Abstract
Obesity has become a global health problem. In recent years, the influence of dietary microbes in the obese population has attracted the attention of scholars. Our study aimed to investigate the link between live microbe intake and obesity in adults. Participants (aged over 20 years) for this study were from the 1999–2018 National Health and Nutrition Examination Survey (NHANES). Participants were categorised into low, medium and high dietary live microbe intake groups. Linear regression was used to analyse the link between live microbe intake and body mass index (BMI) and waist circumference (WC). Logistic regression was used to analyse the link between live microbe intake and obesity and abdominal obesity prevalence. Restricted cubic spline curves (RCS) were used to check whether there was a non-linear relationship between live microbe intake and obesity. A total of 42,749 participants were included in this study and the number of obese reached 15,463. We found that live microbe intake was negatively linked to BMI and WC. In models adjusted for all confounders, the high live microbe intake group had lower obesity (OR = 0.812, 95%CI: 0.754–0.873) and abdominal obesity prevalence (OR = 0.851, 95%CI: 0.785–0.923) than the lowest intake group. Upon further quantification of live microbe intake, we found similar results. RCS analyses showed that live microbe intake was nonlinearly negatively correlated with BMI, WC, obesity, and abdominal obesity prevalence (P for non-linearity < 0.05). This study preliminarily reveals a negative link between live microbe intake and obesity in adults.
Similar content being viewed by others
Introduction
Today, obesity has become a global health challenge, affecting people’s quality of life1. Large population-based studies show that more than 200 million adults worldwide suffer from overweight2. The prevalence of obesity has doubled in more than 70 countries since 19803. Obesity is one of the major risk factors for a wide range of diseases, including cardiovascular disease (CVD), dyslipidemia, and cancers4,5,6. When exploring the factors contributing to the prevalence of obesity, attention is usually focused on dietary habits, lifestyle and genes7. In recent years, the link between obesity and microbial communities has been more thoroughly investigated by the scientific community8. The microbial communities in the body, especially the gut flora, are thought to play an essential part in the regulation of energy metabolism, nutrient absorption, and immune function9. Therefore, attention has been focused on the potential link between dietary intake of live microorganisms and obesity.
The microbial community in our body is the sum of the tiny organisms that inhabit our digestive system, skin, and other parts of the body, and the gut microbiota is one of the most widely studied categories10. The gut microbiota includes a wide variety of bacteria, fungi, and viruses, which are diverse and numerous11. In obesity research, scientists have found that the gut microbiota of obese individuals differs from that of normal-weight individuals12. Obese people’s gut microbiota is often less diverse and has a more particular bacterial composition13. These microbial changes may affect energy metabolism, appetite regulation, and weight control of the host14. The structure of the gut microbiota can be improved by dietary modification, especially by increasing the number of foods rich in live microorganisms, which can help reduce the risk of obesity15. As the understanding of the link between microbial communities and health continues to improve, a growing number of studies have started to draw attention to the link between the intake of live microorganisms and obesity8. Epidemiological studies have found that some foods rich in active probiotics or prebiotics, such as yoghurt, fermented foods, and high-fibre foods, are linked to a lower prevalence of obesity8. These foods not only improve the structure of the intestinal microbiota, but may also have a positive effect on reducing obesity by modulating energy metabolism and appetite regulation pathways16.
Dietary live microorganisms are defined as those microorganisms that continue to survive and exert beneficial effects in the human gastrointestinal tract, including probiotics and other beneficial microorganisms17. There has been a large body of research that demonstrates that probiotics may improve host health through modulation of the intestinal flora, including obesity, chronic kidney disease, and digestive disorders18,19,20. Previous research has focused on the effects of single probiotic supplements on adult obesity, with few studies finding a link between dietary intake of live bacteria and adult obesity. Therefore, we investigated the live microbe intake of U.S. adults who participated in the 1999–2018 National Health and Nutrition Examination Survey (NHANES), and explored the associations between live microbe intake and body mass index (BMI), waist circumference (WC), prevalence of obesity, and prevalence of abdominal obesity. By analysing the association between dietary live microbe intake and obesity, we can provide a scientific basis for the development of more scientific dietary recommendations and obesity prevention and control strategies.
