Abstract
Pain assessment in patients with temporomandibular joint (TMJ) disorders during physiotherapy is a challenging yet desired task. Our study addresses two issues. First, we compare the pain perception in patients with TMJ disorders and a control group. The hand dynamometer is verified as an alternate pain self-report device in the second stage. For the dynamometer validation, we employed a cuff-pressure test to induce pain of various intensities. In the second stage, physiological signals (electrodermal activity and blood volume pulse) have been acquired during the physiotherapy in a control group and patients suffering from TMJ disorder. The linear mixed-effects modeling is carried out to find differences in pain and no pain states, considering subjects’ health condition. The main result of the present pilot study is that physiological reactions to painful physiotherapy do not differ between patients with chronic pain and the control group. This finding concludes that studies on TMJ disorder procedures may also be performed on healthy subjects.
Similar content being viewed by others
Introduction
Pain is a complex, multidimensional, and still not fully understood phenomenon. In 2018, the International Association for the Study of Pain provided the updated definition of pain as follows: an unpleasant sensory and emotional experience associated with, or resembling that associated with actual or potential tissue damage1. This definition emphasizes the physical and psychological aspects, indicating the pain’s dual nature. Indeed, describing pain depends not only on the type of injury suffered, the degree of disability, age, and sex but also on previous pain experiences, environmental factors, personality, and life experience 2.
Considering the large number of variables affecting the perception of pain, an interesting research direction is to look for differences between specific groups. There are several studies on pain feeling in women and men3,4,5 indicating significant sex influence. Many researchers also point to a connection between age and the intensity of experienced pain6,7. The level of health education8 and belonging to racial/ethnic minorities9 were also found to impact pain sensibility.
The pain sensations will also differ for people experiencing chronic pain. Chronic pain describes a state in which the body’s physiological functions have not returned to the level of homeostasis10. Long-term pain can significantly impact daily activities, physical activity, self-esteem, and overall quality of life. Moreover, the chronic pain experience affects the subjective pain rating in acute pain events11. An attempt to find differences in healthy and chronic pain patients was proposed in12. Twenty female patients with myofascial pain dysfunction syndrome (MPDS) and twenty healthy female participants underwent focal pressure stimulation with various intensities. Every five seconds, the subjects were giving the numeric rate scale (NRS) rates until reporting definite pain. The results showed that myofascial pain dysfunction (MPD) patients exhibited significantly lower pain thresholds than healthy subjects. What is more, they also had a greater tendency to report pain than normal control subjects. Peters at al.13 carried out the study where back pain (CLBP) patients and control subjects underwent electrical and pressure pain stimulation. CLBP patients indicated a significantly higher pain perception threshold for both pain tests. The maximal pain tolerance threshold was also found to be higher in the patient group. On the contrary to12, the study outcomes show that subjects with chronic pain are less sensitive to the experimental pain stimuli tests. Mohn et al.14 carried out a study on experimental pain induction in female patients with temporomandibular joint (TMJ) disorder and pain-free controls. Mean arterial pressure (MAP) and heart rate (HR) were acquired during the experiment. Subjects underwent electrocutaneous stimulation and pressure stimulation. The study also included biting on a custom-made U-shaped occlusal force meter for one minute with a certain force. The results showed that patients with TMJ disorders had significantly higher electrocutaneous pain threshold than healthy controls. Compared to normal controls, they also reported increased electrocutaneous and pressure pain sensitivity after isometric contraction of the orofacial region. All presented studies rely on experimental pain induction. Such an attempt brings several benefits, e.g., precise information about stimuli intensity, variability, and occurrence in time, and a repeatable course of the experiment. On the contrary, studies performed in clinical conditions deliver data that is rather challenging to process. Still, pain sensations in real life conditions (e.g., medical procedures) are the target field of research.
Some physiological signals can reflect the body’s response to pain. They result from the autonomic nervous system (ANS) activity and thus can be used to address the problem of possible objective pain response assessment. These include i.a., electrodermal activity (EDA) and blood volume pulse (BVP)15. Since the sympathetic response of the ANS produces, i.e., increased sweat secretion, analysis of the EDA signal is a good method for pain monitoring16. It is also known that pain stimulus increases the heart rate17. Apart from information about HR, BVP also carries a view into blood volume in vessels18. Shelley19 observed that momentary BVP amplitude decreases are related to the body’s response to stress. Introducing physiological signals to studies on differences between healthy and suffering subjects may carry an interesting insight into a problem. We found one study addressing this issue. Evans et al.20 examined 48 children with chronic pain and 104 healthy control children with a series of pain tasks (evoked pressure, cold pressor, focal pressure, and conditioned pain modulation). The post-task electrocardiogram analysis revealed that the heart rate variability (HRV) was unaffected in the chronic pain group in response to pain stimuli. On the other hand, in the healthy children group, the stimulation reduced HRV. The measurements were, however, carried out after the examination and did not follow the pain-feeling dynamics. Again, the pain did not result from a medical procedure yet was induced experimentally.
