Abstract
Thoracic bulky esophageal cancer shrinks during radiotherapy, changing the location and shape of the surrounding heart and lungs. The current study aimed to explore how replanning by volumetric-modulated arc radiotherapy (VMAT) and three-dimensional conformal radiotherapy (3DCRT) influences the target coverage and dose to organs at risk in locally advanced unresectable middle to lower thoracic esophageal cancer. We retrospectively collected CT simulation images of initial and boost radiotherapy plans for locally advanced unresectable thoracic esophageal cancer in 17 consecutive patients. First, we created boost plans of 20 Gy using 3DCRT and VMAT on the initially acquired CT images. Second, we replicated the process on CT images acquired after 20–40 Gy of radiotherapy. We then compared non-replanned boost radiotherapy plans with replanned boost plans. Replanned radiotherapy delivered more conformal doses to the target and reduced heart and lung doses. VMAT reduced more irradiated mean doses to the heart than 3DCRT in the case of replanning (1.7 and 1.1 Gy, p < 0.001). Replanning to accommodate tumor shrinkage during radiotherapy effectively lowers the irradiated doses to the heart and lungs in patients with locally advanced unresectable middle to lower thoracic esophageal cancer, especially those treated with VMAT.
Similar content being viewed by others
Introduction
Definitive chemoradiotherapy is the standard treatment for locally advanced unresectable esophageal cancers1 and takes approximately 6 weeks to complete. The irradiated tumors shrink during chemoradiotherapy sessions and the volume generally shrinks 2–4 weeks after the initial day of radiotherapy in patients with locally advanced esophageal cancer who undergo chemoradiotherapy2. Shrinkage of the targets changes the location of the surrounding organs; initially, the displaced surrounding heart and lungs return to normal. These changes affect the precise dose delivery of radiotherapy, which is likely associated with unexpected local failures or toxicities.
Fitting the irradiation fields to shrinking targets was expected to deliver optimal doses to the target, thereby avoiding unnecessary doses to organs at risk. Replanning using reperformed computed tomography (CT) simulation images during radiotherapy sessions might alleviate unexpected changes in the irradiated doses to the target and surrounding organs associated with tumor shrinkage. However, previous studies did not focus on the association of shrinkage of the target, dose to the target, and organs at risk in thoracic esophageal cancer. They explored the association between daily diaphragm position or gastric filling in each radiotherapy session with target coverage3,4,5,6,7. They did not examine the association of shrinkage of the primary esophageal tumor or radiotherapy techniques with the dose distribution in replanning.
The current study collected data from patients with locally advanced, unresectable middle to lower thoracic esophageal cancer. Subsequently, the patients underwent replanning CT simulations during definitive concurrent chemoradiotherapy. We compared the target coverage and doses to organs at risk in non-replanned radiotherapy plans with those of replanned radiotherapy plans. We also examined the effects of the radiotherapy techniques on the dose distribution. The current study aimed to explore how replanning by volumetric-modulated arc radiotherapy (VMAT) and three-dimensional conformal radiotherapy (3DCRT) influences the target coverage and dose to organs at risk in locally advanced unresectable middle to lower thoracic esophageal cancer.
Materials and methods
The Institutional Review Board of Kyoto University Graduate School and the Faculty of Medicine Ethics Committee approved the current study on August 2, 2021 (R1048-1). This study was conducted in accordance with the Declaration of Helsinki and Japanese Ethical Guidelines for Epidemiological Research and written informed consent has been obtained.
Included patients
The inclusion criteria for the current study were as follows: patients who underwent radiotherapy at our hospital from April 2013 to March 2021; histologically diagnosed esophageal cancer; primary esophageal tumor located in the middle to lower thoracic esophagus; unresectable primary tumor or lymph node metastasis; no distant metastasis except for supraclavicular lymph node metastasis; no fistula formation and atelectasis; and re-CT simulation with exact localization as the initial CT simulation during radiotherapy until 40 Gy irradiation. We assessed tumor shrinkage by cone-beam CT images once or twice a week for patient’s positioning verification or re-CT simulation images. Seventeen patients met these criteria, representing 7% of patients who underwent definitive radiotherapy at our hospital between April 2013 and March 2021 (Fig. 1). The characteristics of the included patients are shown in Table 1.