Materials and methods
Study population
NHANES is an ongoing epidemiological survey conducted by the U.S. Centers for Disease Control and Prevention (NCHS). It describes the health and nutritional status of the U.S. population using a stratified, multistage probability cluster sampling methodology that provides important data for large population-based studies. These data include not only physiological measurements and health questionnaires, but also laboratory tests and nutritional surveys21. The NHANES study was approved by the NCHS Ethics Review Board and all participants provided informed consent.
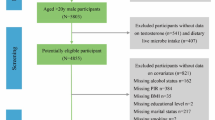
In the 1999–2018 NHANES, we collected basic information on 101,316 participants. We excluded participants with missing dietary information assessing dietary living microbe (n = 11876), participants with missing data on BMI and WC (n = 10187), participants with age < 20 (n = 32532), participants who were pregnant (n = 1343), and participants with extreme energy intake at baseline (n = 2926). A total of 42,452 participants were used for analysis (Fig. S1).
Assessment of live microbe intake
First, 24-hour dietary recall data were collected from the 1999–2018 NHANES. Second, we linked dietary information to the United States Department of Agriculture (USDA) Food Survey Nutrition Database via food codes. According to previous literature, commercially aseptic or pasteurised storage-resistant processed foods contain very low levels of live microbes (< 104 CFU/g), raw, unpeeled fruits and vegetables contain moderate levels of live microbes ((104-107 CFU/g), and unprocessed fermented foods often contain high levels of live microbes (> 107 CFU/g)22,23,24. In addition, we combined the amount of food consumed by each person with a medium and high level of live microbes and defined it as MedHi. Depending on the microbial levels, we categorised participants into three groups: low intake (all foods consumed were low); moderate (any food consumed was moderate, not heavy); and high (any food consumed was high). In order to quantify the intake of live microorganisms, we classified participants into three groups according to MedHi intake: G1, consumers who did not consume any MedHi food; G2, MedHi food intake higher than 0 but lower than the median level of consumers; and G3, those with MedHi food intake higher than the average level of consumers. Table S1 shows the distribution of participants’ live microbe intake in 1999–2018 NHANES.
Assessment of obesity
Individuals’ height (m), weight (kg), and waist circumference (WC, cm) were measured by standardized protocols. Body mass index (BMI, kg/m2) was calculated by dividing weight (kg) by the square of height (m). Obesity was defined as BMI > 30 kg/m225. We defined abdominal obesity as WC > 102 cm for male and WC > 88 cm for female26.
Covariates
Information on age, sex, race, education level, and total energy intakes (kcal/day) was collected. According to the marital status of participants, they are divided into married/living with partner and single/divorced/widowed27. The poverty income ratio (PIR) was calculated by dividing household income by factors specific to household size28. Never smokers were classified as those who reported smoking < 100 cigarettes during their lifetime. Those who smoked > 100 cigarettes in their lifetime were considered as current smokers, and those who smoked > 100 cigarettes and had quit smoking were considered as former smokers29. Drinking status was classified as nondrinker, low-to-moderate drinker (< 2 drinks/day in men and < 1 drink/day in women), or heavy drinker (≥ 2 drinks/day in men and ≥ 1 drinks/day in women)29. Physical activity was divided into three groups: inactive, insufficiently active (moderate activity 1–5 times per week with metabolic equivalents [MET] 3–6 or vigorous activity 1–3 times per week with MET > 6), and active30. We obtained information on the prevalence of hypertension, diabetes, hyperlipidemia, cardiovascular disease (CVD) using self-reported questionnaires. All dietary supplements containing probiotics or specific microorganisms were classified as probiotic dietary supplements, and their intake was estimated by a dietary supplement questionnaire24.
Statistical analysis
All data analyses were data weighted in accordance with the NHANES analysis guidelines. Continuous variables without a normal distribution are presented as medians [interquartile ranges]. Categorical variables are presented as numbers (percentages). Continuous variables were compared using the Mann-Whitney U test, while categorical variables were compared using the chi-square test. Multiple interpolation of missing covariates using the mice package.
