Abstract
LL-37 can inhibit the growth of K562 cancer cells when it is conjugated with iron oxide nanoparticles. In this study, Fe3O4 nanoparticles were synthesized using the co-precipitation method and then modified with the LL-37 peptide through an NH2 bridge. The accuracy of the synthesis process was confirmed through various analytical tests, including FTIR, XRD, FESEM, and EDX. To assess the treatment's effectiveness, a viability test was carried out on K562 leukemia cells and normal peripheral blood mononuclear cells. In addition, flow cytometry and Hoechst staining were used to investigate the mechanism of action of the drug. The expression levels of the Bcl-2, Bax, and TP53 genes in the treated cells and the control group were measured using qRT-PCR. The results indicated that the size of the nanoparticles ranged between 34 and 40 nm. The NH2@LL-37@Fe3O4 nanoparticles more effectively inhibited the growth of cancer cells in a concentration-dependent manner, as compared to Fe3O4 alone. Further analysis revealed that apoptosis occurred through increased expression of TP53 and Bax genes compared to the Bcl-2 gene. Therefore, induction of apoptosis and inhibition of growth in K562 cells was attributed to the impact of iron oxide magnetic nanoparticles conjugated with the LL-37 peptide through the TP53/Bax/Bcl-2 pathway.
Similar content being viewed by others
Introduction
LL-37 is an antimicrobial peptide categorized as a cationic host defense peptide in humans and belonging to the cathelicidin and released from its precursor by serine proteases. LL37 has been shown not only to be effective against bacterial pathogens1, but also to have cancer development potential or anticancer properties. LL-37 is a C-terminal peptide released from the 18 kDa cathelicidin protein through proteolytic mechanisms. It is encoded by the CAMP gene in humans and consists of two leucine residues at the N-terminus, spanning 37 amino acid residues. LL-37, also known as hCAP-18, FALL-39, or CAMP, is a human cationic antimicrobial peptide with potential for cancer development or anticancer properties. It is expressed in various cells and tissues, including circulating neutrophils, bone marrow cells, skin epithelial cells, digestive tract, epididymis of the testes, mouth, tongue, esophagus, and mucosal epithelium of the colon and lung bronchi. Additionally, human cathelicidin plays a crucial role as both a direct and indirect antimicrobial agent in facilitating wound healing2. Studies have shown tumor-promoting effects in organs such as the ovary, lung, and breast, as well as tumor-inhibiting effects in the colon, stomach, blood, and oral malignancies3. The anticancer efficacy of LL-37 peptide fragment analogs has not been explored yet, mainly due to inadequate intracellular translocation4. Many studies have indicated that the membranes of cancerous cells are negatively charged, whereas the membranes of healthy cells are electrically neutral5. Malignant cells differ from non-cancerous cells in their ability to access the negatively charged lipid phosphatidylserine, which is located on the outer leaflet of their cell membranes. In addition, the membrane composition of cancer cells is distinct from that of non-cancerous cells, including differences in cholesterol structure and the presence of various anionic components6. Antimicrobial peptides such as LL37 have a positive charge and can interact with the negatively charged membranes of cancer cells through electrostatic forces. This interaction is made possible by their unique helical structure, which enables them to effectively break down the cancer cells. Numerous scientific studies have highlighted the consistent anti-cancer effects of LL-37 across various cancer models, emphasizing its importance as a versatile tool in cancer treatment strategies7. Recent studies have focused on exploring the anti-cancer properties of this material by attaching the peptide to various polymers and nanoparticles 4. It has been observed that iron oxide nanoparticles can improve the antimicrobial and anticancer activity of cathelicidin LL-37, showing potential for future applications in fighting infections and diseases8. These nanoparticles promote cell destruction by increasing oxidative stress and aiding drug accumulation in cancer cells9. The use of magnetic nanoparticles can help deliver drugs to specific tissues and destroy infected tissue by generating a magnetic field. Furthermore, these nanoparticles can manipulate a magnetic field to induce the formation of blood clots in cancerous blood vessels10. Numerous investigations have suggested different genetic pathways in the development of various cancer types, including leukemia. The regulation of gene expression associated with apoptosis poses significant challenges in the treatment of different cancer types. This change in gene activity can impact the effectiveness of medical treatments, making the treatment of cancer more complex for oncologists. It is crucial to understand these changes to develop more effective strategies for combating cancer. Scientists have observed heightened expression of the anti-apoptotic molecule BCL-2 gene in CML cells, emphasizing its significance as a potential therapeutic target in medical research11. The treatment of chronic myeloid leukemia (CML) is challenging due to the overexpression of pro-survival proteins BCL-2 and low levels of pro-apoptotic proteins. This imbalance causes the persistence of CML stem cells, making it difficult to effectively combat the disease. In CML cells with high levels of BCR-ABL gene expression, the expression of BAX, a protein kinase, is reduced. This reduction in BAX expression reduces the cells' sensitivity to cytotoxic stimuli, making them more resistant to treatment12. The protein p53 plays a crucial role in suppressing tumors. In healthy cells, p53 prevents abnormal cell division or survival when cells are under stress, serving as a reliable defense mechanism against cancer. Mutations in the TP53 gene, which provides instructions for making the p53 protein, can lead to the development and spread of cancer cells. The presence of intronic SNPs (single-nucleotide polymorphisms) linked to chronic myeloid leukemia (CML) progression and response to tyrosine kinase inhibitor (TKI) treatment has confirmed the connection between TP53 mutations and CML13. Several studies have shown an increase in gene expression in K562 leukemia cells14. The primary focus of this research is to investigate the impact of iron oxide nanoparticles that have been modified with cathelicidin LL-37 antimicrobial peptide on the suppression of chronic myeloid leukemia (CML) cell proliferation, and on the regulation of Bcl-2, Bax, and TP53 gene expression.