Details of CT simulation in the included patients
All patients underwent CT simulation in the supine position, with the CT system undergoing regular quality assurance to ensure compliance with the acceptable range specified by the American Association of Physicists in Medicine report8. A vacuum pillow was used to maintain the patient's position. We obtained nonhelical and four-dimensional CT (4D-CT) images using a 16-slice scanner with a 2.0–2.5 mm slice thickness while monitoring respiration using a real-time position management system on the patient's abdomen. We created average-intensity projection images by considering the average of 10 respiratory cycle phases of the 4D-CT images. The current study defined CT images from CT simulation before radiotherapy sessions as CT set 1 and additionally performed images during radiotherapy sessions as CT set 2. The positional reproducibility between CT set 1 and CT set 2 was evaluated by aligning the CT origin marked on the body surface and performing rigid registration based on vertebral bone matching. CT set 1 was performed for 6 days (range, 5–12 days) before the initial day of radiotherapy, and CT set 2 was performed for 23 days (range, 16–31 days) after the initial radiotherapy. The median irradiated dose to the target was 32 Gy (range, 20–40 Gy) on the day of CT set 2.
Delineation of targets and organs at risk in the current study
The current study delineated targets and organs at risk using CT sets 1 and 2, respectively. We (H.I and K.S) contoured the primary esophageal tumor and metastatic lymph nodes as gross tumor volume (GTV). We created the clinical target volume (CTV) of the primary tumor from the GTV of the primary tumor with a 2 cm head-to-tail margin along the esophagus and the CTV of the metastatic lymph nodes, similar to the GTV of the metastatic lymph nodes. We defined the CTV of subclinical regional lymph nodes as follows: mediastinal to perigastric lymph nodes for a middle thoracic esophageal tumor and mediastinal to celiac axis lymph nodes for a lower thoracic esophageal tumor. The planning target volume (PTV) of the primary esophageal tumor, metastatic lymph nodes, and subclinical regional lymph nodes was created by adding a 1 cm head-to-tail margin plus a 0.5 cm margin in the radial direction to the CTV. We defined PTV-boost as the sum of the PTV of the primary esophageal tumors and metastatic lymph nodes. We then created the PTV-initial from the PTV-boost plus the PTV of the subclinical regional lymph nodes. We also considered the spinal cord, heart, and lungs to be organs at risk based on the Radiation Therapy Oncology Group contouring atlas9,10. We defined the GTV, PTV-boost, lungs, and heart delineated by the CT set X (X = 1 or 2) as GTVCTX, PTV-boostCTX, lungsCTX, and heartCTX, respectively. PTV-initial, only delineated using CT set 1, was defined as PTV-initialCT1.
Radiotherapy planning in the current study
The current study created radiotherapy plans for 3DCRT and VMAT for delineated targets. We created radiotherapy plans using a two-step method: the prescribed doses were 40 Gy in 2 Gy per fraction to PTV-initialCT1 and 20 Gy in 2 Gy per fraction to PTV-boostCT1 or PTV-boostCT2. 3DCRT plans used static four fields (0, 135, 180, and 315 degrees) with a leaf margin of 0.5 cm from the PTV-initialCT1, PTV-boostCT1, and PTV-boostCT2. The weight ratio of the anterior–posterior to oblique beams in 3DCRT plan was set to 2:1. The VMAT plans used two coplanar fully rotating arcs, avoiding lateral beam entry from approximately 225–315° and 45–135° to spare the lungs. The beam energy was 6 megavoltage (MV) X-rays in both 3DCRT and VMAT plans. The prescribed dose was D50 (the dose covering 50% of the structure) for PTV-initialCT1, PTV-boostCT1, and PTV-boostCT2. The dose to the spinal cord was limited to < 50 Gy. D95 and D2 of PTV-initialCT1, PTV-boostCT1, and PTV-boostCT2 were mandated to meet ≥ 95% and ≤ 107%, respectively. We adopted a small leaf margin for the PTV, and the field-in-field technique keeping the same gantry angles to spare the spinal cord in the 3DCRT plans to ensure the target doses. The dose calculation algorithm was Acuros XB version 16.1.2 (Eclipse™ Varian, Medical Systems, Palo Alto, CA).
