis of the effect of tDCS without the confounder of the external load.
The novel finding of the present study was that repeated a-tDCS improved technical sport-specific performance, as assessed by the passing test performance. It should be noted that only two previous studies evaluated the effect of a single session of a-tDCS on sport- and exercise-related recovery and they measured only perceived recovery, well-being, and autonomic cardiac control in submaximal exercise23,24. Exercise-induced muscular fatigue might affect technical performance, such as ball skills such as passing and shooting performances55. However, in the present study, three sessions of a-tDCS targeting the left DLPFC improved aspects of the LSPT over time as well as compared to sham. This result indicates that tDCS could be used as a recovery strategy for speeding up the recovery of sport-specific technical performance. Interestingly, Alvino et al.56 demonstrated that one session of tDCS targeting the primary motor cortex (M1) before the tests did not improve the soccer-specific skills of novice soccer players. The difference between studies might be explained by the target of stimulation (M1 vs DLPFC), the number of sessions (1 vs 3 sessions), training status (beginner vs trained), and timing of measurement (before practice vs. after match).
One possible explanation for the improved passing skills after SMS in the a-tDCS condition could be attributed to improved cognitive performance. However, a-tDCS had no effect on cognitive performance as measured by the Stroop test interference, which evaluates selective attention (i.e., ignoring irrelevant information and targeting relevant information), inhibitory control (i.e., inhibiting prepotent/impulsive actions), and processing speed. Previous studies have shown that a-tDCS targeting the left DLPFC improved cognitive performance after a time to exhaustion test in normoxia19 and hypoxia20 and after repeated anaerobic sprints21. However, the timing of measurement might explain the different results. Moreover, it should be noted that we used only one test involving one cognitive domain (executive function) and other cognitive domains were not evaluated. Hence, the effect of tDCS on other cognitive domains during recovery might not be ruled out. It is noteworthy that this was the first study to measure the effect of tDCS on cognitive performance recovery and also to use cognitive performance as a dimension of recovery in sports. A recent review by Perrey57 recommended that the assessment of recovery in sports should be multidimensional and include the cognitive demand, which would be measured with different indicators such as subjective (i.e., self-reported), behavioral (i.e., measurements of performance in cognitive test), physiological and neurophysiological. Therefore, this study demonstrated that, first, a soccer match simulation does not seem to affect inhibitory control and selective attention, and, second, repeated a-tDCS did not improve cognitive performance of this cognitive ability during recovery after an SMS.
Another novel approach of the present study was to measure the effect of a-tDCS on physical performance recovery after an SMS, as measured by CMJ. Interestingly, CMJ remained unchanged from baseline to 48h in both sessions. Despite it would be expected that exercise-induced muscle fatigue would impair the players’ lower limbs explosive performance, it was not found in the present study. Remarkably, Robineau et al.58 found that the fatigue induced by a 90-min soccer game modeling impaired most physical qualities measured (i.e., reduced torque of the quadriceps and hamstrings muscle, sprinting speed, vertical jump) at half time and, most profoundly, after the game in amateur soccer players. However, while squat jump performance decreased, the CMJ was not affected by the soccer match fatigue. Our results align with those findings. Moreover, our results showed that repeated a-tDCS did not affect CMJ. One possible explanation for this finding is that CMJ involves the generation of force not only due to active protein properties, but it also involves the stretch–shortening cycle, in which elastic energy is accumulated on passive structures in the muscle during the stretching phase and released during the shortening phase when a fast transition occurs such as during the CMJ59. In this regard, a-tDCS targeting the left DLPFC might not be able to influence the mechanisms related to the stretch–shortening cycle, and, hence, did not improve performance on this measure. Consistent with our findings, Romero-Arenas reported that a single session of a-tDCS targeting the DLPFC had no effect on the CMJ of healthy young men60. On the other hand, Grosprêtre et al.61 showed that a single session of a-tDCS targeting M1, but not the left DLPFC, improved horizontal jump, squat jump, and CMJ performance in power athletes (i.e., Parkour). Similar results of improved Sargent jump and CMJ performance were found by other studies targeting M1 with a single session of a-tDCS62. Therefore, the lack of an effect of a-tDCS on CMJ depends on the target of stimulation and might be improved by M1 but no DLPFC a-tDCS.
