Abstract
While the negative consequences of particular illnesses have been associated with the neutrophil percentage to albumin ratio (NPAR), its association with cardiovascular disease (CVD) is still lacking in the broader public. This study examines the relationship between NPAR and CVD prevalence using information from the National Health and Nutrition Examination Survey (NHANES), with particular attention to potential nonlinear associations. This analysis included 26,225 adults from NHANES 2011–2020. We investigated the association between NPAR levels and CVD using weighted generalized linear models, subgroup analysis, threshold effects, receiver operating characteristic (ROC) curves, and sensitivity analysis. Restricted cubic splines were used to assess potential nonlinearity in the NPAR-CVD association. Elevated NPAR levels were significantly associated with increased CVD prevalence (P < 0.001). In model 3, individuals in the highest NPAR quartile had a 46% greater CVD prevalence compared to those in the lowest quartile [OR: 1.46 (1.16, 1.83), P = 0.002], a finding consistent across unweighted logistic regression models. This association remained unchanged by various factors (P > 0.05). While restricted cubic spline analysis indicated potential nonlinearity, it did not significantly improve model fit over the linear model (P > 0.05). This study identifies a significant association between NPAR and CVD prevalence among the broader populace, suggesting the potential influence of inflammation on cardiovascular disease.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVD) remained the leading cause of death globally in 2021, responsible for nearly one-third of all fatalities1. The 2023 American Heart Association (AHA) report shows that global CVD mortality rose by 18.7% in 2020 compared to 20102. Given its profound impact on public health and the global economy, early identification and prediction of CVD are essential.
Although conventional risk factors including diabetes, hypertension, and hyperlipidemia are strong predictors of cardiovascular risk, they may not fully account for the variability in CVD risks3,4,5. Recent studies suggest that inflammation and nutritional status are key in atherosclerosis and CVD progression6,7,8.
The neutrophil percentage to albumin ratio (NPAR) is an emerging biomarker that integrates two critical factors: neutrophil percentage, an indicator of systemic inflammation, and albumin, a marker of nutritional status. Neutrophils are a key component of the immune response, and their involvement in inflammation has been linked to coronary artery disease9. Low albumin levels, on the other hand, are associated with poor cardiovascular outcomes and overall mortality10,11. Combining these two markers, NPAR provides a more comprehensive assessment of inflammation and nutrition, integral to cardiovascular health.
Previous studies have linked NPAR with adverse outcomes such as COPD, cancer, and acute kidney injury12,13,14. In addition, NPAR has been shown to outperform either neutrophil percentage or albumin alone in predicting mortality in patients with cardiogenic shock15. However, there is limited research on the association between NPAR and CVD in the overall adults of the United States.
This research aims to evaluate the relationship between NPAR and CVD prevalence using information from the National Health and Nutrition Examination Survey (NHANES). By exploring NPAR’s potential as a predictive biomarker, this research seeks to provide insights into more effective strategies for early CVD prevention.
Methods
Study population
For intricate sample weight computations, we adhered to NHANES rules to guarantee reliable and representative statistical analysis.
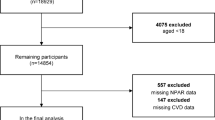
Our study analyzed information from 45,462 NHANES individuals from 2011 to 2020. We excluded 19,182 individuals under 20 years of age, 5 individuals with inadequate cardiovascular disease questionnaire data, and 50 individuals with missing education or smoking status. In total, 26,225 individuals were included in the final analysis.
Figure 1 presents a detailed flow chart illustrating the participant selection process for the study.
Definition of the NPAR and CVD
The neutrophil percentage was obtained using the NHANES CBC Profile. The NHANES Standard Biochemistry Profile was used to calculate the albumin value. The dye bromcresol purple (BCP) is used in the albumin concentration measurement technique. The dye changes color at 600 nm when it binds to albumin preferentially in the pH range of 5.2–6.8. 700 nm is the secondary wavelength. This is an albumin-specific, two-point endpoint reaction.