Linear regression analyses were used to assess the link of live microbe intake with BMI and WC in adults. Logistic regression analyses were used to assess the link of live microbe intake with obesity and abdominal obesity prevalence in adults. A restricted cubic spline curve (RCS) was used to examine whether there was a non-linear link between live microbe intake and obesity-related variables. To ensure the stability of the results, we also performed a stratified analysis according to covariates age (18–39, 40–59, or ≥ 60), sex (male or female), race/ethnicity (Non-Hispanic White, Non-Hispanic Black or Other), marital status (married/living with partner, or single/divorced/widowed), education level (below high school, high school, or above high school), family PIR (≤ 1.0, 1.1–3.0, or > 3.0), drinking status (nondrinker, former drinker, or current drinker), smoking status (never smoker, former smoker, or current smoker), physical activity (inactive, insufficiently active, or active), total energy intakes (in quartiles), hypertension (yes, or no), diabetes (yes, or no), hyperlipidemia (yes, or no), and self-reported CVD (yes, or no) when they were not the strata variables. In sensitivity analyses, we repeated regression analyses of live microbe intake and obesity-related outcomes after further adjustment of nonfood prebiotic and probiotic use. All analyses were performed using R (version 4.3.2) and P-values less than 0.05 were considered statistically significant.
Results
Baseline characteristics of the participants
Table 1 shows the basic characteristics of participants by obesity. We included a total of 42,749 participants and the number of obese reached 15,463. The mean BMI of the participants was 27.65 kg/m2, and the mean WC was 97 cm. Low, moderate, and high live microbe intake were found in 32.57%, 42.00%, and 25.42% of participants, respectively. Further quantifying live microbe intake, 32.57%, 32.94%, and 34.49% of adults in the G1, G2, and G3 groups, respectively. Obese participants were more likely to be middle-aged or older women, non-Hispanic black, not educated beyond tertiary level, of low economic level, infrequent smokers, infrequent drinkers, and physically inactive (P < 0.05). Obese participants were more likely to have hypertension, diabetes, hyperlipidemia, and CVD (P < 0.05). Obese participants had a lower proportion of people with high live microbe intake compared to non-obese participants (P < 0.05). Tables S2 and S3 shows the socio-demographic and health status characteristics of participants according to the low, medium and high dietary live microbe intake groups and the G1, G2 and G3 groups, respectively.
Associations between live microbe intake and BMI and WC
Firstly, we explored the link between live microbe intake and BMI and WC (Tables 2 and 3). Linear regression analysis showed that live microbe intake was negatively linked to BMI (β = -0.246, 95%CI: -0.300, 0.193) and WC (β = -0.576, 95%CI: -0.706, 0.447). The negative correlation between live microbe intake and BMI (β=-0.239, 95%CI: -0.293, 0.186) and WC (β=-0.641, 95%CI: -0.767, 0.515) persisted after adjusting for age, sex and race. In the model adjusted for all confounders, each 1% increase in live microbe intake was linked to a decrease in BMI (β=-0.202, 95%CI: -0.253, 0.150) and WC (β=-0.497, 95%CI: -0.617, 0.377), respectively. RCS analyses showed a non-linear and negative correlation between live microbe intake and BMI and WC, with inflection points of 177.16 and 177.19 (P for non-linearity < 0.05) (Fig. 1A, B).
The exposure-response association of live microbe intake with BMI (A), WC (B), obesity (C) and abdominal obesity prevalence (D) among adults in NHANES 1999–2018 by restricted cubic spline (RCS). Analyses were adjusted for covariates age (18–39, 40–59, or ≥ 60), sex (male or female), race/ethnicity (Non-Hispanic White, Non-Hispanic Black or Other), marital status (married/living with partner, or single/divorced/widowed), education level (below high school, high school, or above high school), family PIR (≤ 1.0, 1.1–3.0, or > 3.0), drinking status (nondrinker, former drinker, or current drinker), smoking status (never smoker, former smoker, or current smoker), physical activity (inactive, insufficiently active, or active), total energy intakes (in quartiles), hypertension (yes, or no), diabetes (yes, or no), hyperlipidemia (yes, or no), and self-reported CVD (yes, or no) when they were not the strata variables.