Concerning the neuromuscular activity measurement during the CMJ, there was only the main effect of the condition, indicating that in the a-tDCS condition (irrespective of any time points) the EMG amplitude of the VL and RF was higher than sham tDCS, but there was no specific difference at specified time points between conditions. Studies have shown that soccer-induced neuromuscular fatigue may not alter the EMG amplitude in the short term, depending on the muscle and the match duration. For instance, Goodal et al.63 showed that VL and RF EMG were unaltered during the 90-min match, but the RF EMG decreased during the extra time. Robineau et al.58 found that the EMG of the quadriceps muscle decreased at half-time and the end of a 90-min soccer game modeling. It should be noted, however, that we could not find studies that measure the EMG beyond the day of the soccer match, which limits direct comparison. In addition, the EMG was measured during the CMJ test in the present study, while in the aforementioned studies it was measured during isometric or isokinetic muscle actions, which could also impact comparison. The fact that the EMG amplitude was higher under a-tDCS condition might indicate that tDCS targeting the left DLPFC might have increased the volitional drive to M1, which in turn increased its output to the active muscle, reflecting in increased EMG amplitude. A similar increase in EMG amplitude of the VL after an acute session of a-tDCS targeting the left DLPFC was demonstrated in a previous study with cycling exercise in hypoxia20. However, greater EMG amplitude under a-tDCS condition was not reflected in greater CMJ performance in the present study. Moreover, these results should be interpreted with caution since no interaction effect or specific time points differences between conditions were found.
Repeated sessions a-tDCS targeting the left DLPFC improved well-being at 24 and 48 h post SMS in the present study compared to sham. This result is in line with Moreira et al.24 that showed that an acute session of a-tDCS, using the same tDCS parameters as in the present study, significantly improved well-being and autonomic cardiac control during a submaximal running test ~ 12 h after an official soccer match in professional male soccer players. In a subsequent study, the same authors showed that a-tDCS with the same protocol in addition to active recovery improved the well-being of female soccer players ~ 14–16 h after official soccer matches23. It should be noted that in those studies tDCS was applied in a single session and less than 24h after the soccer match and the outcomes were measured immediately after tDCS, while in the present study, tDCS was applied three times (immediately after, 24h, and 48h after the SMS) and the outcomes measured at baseline, 24 and 48h post-match, which approximate to the reality to the post-exercise recovery dynamics. On the other hand, there was no effect of a-tDCS on perceived recovery in the present study. These results are in line with Moreira et al.64 who found no acute effect of a-tDCS with the same tDCS protocol as in the present study combined with an active recovery on TQR in female soccer players ~ 14–16 h after official soccer matches. Taken together, these results indicate that a single or repeated session a-tDCS targeting the left DLPFC improves well-being but not perceived recovery after a soccer match.
The limitations of the present study include the fact that a relatively small sample size was evaluated. However, the study still had enough power to detect some significant changes in well-being and sport-specific performance. Moreover, evaluating professional soccer players is difficult because athletes and their teams are not normally willing to participate in research projects, especially those of relatively long duration such as the present study. In addition, all measures were performed immediately after tDCS at 24 and 48 h, which might reflect both the acute effect of the last session as well as the summed effect of all previous tDCS sessions. However, it was performed in this way to decrease the duration of the experiment and avoid interfering with the players’ schedules. Only male soccer players were included in the sample, and the results might not be generalized for female athletes. Furthermore, the fact that we evaluated a soccer match simulation rather than an official match should be taken into account when generalizing the current results. However, it should be noted that a validated protocol that simulates the physiological and technical demands of a soccer match was used, which allowed for control of the external load (e.g., distance covered, intensities) and technical actions of the players, as well as the fact that all players performed the same amount of work in both conditions independent of their playing position, which would be impossible to control in an official soccer match. Finally, the lack of measurement of neurophysiological parameters and central fatigue might be seen as a limitation of the present study. These limitations should be considered when interpreting the present results. Future studies should consider these limitations as well as broaden the range of recovery-related variables analyzed, increase the time between stimulation and measurement of the outcome, and consider targeting concomitantly the left DLPFC and M1, as both regions could improve psychological, technical, and physical performance.
The strengths of the present study included the fact that we used a double-blinded, randomized, counterbalanced, crossover design, which makes a robust research design to test the effectiveness of tDCS. In addition, we expanded the range of variables analyzed compared to previous studies, which included neuromuscular activity and performance, cognitive and sport-specific technical performance, providing a more comprehensive picture of soccer match recovery. In addition, this is the first study testing the effectiveness of repeated session tDCS on recovery and one of the few studies using repeated tDCS in soccer players.