NPAR is calculated by the formula: Neutrophil percentage (%) × 100/Albumin (g/dL).
The survey questionnaire responses from the participants in this study were used for defining CVD. If a participant reported having angina, myocardial infarction (MI), congestive heart failure (CHF), coronary heart disease (CHD), or stroke, a diagnosis of CVD was taken into consideration. As a result, any positive response to these particular requirements was taken to mean that CVD was present16,17.
Covariates
This study examined a range of variables and clinical conditions. Three levels of education were distinguished: underhigh school, secondary school or its equivalent, and colleges or higher. BMI was classified as ordinary (< 25 kg/m2), overweight (25–29.9 kg/m2), and obese (≥ 30 kg/m2). The family income-to-poverty ratio (PIR) was divided into three groups: poor, intermediate, and wealthy18. eGFR was calculated using creatinine-based equations19. Excessive alcohol consumption was defined as ≥ 4 drinks/day for men and ≥ 3 drinks/day for women. Moderate drinking was 3 drinks/day for men and 2 drinks/day for women, while lower intake was classified as minimal (< 3 drinks/day for men, < 2 drinks/day for women)20. The criterion for determining smoking status was a history of smoking more than 100 cigarettes in a year21. Total cholesterol > 200 mg/dL, triglycerides ≥ 150 mg/dL, high-density lipoprotein (HDL) ≤ 40 mg/dL in males and < 50 mg/dL in females, or low-density lipoprotein (LDL) ≥ 130 mg/dL were considered hyperlipidemia22,23. Hypertension was defined as an average systolic blood pressure (SBP) ≥ 140 mmHg, diastolic blood pressure (DBP) ≥ 90 mmHg, use of antihypertensive medication, or a documented history of hypertension. Diabetes was diagnosed if fasting plasma glucose was ≥ 126 mg/dL, glycated hemoglobin (HbA1c) was ≥ 6.5%, or if the participant used insulin or hypoglycemic medications.
Statistical analysis
Constant variables were presented as means with 95% confidence intervals (CIs), and classified variables as proportions with 95% CIs. For the study of baseline characteristics, individuals were separated into NPAR quartiles. Weighted generalized linear models were used to evaluate the association between NPAR and CVD. Model 1 was unadjusted, while Model 2 adjusted for demographic factors such as sex, age, and ethnicity. Model 3 further adjusted for sex, age, ethnicity, BMI, PIR, education, habit of smoking, drinking habits, hypertension, hyperlipidemia, and diabetes. Model 4 was adjusted for sex, age, ethnicity, BMI, PIR, education, habit of smoking, drinking habits, hypertension, LDL-C, and diabetes. Restricted cubic splines were used to analyze nonlinear associations, threshold effects investigation of inflection points and the association of NPAR and CVD. To assess the robustness of the NPAR-CVD association, weighted subgroup analyses were conducted, with unweighted logistic regression used for validation. Model discrimination was evaluated using receiver operating characteristic (ROC) curves, and area under curves (AUCs) comparisons were made with the DeLong test. All statistical analyses were conducted in R Studio (v4.2.2) and Empower Stats (v4.1), with statistical significance set at a two-sided P-value < 0.05.
Results
Characteristics of individuals
All of the 26,225 participants in this research, 51.59% were female and 48.41% were male, with an average age of 49.9 ± 17.7 years. The median NPAR was 1372.7, and the prevalence of CVD was 11.4%. Individuals were grouped into quartiles depending on NPAR. A noteworthy pattern revealed rising CVD prevalence throughout NPAR quartiles. (P < 0.001). Important differences in Demographic, lifestyle, and clinical factors were noted among the quartiles (P < 0.05) (Table 1).