The need for pain assessment during the treatment applies strongly to manual therapy. Most physiotherapy procedures focus on reducing pain to improve motor function2. Meanwhile, since temporomandibular disorders (TMD) produce much tension in tissues21, TMJ manual therapy is considered painful for the patient. One-third of adults report having one or more symptoms of TMD22. These include head, temple, and shoulder pain, limited mobility, or crackling sounds when moving the joint23,24, which significantly impact the life quality. Indeed, some studies point to an association between TMJ pain and depression, anxiety, and post-traumatic stress disorders25. Therefore, employing appropriate therapy is much needed. It assumes masseter muscle relaxation and joint mobilization26,27,28. The palpation pain and increased tension are observed during the initial phase of therapy29,30,31. Hence, the intensity of the procedure changes over time and produces dynamic pain sensations, including a drop in pain. Continuous information about pain level is essential for the therapist because there is a narrow margin between a safe and effective procedure. Therapy performed with too low intensity may not be effective enough, while too much force may cause permanent tissue deformation32,33. This motivates our study towards the problem of how patients react in ongoing painful procedures and, thus, if it may affect the course of the treatment.
This pilot study aims to compare pain perception in patients with TMJ disorders and in a control group during TMJ therapy. A statistical analysis for both groups and a combined analysis highlight the issue. To our knowledge, this is the first attempt to analyze differences in physiological responses in healthy and suffering subjects during physiotherapy. Additionally, the ground truth issue is addressed. The analysis of dynamometer reliability as a pain-feeling indicator has been verified in another group.
Materials and methods
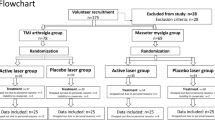
In our study, we present two experiments (Fig. 1). The first one aimed to verify the hand dynamometer as a device for pain feeling self-reporting. Hence, we performed a cuff-pressure test to induce pain of various intensities. The outcomes were the basis for introducing the dynamometer in the second stage of the study. Therefore, in the second experiment, we analyzed EDA and BVP responses to painful physiotherapy in healthy and suffering subjects. The statistical analysis was carried out to find short-term differences in pain and no pain states, taking into account subjects’ health condition.
All procedures in the study were performed in accordance with the Declaration of Helsinki and approved by University Bioethics Committee for Scientific Research at the Academy of Physical Education in Katowice, decision number 1/2019. Informed consent was obtained from all subjects involved in the study.
Dynamometer validation
In clinical practice, several pain level scales enable a patient to describe numerically, visually, or verbally the pain intensity: numeric rate scale (NRS, where 0 stands for no pain and 10 for maximum imaginable pain), visual analogue scale (VAS, with boundaries as in NRS, yet with continuous values), verbal rating scale (VRS), and others34. Some clinical procedures limit the use of described scales, especially during ongoing treatment. It refers to the situation where a patient remains in a supine position and cannot point any value at the range or when the procedures are performed around the face (e.g., TMJ therapy), which precludes verbal communication. Therefore, we carried out a study on a hand dynamometer employed as an alternate pain level indicator.
Participants and procedure
For this experiment, we recruited 16 healthy subjects aged 20–27, who were informed about the course of the study and the possibility of canceling it at any time. The experiment was carried out in a quiet, isolated room. Subjects were in a sitting position with a cuff placed on their left arm above the elbow and the KForce hand dynamometer (Kinvent35) in the right hand. Pressure in a cuff stood as a pain stimulus, whereas the dynamometer was used to indicate the pain level. The dynamometer signal was acquired with a sampling frequency of 75 Hz and synchronized in time with the pressure value data. We asked participants to press the device with the force corresponding to experienced pain. The pressure stimulation was induced automatically by a device programmed with dedicated software. The setup included the emergency button, which, when pressed, stopped the procedure immediately. The experiment started with applying the initial pressure at 60 mmHg as a reference value, and this measurement was not included in the analysis. Then, a subject was given eight pain stimuli in randomized order (the same for everyone), where the pressure varied from 60 to 250 mmHg. Each stimulus intensity arose from 0 mmHg with a speed around 50 \(\frac{{{\text{mmHg}}}}{{\text{s}}}\), the plateau segment was set to 1.5 seconds, and then the pressure dropped to a baseline value (0 mmHg) (Fig. 2). An around 50-second break separated subsequent pain events. The stimulation phase was 10 minutes, and the whole procedure took about 15 minutes. Since four patients claimed they experienced no pain at all during the procedure, they were excluded from the analysis.