From a practical standpoint, given that players’ well-being is critical to maintaining the quality of training and physical performance of athletes, and that during congested fixtures, recovery time may be insufficient, three sessions of a-tDCS targeting the left DLPFC could be considered as an alternative option to speed up recovery after soccer matches or training sessions, as it improves technical passing performance and well-being. Furthermore, while we only evaluated soccer matches in this study, the findings may apply to other team sports with similar characteristics to soccer (e.g., futsal, handball, basketball, etc.).
Conclusion
Our findings suggest that three sessions of a-tDCS targeting the left DLPFC may improve professional male soccer players' well-being and sport-specific technical performance following a soccer match simulation while having no effect on perceived recovery, cognitive performance, jump height, or EMG of the thigh muscle. Thus, tDCS could be used as an adjunct method to accelerate soccer players' well-being and technical performance recovery, particularly after congested matches and/or training sessions.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ispirlidis, I. et al. Time-course of changes in inflammatory and performance responses following a soccer game. Clin. J. Sport Med. 18(5), 423–431 (2008).
Brownstein, C. G. et al. Etiology and recovery of neuromuscular fatigue following competitive soccer match-play. Front. Physiol. 8, 831. https://doi.org/10.3389/fphys.2017.00831 (2017).
Altarriba-Bartes, A., Peña, J., Vicens-Bordas, J., Milà-Villaroel, R. & Calleja-González, J. Post-competition recovery strategies in elite male soccer players. Effects on performance: A systematic review and meta-analysis. PLoS One. 15(10), e0240135. https://doi.org/10.1371/journal.pone.0240135 (2020).
Baghaei, S., Tadibi, V., Amiri, E. & Giboin, L.-S. Subjective and objective variables of overtraining syndrome in female soccer players: A longitudinal study. Sci. Sports 37(5–6), 459–467. https://doi.org/10.1016/j.scispo.2021.05.005 (2022).
Kargarfard, M., Amiri, E., Shaw, I., Shariat, A. & Shaw, B. S. Salivary testosterone and cortisol concentrations, and psychological societe francaise de medecine du sport overtraining scores as indicators of overtraining syndromes among elite soccer players. J. Sport Psuchol. 27(1), 155–160 (2018).
Meeusen, R. et al. Prevention, diagnosis, and treatment of the overtraining syndrome: Joint consensus statement of the European College of Sport Science and the American College of Sports Medicine. Med. Sci. Sports Exerc. 45(1), 186–205. https://doi.org/10.1249/MSS.0b013e318279a10a (2013).
Kinugasa, T. & Kilding, A. E. A comparison of post-match recovery strategies in youth soccer players. J. Strength Cond. Res. 23(5), 1402–1407 (2009).
Rey, E., Padrón-Cabo, A., Barcala-Furelos, R., Casamichana, D. & Romo-Pérez, V. Practical active and passive recovery strategies for soccer players. Strength Condition. J. 40(3), 45–57 (2018).
Van Cutsem, J. et al. The effects of mental fatigue on physical performance: A systematic review. Sports Med. 47(8), 1569–1588. https://doi.org/10.1007/s40279-016-0672-0 (2017).
Reilly, T. & Ekblom, B. The use of recovery methods post-exercise. J. Sports Sci. 23(6), 619–627 (2005).
Sun, H. et al. Effects of mental fatigue on technical performance in soccer players: A systematic review with a meta-analysis. Front. Public Health. 10, 922630. https://doi.org/10.3389/fpubh.2022.922630 (2022).
Smith, M. R. et al. Mental fatigue impairs soccer-specific physical and technical performance. Med. Sci. Sports Exerc. 48(2), 267–276. https://doi.org/10.1249/mss.0000000000000762 (2016).
Smith, M. R. et al. Mental fatigue impairs soccer-specific decision-making skill. J. Sports Sci. 34(14), 1297–1304. https://doi.org/10.1080/02640414.2016.1156241 (2016).
Badin, O. O., Smith, M. R., Conte, D. & Coutts, A. J. Mental fatigue: Impairment of technical performance in small-sided soccer games. Int. J. Sports Physiol. Perform. 11(8), 1100–1105. https://doi.org/10.1123/ijspp.2015-0710 (2016).