Association of the NPAR with CVD
Table 2 demonstrates a strong positive association between CVD and NPAR. In every model, the results were statistically significant (P < 0.05), this association holds consistent whether NPAR is viewed as a constant variable or as a classification variable. The CVD prevalence was consistently greater in the fourth NPAR quartile than in the first in all models. The CVD prevalence in the fourth quartile of the fully adjusted Model 3 was 46% greater than that in the first quartile [OR 1.46, 95 %CI (1.16, 1.83), P = 0.002]. Even after replacing hyperlipidemia with LDL-C in model 4, cardiovascular disease prevalence remained 40% higher in the fourth quartile of NPAR compared to the first quartile [1.40 (1.12, 1.75), P = 0.005]. Additionally, every model’s trend test has statistical significance (P < 0.05).
Furthermore, a nonlinear association between NPAR and CVD prevalence is depicted in Fig. 2.
Subgroup investigation of the association between the NPAR and CVD
A subgroup analysis based on clinical, lifestyle, and demographic variables was planned to evaluate the association between NPAR and CVD.
Interaction analyses revealed statistically significant differences in NPAR across subgroups defined by PIR and alcohol consumption (P < 0.05). In contrast, other factors did not significantly modify the positive association between NPAR and CVD (P > 0.05) (Table 3).
Sensitivity analysis
The positive association between NPAR and the prevalence of CVD in all models is demonstrated by the sensitivity analysis employing unweighted logistic models in Table 4 (P < 0.05). The outcomes in Table 2 agree with these findings.
Log-likelihood ratios indicated no significant improvement in model fit (Table S1).
Incremental value of the NPAR in predicting CVD
Figure 3 evaluates the added predictive value of albumin, neutrophil percentage, and NPAR to a baseline model with conventional CVD risk factors (age, sex, hypertension, diabetes, hyperlipidemia). Adding NPAR increased the AUC from 0.825 to 0.830, though there was no statistically significant difference. (P = 0.349)
Discussion
To our knowledge, this is the first research to investigate the association between cardiovascular disease prevalence and the novel biomarker NPAR. In this study, which used data from 26,225 participants in the NHANE database from 2011 to 2020, high NPAR was associated with a significant increase in cardiovascular prevalence even as a classification and constant variable (P < 0.05) after basic clinical, lifestyle, and demographic variables were taken into account. The stability of our results was further verified by the sensitivity analysis results, which were identical to the weighted results (P < 0.05).
Neutrophils are involved in every stage of the atherosclerotic process, they promote platelet adhesion, activate macrophages and endothelial cells, increase monocyte recruitment, and start the plaque formation process9,24. Furthermore, risk factors like obesity and diabetes raise the risk of cardiovascular disease by boosting the inflammatory response25,26. Research indicates that hypoalbuminemia is a separate risk factor for CVD. Albumin can sustain plasma colloid osmotic pressure and has anti-inflammatory, antioxidant, and anticoagulant properties10,27. NPAR is a new biomarker that combines albumin and neutrophil percentage. It is derived from routine blood and biochemical tests and is easy to use and repeat. Compared to albumin and neutrophil percentage alone, NPAR has a synergistic amplification effect that enhances the ability to evaluate a patient’s risk for cardiovascular disease. According to a study, NPAR is a separate risk factor for stroke-associated pneumonia and spontaneous intracerebral hemorrhage28. A retrospective cohort analysis revealed that among patients undergoing peritoneal dialysis, the risk of cardiovascular death was 1.57 times higher and the possibility of death of all causes was 1.51 times higher in the highest NPAR quartile than in the lowest quartile29. A follow-up study of 1,141 atrial fibrillation patients older than 80 years found a positive relationship between NPAR and the rates of cardiovascular and all-cause death after 28 days30.