Data analysis
The raw dynamometer signal was preprocessed in several steps. Firstly, data was normalized to the 0–1 range with the formula:
where \(x_{min}\) and \(x_{max}\) stand for the minimum and maximum values collected during the whole pressure stimulation test. It may be argued that the study protocol does not assume applying a wide range of pain intensities, and thus collected dynamometer data may not reflect expected \(x_{min}\) and \(x_{max}\) indications. Nevertheless, the experiment aimed to verify whether a setup is credible for gradual pain self-reporting rather than to prove its precision in reflecting boundary feelings (i.e., no pain and worst imaginable pain). In the next step, the signal was filtered with the median filter with a window width of 10 samples. Similarly to dynamometer data, we also scaled the pressure values to the 0–1 range. Next, the Pearson correlation coefficient was determined for pressure and dynamometer data. We also analyzed data diversity in the following pain stimulations.
Physiotherapy sessions
Measurement setup
The measurement setup consists of two wireless devices acquiring data with a synchronized timestamp. The bioplux (Plux) device36 is a wearable central unit that collects physiological signals from peripheral sensors. In this study, we used EDA (sampling frequency at 8 Hz) and BVP (sampling frequency at 64 Hz) signals. EDA electrodes were placed on the middle phalanx of the index and middle fingers of the left hand, whereas the photoplethysmograph acquired data from the ring finger of the same hand. Subjects were holding the hand dynamometer (KForce) in the right hand. Figure 3 presents the used sensors. Since devices are wireless and wearable, the setup does not limit the therapeutic procedure. More about the measurement platform can be found in37.
Participants and procedure
The dataset contains data from 22 subjects split into two subgroups. Ten subjects (women) aged 15–45 (mean 27 ± 1) have been diagnosed with TMJ dysfunction of muscular origin (related to bruxism, degenerative changes of the TMJ, or facial nerve palsy). The other 12 subjects (8 women, 4 men, aged 21–47, mean 24 ± 5) were healthy and stood for the control group. Participants from the suffering group included in the study had to be diagnosed by a physician as experiencing long-term pain in the TMJ area (inclusion criteria). The pain was localized in the masseter and temporalis muscles area. Ailments resulted mainly from chronic, increased muscle tension. The control group subjects had to be in overall good health and have no chronic pain feelings. None of the participants (both suffering and healthy) took anti-inflammatory medications, reported depression, or took serotonergic medications. None of the participants took painkillers regularly. They also did not take painkillers on the day of the procedure.
At the beginning of the procedure, participants were informed about the course of the study, its purpose, and the possibility of canceling it at any time. Then, a personal questionnaire was completed. It included questions about sex, age, and overall health condition. In the next step, we disinfected the subject’s skin and put on the measuring devices. Before starting the measurements, the dynamometer calibration was carried out. The subject was asked to squeeze the dynamometer with a force reflecting the worst imaginable pain. The obtained value served as a reference in subsequent data analysis. During the therapy, the patient was in a supine position while the physiotherapist sat behind his/her head. The protocol assumed treating trigger points occurring in the TMJ area. Once the patient indicated painful areas, the therapist located points that were particularly sensitive to touch. Deep rubbing of the stretched muscle was performed in the first stage, lasting about one minute. The movements were carried out slowly along the tissue. Then, the therapist applied increased pressure along with minimal movement until pain occurred. This phase lasted about 10-15 seconds and was followed by 10-15 seconds of relaxation. Such action was repeated until the muscle tension significantly decreased. Since pressure placed on trigger points may produce severe pain, the therapist adjusted the stimulus depending on the patient’s predispositions. In some cases, the procedure was also performed inside the oral cavity. The therapy time was approximately six minutes. In the described study protocol, the hand dynamometer served as an indicator of the pain level: the patient’s task was to squeeze the device in his hand with the force corresponding to the experienced pain. After completing the therapy, the measurement equipment was removed. The diagram of the test protocol is shown in Fig. 1. One specialist performed all therapy sessions.