Associations between live microbe intake and obesity and abdominal obesity prevalence
We explored the link between live microbe intake and obesity and abdominal obesity prevalence (Tables 4 and 5). Logistic regression analyses showed that obesity (OR = 0.755, 95%CI: 0.707–0.806) and abdominal obesity prevalence (OR = 0.880, 95%CI: 0.820–0.945) were significantly lower in the high dietary intake group compared with the low dietary intake group. After adjusting for age, sex, and race, obesity and abdominal obesity prevalence were 22.5% (OR = 0.775, 95%CI: 0.724–0.829) and 21.3% (OR = 0.787, 95%CI: 0.729–0.850) lower in the high live microbe intake group. In models adjusted for all confounders, the high live microbe intake group had lower obesity (OR = 0.812, 95%CI: 0.754–0.873) and abdominal obesity prevalence (OR = 0.851, 95%CI: 0.785–0.923) than the lowest intake group. After further quantifying dietary live microbe intake, we found that obesity and abdominal obesity prevalence were 26.3% (OR = 0.737, 95%CI: 0.683–0.796) and 23.2% lower in the G3 group compared to the G1 group (OR = 0.758, 95%CI: 0.712–0.829). RCS analyses showed that live microbe intake was negatively and non-linearly linked to obesity and abdominal obesity prevalence, with inflection points of 173.35 and 170.84 (P for non-linearity < 0.05) (Fig. 1C, D).
Stratified analyses
To test the stability of the link between live microbe intake and obesity-related outcomes, we conducted subgroup analyses among age, sex, race, marital status, education level, family PIR, smoking status, drinking status, physical activity, hypertension, diabetes, hyperlipidemia, and self-reported CVD. In the study of MedHi per 100 unit increase and obesity prevalence, age, race, education level, physical activity, and diabetes mellitus had some effect on the correlation (Fig. 2A). The negative link between MedHi per 100 unit increase and obesity prevalence was more pronounced in participants who were young, non-Hispanic white, with higher education, physically active, and non-diabetic. In the study of MedHi per 100 unit increase and abdominal obesity prevalence, age, sex, education level, physical activity, hypertension, and diabetes mellitus had some effect on the correlation (Fig. 2B). The negative link between MedHi per 100 unit increase and abdominal obesity prevalence was more pronounced in participants who were young males, had higher education, were physically active, non-hypertensive, and non-diabetic. In addition, age, sex, race, education level, physical activity, diabetes, and self-reported CVD had some effect on the correlation in the study of MedHi per 100 unit increase with BMI and WC (Fig. S2A, B).
Stratified analyses of the associations of live microbe intake with obesity (A) and abdominal obesity prevalence (B) among adults in NHANES 1999–2018. Analyses were adjusted for covariates age (18–39, 40–59, or ≥ 60), sex (male or female), race/ethnicity (Non-Hispanic White, Non-Hispanic Black or Other), marital status (married/living with partner, or single/divorced/widowed), education level (below high school, high school, or above high school), family PIR (≤ 1.0, 1.1–3.0, or > 3.0), drinking status (nondrinker, former drinker, or current drinker), smoking status (never smoker, former smoker, or current smoker), physical activity (inactive, insufficiently active, or active), total energy intakes (in quartiles), hypertension (yes, or no), diabetes (yes, or no), hyperlipidemia (yes, or no), and self-reported CVD (yes, or no) when they were not the strata variables.