Nonetheless, little study has examined the association between NPAR and CVD among the broader populace. Our results demonstrate that the association between cardiovascular disease prevalence and increasing NPAR is stable across various subgroup analyses, indicating that these factors do not influence the observed association (P > 0.05). In contrast, results from the PIR and drinking subgroups suggest that both factors may affect the NPAR-CVD association. A low-income poverty ratio was significantly associated with higher CVD prevalence, while the middle-income group also showed increased prevalence. No significant association was found for the high-income group, indicating that low-income individuals may be more vulnerable to adverse socioeconomic factors such as limited healthcare access and unhealthy lifestyles, increasing their CVD risk31. Thus, future interventions should target reducing the socioeconomic impact of CVD, particularly in low-income populations. Additionally, moderate alcohol consumption was significantly associated with increased CVD prevalence, while light and heavy drinking showed no significant link. This may be due to the cardiovascular effects of exceeding moderate alcohol intake, such as acute hypertension, arrhythmias, and chronic inflammation32. Additionally, drinking patterns, frequency, and unadjusted confounders could influence these outcomes. Light drinking may not reach the threshold for harm, and the sample size for heavy drinkers may have been insufficient to detect an effect. Future studies should explore the long-term impact of different drinking behaviors on CVD risks. And, threshold effect analysis indicated that, although a significant association was observed below the point, the log-likelihood ratio test (P > 0.05) suggests that the nonlinear effect between NPAR and CVD may be limited to specific subgroups or NPAR levels. Future studies should explore the underlying mechanisms between varying NPAR levels and CVD.
Among the many advantages of our study is that it is the first to investigate the association between NPAR and CVD in the general public. Second, our model fully accounts for any confounders. Third, we utilized information from the NHANES database, which includes information from 26,255 participants, strengthening the robustness of our findings. Nevertheless, there are several restrictions in our research. First, as a cross-sectional study, it is difficult to determine the cause relationships between NPAR and CVD, and prospective studies are needed to validate these findings. Second, NPAR changes could not be tracked over time, and relying on a single hematologic measurement may introduce errors. Additionally, CVD diagnoses were based on self-report questionnaires, which may lead to bias and limit the accuracy of disease classification. Furthermore, we lacked key systemic inflammation markers (e.g., CRP, ESR) and cardiovascular biomarkers (e.g., troponin, natriuretic peptides), which may affect the precision of the NPAR-CVD association. Future research should include more comprehensive diagnostic tools and biomarkers to confirm these results.
Conclusion
In conclusion, our study confirms the positive association between NPAR and CVD prevalence in the general public. Our research emphasizes the significance of the association between NPAR and CVD prevalence and points out its potential as a new biomarker for assessing CVD risk.
Data availability
The data analyzed during the current study are available in the NHANES study (https://www.cdc.gov/nchs/nhanes/index.htm).
References
GBD 2019 Risk Factors Collaborators, Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1223–1249. https://doi.org/10.1016/s0140-6736(20)30752-2 (2020).
Tsao, C. W. et al. Heart Disease and Stroke Statistics-2023 update: A report from the American Heart Association. Circulation 147, e93–e621. https://doi.org/10.1161/cir.0000000000001123 (2023).
Glovaci, D., Fan, W. & Wong, N. D. Epidemiology of diabetes mellitus and cardiovascular disease. Curr. Cardiol. Rep. 21, 21. https://doi.org/10.1007/s11886-019-1107-y (2019).
Fuchs, F. D. & Whelton, P. K. High blood pressure and cardiovascular disease. Hypertension 75, 285–292. https://doi.org/10.1161/hypertensionaha.119.14240 (2020).
Sandesara, P. B., Virani, S. S., Fazio, S. & Shapiro, M. D. The forgotten lipids: triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk. Endocr. Rev. 40, 537–557. https://doi.org/10.1210/er.2018-00184 (2019).
North, B. J. & Sinclair, D. A. The intersection between aging and cardiovascular disease. Circ. Res. 110, 1097–1108. https://doi.org/10.1161/circresaha.111.246876 (2012).
De Backer, G. Epidemiology and prevention of cardiovascular disease: Quo Vadis? Eur. J. Prev. Cardiol. 24, 768–772. https://doi.org/10.1177/2047487317691875 (2017).
Kivimäki, M. & Steptoe, A. Effects of stress on the development and progression of cardiovascular disease. Nat. Rev. Cardiol. 15, 215–229. https://doi.org/10.1038/nrcardio.2017.189 (2018).