Preprocessing and feature extraction
Firstly, collected data were subjected to the preprocessing step. A moving average filter with a Gaussian window was applied to the EDA signal. BVP data was subjected to the median filter of the 10-sample window. Next, we employed the 2nd-order Butterworth bandpass filter with a lower cutoff at 0.6875 Hz and a higher cutoff at 10 Hz. As the subjects were in a supine position with arms along the body, motion artifacts did not affect the data. Therefore, no further steps were required.
On the contrary, the hand dynamometer signal (GRIP) is sensitive to artifacts and needed a few-step preprocessing. First, the signal was normalized according to (1), where minimum and maximum values were derived from the calibration phase. Next, the moving-median filter was applied to remove the high-frequency impulse noise. Finally, we determined the signal envelope using spline over local maxima method. The raw (yet normalized) and preprocessed dynamometer data are presented in Fig. 4. The signal was thresholded at 2/3 to obtain pain and no pain indications: the values above the threshold were marked as pain, whereas lower or equal were treated as no pain indications. According to38, scores of 6–7 correspond to moderate pain and scores \(\ge\) 8 to severe pain. Therefore, the determined cut-off value at 2/3 in a continuous 0–1 scale approximately divided data into binary pain/no pain observations.
Physiological signals stand for the continuous body’s response to a given stimulus (pressure applied to the tissue). Since single data recording may comprise acute pain followed by rapid release, long, dull pain periods, or the body’s resting state, a short-term analysis is desired. Thus, EDA and BVP data were divided into four-second frames with a 50% overlap. Then, 16 features were extracted from EDA and BVP signals for each frame. Median, standard deviation, minimum, and maximum values were derived per each frame of EDA, its first (EDA’) and second derivative (EDA”), producing a total of 12 features. Consecutive BVP signal periods (\(P_{BVP}\), i.e., heart rate) were computed, and its median and standard deviation within each frame served as the next two features. Similarly, the median and standard deviation of the amplitude (\(A_{BVP}\)) were determined. All features were passed through the z-standardization in a subject-dependent mode.
Since the dynamometer outcome serves as the ground truth, this data was also divided into four-second frames with a 50% overlap. In frames including both no pain and pain labels, the maximum (therefore pain) value accounted. Finally, with derived labels, all data has been aggregated into no pain or pain class for certain groups (healthy, suffering, and all). Table 1 shows detailed numerical distribution of determined observations.
Statistical analysis
The analysis seeks differences between pain and no pain frames and the possible influence of the health state on feature values. Since the reaction to pain is highly subject-dependent and several observations were obtained per subject, we employed the linear mixed effects (LME) modeling approach for each feature. The normal distribution of the data was checked with the one-sample Student’s t-test. All tests performed in the study were two-tailed, and a p-value less than 0.05 was considered statistically significant.
In the first stage, we specified the random effects term best fitting the data. The following structures were tested: (1) random intercepts and slopes for subjects: (1|subjects), (2) random intercepts for pain occurrence (categorical variable no pain/pain), and random slopes for subjects influenced by pain occurrence: (1+painOccurrence|subjects), as the effect of pain occurrence may vary between subjects. We compared the models’ performance and found that structure 2) better fits the data.
Next, we introduced the fixed effects to the model. First, the pain occurrence stood as the only predictor of feature value:
featureValue~1+painOccurence+(1+painOccurrence|subjects)
The model is further called \(\text {M}_{{1}}\). The effect of health condition (categorical variable healthy/suffering) was included in model \(\text {M}_{{2}}\):
featureValue~1+painOccurrence+healthCondition+(1+painOccurrence|subjects)
Finally, model \(\text {M}_{{3}}\) also seeks the interaction between pain occurrence and health condition:
featureValue~1+painOccurrence*healthCondition+(1+painOccurrence|subjects)
The improvement in model fit was determined with the likelihood ratio test. For all significant results, the 80% statistical power of the post-hoc test was provided. We carried out the analysis with Matlab R2022a and R (version 4.4.0, R Core Team 2021). The power of the test was verified with the simr package.