Machado, D. & Amiri, E. Letter to the editor regarding “Single-session anodal transcranial direct current stimulation to enhance sport-specific performance in athletes: A systematic review and meta-analysis”. Brain Stimul. 16(5), 1549–1550. https://doi.org/10.1016/j.brs.2023.08.008 (2023).
Maudrich, T., Ragert, P., Perrey, S. & Kenville, R. Single-session anodal transcranial direct current stimulation to enhance sport-specific performance in athletes: A systematic review and meta-analysis. Brain Stimul. 15(6), 1517–1529. https://doi.org/10.1016/j.brs.2022.11.007 (2022).
Machado, D. et al. Effect of transcranial direct current stimulation on exercise performance: A systematic review and meta-analysis. Brain Stimul. 12(3), 593–605. https://doi.org/10.1016/j.brs.2018.12.227 (2019).
Alix-Fages, C. et al. Anodal transcranial direct current stimulation enhances strength training volume but not the force-velocity profile. Eur. J. Appl. Physiol. 120(8), 1881–1891. https://doi.org/10.1007/s00421-020-04417-2 (2020).
Angius, L., Santarnecchi, E., Pascual-Leone, A. & Marcora, S. M. Transcranial direct current stimulation over the left dorsolateral prefrontal cortex improves inhibitory control and endurance performance in healthy individuals. Neuroscience. 419, 34–45. https://doi.org/10.1016/j.neuroscience.2019.08.052 (2019).
Etemadi, M. et al. Anodal tDCS over the left DLPFC but not M1 increases muscle activity and improves psychophysiological responses, cognitive function, and endurance performance in normobaric hypoxia: A randomized controlled trial. BMC Neurosci. 24(1), 25. https://doi.org/10.1186/s12868-023-00794-4 (2023).
Teymoori, H. et al. Effect of tDCS targeting the M1 or left DLPFC on physical performance, psychophysiological responses, and cognitive function in repeated all-out cycling: A randomized controlled trial. J. Neuroeng. Rehabil. 20(1), 97. https://doi.org/10.1186/s12984-023-01221-9 (2023).
Banaei, P., Tadibi, V., Amiri, E. & Machado, D. Concomitant dual-site tDCS and dark chocolate improve cognitive and endurance performance following cognitive effort under hypoxia: A randomized controlled trial. Sci. Rep. 13(1), 16473. https://doi.org/10.1038/s41598-023-43568-y (2023).
Moreira, A. et al. Effect of transcranial direct current stimulation on professional female soccer players’ recovery following official matches. Percept. Mot. Skills. 128(4), 1504–1529. https://doi.org/10.1177/00315125211021239 (2021).
Moreira, A. et al. Effect of tDCS on well-being and autonomic function in professional male players after official soccer matches. Physiol. Behav. 233, 113351. https://doi.org/10.1016/j.physbeh.2021.113351 (2021).
Alix-Fages, C. et al. Short-term effects of anodal transcranial direct current stimulation on endurance and maximal force production. A systematic review and meta-analysis. J. Clin. Med. 8, 4. https://doi.org/10.3390/jcm8040536 (2019).
Ke, Y., Liu, S., Chen, L., Wang, X. & Ming, D. Lasting enhancements in neural efficiency by multi-session transcranial direct current stimulation during working memory training. NPJ Sci. Learn. 8(1), 48. https://doi.org/10.1038/s41539-023-00200-y (2023).
Guo, Z., Qiu, R., Qiu, H., Lu, H. & Zhu, X. Long-term effects of repeated multitarget high-definition transcranial direct current stimulation combined with cognitive training on response inhibition gains. Front. Neurosci. 17, 1107116. https://doi.org/10.3389/fnins.2023.1107116 (2023).
Ljubisavljevic, M., Maxood, K., Bjekic, J., Oommen, J. & Nagelkerke, N. Long-term effects of repeated prefrontal cortex transcranial direct current stimulation (tDCS) on food craving in normal and overweight young adults. Brain Stimul. 9(6), 826–833. https://doi.org/10.1016/j.brs.2016.07.002 (2016).
Robertson, C. V. & Marino, F. E. A role for the prefrontal cortex in exercise tolerance and termination. J. Appl. Physiol. (1985) 120(4), 464–466. https://doi.org/10.1152/japplphysiol.00363.2015 (2016).
McMorris, T., Barwood, M. & Corbett, J. Central fatigue theory and endurance exercise: Toward an interoceptive model. Neurosci. Biobehav. Rev. 93, 93–107. https://doi.org/10.1016/j.neubiorev.2018.03.024 (2018).