Materials and methods
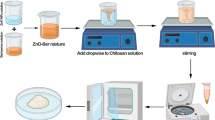
The graphical summary of the method is depicted in Fig. 1
Synthesis of Fe3O4 nanoparticles
Fe3O4 nanoparticles were synthesized using the co-precipitation method outlined in the study by Silveira et al.15. To begin, a solution was created by combining FeCl3.6H2O and FeCl2.4H2O salts at a 2 to 1 concentration ratio. This mixture was then added to 300 ml of distilled water and stirred under a nitrogen (N2) atmosphere at room temperature for ten minutes to remove any excess oxygen. Following this, NH4OH was gradually added drop by drop until the pH reached 9.5. The successful formation of iron oxide nanoparticles was indicated by the appearance of a black sediment. The system was then stirred at a speed of 12,000 RPM while being exposed to N2 bubbles for one hour. Subsequently, the nanoparticles were separated using a magnetic field and washed with double distilled water until the pH approached 7.0. Finally, the nanoparticles were dried in an oven at a temperature of 60 °C for three hours.
The Functionalization of iron oxide nanoparticles with cathelicidin antimicrobial peptide
The amidation reaction between the carboxyl group of the peptide and the primary amine group of MNP was performed using the Piktel method. This resulted in the stabilization of the LL-37 peptide on the nanoparticles with the primary amide group (Fe3O4@NH2)16. To achieve this objective, a solution was prepared by adding 0.6 g of Fe3O4@NH2 nanoparticles to a 100 ml aqueous solution of glutaraldehyde (25%). The mixture was then suspended using a shaker for 15 min to ensure a uniform solution. Following this, the magnetic nanoparticles were functionalized with glutaraldehyde through continuous stirring at room temperature for 6 h. The separation of the sediment was accomplished using a magnetic method. The resulting precipitate was subsequently washed three times with ethanol. Moving forward, the obtained precipitate and 1 mg/ml of cathelicidin LL-37 were combined with 10 ml of ethanol and stirred overnight. The particles were then magnetically separated, washed three times with ethanol, and finally dried at a temperature of 60 °C. To assess the synthesized compounds, confirmatory tests including FT-IR, XRD, SEM, and EDX were conducted.
Cell culture
The K562 cell line was sourced from the Iranian Center for Genetic Resources (ACECR). The cells were cultured according to the protocol detailed in the work of Ahmadi et al.17 in RPIM1640 medium (Biosera, England) supplemented with 10% fetal bovine serum (FBS; Biosera, England), 0.05 mg/ml penicillin G, and 0.05 mg/ml streptomycin. The cells were incubated at 37 °C and 5% carbon dioxide (CO2) atmosphere for 24 h.
Cell viability assay
The viability of cancer and normal cells after treatment with synthesized compounds was evaluated following the procedure outlined in recent research1,18,19,20. First, a DMSO solution was added to the RPMI-1640 medium and stored at − 20 °C for future use. Then, 5 × 103 K562 and PBMC cells were cultured in separate wells of a 96-well plate. After a 24-h incubation period, the MTT assay was performed to determine the most effective concentration of the substance for inhibiting cancer cells and to evaluate its toxicity towards peripheral blood mononuclear cells. Different concentrations (1500, 500, 250, 125, and 62.5 µ/mL) of the synthesized substances were tested on both cancer and normal cells to establish the range of drug efficacy. The optical density (OD) of the samples was measured at 540 nm, and the IC50 value was then calculated using GraphPad Prism V5 software. Cell viability was assessed based on the formation of blue formazan crystals, with the experiments being conducted in triplicate.