Silvestre-Roig, C., Braster, Q., Ortega-Gomez, A. & Soehnlein, O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 17, 327–340. https://doi.org/10.1038/s41569-019-0326-7 (2020).
Manolis, A. A., Manolis, T. A., Melita, H., Mikhailidis, D. P. & Manolis, A. S. Low serum albumin: A neglected predictor in patients with cardiovascular disease. Eur. J. Intern. Med. 102, 24–39. https://doi.org/10.1016/j.ejim.2022.05.004 (2022).
Ronit, A. et al. Plasma albumin and Incident Cardiovascular Disease: Results from the CGPS and an updated meta-analysis. Arterioscler. Thromb. Vasc Biol. 40, 473–482. https://doi.org/10.1161/atvbaha.119.313681 (2020).
Ferro, M. et al. Neutrophil percentage-to-albumin ratio predicts mortality in bladder cancer patients treated with neoadjuvant chemotherapy followed by radical cystectomy. Future Sci OA 7, Fso709. https://doi.org/10.2144/fsoa-2021-0008 (2021).
Wang, B., Li, D., Cheng, B., Ying, B. & Gong, Y. The Neutrophil percentage-to-albumin ratio is associated with all-cause mortality in critically ill patients with acute kidney Injury. Biomed. Res. Int. 2020, 5687672. https://doi.org/10.1155/2020/5687672 (2020).
Lan, C. C., Su, W. L., Yang, M. C., Chen, S. Y. & Wu, Y. K. Predictive role of neutrophil-percentage-to-albumin, neutrophil-to-lymphocyte and eosinophil-to-lymphocyte ratios for mortality in patients with COPD: Evidence from NHANES 2011–2018. Respirology 28, 1136–1146. https://doi.org/10.1111/resp.14589 (2023).
Peng, Y. et al. Association between neutrophil-to-albumin ratio and mortality in patients with cardiogenic shock: A retrospective cohort study. BMJ Open.10, e039860. https://doi.org/10.1136/bmjopen-2020-039860 (2020).
Shen, R. et al. The mediating role of depression in the association between socioeconomic status and cardiovascular disease: A nationwide cross-sectional study from NHANES 2005–2018. J. Affect. Disord. https://doi.org/10.1016/j.jad.2024.08.145 (2024).
Liu, A. B. et al. Metabolic syndrome and cardiovascular disease among adult cancer patients: Results from NHANES 2007–2018. BMC Public. Health 24, 2259. https://doi.org/10.1186/s12889-024-19659-4 (2024).
Zhou, H. et al. Linear association of compound dietary antioxidant index with hyperlipidemia: A cross-sectional study. Front. Nutr. 11, 1365580. https://doi.org/10.3389/fnut.2024.1365580 (2024).
Inker Lesley, A. et al. New Creatinine- and cystatin C–Based equations to Estimate GFR without Race. N. Engl. J. Med. 385, 1737–1749. https://doi.org/10.1056/NEJMoa2102953 (2021).
Phillips, J. A. Dietary guidelines for americans, 2020–2025. Workplace Health Saf. 69, 395. https://doi.org/10.1177/21650799211026980 (2021).
Paul, I. Update on prevalence of periodontitis in adults in the United States: NHANES 2009 to 2012. J. Periodontol. https://doi.org/10.1902/jop.2015.140520 (2015).
Nayili, M. et al. Association between systemic immunity-inflammation index and hyperlipidemia: A population-based study from the NHANES (2015–2020). Nutrients. https://doi.org/10.3390/nu15051177 (2023).
Third Report of the National Cholesterol Education Program (NCEP). Expert Panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III) final report. Circulation 106, 3143–3421 (2002).
Klopf, J., Brostjan, C., Eilenberg, W. & Neumayer, C. Neutrophil extracellular traps and their implications in cardiovascular and inflammatory disease. Int. J. Mol. Sci. https://doi.org/10.3390/ijms22020559 (2021).