Results
Dynamometer validation
The median value of the Pearson correlation coefficient for raw dynamometer data and the pressure data yielded 0.56±0.18. In this study, signals were not subjected to any segmentation process, yet we analyzed the whole recording. Therefore, the dynamometer signal affected by motion artifacts (see Fig. 2) at no stimuli periods was also considered. Indeed, relatively low correlation is due to high-frequency noise produced by the dynamometer, especially in no-pressure periods. On the other hand, considering extracted values, i.e., eight consecutive pressure values and corresponding GRIP data samples (per subject), results in a median value of 0.80±0.17. Such an approach better reflects the accuracy of pain level indications yet neglects the time dimension.
Next, we analyzed the variability of pain indications for consecutive pressure stimuli between subjects (Fig. 5). The highest standard deviation at 0.31 was noted for the fifth stimulation, i.e., the pressure induction at 60 mmHg, and for the last stimuli (250 mmHg). Interestingly, we observed the smallest dispersion for the third one, i.e., the first occurrence of 60 mmHg pressure (0.23). Inconsistent GRIP indications are also found for other repeated pressures. In the case of secondary 125 Hg induction, pain level perception was lower, while for secondary 190 and 250 mmHg, it was higher.
We also employed the Bland-Altman plot39 to assess the degree of agreement between pressure magnitude and GRIP indications (Fig. 6). Before the plot was determined, normal distribution of pressure and GRIP values differences was verified with the Kolmogorov-Smirnov test40. The null hypothesis that the data comes from a standard normal distribution has not been rejected at significance level \(\alpha\) = 0.05. The mean difference between normalized pressure and GRIP at 0.1 shows that, in general, GRIP indications were lower than corresponding pressure values. A positive trend along the graph reflects a slight systematic shift of mean GRIP indications towards lower values. Nonetheless, the range of variability is consistent across the graph. The paired-sample t-test with the significance level at \(\alpha\) = 0.05 rejected the null hypothesis that the pairwise difference between pressure and GRIP data has a mean equal to zero (\(p\) < 0.05), which also suggests a consistent bias. Indeed, the t-test for GRIP and pressure reduced by the mean difference (pressure − 1) resulted in \(p\) = 0.99. The diagonal line pattern in the plot results from the assignment to the discrete pressure values.
The Bland-Altman plot for 0-1 normalized cuff pressure stimuli (pressure) and corresponding dynamometer indications (GRIP). Black solid line presents the mean difference, and the green solid lines stand for limits of agreement determined at 0.619 and − 0.419. Dashed lines are the 95% confidence intervals (black and green for the mean difference and limits of agreement, respectively).
Statistical analysis
The series of determined LME models showed a significant effect on most features for pain occurrence condition. The set of 11 features and its subset of 10 features for \(\text {M}_{\text {1}}\) and \(\text {M}_{2}\), respectively, were found influenced by no pain/pain state. The significance level \({\alpha}\) = 0.05 was applied to all models. Figure 7 presents box plots of sample features in analyzed groups, and detailed modeling results can be found in Tables 2 and 3. Note that both EDA and BVP features showed significant effects in all experiments.
Model investigating the impact of health condition (\(\text {M}_{\text {2}}\)) indicated no significant effect on any of the considered features. Moreover, \(\text {M}_{\text {3}}\) revealed no significant interaction between pain occurrence and health condition. Table 4 summarizes \(\text {M}_{\text {3}}\) model performance. We found four features influenced by the pain occurrence in all tested models (\(EDA_{med}\), \(EDA_{max}\), \(P_{BVP,med}\), \(A_{BVP,med}\)). Similarly, five features did not differentiate either pain occurrence or health condition in any model (\(EDA'_{med}\), \(EDA''_{med}\), \(EDA''_{min}\), \(P_{BVP,std}\), and \(A_{BVP,std}\)).
The likelihood ratio test showed that neither \(\text {M}_{\text {2}}\) nor \(\text {M}_{\text {3}}\) were significantly better than the simpler model structure \(\text {M}_{\text {1}}\) for all of the determined features. Therefore, we conclude that pain occurrence influences physiological signals after controlling for random effects, but there is neither effect nor interaction with the subject’s health condition.
Discussion and conclusion
The pain assessment research in clinical setups carries some challenges. First, collecting trustworthy time-continuous pain rates during medical procedures seems demanding. Data labeling can be easily handled in controlled experimental studies, yet the multiple factors influencing the patient’s state during treatment significantly limit the use of standard solutions. In this work, we tackled the issue of subjective pain assessment during physiotherapy, showing that the hand dynamometer can be applied as a valid pain rating device. The study also addressed another matter: obtaining a representative group of subjects. Our analysis found that healthy subjects perceive manual therapy-related pain similarly to suffering ones, and thus, both groups may be treated as one cohort. Still, we are aware that with a relatively limited number of subjects, the report presents a pilot study.