Evaluating the mechanism of cell death
Using the flow cytometry protocol, we investigated how drugs impact cancer cells21. The K562 cells were seeded at a concentration of 5 × 105 cells per ml in a six-well plate. The cells were then exposed to Fe3O4 nanoparticles, as well as a combination of nanoparticles and cathelicidin LL-37 at the IC50 concentration. After a 24-h incubation period, 200 µl of cells were harvested, centrifuged at 2000 g for 5 min at 4 °C, and the supernatant was removed. The cells were washed with 500 μL of cold PBS, centrifuged again, and the supernatant was discarded. Afterward, the cells were resuspended in 150 μl of Annexin V binding buffer and 5 μl of Annexin V-FITC in the dark for 15 min at room temperature. Propidium iodide (10 μl) was added to the samples and incubated for an additional 2 min at room temperature. Then, 350 μl of Annexin V binding buffer was added, and the samples were transferred to Falcon tubes. Dexamethasone and hydrogen peroxide were used as positive controls for apoptosis and necrosis, respectively. Finally, the samples were analyzed using a flow cytometry device from BD Inc., USA.
Hoechst 33,342 staining
The morphological features of apoptotic and necrotic cells were examined using Hoechst 33,342 dye following the Niemirowicz and C. Zhang technique1,22. A density of 5 × 104 K562 cells per well was seeded in 6-well plates. After the cells adhered to the wall, varying concentrations of TSN (0, 50, 100, and 200 μM) were added to each well. The cells were stained with Hoechst 33,342 (5 μg/mL) for 15 min in a light-proof environment after 24 h and then observed under a fluorescence microscope at 200× magnification. Afterward, the apoptotic cells in each field were counted and analyzed.
Quantitative reverse-transcriptase PCR assay (qRT-PCR)
The Bcl-2, Bax, and TP53 gene expression levels in the K562 cell line were evaluated according to the methodology by Chamani et al., following exposure to Fe3O4 nanoparticles and cathelicidin ll-37 conjugates23. In brief, K562 cells were collected after being exposed to synthesized compounds. The total RNA was extracted using the manufacturer's TRIZol reagent protocol (Invitrogen, USA). To create the first strand of cDNA, reverse transcriptase enzyme (Thermo Scientific) was used. The final sample volume of 20 µl consisted of 5 µl of deionized water, 10 µl of SYBR Green Master Mix (Thermo Fisher, England), 1 µl of the specific gene primer, and 4 µl of cDNA. These samples were then subjected to quantitative PCR using the StepOne™ Real-Time PCR System with the following thermal cycling conditions: initial denaturation at 95 °C for 5 min, followed by 45 cycles of denaturation at 95 °C for 10 s, annealing at 56 °C for 10 s, and extension at 72 °C for 10 s for PCR amplification. A final melting step was performed at 65 °C for 1 min. β-actin was used as an internal control to evaluate gene expression. The 2-ΔΔCt method was employed for quantitative analysis of gene expression. All reactions were performed in triplicate to validate the results. The primer sequence used is provided.:
Primers used for Bcl-2 gene:
Forward: 5′- CGGTTCAGGTACTCAGTCATCC -3′ Reverse: 5′- GAAATTGTGGAATTGATGCGTG -3′
Primers used for Bax gene:
Forward: 5′- CCCGAGAGGTCTTTTTCCGAG -3′ Reverse: 5′- CCAGCCCATGATGGTTCTGAT -3′
Primers used for TP53 gene:
Forward: 5′- CCCCTCCTGGCCCCTGTCATCTT -3′ Reverse: 5′- GCAGCGCCTCACAACCTCCGTC -3′
Primers used for β-ACT gene:
Forward: 5′- CTCCTTAATGTCACGCACGAT -3′ Reverse: 5′- CATGTACGTTGCTATCCAGGC -3′
Statistic analysis
The data analysis was performed using GraphPad Prism V5 software, and the results were presented as the mean ± standard deviation. Statistical differences between the treatment and control groups were assessed using an unpaired t-test24, with significance established at a level of p < 0.05.
Results
Characterization of magnetic nanoparticles modified with cathelicidin LL-37
The synthesized compound exhibited a burnt brown hue owing to the presence of iron oxide nanoparticles. After successfully identifying and confirming chemical tests, the compound was named NH2@LL-37@Fe3O4.