Koenen, M., Hill, M. A., Cohen, P., Sowers, J. R. & Obesity Adipose tissue and vascular dysfunction. Circ. Res. 128, 951–968. https://doi.org/10.1161/circresaha.121.318093 (2021).
Poznyak, A. et al. The diabetes Mellitus-Atherosclerosis connection: The role of lipid and glucose metabolism and chronic inflammation. Int. J. Mol. Sci. https://doi.org/10.3390/ijms21051835 (2020).
Arques, S. Human serum albumin in cardiovascular diseases. Eur. J. Intern. Med. 52, 8–12. https://doi.org/10.1016/j.ejim.2018.04.014 (2018).
Lv, X. N. et al. Neutrophil percentage to albumin ratio is associated with stroke-associated pneumonia and poor outcome in patients with spontaneous intracerebral hemorrhage. Front. Immunol. 14, 1173718. https://doi.org/10.3389/fimmu.2023.1173718 (2023).
Yu, Y. et al. Neutrophil percentage-to-albumin ratio and risk of mortality in patients on peritoneal dialysis. J. Inflamm. Res. 16, 6271–6281. https://doi.org/10.2147/jir.S437256 (2023).
Jiasheng, C. et al. The Relationship between the Neutrophil percentage-to-albumin ratio and rates of 28-Day mortality in Atrial Fibrillation patients 80 years of age or older. J. Inflamm. Res. https://doi.org/10.2147/jir.S400924 (2023).
Stringhini, S. et al. Socioeconomic status and the 25 × 25 risk factors as determinants of premature mortality: A multicohort study and meta-analysis of 1·7 million men and women. Lancet 389, 1229–1237. https://doi.org/10.1016/s0140-6736(16)32380-7 (2017).
Biddinger, K. J. et al. Association of habitual alcohol intake with risk of cardiovascular disease. JAMA Netw Open 5, e223849. https://doi.org/10.1001/jamanetworkopen.2022.3849 (2022).
Acknowledgements
We are thankful to the NHANES for providing the database that enriched our research. Their valuable data contribution was a cornerstone in the development of our study.
Funding
This research was funded by the Anhui Medical University Research of China (Grant no. 2023xkj238), the Municipal Health and Wellness Committee 2023 Applied Medical Research Program of Hefei (Grant no. Hwk2023yb024) and The Sixth Batch of Renowned Physician Studios in Hefei City.
Author information
Authors and Affiliations
Contributions
R.W. and WJ.T. conceived the study design and are responsible for the overall content. R.W. and XB.C. analyzed and interpreted the data. HL.C. and TY.M. acquired the data. R.W. prepared the manuscript. All authors approved the submitted and final versions.
Consent for publication
All participants in the NHANES study provided consent for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
According to local laws and institutional standards, there was no need for ethical review or clearance for the study involving human subjects. To take part in this study, the patients/participants gave their written, informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, R., Tao, W., Chen, H. et al. Investigating nonlinear associations between neutrophil percentage to albumin ratio and cardiovascular disease: a nationally representative cross-sectional study. Sci Rep 14, 23632 (2024). https://doi.org/10.1038/s41598-024-75111-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-75111-y
Keywords
This article is cited by
-
Association between neutrophil percentage-to-albumin ratio and all-cause mortality in patients post-cardiac surgery: a retrospective cohort study
Molecular and Cellular Biochemistry (2026)
-
Association between neutrophil percentage to albumin ratio and chronic obstructive pulmonary disease in adult population: a cross-sectional study and verification
The Egyptian Journal of Bronchology (2025)
-
Correlation of neutrophil percentage-to-albumin ratio with lung function in American adults: a population study
BMC Pulmonary Medicine (2025)
-
Predictive role of neutrophil-percentage-albumin ratio (NPAR) in overactive bladder (OAB) in adults in the United States: a cross-sectional study
Journal of Health, Population and Nutrition (2025)
-
Neutrophil to albumin ratio predicts cardiovascular and all cause mortality in CVD patients with abnormal glucose metabolism
Scientific Reports (2025)