The main result of the present study is that short-term physiological reactions to painful TMJ physiotherapy do not differ between patients with chronic pain and healthy subjects. Thus, outcomes of pain assessment studies performed in healthy controls may also apply to suffering patients. It gives a chance to obtain a broader group in future research. Our finding is based on multimodal data, which impacts its reliability. Moreover, the LME modeling showed the effectiveness of the proposed data analysis method. Since physiological signals’ features significantly differed no pain and pain frames in all experiments, we assume that studies on automatical pain assessment in physiotherapy are a promising direction. An approach to the problem can be found in our previous studies41,42.
The validation experiment for the hand dynamometer showed that the device is credible for self-reporting and thus can be used in pain assessment studies. A strong argument for employing a dynamometer is the possibility of using it in a broader group of patients. It does not limit including patients with mental impairments, children, or older adults, who may find challenging precise pain rating with, e.g., NRS. Moreover, the hand dynamometer is applicable to any procedure that demands a patient’s position other than sitting. It also applies to TMJ therapy, where verbal communication between the patient and therapy is inaccessible. Though the results of our study on dynamometer indications are dispersed (see Fig. 5), the pain rating consistently follows the intensity of the stimulus. Our previous studies showed that the exact pain rating, i.e., by NRS, in the case of physiotherapeutic procedures is pointless. The reference pain value at the beginning of the manual therapy cannot be provided as it may cause tissue damage. In this case, analyzing pain feeling dynamics seems more suitable than considering precise numerical values. Therefore, we propose an alternate method for pain self-report interpretation, especially for clinical condition studies.
To our knowledge, this is the first study on differences in chronic pain patients and healthy control group where pain stimulus was not induced experimentally. Vast majority of works relayed mostly on controlled/predefined pressure (12,13,14,43) and electrical stimuli13,14. Moreover, pain assessment was based on pain thresholds and questionnaires (numeric rating scale—NRS12, pain perception threshold—PPT, maximal pain tolerance—MPT13, State-Trait Anxiety Inventory—STAI43). On the contrary, our study employed time-continuous physiological data, such as EDA and BVP. We found the only study where the physiological signal was analyzed20. The authors found that patients with chronic pain have a sustained stress response (measured with HRV) and minimal variability in response to new acute pain stressors. Nevertheless, the outcomes were based on a single modality. Other physiological data may need to be analyzed to reflect pain response. Our study relies on multimodal data, what makes our findings more relevant. Table 5 presents a summary of studies on pain feeling differences in healthy and suffering subjects. While our results suggest that healthy and suffering subjects do not significantly differ, some studies found that chronic pain patients indicate lower pain threshold12,43. On the contrary, in13 and14, higher pain thresholds were noted for the suffering group. Since results are ambiguous, there is a need for further research in this direction, especially for clinical setups.
Studies on pain assessment during manual therapy are burdened with some more issues. First of all, the intensity of pain stimulation is unknown. Thus, no measurable pain thresholds can be determined and used in further steps. Next, predicting the course of pressure induction (transduced by the therapist’s hands) is impossible. Each procedure is individual and adjusted to the patient’s current condition. Therefore, alternate analysis methods are desired in future research. Analysis of physiological responses to pain directs our efforts to develop an automatic pain assessment system dedicated to physiotherapy.
Data availability
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
References
Treede, R.-D. The international association for the study of pain definition of pain: As valid in 2018 as in 1979, but in need of regularly updated footnotes. Pain Rep.3, e643 (2018).
Strong, J., Unruh, A., Wright, A. & Baxter, G. Pain: A Textbook for Therapists (Elsevier Churchill Livingstone, 2002).
Riley, J. L. III., Robinson, M. E., Wise, E. A. & Price, D. A meta-analytic review of pain perception across the menstrual cycle. Pain81, 225–235 (1999).
Paller, C. J., Campbell, C. M., Edwards, R. R. & Dobs, A. S. Sex-based differences in pain perception and treatment. Pain Med.10, 289–299 (2009).
Rovner, G. S. et al. Chronic pain and sex-differences; women accept and move, while men feel blue. PLoS ONE12, e0175737 (2017).