FTIR data analysis for LL-37@NH2@Fe3O4
Figure 2 shows the vibration peaks of Fe3O4 nanoparticles. The peaks at 462.25 cm−1 and 619.28 cm−1 indicate the presence of vibration in the Fe–O bond within the Fe3O4 nanoparticles25,26. The bands at 1418.92 cm−1 and 3440.36 cm−1 can be attributed to the O–H and N–H groups26, respectively. Moreover, the peak observed at 1577.86 cm-1 indicates the presence of the N–O group within the iron oxide nanoparticles27. Bands observed at 924.31 cm−1, 1012.96 cm−1, and 1047.23 cm−1 in the composition covering iron oxide nanoparticles provide evidence for the existence of C–H, C=C, and CO–O carbon groups attributed to the cathelicidin LL-37 peptide16.
The results of the XRD analyses for LL-37@NH2@Fe3O4 nanoparticles
The XRD spectrum of LL-37@NH2@Fe3O4 nanoparticles is shown in Fig. 3. The spectrum indicates the presence of reflective plates (220), (311), (400), (422), (511), and (440) at 2θ° angles of 18.15, 30.20, 35.50, 43.44, 57.6, and 63.1, respectively. These findings suggest that the nanoparticles have a cubic phase with filled faces at the centers. The diffraction patterns of these peaks match the data found in the JCPDS -750,033 code. To determine the size of the nanoparticles, Scherer's equation was used, which resulted in an estimated size of about 30 nm. These results align with the findings reported by Niemirowicz et al. 1.
FESEM results for LL-37@NH2@Fe3O4 nanoparticles
The morphology of LL-37@NH2@Fe3O4 nanoparticles was analyzed using a field emission scanning electron microscope (FESEM). The resulting images in Fig. 4 confirmed that the nanoparticles are spherical in shape. These nanoparticles showed a consistent morphology, with a size range of 34–40 nm. The variation in size, determined by the XRD method and Scherer's equation, compared to the sizes observed in the SEM images, can be attributed to the relative accumulation of the synthesized nanoparticles.
The size distribution of LL-37@NH2@Fe3O4 particles is shown in Fig. 5 with a histogram that follows a normal distribution curve. The close resemblance of the histogram curve to a Gaussian shape provides insight into the nanoparticles' structure, which resembles that of an octahedron28.
The EDX Analysis results to determine the constituent elements of LL-37@NH2@Fe3O4
Figure 6 shows the results of the EDAX analysis conducted on LL-37@NH2@Fe3O4 nanoparticles. The analysis revealed the presence and relative proportions of Fe, C, N, and O elements within the nanoparticles' structure. The presence of the Si element is likely due to APTMS, which was used during the synthesis of NH2@LL-37@Fe3O4. Additionally, the presence of the P element confirms the existence of phosphate groups in certain amino acids that make up cathelicidin. The absence of any other elements indicates the high purity of the synthesized compound.
LL-37@NH2@Fe3O4 inhibits the growth of leukemia K562 cells in a concentration-dependent manner
The MTT assay was used to evaluate the cytotoxicity and cell viability of K562 cells after being treated with LL-37@NH2@Fe3O4 and Fe3O4 nanoparticles separately (Fig. 7). The IC50 values for these compounds were determined based on the MTT results. The initial signs of the impact of both agents on K562 cells were observed at a concentration of 250 µg/ml. However, at concentrations ranging from 500 to 1500 µg/ml, the toxicity of LL-37@NH2@Fe3O4 compared to Fe3O4 escalated. Essentially, in these three concentrations, the efficacy of the composite compound was superior to that of Fe3O4 alone. The results of this assay indicated that both substances could demonstrate anti-proliferative effects on leukemia cells, but when iron oxide nanoparticles are combined with cathelicidin LL-37, their efficacy is enhanced against K562 cells. The IC50 values for LL-37@NH2@Fe3O4 and Fe3O4 were 1154 and 1929 μg/ml, respectively. The significant difference (P < 0.05) between the IC50 values of these two substances underscored the impact of LL-37 in augmenting cytotoxicity and reducing the survival of cancer cells. The data were expressed as the mean ± standard error (n = 0.05–0.1).