Sensitivity analyses
In models further adjusted for non-food prebiotic and probiotic use, we found live microbe intake to be significantly and negatively linked to BMI (β = -0.206, 95%CI: -0.268, 0.144) and WC (β = -0.5, 95%CI: -0.648, 0.353) (Table S4). Using the low dietary live microbe intake group as the reference group, the high live microbe intake group was negatively linked to BMI (β=-0.616, 95%CI: -0.895, 0.337) and WC (β=-1.443, 95%CI: -2.119, 0.767). Using group G1 as the reference group, group G3 was negatively linked to BMI (β=-0.798, 95%CI: -1.066, 0.531) and WC (β=-2.008, 95%CI: -2.672, 1.344). Obesity (OR = 0.793, 95%CI: 0.715–0.878) and abdominal obesity prevalence (OR = 0.837, 95%CI: 0.747–0.939) were significantly lower in the high live microbe intake group compared to the low dietary live microbe intake group (Table S5). Participants in the G3 group had a 24.3% (OR = 0.757, 95%CI: 0.683–0.840) and 22.1% (OR = 0.779, 95%CI: 0.701–0.865) reduction in obesity and abdominal obesity prevalence compared to the G1 group. The results of the sensitivity analyses were similar to the results of our main analyses, suggesting that the stability of our study.
Discussion
We included a total of 42,749 participants, reaching 15,463 obese. The analyses showed a negative association between dietary live microbe intake and obesity-related outcomes. Specifically, obesity and abdominal obesity prevalence were lower in the high microbe intake group than in the lowest intake group. RCS analyses showed that live microbe intake was nonlinearly and negatively linked to obesity-related outcomes prevalence. Age, education level, and prevalence of diabetes may had some effect on the correlation between microbe intake and obesity-related outcomes.
Obesity has become a global issue as living standards have improved and eating patterns have changed. Previous research suggests that the global prevalence of obesity will be 18% in men and 21% in women by 202531. Obesity is not just a personal health issue, but also a significant risk factor for numerous chronic conditions32. Therefore, the study of obesity and its related factors has become more important. Obesity is a complex multifactorial disease, and its pathogenesis involves genetic, environmental and lifestyle aspects33. The role of the microbial community has received increasing attention when exploring strategies for the prevention obesity34. Over the past few decades, studies have demonstrated a strong link between the microbial community (gut flora) in the body and obesity35. Increased intake of live microorganisms is linked with the diversity of the gut flora, which in turn is strongly linked to the development of obesity. Consumption of foods rich in live microorganisms, such as yoghurt, promotes the diversity of the gut flora and reduces obesity prevalence8.
A systematic review that pooled 15 studies showed that probiotic supplementation reduced body weight and treated obesity-related complications compared to a placebo group19. A study evaluating the anti-obesity effects of Lactobacillus gasseri BNR17 in humans found that daily intake of BNR17 helped to reduce the amount of visceral fat in obese adult36. Hadi et al. showed that after eight weeks of continuous supplementation with probiotic preparations in obese or overweight adults, their TG, TC, LDL-C, and body weight were significantly reduced37. Kadooka et al. found that after supplementation with fermented milk containing Lactobacillus gasseri SBT2055, obese subjects had a significant reduction in visceral fat, subcutaneous fat, and body weight38. Sharafedtinov et al. found that supplementation of cheese containing the probiotic Lactobacillus plantarum improved BMI in obese hypertensive patients39. Mohammadi-Sartang et al. concluded that consumption of fortified yoghurt for 10 consecutive weeks reduced WC and body fat percentage and increased insulin sensitivity in obese subjects40. Although most studies have demonstrated the potential value of probiotics in the treatment of obesity, further trials are needed to ensure their anti-obesity effects in the population. In addition, food delivery matrices of probiotic formulations are more advantageous compared to direct administration of extracted microorganisms41. Gomes et al. found that supplementation with a probiotic mixture reduced abdominal obesity in adult females compared to dietary interventions alone42. Similarly, our study showed that dietary live microbe intake was negatively correlated with BMI and WC. In addition, obesity and abdominal obesity prevalence were significantly lower in the high microbe intake population.