First, we created two initial plans and two boost plans for each patient as follows and calculated the dose distributions of these plans for CT set 1: 3DCRT plans for PTV-initialCT1; 3DCRT plans for PTV-boostCT1; VMAT plans for PTV-initialCT1; and VMAT plans for PTV-boostCT1. Second, we replicated the irradiation fields and arcs of the 3DCRT and VMAT plans for PTV-boostCT1 in CT set 2 and calculated their dose distributions in CT set 2, respectively (non-replanned 3DCRT and VMAT). Finally, we created 3DCRT and VMAT plans for PTV-boostCT2 and calculated the dose distributions in CT set 2 (replanned 3DCRT and VMAT). Figure 2 shows the flow of creating the above dose distributions.
Quantitative variables
Quantitative variables for dose-volume metrics were as follows: D95, D50, D2, homogeneity index, and conformity index of PTV-boostCT1 and PTV-boostCT2; the mean dose and volume receiving 30 Gy (V30 Gy) of the heart; and the mean dose and V20 Gy of the lungs. The conformity index and homogeneity index were defined as follows: the volume of the body receiving at least 95% of the prescribed dose divided by the volume of the PTV-boost and (D2–D98)/D5011. In comparing boost plans, the volume receiving 50% of the prescribed dose (V50) of the heart and V33.3 of the lungs were used as alternative indices for V30 Gy of the heart and V20 Gy of the lungs.
Evaluation
First, we assessed the change in the target volume from CT set 1 to 2, and the change in the overlapping volume about PTV-boostCT1 and heartCT1, PTV-boostCT1 delineated into CT set 2 and heartCT2, and PTV-boostCT2 and heartCT2. Additionally, we performed a similar analysis on the overlapping volumes between PTV-boost and lungs. Second, we compared the dose-volume metrics of PTV-boostCT1, heartCT1, and lungsCT1 in 3DCRT plans for PTV-initialCT1 plus 3DCRT plans for PTV-boostCT1 with those in VMAT plans for PTV-initialCT1 plus VMAT plans for PTV-boostCT1. Their doses were calculated using CT set 1. Third, we compared the dose-volume metrics of heartCT2 and lungsCT2 in the non-replanned 3DCRT plans with those in the replanned 3DCRT plans. Similar comparisons were performed for the VMAT plans. Finally, we compared the differences in these dose-volume metrics in 3DCRT plans with those in VMAT plans to assess the effects of different radiotherapy techniques on dose reduction to the organs at risk by replanning for shrunken tumors.
Statistical analysis
We compared the volumes of targets and organs at risk between CT sets 1 and 2, and the dose-volume metrics and changes between 3DCRT plans and VMAT plans using paired t-tests. To assess the statistical power of our study, we conducted a post-hoc power analysis (PA) based on an alpha level of 0.05. The effect size (Cohen’s d) was calculated from the observed means and standard deviations of the reduction of dose-volume metrics in the replanned 3DCRT and VMAT plan. All statistical analyses were performed using EZR, version 1.61 (Saitama Medical Center, Jichi Medical University, Saitama, Japan)12, which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria).
Results
The tumors in all patients shrank between CT set 1 and CT set 2. The tumor volumes decreased by approximately 50% from CT set 1 to CT set 2: the mean volume was 95 cc (95% confidence interval [CI] 70–121 cc) in GTVCT1, 53 cc (95% CI 41–65 cc) in GTVCT2 (p < 0.001), 266 cc (95% CI 216–315 cc) in PTV-boostCT1, and 187 cc (95% CI 158–217 cc) in PTV-boostCT2 (p < 0.001). As the tumor shrank, the overlapping volume of PTV-boostCT1 with the heart and lungs increased (p = 0.014 and 0.066). Re-contouring in CT set 2, the overlap with the PTV-boostCT2 decreased (p = 0.002 and 0.004) (Table 2).