In the study of the effects of these two compounds on peripheral blood mononuclear cells (PBMC), it was observed that after 24-h treatment, there was a significant decrease in the survival rate once the concentration of LL-37@NH2@Fe3O4 reached 500 µg/ml (Fig. 8). Interestingly, the impact on cancer cells was less pronounced. Additionally, the IC50 value for this compound was found to be 1675 μg/ml, indicating lower toxicity in these cells compared to cancer cells. Notably, the results of the test carried out on normal cells treated with Fe3O4 showed a significant difference (P < 0.05). Accordingly, the IC50 value for these cells about this substance was determined to be 1802μg/ml. Essentially, it can be concluded that at higher concentrations, Fe3O4 can have a toxic effect on normal cells compared to NH2@LL-37@Fe3O4.
LL-37@NH2@Fe3O4 can induce apoptosis in cancer cells
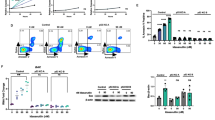
The confirmation of apoptosis induction by LL-37@NH2@Fe3O4 was achieved using Annexin V/PI staining. A comparison of the results obtained from the treated cells (IC50 concentration) (Fig. 9A) and the control group (Fig. 9B) showed that LL-37@NH2@Fe3O4 caused 13.39% early apoptosis (Q3), 22.61% late apoptosis (Q2), and 10.02% necrosis (Q1). Also, the survival rate of K562 cells was found to be 54.08%. It's worth noting that the combined percentage of apoptosis (Q2 + Q3) was 36%, as confirmed by the data presented in Fig. 9A
LL-37@NH2@Fe3O4 leads to morphological changes in K562 leukemia cells
In the process of apoptosis, we observed changes in cell morphology such as reduced volume, increased brightness and transparency, heightened density, and gradual nucleus fragmentation using Hoechst staining. Comparing the control group with treated cells (Fig. 10) revealed that cells in the control group were distinctly separated and strongly adhered to the culture plate, while treated cells showed distorted shapes and some displayed decreased volume and brightness. These changes became more pronounced with increased treatment.
LL-37@NH2@Fe3O4 nanoparticles increase the expression of the TP53 gene
The qRT-PCR test results showed that the LL-37@NH2@Fe3O4 magnetic nanoparticles caused a 1.184-fold increase in tp53 gene expression in the treated cells (IC50 concentration) relative to the control group, as illustrated in Fig. 11.
Bax gene expression is increased compared to BCL-2 in cells treated with LL-37@NH2@Fe3O4 nanoparticles
The results of the qRTPCR analysis are shown in Fig. 12. The figure illustrates the expression levels of the Bax and Bcl-2 genes in both the treated cells and the control group (IC50 concentration). The data analysis shows that the expression of the Bax gene is significantly higher compared to the Bcl-2 gene in the treated cells.
Discussion
Despite the use of treatment modalities such as chemotherapy, radiation therapy, and bone marrow transplantation, patients with blood leukemia continue to experience high rates of complications and mortality. As a result, researchers have shifted their focus towards creating more targeted and less harmful pharmaceuticals to improve the effectiveness of leukemia treatment.
Several studies have shown that using functionalized nanoparticles along with specific chemical compounds has been successful in treating blood leukemia. In a study by Loos et al., it was demonstrated that functionalizing polystyrene nanoparticles with amine groups (PS-NH2) halted the G2 phase of the cell cycle and inhibited the growth of certain blood leukemia cell lines29. Mori et al. showed that combining cathelicidin antimicrobial peptide LL-37 with lactic acid/glycolic acid copolymer (PLGA) enhanced its efficacy. This combination inhibited the proliferation, invasion, and migration of HM-1, B16/BL6, HeLa, and HepG2 cancer cell lines4. The research conducted by Ewelina Piktel emphasized the synergistic influence of seragenin CSA-131, an antimicrobial peptide, and iron oxide nanoparticles in promoting apoptosis in lung and colon cancer cells16. In another study, Mader et al. noted that LL-37 triggered apoptosis in Jurkat T leukemia cells by decreasing the levels of Bcl-2, while simultaneously increasing the levels of Bax and Bak30,31. In their research, Guo et al. found that using nickel nanoparticles functionalized with tetraheptylammonium, which carries a positive charge, led to significant cytotoxic effects by inducing apoptosis and necrosis in leukemia cells K562. The study also showed that these positively charged nickel nanoparticles could enhance cell membrane permeability, making it easier for target molecules to enter cancer cells6,32. Chen and colleagues showed that the antimicrobial peptide cathelicidin has inhibitory effects on the growth, movement, and spread of oral carcinoma HSC-3 cells. These effects are achieved by triggering cell death through caspase 3 and involving the TP53-Bcl-2/BAX signaling