Rustøen, T. et al. Age and the experience of chronic pain: Differences in health and quality of life among younger, middle-aged, and older adults. Clin. J. Pain21, 513–523 (2005).
Lautenbacher, S., Peters, J. H., Heesen, M., Scheel, J. & Kunz, M. Age changes in pain perception: A systematic-review and meta-analysis of age effects on pain and tolerance thresholds. Neurosci. Biobehav. Rev.75, 104–113 (2017).
Köppen, P. J., Dorner, T. E., Stein, K. V., Simon, J. & Crevenna, R. Health literacy, pain intensity and pain perception in patients with chronic pain. Wien. Klin. Wochenschr.130, 23–30 (2018).
Kim, H. J. et al. Racial and ethnic differences in experimental pain sensitivity: Systematic review and meta-analysis. Pain158, 194–211 (2017).
Loeser, J. D. & Melzack, R. Pain: An overview. Lancet353, 1607–1609 (1999).
Nielsen, C. S., Staud, R. & Price, D. D. Individual differences in pain sensitivity: Measurement, causation, and consequences. J. Pain10, 231–237 (2009).
Malow, R. M., Grimm, L. & Olson, R. E. Differences in pain perception between myofascial pain dysfunction patients and normal subjects: A signal detection analysis. J. Psychosom. Res.24, 303–309 (1980).
Peters, M. L. & Schmidt, A. J. Differences in pain perception and sensory discrimination between chronic low back pain patients and healthy controls. J. Psychosom. Res.36, 47–53 (1992).
Mohn, C., Vassend, O. & Knardahl, S. Experimental pain sensitivity in women with temporomandibular disorders and pain-free controls: The relationship to orofacial muscular contraction and cardiovascular responses. Clin. J. Pain24, 343–352 (2008).
Burton, A. R., Fazalbhoy, A. & Macefield, V. G. Sympathetic responses to noxious stimulation of muscle and skin. Front. Neurol.7, 109 (2016).
Setz, C. et al. Discriminating stress from cognitive load using a wearable EDA device. IEEE Trans. Inf Technol. Biomed.14, 410–417 (2010).
Terkelsen, A. J., Mølgaard, H., Hansen, J., Andersen, O. K. & Jensen, T. S. Acute pain increases heart rate: Differential mechanisms during rest and mental stress. Auton. Neurosci.121, 101–109 (2005).
Peper, E., Harvey, R., Lin, I.-M., Tylova, H. & Moss, D. Is there more to blood volume pulse than heart rate variability, respiratory sinus arrhythmia, and cardiorespiratory synchrony?. Biofeedback35, 54–61 (2007).
Shelley, K. H. Photoplethysmography: Beyond the calculation of arterial oxygen saturation and heart rate. Anesth. Analg.105, S31–S36 (2007).
Evans, S. et al. Heart rate variability as a biomarker for autonomic nervous system response differences between children with chronic pain and healthy control children. J. Pain Res.6, 449–457 (2013).
Herb, K., Cho, S. & Stiles, M. A. Temporomandibular joint pain and dysfunction. Curr. Pain Headache Rep.10, 408–414 (2006).
Buescher, J. J. Temporomandibular joint disorders. Am. Fam. Physician76, 1477–1482 (2007).
Cuccia, A. & Caradonna, C. The relationship between the stomatognathic system and body posture. Clinics64, 61–66 (2009).
Gauer, R. L. & Semidey, M. J. Diagnosis and treatment of temporomandibular disorders. Am. Fam. Physician91(6), 378–86 (2015).
Kindler, S. et al. Depressive and anxiety symptoms as risk factors for temporomandibular joint pain: A prospective cohort study in the general population. J. Pain13, 1188–1197 (2012).
Ariji, Y. et al. Potential clinical application of masseter and temporal muscle massage treatment using an oral rehabilitation robot in temporomandibular disorder patients with myofascial pain. Cranio33, 256–262 (2015).
Olchowy, C. et al. Potential of using shear wave elastography in the clinical evaluation and monitoring of changes in masseter muscle stiffness. Pain Res. Manag.2020, 4184268 (2020).
Wilmont, P., Saczuk, K., Pawlak, Ł & Łukomska-Szymańska, M. The most commonly used methods of treatment for bruxism-a literature review. J. Stomatol.71, 350–355 (2018).