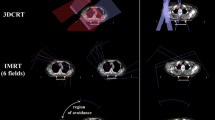
The VMAT plans delivered more conformal doses to the PTV-boostCT1 than the 3DCRT plans (Fig. 3a and b). The VMAT plans reduced the medium- to high-doses irradiated to the heart and lungs compared to the 3DCRT plans, increasing the low doses irradiated to the heart and lungs (Fig. 4). VMAT showed better target conformity than the 3DCRT plans (p < 0.001) without compromising target homogeneity (p = 0.16) (Table 3). VMAT plans reduced clinically relevant dose-volume parameters in organs at risk: the mean dose to the heart (p < 0.001), V30 Gy to the heart (p < 0.001), the mean dose to the lungs (p < 0.001), and V20 Gy to the lungs (p < 0.001); V5 Gy to the lungs was smaller in 3DCRT plans (p < 0.001) (Table 3).
Dose distributions of (a) 3DCRT plan created for PTV-boostCT1 calculated in CT set 1, (b) VMAT plan created for PTV-boostCT1 calculated in CT set 1, (c) 3DCRT plan created for PTV-boostCT1 calculated in CT set 2, (d) VMAT plan created for PTV-boostCT1 calculated in CT set 2, (e) 3DCRT plan created for PTV-boostCT2 calculated in CT set 2, and (f) VMAT plan created for PTV-boostCT2 calculated in CT set 2.
Replanned radiotherapy plans delivered more conformal doses to PTV-boostCT2 than non-replanned radiotherapy plans, irrespective of 3DCRT (Fig. 3c and e) and VMAT (Fig. 3d and f). The doses to the surrounding heart and lungs were reduced by replanning using 3DCRT and VMAT (Fig. 5a and b). The mean dose and V50 of the heart changed from 11.1 to 10.1 Gy and 59 to 54%, and the mean dose and V33.3 of the lungs from 3.6 to 3.1 Gy and 23 to 20% in replanned 3DCRT plans. The mean dose and V50 of the heart changed from 8.8 to 7.1 Gy and 36 to 23%, and the mean dose and V33.3 of the lungs from 3.2 to 2.7 Gy and 15 to 10% in replanned VMAT plans (Table 4).
VMAT reduced the larger volume of irradiated heart volumes more than 3DCRT in the case of replanning. However, the reduced irradiated volumes of the lungs were similar between 3DCRT and VMAT (Fig. 5c). The mean dose of the heart was 1.7 Gy (95% CI 1.3–2.1) and 1.1 Gy (95% CI 0.62–1.5) (p < 0.001, PA = 0.85); V50 of the heart was13.6% (95% CI 10.9–16.2) and 5.4% (95% CI 2.8–8.0) (p < 0.001, PA > 0.99); the mean dose of the lungs was 0.54 Gy (95% CI 0.42–0.66) and 0.48 Gy (95% CI 0.36–0.60) (p = 0.037, PA = 0.17); V33.3 of the lungs was 4.7% (95% CI 3.6–5.9) and 3.3% (95% CI 2.5–4.1) (p = 0.003, PA = 0.80).
Discussion
VMAT plans delivered more conformal doses to the targets and reduced doses to the heart and lungs compared to 3DCRT plans. Compared with non-replanned radiotherapy, replanned radiotherapy plans achieved more conformal dose delivery, sparing the heart and lungs in 3DCRT and VMAT; however, VMAT reduced the doses to the heart more than 3DCRT in the case of replanned radiotherapy.