pathway22,33. The study aimed to achieve two main objectives: first, to observe the impact of treating cells with LL-37@NH2@Fe3O4 on cell growth inhibition, and second, to assess the changes in the expression of Bcl-2, Bax, and TP53 genes. The results of the study indicated that Fe3O4 and NH2@LL-37@Fe3O4, at a concentration of 250 µg/ml or higher, can hinder the growth of K562 cells. Notably, LL-37@NH2@Fe3O4 showed higher toxicity toward these cells compared to Fe3O4 at concentrations of 1000 and 1500 µg/ml, likely due to the presence of iron oxide nanoparticles. Moreover, the study confirmed the growth-inhibiting effects of both substances on blood leukemia cells, with the combination of cathelicidin LL-37 and iron oxide enhancing nanoparticle toxicity. Comparing the IC50 values between LL-37@NH2@Fe3O4 and Fe3O4 demonstrated the superior efficacy of the conjugated compound in increasing toxicity and reducing the survival of cancer cells. These findings are consistent with the research by K Niemirowicz et al., which highlighted the enhanced anticancer properties of the antimicrobial peptide LL-37 in conjunction with iron oxide nanoparticles in colon cancer cells1. The selectivity index of the synthesized compound was determined by dividing the IC50 value of normal cells by the IC50 value of cancer cells, as demonstrated by Habibi et al.34. A selectivity index greater than one suggests the efficacy of the drug. In the case of NH2@LL-37@Fe3O4, the selectivity index was determined to be 1.45, indicating that the necessary concentration of the drug to induce toxicity in healthy cells is 1.45 times higher than in cancer cells. A 36% increase in apoptosis was observed when comparing the effects of LL-37@NH2@Fe3O4 on treated cells rather than control cells. The intensified penetration of cathelicidin, facilitated by iron oxide nanoparticles, and the subsequent activation of the apoptotic pathway, are the key factors responsible for the enhanced toxicity of LL-37 on cancer cells. These findings were also corroborated by Piktel et al. in their investigation of colon cancer16,35. The second objective of the present study showed that cells treated with LL-37@NH2@Fe3O4 exhibited a 1.184-fold increase in TP53 gene expression compared to the control group. Additionally, cells treated with the combination showed a Bax to Bcl-2 gene expression ratio of 2.4 to 1.91, indicating higher Bax gene expression. The findings of Ren et al. indicate that cathelicidin (LL-37) induces the apoptosis pathway in Fk-16 colon cancer cells by increasing the transcriptional expression of TP53 and Bax genes, rather than affecting the level of Bcl-231,36. From a molecular and signaling perspective, TP53 acts as a transactivator in transcription-dependent pathways. It promotes the upregulation of pro-apoptotic genes like Bax, while concurrently inhibiting the increased expression of anti-apoptotic genes such as Bcl-2, thereby inducing apoptosis35,37. Therefore, the increase in TP53 levels in human malignancies can impede the abnormal expression of the Bcl-2 protein in various cancer types, leading to apoptosis by activating signals that promote an increase in Bax expression. The reduced levels of Bcl-2 expression observed in this study enhance our understanding of the mechanism behind cell death. The difference in the expression of Bax and Bcl-2 genes further confirms the induction of apoptosis in cancer cells K562 upon treatment with NH2@LL-37@Fe3O4.
Conclusion
This investigation aimed to evaluate the effectiveness of a new combination LL-37@NH2@Fe3O4 in inhibiting the growth of the k562 blood leukemia cell line and affecting the activity of TP53, Bax, and Bcl-2 genes. The results showed that the combination of iron oxide magnetic nanoparticles and cathelicidin LL-37 antimicrobial peptide could trigger apoptotic signaling pathways involving TP53/Bax/Bcl-2, leading to the suppression of K562 cell proliferation. While the study was limited to in vitro-based analyses, it is possible that these findings could be applied to in vivo studies on laboratory animals. Therefore, further research involving robust experiments in in vivo conditions is recommended.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Niemirowicz, K., Prokop, I., Wilczewska, A. Z., Wnorowska, U., Piktel, E., Wątek, M., et al. Magnetic nanoparticles enhance the anticancer activity of cathelicidin LL-37 peptide against colon cancer cells. International journal of nanomedicine. 3843–53 (2015).
Bowdish, D. M., Davidson, D. J., Scott, M. G. & Hancock, R. E. Immunomodulatory activities of small host defense peptides. Antimicrob. Agents Chemother. 49(5), 1727–1732 (2005).
Chen, X. et al. Human cathelicidin antimicrobial peptide suppresses proliferation, migration and invasion of oral carcinoma HSC-3 cells via a novel mechanism involving caspase-3 mediated apoptosis. Mol. Med. Rep. 22(6), 5243–5250 (2020).