Machado, D. & Amiri, E. Critical considerations on tDCS-induced changes in corticospinal excitability and exercise performance: Should we go beyond M1?. J. Physiol. 601(23), 5453–5455. https://doi.org/10.1113/JP285507 (2023).
Bell, M. L., Kenward, M. G., Fairclough, D. L. & Horton, N. J. Differential dropout and bias in randomised controlled trials: When it matters and when it may not. BMJ. 346, e8668. https://doi.org/10.1136/bmj.e8668 (2013).
Russell, M., Rees, G., Benton, D. & Kingsley, M. An exercise protocol that replicates soccer match-play. Int. J. Sports Med. 32(7), 511–518. https://doi.org/10.1055/s-0031-1273742 (2011).
Shiravand, F., Amani-Shalamzari, S., Motamedi, P., Amiri, E. & Machado, D. The effect of transcranial direct current electrical stimulation on the recovery of neuromuscular and technical performance of young soccer players after a simulated soccer match. J. Appl. Exerc. Physiol. 18(36), 29–45. https://doi.org/10.22080/jaep.2023.25827.2151 (2023).
Jurcak, V., Tsuzuki, D. & Dan, I. 10/20, 10/10, and 10/5 systems revisited: Their validity as relative head-surface-based positioning systems. Neuroimage. 34(4), 1600–1611 (2007).
Thielscher, A., Antunes, A. & Saturnino, G. B. Field modeling for transcranial magnetic stimulation: A useful tool to understand the physiological effects of TMS?. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2015, 222–225. https://doi.org/10.1109/EMBC.2015.7318340 (2015).
Opitz, A., Paulus, W., Will, S., Antunes, A. & Thielscher, A. Determinants of the electric field during transcranial direct current stimulation. Neuroimage. 109, 140–150. https://doi.org/10.1016/j.neuroimage.2015.01.033 (2015).
Rahman, A. et al. Cellular effects of acute direct current stimulation: Somatic and synaptic terminal effects. J. Physiol. 591(10), 2563–2578. https://doi.org/10.1113/jphysiol.2012.247171 (2013).
Neri, F. et al. A novel tDCS sham approach based on model-driven controlled shunting. Brain Stimul. 13(2), 507–516. https://doi.org/10.1016/j.brs.2019.11.004 (2020).
Fertonani, A., Ferrari, C. & Miniussi, C. What do you feel if I apply transcranial electric stimulation? Safety, sensations and secondary induced effects. Clin. Neurophysiol. 126(11), 2181–2188. https://doi.org/10.1016/j.clinph.2015.03.015 (2015).
Fassi, L. & Cohen-Kadosh, R. Letter to the editor: How some brain stimulation studies fail to evaluate blinding adequately. J. Psychiatr. Res. 137, 452–453. https://doi.org/10.1016/j.jpsychires.2021.03.020 (2021).
Fassi, L. K. R. Is It All In Our Head? When Sunjective Beliefs About Receiving An Intervention Are Better Predictors Of Experimental Results Than the Intervention Itself (Springer, 2020). https://doi.org/10.1101/2020.12.06.411850.
McLean, B. D., Coutts, A. J., Kelly, V., McGuigan, M. R. & Cormack, S. J. Neuromuscular, endocrine, and perceptual fatigue responses during different length between-match microcycles in professional rugby league players. Int. J. Sports Physiol. Perform. 5(3), 367–383 (2010).
MacLeod, C. M. Half a century of research on the Stroop effect: An integrative review. Psychol. Bull. 109(2), 163 (1991).
Scarpina, F. & Tagini, S. The Stroop color and word test. Front. Psychol. 8, 557. https://doi.org/10.3389/fpsyg.2017.00557 (2017).
Balsalobre-Fernández, C., Glaister, M. & Lockey, R. A. The validity and reliability of an iPhone app for measuring vertical jump performance. J. Sports Sci. 33(15), 1574–1579. https://doi.org/10.1080/02640414.2014.996184 (2015).
Gallardo-Fuentes, F. et al. Intersession and intrasession reliability and validity of the my jump app for measuring different jump actions in trained male and female athletes. J. Strength Cond. Res. 30(7), 2049–2056. https://doi.org/10.1519/jsc.0000000000001304 (2016).
Cormie, P., McGuigan, M. R. & Newton, R. U. Developing maximal neuromuscular power: Part 1—biological basis of maximal power production. Sports Med. 41, 17–38 (2011).