The current trial showed that replanning, reflecting changes in the target volume, reduced the dose to the heart and maintained target coverage. In this study, the target volume decreased by half between CT1 and CT2 during radiotherapy sessions. The target volume was composed of the primary esophageal tumor and lymph node metastasis. Compared with previous studies, the current study included patients with bulky primary esophageal tumors and had massive tumor shrinkage during radiotherapy sessions2,13. Bulky primary esophageal tumors were dislocated from the heart before radiotherapy. Subsequently, the location of the heart changes to a normal position as the esophageal tumors diminish during radiotherapy. A pilot study reported that the heart volume was closer to the irradiated volume than the baseline when the target volume decreased during radiotherapy for thoracic esophageal cancer2. This is consistent with our study's findings, which show that as the tumor shrinks, the volume of the heart within the initially created PTV-boost increase. The movement of the heart to the target volume indicates that the irradiated dose to the heart potentially became larger than the baseline dose as the radiotherapy sessions progressed. The current study showed that replanning radiotherapy plans spared the heart owing to the interfractional movement of the heart resulting from the reduction of the primary esophageal tumor during radiotherapy sessions. Replanning radiotherapy for locally advanced esophageal cancer potentially avoids an unexpected increase in the irradiation dose to the heart.
The current study demonstrated that VMAT reduced medium- to high-dose irradiation to the heart more than 3DCRT in the case of replanning; V50 of the heart was reduced from 57 to 52% in 3DCRT, whereas it was reduced from 35 to 22% in VAMT. In 3DCRT, the irradiation fields can be reduced to fit the shrunken targets and avoid the moved heart; however, the dose to the heart cannot be reduced along the anteroposterior beam paths (Fig. 3c and e). In contrast, VMAT delivered a conformal dose to the target, sparing the heart (Fig. 3d and f); the reduction of medium- to high-dose irradiation to the heart by replanning was remarkable. In the areas of the heart irradiated with high doses, myocardial changes reflecting myocardial damage and fibrosis appear14,15. Reducing the volume of high-dose irradiation to the heart was significant. VMAT is the preferable radiotherapy technique to 3DCRT for sparing the heart when replanning.
In the current study, the lung volume receiving low doses increased in VMAT; however, the lung volume receiving medium to high doses decreased, compared with 3DCRT. Increased low-dose exposure in the lungs may not always relate to radiation pneumonitis. A retrospective study on esophageal cancer reported that the incidence of radiation pneumonitis did not increase in patients treated with IMRT, whose V5 Gy of lungs was higher than 3DCRT16. A secondary analysis of the randomized controlled trial for non-small cell lung cancer reported that the incidence of radiation pneumonitis was lower in the IMRT group than in the 3DCRT one, although patients treated with IMRT suffered larger target volumes with higher V5 Gy in lungs than those treated with 3DCRT17. A retrospective study reported that the volume of lungs ≥ 20 Gy is related to radiation pneumonitis rather than low-dose exposure in radiotherapy for esophageal cancer18. The increased low-dose exposure in the lungs may be counterbalanced by the decreased medium- to high-dose exposure in the lungs in the incidence of radiation pneumonitis.
Radiotherapy modalities affect dose-volume metrics for the heart and lungs. The improvement in dose-volume metrics for the heart and lungs using intensity-modulated radiotherapy is consistent with previous findings in esophageal cancer radiotherapy in non-replanning comparisons19,20. Retrospective analyses of radiotherapy for esophageal cancer reported that intensity-modulated radiotherapy reduced the incidence of adverse cardiopulmonary events and improved overall survival compared to 3DCRT20,21. Cardiopulmonary complications in long-term survivors are a significant issue in radiotherapy for thoracic esophageal cancer. Replanning for tumor shrinkage using VMAT for locally advanced esophageal cancer is beneficial considering late cardiopulmonary complications. Without patient burden, only one replanning afforded the benefit of reduced radiation exposure to the heart and lungs, while maintaining target coverage. The effect of replanning on tumor shrinkage in radiotherapy for non-small-cell lung cancer was reported to be 85% with weekly replanning and 65% with only one replanning in the middle of treatment, compared with daily replanning22. The appropriate timing and frequency of replanning for tumor shrinkage during radiotherapy for esophageal cancer is unknown; however, our study supports the usefulness of adaptive radiotherapy and one replanning during radiotherapy sessions should be considered first.