Olchowy, A., Seweryn, P., Olchowy, C. & Wieckiewicz, M. Assessment of the masseter stiffness in patients during conservative therapy for masticatory muscle disorders with shear wave elastography. BMC Musculoskelet. Disord.23, 1–9 (2022).
Lee, K. & Chon, S. Assessments of muscle thickness and tonicity of the masseter and sternocleidomastoid muscles and maximum mouth opening in patients with temporomandibular disorder. Healthcare9, 1640 (2021).
Çebi, A. T. Ultrasonographic evaluation of masseter muscle thickness in patients with disk displacement with reduction. Oral Radiol. 1–6 (2018).
Chaudhry, H. et al. Three-dimensional mathematical model for deformation of human fasciae in manual therapy. J. Am. Osteopath. Assoc.108, 379–390 (2008).
Threlkeld, A. J. The effects of manual therapy on connective tissue. Phys. Ther.72, 893–902 (1992).
Karcioglu, O., Topacoglu, H., Dikme, O. & Dikme, O. A systematic review of the pain scales in adults: Which to use?. Am. J. Emerg. Med.36, 707–714. https://doi.org/10.1016/j.ajem.2018.01.008 (2018).
KFORCE User Manual. https://k-invent.com/wp-content/uploads/2020/09/K-FORCE-manual.En21_04_20.pdf. Accessed 28 Sept 2023.
biopluxsignals User Manual. https://bio-medical.com/media/support/biosignalsplux_explorer_user_manual_v.1.0.pdf. Accessed 03 Aug 2023.
Badura, A. et al.Multimodal Signal Acquisition for Pain Assessment in Physiotherapy 227–237 (Springer International Publishing, 2021).
Boonstra, A. M. et al. Cut-off points for mild, moderate, and severe pain on the numeric rating scale for pain in patients with chronic musculoskeletal pain: Variability and influence of sex and catastrophizing. Front. Psychol.7, 1466 (2016).
Bland, J. M. & Altman, D. G. Measuring agreement in method comparison studies. Stat. Methods Med. Res.8, 135–160 (1999).
Giavarina, D. Understanding bland Altman analysis. Biochem. Med.25, 141–151 (2015).
Badura, A., Masłowska, A., MyŚliwiec, A. & Piętka, E. Multimodal signal analysis for pain recognition in physiotherapy using wavelet scattering transform. Sensors21, 1311 (2021).
Bieńkowska, M., Badura, A., Myśliwiec, A. & Piętka, E. Head pose and biomedical signals analysis in pain level recognition. In Information Technology in Biomedicine (eds Pietka, E. et al.) 345–355 (Springer International Publishing, 2022).
Muñoz-García, D., López-de Uralde-Villanueva, I., Beltran-Alacreu, H., La Touche, R. & Fernández-Carnero, J. Patients with concomitant chronic neck pain and myofascial pain in masticatory muscles have more widespread pain and distal hyperalgesia than patients with only chronic neck pain. Pain Med.18, 526–537 (2017).
Acknowledgements
This research was funded by the Polish Ministry of Science, Poland, statutory financial support No. 07/010/BK_24/1034, and by The National Center for Research and Development, Poland, grant number WPN-3/1/2019 and by European Funds for Silesia 2021–2027 Program co-financed by the Just Transition Fund Project “Development of the Silesian biomedical engineering potential in the face of the challenges of the digital and green economy (BioMeDiG)”, grant number FESL.10.25-IZ.01-07G5/23.
Author information
Authors and Affiliations
Contributions
All authors are justifiably credited with authorship. The detailed contribution is as follows: Conceptualization, E.P., A.M., A.B., and M.B.; methodology, A.B., M.B., A.O., and E.P.; software, A.B., M.B., and A.O.; validation, A.B., M.B., and A.O.; formal analysis, A.B. and M.B.; investigation, M.B., A.B., E.P., and A.M.; resources, A.M., A.B., and M.B.; data curation, A.B. and M.B.; writing—original draft preparation, A.B., M.B., and E.P.; writing—review and editing, E.P., A.M., A.B., and M.B.; visualization, A.B. and M.B.; supervision, E.P. and A.M.; project administration, E.P. and A.M.; funding acquisition, E.P. and A.M. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Badura, A., Bieńkowska, M., Myśliwiec, A. et al. Physiological response to pain in patients with temporomandibular joint disorders during physiotherapy. Sci Rep 14, 27861 (2024). https://doi.org/10.1038/s41598-024-74084-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-74084-2