The mechanisms underlying the link between live microbe intake and obesity are not fully understood, but there are several possible explanations. First, the intake of abundant active microbes helps to maintain the balance of gut flora helping to control obesity43.Quigley and other scholars have suggested that certain probiotics can produce bacteriocins or antitoxins, which inhibit the growth of pro-inflammatory microbes while promoting the growth of beneficial strains of microbes44. Second, active microbes influence host energy absorption and utilisation by participating in the metabolism of nutrients in the gut45. Turnbaugh et al. showed that the microbiota of the obese group had a significantly increased ability to derive energy from the diet35. Third, active microbes may reduce the incidence of obesity by reducing inflammatory responses46. Finamore et al. showed that supplementation with food-borne microbes can inhibit obesity-associated inflammation and reduce blood lipid levels47. Fourth, active microbes may reduce the incidence of obesity by affecting insulin sensitivity48. Insulin is a key hormone for controlling blood glucose levels, and obesity is often accompanied by insulin resistance. Kang et al. found that Lactobacillus acidophilus treatment reduced insulin resistance in mice on a high-fat diet while improving glucose and lipid metabolism49.
This study has several strengths. First, the data used in this study are from NHANES, a large, population-representative study that is highly robust. Second, we used a variety of classification methods and the direction of all results remained consistent, indicating that our results are stable and reliable. Third, we analysed the effect of food-based intake of live microbes on obesity-related outcomes, rather than the use of a particular probiotic supplement. However, there are some limitations of this study. First, dietary data were collected using the 24-hour recall method, which does not reflect daily dietary intake and may be subject to recall bias. Second, the estimation method of dietary microbiological content was based on previous studies and expert opinions rather than direct testing, which lacked some accuracy. Third, this was a cross-sectional study and could not provide a causal association between live microbe intake and obesity-related outcomes. Finally, our study is the lack of data on medication use, which is known to influence the gut microbiome and could potentially confound the observed associations between live microbe intake and obesity. Future research should consider the impact of medication intake to provide a more comprehensive understanding of this relationship.
Conclusion
We found a non-linear negative correlation between live microbe intake and BMI, WC, obesity and abdominal obesity prevalence in adults. Increased intake of dietary live microbes can help improve obesity-related outcomes in the population.
Data availability
NHANES data described in this manuscript are available at https://wwwn.cdc.gov/nchs/nhanes/.
References
Perdomo, C. M., Cohen, R. V., Sumithran, P., Clement, K. & Fruhbeck, G. Contemporary medical, device, and surgical therapies for obesity in adults. Lancet. 401(10382), 1116–1130 (2023).
Caballero, B. Humans against obesity: Who will win? Adv. Nutr. 10(suppl_1), S4–S9 (2019).
Collaborators, G. B. D. O. et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl. J. Med. 377(1), 13–27 (2017).
Owei, I., Umekwe, N., Provo, C., Wan, J. & Dagogo-Jack, S. Insulin-sensitive and insulin-resistant obese and non-obese phenotypes: Role in prediction of incident pre-diabetes in a longitudinal biracial cohort. BMJ Open. Diabetes Res. Care. 5(1), e000415 (2017).
Flegal, K. M., Panagiotou, O. A. & Graubard, B. I. Estimating population attributable fractions to quantify the health burden of obesity. Ann. Epidemiol.25(3), 201–207 (2015).
Fruhbeck, G. et al. Obesity: The gateway to ill health—An EASO position statement on a rising public health, clinical and scientific challenge in Europe. Obes. Facts. 6(2), 117–120 (2013).
Kim, D., Hou, W., Wang, F. & Arcan, C. Factors affecting obesity and Waist Circumference among US adults. Prev. Chronic Dis.16, E02 (2019).
Green, M., Arora, K. & Prakash, S. Microbial medicine: Prebiotic and probiotic functional foods to target obesity and metabolic syndrome. Int. J. Mol. Sci.21(8), 2890 (2020).
Backhed, F., Ley, R. E., Sonnenburg, J. L., Peterson, D. A. & Gordon, J. I. Host-bacterial mutualism in the human intestine. Science. 307(5717), 1915–1920 (2005).
Chen, Y., Zhou, J. & Wang, L. Role and mechanism of Gut Microbiota in Human Disease. Front. Cell. Infect. Microbiol.11, 625913 (2021).
Adak, A. & Khan, M. R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci.76(3), 473–493 (2019).