The current study had several limitations. The study exclusively included patients who underwent re-CT scans during radiotherapy sessions until 40 Gy irradiation (25% of patients who underwent radiotherapy for locally advanced unresectable middle to lower thoracic esophageal cancer), and the timing of CT scans varied between 20 and 40 Gy during radiotherapy; therefore, these results may not be readily generalizable to all esophageal cancer patients undergoing radiotherapy. This study fixed beam energy to 6 MV X-rays; however, more high-energy X-rays might improve the dose distribution in the target, especially in 3DCRT. The beam angles were fixed in this current study; however, the more optimal beam angles may exist in each patient. The impact of replanning to reduce doses to the heart and lungs on clinical outcomes needs to be examined in future studies. Nevertheless, despite these limitations, the current study reports that replanning for tumor shrinkage during radiotherapy for esophageal cancer effectively decreases the irradiated doses to the heart and lungs. These findings can inform the implementation of replanning during radiotherapy for locally advanced unresectable middle to lower thoracic esophageal cancer and support the consideration of adaptive radiotherapy.
In conclusion, the current study showed that replanning radiotherapy plans reflecting the shrinkage of tumors during radiotherapy reduced irradiated doses to the heart and lungs in patients with locally advanced unresectable middle to lower thoracic esophageal cancer. VMAT is beneficial in sparing the heart and lungs compared with 3DCRT when replanning is performed in this population. Replanning using VMAT is preferable to 3DCRT to reduce late cardiopulmonary toxicities; however, a long preparation time is required to conduct VMAT.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Network, N. C. C. Esophageal and Esophagogastric Junction Cancers (Version 3. 2023). https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf.
Liu, Y. M. et al. Computed tomography-based evaluation of volume and position changes of the target region and organs at risk during radiotherapy for esophageal cancer: A pilot study. Front. Oncol. 11, 702400. https://doi.org/10.3389/fonc.2021.702400 (2021).
Hafeez, S., Bedford, J. L., Tait, D. M. & Hawkins, M. A. Normal tissue sparing with respiratory adapted volumetric modulated arc therapy for distal oesophageal and gastro-oesophageal tumours. Acta Oncol. 53, 149–154. https://doi.org/10.3109/0284186X.2013.776174 (2014).
Nyeng, T. B., Nordsmark, M. & Hoffmann, L. Dosimetric evaluation of anatomical changes during treatment to identify criteria for adaptive radiotherapy in oesophageal cancer patients. Acta Oncol. 54, 1467–1473. https://doi.org/10.3109/0284186X.2015.1068449 (2015).
Moller, D. S. et al. Validation of a robust strategy for proton spot scanning for oesophageal cancer in the presence of anatomical changes. Radiother. Oncol. 131, 174–178. https://doi.org/10.1016/j.radonc.2018.09.018 (2019).
Anakotta, R. M. et al. Weekly robustness evaluation of intensity-modulated proton therapy for oesophageal cancer. Radiother. Oncol. 151, 66–72. https://doi.org/10.1016/j.radonc.2020.07.015 (2020).
Visser, S. et al. Robustness assessment of clinical adaptive proton and photon radiotherapy for oesophageal cancer in the model-based approach. Radiother. Oncol. 177, 197–204. https://doi.org/10.1016/j.radonc.2022.11.001 (2022).
Davis, A. T., Palmer, A. L. & Nisbet, A. Can CT scan protocols used for radiotherapy treatment planning be adjusted to optimize image quality and patient dose? A systematic review. Br. J. Radiol. 90, 20160406. https://doi.org/10.1259/bjr.20160406 (2017).
Feng, M. et al. Development and validation of a heart atlas to study cardiac exposure to radiation following treatment for breast cancer. Int. J. Radiat. Oncol. Biol. Phys. 79, 10–18. https://doi.org/10.1016/j.ijrobp.2009.10.058 (2011).
Kong, F. M. et al. Consideration of dose limits for organs at risk of thoracic radiotherapy: atlas for lung, proximal bronchial tree, esophagus, spinal cord, ribs, and brachial plexus. Int. J. Radiat. Oncol. Biol. Phys. 81, 1442–1457. https://doi.org/10.1016/j.ijrobp.2010.07.1977 (2011).