Tankisi, H. et al. Standards of instrumentation of EMG. Clin. Neurophysiol. 131(1), 243–258 (2020).
Verdugo, R. J. & Matamala Cappon, J. Clinical Neurophysiology Standards of Emg Instrumentation: Twenty Years of Changes (Springer, 2020).
Rouffet, D. M. & Hautier, C. A. EMG normalization to study muscle activation in cycling. J. Electromyogr. Kinesiol. 18(5), 866–878. https://doi.org/10.1016/j.jelekin.2007.03.008 (2008).
Ali, A. et al. Reliability and validity of two tests of soccer skill. J. Sports Sci. 25(13), 1461–1470 (2007).
Impellizzeri, F. M., Rampinini, E., Coutts, A. J., Sassi, A. & Marcora, S. M. Use of RPE-based training load in soccer. Med. Sci. Sports Exerc. 36(6), 1042–1047 (2004).
Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 4, 863. https://doi.org/10.3389/fpsyg.2013.00863 (2013).
Russell, M., Rees, G., Benton, D. & Kingsley, M. An exercise protocol that replicates soccer match-play. Int. J. Sports Med. 2011, 511–518 (2011).
Alvino, J. M. Neuromodulação por meio da Estimulação Transcraniana por Corrente Contínua e o desempenho no futebol (Springer, 2023).
Perrey, S. Training monitoring in sports: It is time to embrace cognitive demand. Sports Basel. 10, 4. https://doi.org/10.3390/sports10040056 (2022).
Robineau, J., Jouaux, T., Lacroix, M. & Babault, N. Neuromuscular fatigue induced by a 90-minute soccer game modeling. J. Strength Condition. Res. 26(2), 555–562. https://doi.org/10.1519/JSC.0b013e318220dda0 (2012).
Turner, A. N. & Jeffreys, I. The stretch-shortening cycle: Proposed mechanisms and methods for enhancement. Strength Condition. J. 32(4), 87–99. https://doi.org/10.1519/SSC.0b013e3181e928f9 (2010).
Romero-Arenas, S. et al. Transcranial direct current stimulation does not improve countermovement jump performance in young healthy men. J. Strength Condition. Res. 35(10), 2918–2921 (2021).
Grosprêtre, S. et al. Effect of transcranial direct current stimulation on the psychomotor, cognitive, and motor performances of power athletes. Sci. Rep. 11(1), 9731 (2021).
Lattari, E. et al. Can transcranial direct current stimulation improve muscle power in individuals with advanced weight-training experience?. J. Strength Condition. Res. 34(1), 97–103 (2020).
Goodall, S. et al. The assessment of neuromuscular fatigue during 120 min of simulated soccer exercise. Eur. J. Appl. Physiol. 117(4), 687–697. https://doi.org/10.1007/s00421-017-3561-9 (2017).
Moreira, A. et al. Effect of transcranial direct current stimulation on professional female soccer players’ recovery following official matches. Percept. Motor Skills. 128(4), 1504–1529 (2021).
Acknowledgements
We would like to appreciate the participants who collaborated with us to do this study.
Funding
This research supported by Kharazmi University.
Author information
Authors and Affiliations
Contributions
FSH, PM and SA-S designed the study. FSH and SA-S conducted the experiments. SA-S, DG-SM and EA analyzed the study. EA and DG-SM interpreted the data for the study. All authors made substantial contributions to the design of the work, drafted the work and revise it. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shiravand, F., Motamedi, P., Amani-Shalamzari, S. et al. Effect of repeated sessions of transcranial direct current stimulation on subjective and objective measures of recovery and performance in soccer players following a soccer match simulation. Sci Rep 14, 20809 (2024). https://doi.org/10.1038/s41598-024-71701-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71701-y
Keywords
This article is cited by
-
Multisession transcranial direct current stimulation and aerobic exercise synergistically improve food craving symptoms, impulsivity, and cognitive flexibility in women with overweight and obesity: a randomized controlled trial
International Journal of Behavioral Nutrition and Physical Activity (2025)
-
Effect of low-intensity resistance training at different levels of blood flow restriction on pain, proprioception and muscle strength in patients with knee osteoarthritis: a randomized controlled trial
BMC Musculoskeletal Disorders (2025)
-
Machine learning-guided high-definition transcranial direct current stimulation prevents cybersickness
Virtual Reality (2025)