Bell, D. S. Changes seen in gut bacteria content and distribution with obesity: Causation or association? Postgrad. Med.127(8), 863–868 (2015).
Ley, R. E. et al. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. U S A. 102(31), 11070–11075 (2005).
Cani, P. D., Osto, M., Geurts, L. & Everard, A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes. 3(4), 279–288 (2012).
Dahiya, D. K. et al. Gut microbiota modulation and its relationship with obesity using prebiotic fibers and probiotics: A review. Front. Microbiol.8, 563 (2017).
Kobyliak, N. et al. Probiotics in prevention and treatment of obesity: A critical view. Nutr. Metab. (Lond). 13, 14 (2016).
Hill, C. et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol.11(8), 506–514 (2014).
Cooper, T. E. et al. Synbiotics, prebiotics and probiotics for people with chronic kidney disease. Cochrane Database Syst. Rev.10(10), CD013631 (2023).
Roshanravan, N. et al. A comprehensive systematic review of the effectiveness of Akkermansia muciniphila, a member of the gut microbiome, for the management of obesity and associated metabolic disorders. Arch. Physiol. Biochem.129(3), 741–751 (2023).
Quigley, E. M. M. Prebiotics and Probiotics in Digestive Health. Clin. Gastroenterol. Hepatol.17(2), 333–344 (2019).
Gu, W. et al. Association of rest-activity circadian rhythm with chronic respiratory diseases, a cross-section survey from NHANES 2011–2014. Respir Med.209, 107147 (2023).
Huo, X. et al. Association of dietary live microbe intake with abdominal aortic calcification in US adults: A cross-sectional study of NHANES 2013–2014. Front. Nutr.10, 1267607 (2023).
Wang, X. et al. High dietary live microbe intake is correlated with reduced risk of depressive symptoms: A cross-sectional study of NHANES 2007–2016. J. Affect. Disord. 344, 198–206 (2024).
Marco, M. L. et al. A classification system for defining and estimating Dietary Intake of Live microbes in US adults and children. J. Nutr.152(7), 1729–1736 (2022).
American College of Cardiology/American Heart Association Task Force on Practice & Guidelines, O. E. P. Executive summary: Guidelines (2013) for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Obesity Society published by the Obesity Society and American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Based on a systematic review from the the obesity Expert Panel, 2013. Obes. (Silver Spring). 22(Suppl 2), S5–39 (2014).
Expert Panel on Detection E. Treatment of high blood cholesterol in A: Executive summary of the third report of the National Cholesterol Education Program (NCEP) Expert Panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA. 285(19), 2486–2497 (2001).
Wang, L. & Yi, Z. Marital status and all-cause mortality rate in older adults: A population-based prospective cohort study. BMC Geriatr.23(1), 214 (2023).
Fadeyev, K., Nagao-Sato, S. & Reicks, M. Nutrient and Food Group Intakes among U.S. Children (2–5 Years) Differ by Family Income to Poverty Ratio, NHANES 2011–2018. Int J Environ Res Public Health 18(22). (2021).
Qiu, Z. et al. Associations of serum carotenoids with risk of Cardiovascular Mortality among individuals with type 2 diabetes: Results from NHANES. Diabetes Care. 45(6), 1453–1461 (2022).
Liang, J. et al. Association between Joint Physical Activity and Dietary Quality and Lower Risk of Depression symptoms in US Adults: Cross-sectional NHANES Study. JMIR Public. Health Surveill. 9, e45776 (2023).
Collaboration, N. C. D. R. F. Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet. 387(10026), 1377–1396 (2016).
Conway, B. & Rene, A. Obesity as a disease: No lightweight matter. Obes. Rev.5(3), 145–151 (2004).
Mohammed, M. S., Sendra, S., Lloret, J. & Bosch, I. Systems and WBANs for Controlling obesity. J. Healthc. Eng.2018, 1564748 (2018).
Castaner, O. et al. The gut Microbiome Profile in obesity: A systematic review. Int. J. Endocrinol.2018, 4095789 (2018).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 444(7122), 1027–1031 (2006).