Mori, T. et al. Enhancing the anticancer efficacy of a LL-37 peptide fragment analog using peptide-linked PLGA conjugate micelles in tumor cells. Int. J. Pharm. 606, 120891 (2021).
Rashid, K. & Ahmad, A. In vitro selective suppression of tumor cells by an oncolytic peptide in pancreatic ductal adenocarcinoma. Int. J. Pept. Res. Ther. 27, 863–873 (2021).
Wang, L., Dong, C., Li, X., Han, W. & Su, X. Anticancer potential of bioactive peptides from animal sources. Oncol. Rep. 38(2), 637–651 (2017).
Ruan, K., Song, G. & Ouyang, G. Role of hypoxia in the hallmarks of human cancer. J. Cell. Biochem. 107(6), 1053–1062 (2009).
Wnorowska, U. et al. Nanoantibiotics containing membrane-active human cathelicidin LL-37 or synthetic ceragenins attached to the surface of magnetic nanoparticles as novel and innovative therapeutic tools: Current status and potential future applications. J. Nanobiotechnol. 18, 1–18 (2020).
Srivastava, P., Sharma, P. K., Muheem, A. & Warsi, M. H. Magnetic nanoparticles: A review on stratagems of fabrication an d its biomedical applications. Recent Pat. Drug Deliv. Formul. 11(2), 101–113 (2017).
Gallo, J., Long, N. J. & Aboagye, E. O. Magnetic nanoparticles as contrast agents in the diagnosis and treatment of cancer. Chem. Soc. Rev. 42(19), 7816–7833 (2013).
Burger, J. A. Treatment of chronic lymphocytic leukemia. N. Engl. J. Med. 383(5), 460–473 (2020).
Narissa, P., Wheadon, H., Mhairi, C. The application of BH3 mimetics in myeloid leukemias. Cell Death Dis. 12(2), (2021).
Abdulmawjood, B., Costa, B., Roma-Rodrigues, C., Baptista, P. V. & Fernandes, A. R. Genetic biomarkers in chronic myeloid leukemia: What have we learned so far?. Int. J. Mol. Sci. 22(22), 12516 (2021).
Eslami, F. et al. Down-regulation of Survivin and Bcl-2 concomitant with the activation of caspase-3 as a mechanism of apoptotic death in KG1a and K562 cells upon exposure to a derivative from ciprofloxacin family. Toxicol. Appl. Pharmacol. 409, 115331 (2020).
Silveira, M., Silva, I. & Magdalena, A. Synthesis and characterization of Fe 3 O 4-NH 2 and Fe 3 O 4-NH 2-chitosan nanoparticles. Cerâmica 67, 295–300 (2021).
Piktel, E. et al. Recent insights in nanotechnology-based drugs and formulations designed for effective anti-cancer therapy. J. Nanobiotechnol. 14, 1–23 (2016).
Kafi-Ahmadi, L. et al. Co-precipitation synthesis, characterization of CoFe2O4 nanomaterial and evaluation of its toxicity behavior on human leukemia cancer K562 cell line. J. Chil. Chem. Soc. 65(2), 4845–4848 (2020).
Abbas, Z. S. et al. Galangin/β-cyclodextrin inclusion complex as a drug-delivery system for improved solubility and biocompatibility in breast cancer treatment. Molecules. 27(14), 4521 (2022).
Ibrahim, A. A. et al. Pt (II)-thiocarbohydrazone complex as cytotoxic agent and apoptosis inducer in Caov-3 and HT-29 Cells through the P53 and caspase-8 pathways. Pharmaceuticals. 14(6), 509 (2021).
Kadhim, R. J., Karsh, E. H., Taqi, Z. J. & Jabir, M. S. Biocompatibility of gold nanoparticles: In-vitro and In-vivo study. Mater. Today: Proc. 42, 3041–3045 (2021).
Kamaludin, N. F., Ismail, N., Awang, N., Mohamad, R. & Pim, N. U. Cytotoxicity evaluation and the mode of cell death of K562 cells induced by organotin (IV)(2-methoxyethyl) methyldithiocarbamate compounds. J. Appl. Pharm. Sci. 9(6), 010–015 (2019).
Zhang, C. et al. Mechanisms involved in the anti-tumor effects of Toosendanin in glioma cells. Cancer Cell Int. 21, 1–13 (2021).
Chamani, E. et al. Evaluation of some genes and proteins involved in apoptosis on human chronic myeloid leukemia cells (K562 cells) by datura innoxia leaves aqueous extract. J. Biomol. Struct. Dynam. 38(16), 4838–4849 (2020).