International Commission on Radiation, U. & Measurements. Prescribing, recording, and reporting photon-beam intensity-modulated radiation therapy (IMRT), Vol 83 (Oxford University Press, 2010).
Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 48, 452–458. https://doi.org/10.1038/bmt.2012.244 (2013).
Defize, I. L. et al. Tumor volume regression during neoadjuvant chemoradiotherapy for esophageal cancer: a prospective study with weekly MRI. Acta Oncol. 59, 753–759. https://doi.org/10.1080/0284186X.2020.1759819 (2020).
de Groot, C. et al. Radiation-induced myocardial fibrosis in long-term esophageal cancer survivors. Int. J. Radiat. Oncol. Biol. Phys. 110, 1013–1021. https://doi.org/10.1016/j.ijrobp.2021.02.007 (2021).
Ishida, Y. et al. Effect of long fasting on myocardial accumulation in 18F-fluorodeoxyglucose positron emission tomography after chemoradiotherapy for esophageal carcinoma. J. Radiat. Res. 59, 182–189. https://doi.org/10.1093/jrr/rrx076 (2018).
Inoo, H., Sakanaka, K., Fujii, K., Ishida, Y. & Mizowaki, T. Association of volumetric-modulated arc therapy with radiation pneumonitis in thoracic esophageal cancer. J. Radiat. Res. https://doi.org/10.1093/jrr/rrac021 (2022).
Chun, S. G. et al. Impact of intensity-modulated radiation therapy technique for locally advanced non-small-cell lung cancer: A secondary analysis of the NRG oncology RTOG 0617 randomized clinical trial. J. Clin. Oncol. 35, 56–62. https://doi.org/10.1200/JCO.2016.69.1378 (2017).
Du, F., Liu, H., Wang, W., Zhang, Y. & Li, J. Correlation between lung density changes under different dose gradients and radiation pneumonitis-based on an analysis of computed tomography scans during esophageal cancer radiotherapy. Front. Oncol. 11, 650764. https://doi.org/10.3389/fonc.2021.650764 (2021).
Kole, T. P., Aghayere, O., Kwah, J., Yorke, E. D. & Goodman, K. A. Comparison of heart and coronary artery doses associated with intensity-modulated radiotherapy versus three-dimensional conformal radiotherapy for distal esophageal cancer. Int. J. Radiat. Oncol. Biol. Phys. 83, 1580–1586. https://doi.org/10.1016/j.ijrobp.2011.10.053 (2012).
Lan, K. et al. Propensity score-based comparison of survival and radiation pneumonitis after definitive chemoradiation for esophageal cancer: Intensity-modulated radiotherapy versus three-dimensional conformal radiotherapy. Radiother. Oncol. https://doi.org/10.1016/j.radonc.2020.05.036 (2020).
Lin, S. H. et al. Propensity score-based comparison of long-term outcomes with 3-dimensional conformal radiotherapy vs intensity-modulated radiotherapy for esophageal cancer. Int. J. Radiat. Oncol. Biol. Phys. 84, 1078–1085. https://doi.org/10.1016/j.ijrobp.2012.02.015 (2012).
Dial, C., Weiss, E., Siebers, J. V. & Hugo, G. D. Benefits of adaptive radiation therapy in lung cancer as a function of replanning frequency. Med. Phys. 43, 1787. https://doi.org/10.1118/1.4943564 (2016).
Acknowledgements
This work was supported by JSPS KAKENHI Grant Number 24K02401. We would like to thank Editage (http://www.editage.jp) for English language editing.
Author information
Authors and Affiliations
Contributions
H.I. and K.S. made substantial contributions to the conception and the design of the work and the data analysis of the current study. H.I., K.S. and T.M. made significant contributions to the interpretation of data and the revision of the manuscript drafts. All authors had final approval of the submitted version of the manuscript and agreed to be accountable for any part of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Inoo, H., Sakanaka, K. & Mizowaki, T. Effect of replanning boost radiotherapy plan in locally advanced unresectable middle to lower thoracic esophageal cancer. Sci Rep 14, 23337 (2024). https://doi.org/10.1038/s41598-024-74615-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-74615-x