Kim, J., Yun, J. M., Kim, M. K., Kwon, O. & Cho, B. Lactobacillus gasseri BNR17 supplementation reduces the visceral Fat Accumulation and Waist circumference in obese adults: A Randomized, Double-Blind, placebo-controlled trial. J. Med. Food. 21(5), 454–461 (2018).
Hadi, A., Sepandi, M., Marx, W., Moradi, S. & Parastouei, K. Clinical and psychological responses to synbiotic supplementation in obese or overweight adults: A randomized clinical trial. Complement. Ther. Med.47, 102216 (2019).
Kadooka, Y. et al. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur. J. Clin. Nutr.64(6), 636–643 (2010).
Sharafedtinov, K. K. et al. Hypocaloric diet supplemented with probiotic cheese improves body mass index and blood pressure indices of obese hypertensive patients–a randomized double-blind placebo-controlled pilot study. Nutr. J.12, 138 (2013).
Mohammadi-Sartang, M. et al. The effect of daily fortified yogurt consumption on weight loss in adults with metabolic syndrome: A 10-week randomized controlled trial. Nutr. Metab. Cardiovasc. Dis.28(6), 565–574 (2018).
Flach, J., van der Waal, M. B., van den Nieuwboer, M., Claassen, E. & Larsen, O. F. A. The underexposed role of food matrices in probiotic products: Reviewing the relationship between carrier matrices and product parameters. Crit. Rev. Food Sci. Nutr.58(15), 2570–2584 (2018).
Gomes, A. C. et al. The additional effects of a probiotic mix on abdominal adiposity and antioxidant status: A double-blind, randomized trial. Obes. (Silver Spring). 25(1), 30–38 (2017).
Markowiak, P. & Slizewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients9(9), 1021 (2017).
Quigley, E. M. M. Nutraceuticals as modulators of gut microbiota: Role in therapy. Br. J. Pharmacol.177(6), 1351–1362 (2020).
Sanmiguel, C., Gupta, A. & Mayer, E. A. Gut microbiome and obesity: A plausible explanation for obesity. Curr. Obes. Rep.4(2), 250–261 (2015).
Geurts, L., Neyrinck, A. M., Delzenne, N. M., Knauf, C. & Cani, P. D. Gut microbiota controls adipose tissue expansion, gut barrier and glucose metabolism: Novel insights into molecular targets and interventions using prebiotics. Benef Microbes. 5(1), 3–17 (2014).
Roselli, M. et al. Impact of supplementation with a food-derived microbial community on obesity-associated inflammation and gut microbiota composition. Genes Nutr.12, 25 (2017).
Plaza-Diaz, J., Ruiz-Ojeda, F. J., Gil-Campos, M. & Gil, A. Mechanisms of action of Probiotics. Adv. Nutr.10(suppl_1), S49–S66 (2019).
Kang, Y. et al. Lactobacillus acidophilus ameliorates obesity in mice through modulation of gut microbiota dysbiosis and intestinal permeability. Pharmacol. Res.175, 106020 (2022).
Acknowledgements
We appreciate the people who contributed to the NHANES data we studied.
Funding
This work was supported by Funding: grants from the Traditional Chinese Medicine Science and Technology Plan of Zhejiang (2022ZB102 to HZ) and the National Natural Science Foundation of China (82304940 to HZ).
Author information
Authors and Affiliations
Contributions
The authors’ responsibilities were as follows—HZ: designed the research, and had primary responsibility for the final content; YC: conducted analyses and wrote the first draft of the paper; HN: revised the manuscript; and all authors: read and approved the final manuscript and approved the final submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics, consent and permissions
All participants provided written informed consent and study procedures were approved by the National Center for Health Statistics Research Ethics Review Board (Protocol Number: Protocol #98 -2, Protocol #2005-06, and Protocol #2011-17).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Y., Ni, H. & Zhang, H. Exploring the relationship between live microbe intake and obesity prevalence in adults. Sci Rep 14, 21724 (2024). https://doi.org/10.1038/s41598-024-72961-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-72961-4