Jabir, M. S. et al. Inhibition of Staphylococcus aureus α-hemolysin production using nanocurcumin capped Au@ ZnO nanocomposite. Bioinorg. Chem. Appl. 2022(1), 2663812 (2022).
Granados Oliver, J., Reyes Pérez, M., Teja Ruiz, A., Palacios Beas, E., Pérez Labra, M., Barrientos Hernández, F. et al., editors. Characterization by FTIR of sphalerite obtained in the flotation without collector in the presence of ferric iron. Characterization of Minerals, Metals, and Materials 2020; (Springer: 2020).
Jabir, M. S., Nayef, U. M. & Kadhim, W. K. A. Polyethylene glycol-functionalized magnetic (Fe3O4) nanoparticles: A novel DNA-mediated antibacterial agent. Nano Biomed. Eng. 11(1), 18–27 (2019).
Smiri M, Guey F, Chemingui H, Dekhil A, Elarbaoui S, Hafiane A. Remove of humic acid from water using magnetite nanoparticles. European Journal of Advanced Chemistry Research. 2020;1(4).
Ooi, F., DuChene, J. S., Qiu, J., Graham, J. O., Engelhard, M. H., Cao, G., et al. A facile solvothermal synthesis of octahedral Fe3O4 nanoparticles. Pacific Northwest National Laboratory (PNNL), Richland, WA (United States; 2015).
Loos, C. et al. Amino-functionalized nanoparticles as inhibitors of mTOR and inducers of cell cycle arrest in leukemia cells. Biomaterials. 35(6), 1944–1953 (2014).
Mader, J. S., Mookherjee, N., Hancock, R. E. & Bleackley, R. C. The human host defense peptide LL-37 induces apoptosis in a calpain-and apoptosis-inducing factor–dependent manner involving bax activity. Mol. Cancer Res. 7(5), 689–702 (2009).
Ahmad, A. & Fawaz, M. A. M. The anticancer mechanism of human antimicrobial peptide LL-37. Neuropharmac. J. 6(3), 261 (2021).
Guo, D. et al. In vitro cellular uptake and cytotoxic effect of functionalized nickel nanoparticles on leukemia cancer cells. J. Nanosci. Nanotechnol. 8(5), 2301–2307 (2008).
Chen, X. et al. Roles and mechanisms of human cathelicidin LL-37 in cancer. Cell. Physiol. Biochem. 47(3), 1060–1073 (2018).
Habibi, A., Sadat Shandiz, S. A., Salehzadeh, A. & Moradi-Shoeili, Z. Novel pyridinecarboxaldehyde thiosemicarbazone conjugated magnetite nanoparticulates (MNPs) promote apoptosis in human lung cancer A549 cells. JBIC J. Biol. Inorg. Chem. 25, 13–22 (2020).
Ren, S. X. et al. Host immune defense peptide LL-37 activates caspase-independent apoptosis and suppresses colon cancer. Cancer Res. 72(24), 6512–6523 (2012).
Ren, S. X. et al. Correction: FK-16 derived from the anticancer peptide LL-37 induces caspase-independent apoptosis and autophagic cell death in colon cancer cells. PLoS One. 10(6), e0131750 (2015).
Soares, N. D. C. P. et al. Lycopene extracts from different tomato-based food products induce apoptosis in cultured human primary prostate cancer cells and regulate TP53, Bax and Bcl-2 transcript expression. Asian Pac. J. Cancer Prev.: APJCP. 18(2), 339 (2017).
Acknowledgements
The authors would like to thank to Dr. Vahid Askari for his help in molecular cell tests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
A.R.H: Supervisor and author of the introduction, working method, discussion and final review of the manuscript A.D: Performed molecular cell tests and contributed to writing the results section K. I: Supervisor and author of the introduction, working method, discussion and final review of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors agree for publication.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Habibi, A., Davari, A. & Isazadeh, K. A novel LL-37@NH2@Fe3O4 inhibits the proliferation of the leukemia K562 cells: in-vitro study. Sci Rep 14, 22245 (2024). https://doi.org/10.1038/s41598-024-71946-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71946-7
Keywords
This article is cited by
-
Cathelicidin LL37-loaded extracellular vesicles from Edwardsiella piscicida promote antibacterial and wound-healing activity
Scientific Reports (2025)
-
Enhanced therapeutic efficacy of silibinin loaded silica coated magnetic nanocomposites against Pseudomonas aeruginosa in Combination with Ciprofloxacin and HepG2 cancer cells
Scientific Reports (2025)